Abstract
In this chapter, we synthesize the state of the science regarding ecological subsidies and contaminants at the land-water interface and suggest research and management approaches for linked freshwater-terrestrial ecosystems. Specifically, we focus on movements of animals with complex life histories and the detrital inputs associated with animal and plant matter delivered to freshwaters. We present a framework based on the physicochemical parameters of contaminants and how they shape the relationship between contaminant persistence within resource subsidies (“dark side” of subsidies) and movement of resource subsidies (“bright side” of subsidies) across ecosystem boundaries. This relationship between the “dark side” and “bright side” of subsidies defines an important parameter space that allows researchers and practitioners to predict the potential impacts of aquatic contaminants on resource subsidies and their interaction with other stressors on consumers. Ecological factors such as ecosystem productivity, community composition, and consumer prey preference shape the ecotoxicological outcomes of aquatic contamination on subsidies. Landscape factors such as lithology, hydrogeomorphology, hydroperiod, and land use underlie chemical, toxicological, and ecological patterns and provide the context within which effects of contaminants play out. Finally, effects of contaminants combine with effects of other global stressors on timing, quality, and quantity of subsidies that drive responses to contaminants at the land-water interface. Understanding the “dark side” of ecological subsidies requires expertise from multiple disciplines. We attempt to synthesize current knowledge from those disciplines and generate conceptual models that ecologists can use to guide future research in understanding cross-ecosystem subsidies and contaminant fate and effects.
Creative thought on [food] webs…supports my optimism that the holy trinity of field observation, controlled experimentation and mathematical exploration can discover some common unity, and in the process provide important guidelines on how to save and even resurrect our increasingly battered natural world.
Paine (1996)
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Chemical contaminants influence ecological systems and patterns of species distribution at a global scale (Nriagu and Pacyna 1988; Rockstrom et al. 2009; Stehle and Schulz 2015; Beketov et al. 2013). Well-documented examples include pesticides that directly reduce insect biomass and contribute to declines in insectivorous birds (Carson 1962; Hallmann et al. 2014, 2017; Eng et al. 2019), and methylmercury that biomagnifies up the food chain to poison higher-order consumers, including humans (Harada 1995; Lavoie et al. 2010). In fact, chemical pollution resulting from contaminant inputs has such potentially large effects on ecosystem structure and health that it is identified as one of the planetary boundaries needed to maintain a safe operating space for humanity (Rockstrom et al. 2009). Despite these concerns, the use and manufacture of chemicals used in agriculture, manufacturing, medications, and cosmetics have increased exponentially over the past 60 years (Bernhardt et al. 2017). Furthermore, the ecological effects of these inputs of contaminants into the environment have been grossly understudied (Bernhardt et al. 2017). Thus, understanding and managing the extent and magnitude of the effects of contaminants on ecosystems will continue to be one of the major challenges of this century.
In this book, we identify two main pathways by which chemical contaminants affect ecosystems. First, contaminants affect ecosystems in contaminant bioaccumulation and biomagnification in food webs (Carson 1962). Contaminants that bioaccumulate within food webs, such as persistent organic pollutants (POPs) and organometals, can increase exposure in higher-order predators. Furthermore, the fate of these compounds may not be limited to the contaminated ecosystem, because dispersing organisms can transport these contaminants to other environments (Walters et al. 2008). The second pathway governs contaminant effects on ecosystems by increasing mortality and altering behavior of specific taxa (Rohr et al. 2006; Clements and Rohr 2009; Halstead et al. 2014), which can indirectly affect other members of the community. For example, contaminants such as trace metals and current-use pesticides reduce prey abundances, alter competitive dominance of sensitive taxa, and cause trophic cascades (Halstead et al. 2014; Rogers et al. 2016). Therefore, both ecological factors and physicochemical characteristics of contaminants determine effects of contaminants on aquatic ecosystems.
Aquatic ecosystems are particularly likely to receive inputs of chemical pollutants as well as allochthonous resources because of their relatively low topographical position in the landscape (Allan 2004). Water, sediment, and detritus tend to move downhill with gravity, along with associated contaminants. This transport of materials from land to water is long recognized to have important effects on ecosystem function, community dynamics, and species distributions. More recently, the movement of animals from water to land as part of their ontogeny (i.e., complex life cycles), or other means of dispersal (i.e., movement by predators or flooding), has been identified as an important conduit of energy and nutrients back to land (Muehlbauer et al. 2014; Schindler and Smits 2017). Theory describing these linkages, which merge paradigms from both landscape and food web ecology, is now being used to answer questions regarding the ecological effects of contaminants and food web structure on contaminant flux (Kraus 2019a, b; Chumchal and Drenner this volume; Sullivan and Cristol this volume).
In this chapter, we review heuristic models useful for predicting the effects of chemical contaminants on ecological subsidies at the land-water interface. These models allow us to generalize empirical findings to new environments; more efficiently utilize time and monetary resources by providing information about what, when, and where to measure effects; and to derive and test hypotheses that separate causal versus correlative agents of ecological change. We focus on effects of contaminants on production of animals with complex life histories, use and quality of terrestrial detritus to aquatic ecosystems, and the flux of contaminants through food webs both within and across ecosystem boundaries. We then construct a synthetic framework using conceptual control (a.k.a. programmatic) and food web models that pull together many of the factors and processes reviewed, including landscape, chemical/ecotoxicological, and ecological factors. Here we define control models as describing how subsidy endpoints are influenced by specific drivers and stressors, while conceptual food web models describe trophic relationships among organisms that drive resource and contaminant flux across ecosystem boundaries (Gentile et al. 2001; Miller et al. 2010). Finally, we use this framework to identify (1) patterns of contaminant effects on ecosystems, and (2) data gaps and areas for future research.
2 Review of Conceptual Models
One of the major patterns evident from efforts to predict effects of aquatic contaminants on linked aquatic-terrestrial food webs is that the physicochemical characteristics of contaminants determine the mechanism of potential effects on consumers (Kraus 2019a). For example, organometals and persistent organic pollutants like polychlorinated biphenyls (PCBs) are more likely to affect aquatic-terrestrial linkages by magnifying in bodies of adult insects and amphibians (i.e., “exposure” pathway, Kraus et al. 2014a; “subsidy drives exposure” sensu Walters et al. this volume). On the other hand, trace metals and some pesticides are more likely to sever aquatic-terrestrial linkages through direct effects on survival of animals with complex life histories through metamorphosis (i.e., “biomass” pathway, sensu Kraus et al. 2014a; “exposure drives subsidy” sensu Walters et al. this volume). As in the organization of this book, we invoke the physicochemical dichotomy of biomagnifying versus toxic contaminants as a heuristic starting point for reviewing the complex suite of factors identified as drivers of contaminant effects on ecological subsidies in linked aquatic-terrestrial ecosystems (Walters et al. this volume).
2.1 Ecology Predicts Ecotoxicology: Subsidy Drives Exposure
Contaminants that accumulate in resource subsidies and biomagnify in food webs can lead to potentially harmful exposures in subsidized consumers. The effects of these contaminants ripple through linked aquatic-terrestrial food webs by disrupting reproduction, endocrine functioning, and metabolism of higher-order consumers, as well as reducing survival of their young (e.g., Custer et al. 1998, 2003, 2005). Aquatic contaminants that biomagnify and persist in subsidies (e.g., adult aquatic insects) but do not tend to directly lower their production include methylmercury as well as many other persistent organic pollutants. For example, Chumchal and colleagues found that adult aquatic insects export methylmercury from ponds to riparian spiders (Speir et al. 2014; Chumchal and Drenner this volume). Methylmercury flux to riparian spiders was mediated by the emergence of small-bodied aquatic insect taxa (Tweedy et al. 2013). Furthermore, the maximum concentration of mercury detected in odonates emerging from these small ponds exceeded wildlife health values for red-wing blackbird nestlings (Williams et al. 2017). Similarly, Walters and colleagues found that PCBs accumulated in a diverse assemblage of riparian insectivores (spiders and herptiles) in proportion to their reliance on aquatic prey (Walters et al. 2008) and that these concentrations reflected PCB concentrations and congener profiles found in nearby river sediment (Walters et al. 2010;Kraus et al. 2017). Maximum total PCB concentrations found in riparian spiders were 14-times higher than risk values protective of bird health for chickadee nestlings (Walters et al. 2010). Finally, Christine and Tom Custer and colleagues have found bioaccumulation of multiple POPs including PCBs, dioxins and dichlorodiphenyl trichloroethane (DDT) and derivatives in diet, eggs, nestlings, tissues, and blood of tree swallows foraging for aquatic insects near industrially polluted rivers (Custer et al. 1998, 2003, 2005). Hatching success declined with summed concentration of dioxins and furans in eggs and newly hatched chicks (Custer et al. 2003).
Contaminants in aquatic ecosystems can be strong drivers of terrestrial food web exposure. However, other factors, besides contaminant type and concentration, can influence subsidy production, contaminant flux, and impacts on recipient food webs. Approaches including field surveys (Walters et al. 2008; Raikow et al. 2011), experiments (Chumchal and Drenner this volume), conceptual models (Chumchal and Drenner this volume; Otter et al. this volume), network models (Sullivan and Manning 2019; Sullivan and Cristol this volume), and meta-community frameworks (Schiesari et al. 2018) have explicitly incorporated ecological factors in predicting exposure resulting from subsidy-mediated contaminant flux. Specifically, these approaches have revealed that organism life history, community structure, ecosystem productivity, landscape structure, and disturbance are important predictors of subsidy-mediated exposure for contaminants that biomagnify and do not strongly reduce subsidy flux. For example, Chumchal and colleagues have shown that adult aquatic insect emergence from ponds and concomitant mercury flux is increased by nutrient enrichment (bottom-up control) but decreased by presence of top predators like fish (top down control; Jones et al. 2013; Tweedy et al. 2013; Chumchal and Drenner this volume). Community structure and composition was also altered by these factors (i.e., nutrient enrichment and predation), which combine with concentration of methylmercury at the base of the food web to influence the flux of methylmercury by adult aquatic insect prey (Tweedy et al. 2013). Sullivan and colleagues found that mercury and selenium body burdens of riparian spiders were more affected by riparian zone width, vegetation structure, and distance from hydrological disturbance than flux of these contaminants in adult aquatic insects (Alberts and Sullivan 2016). In the case of persistent organic pollutants, Walters and colleagues demonstrated that the distance that PCBs penetrate riparian habitats tracks patterns of insect dispersal away from water (Raikow et al. 2011). Thus, ecological factors strongly influence the patterns of contaminant flux, and thus terrestrial food web exposure, for these contaminants.
Certainly, study of the movement of contaminants through food webs is not new. American bald eagle populations famously crashed as a result of DDT accumulation from contaminated fish that caused egg shell thinning and population collapse (Grier 1982). Vultures across the Indian subcontinent met a similar fate as a result of renal failure after exposure to the anti-inflammatory drug diclofenac through eating contaminated dead livestock (Oaks et al. 2004; Green et al. 2004). In fact, the direct exposure effect of biomagnifying contaminants on higher-order consumers has often been the focus of studies on food web–related effects of contaminants, likely because of the similar position of these animals to humans in the food web. However, knowledge of subsidy-mediated exposure to contaminants has lagged. Recent efforts to fill this data gap have borne several conceptual models aimed at synthesizing findings.
Several approaches have been taken to predict the effects of biomagnifying contaminants on cross-ecosystems linkages. Sullivan and colleagues enumerated multiple food web pathways of biologically mediated flux of contaminants across the aquatic-terrestrial boundary (Sullivan and Rodewald 2012). They also tested factors driving the effects of this flux and illustrate how to model these factors using network theory (Sullivan and Rodewald 2012; Alberts and Sullivan 2016; Sullivan and Cristol this volume). For example, they show how changes in fish species composition caused by dam removal can reduce fish-mediated transfer of aquatic contaminants to terrestrial food webs (Sullivan and Cristol this volume). Kraus and colleagues used meta-analysis and qualitative models to look for patterns of transfer of contaminants across insect metamorphosis based on contaminant physicochemical properties (Kraus et al. 2014b, Kraus 2019. Persistent organic contaminants and organometals that are not highly toxic to aquatic insects at concentrations typically found in the environment are likely to affect terrestrial food webs via an “exposure” pathway (sensu Kraus et al. 2014a; Kraus 2019a, b). These compounds accumulate in larval insects and are retained through metamorphosis, in some cases bioamplifying to higher concentrations in adults as they lose body mass during metamorphosis (Kraus et al. 2014b). These increased contaminant concentrations can lead to higher contaminant flux mediated by resource subsidies and to greater toxic effects on consumers (Walters et al. 2010; Chumchal and Drenner 2015; Kraus 2019a, b).
Chumchal and Drenner (this volume) present a conceptual model based on their empirical work showing the preeminence of ecological factors (top predators, nutrient enrichment, and drying disturbance) in driving aquatic-insect-mediated flux of methylmercury from ponds to terrestrial predators. Schiesari et al. (2018) built on these findings using a meta-community perspective to model the role of dispersing organisms in directly or indirectly mediating contaminant flux across ecosystem boundaries. Multiple factors, including other contaminants, can modulate dispersal and alter contaminant fluxes (Schiesari et al. 2018). This pattern has led to several conceptual models showing insect-mediated contaminant flux as being driven by factors altering contaminant retention through metamorphosis and survival to metamorphosis (Chumchal and Drenner 2015; Kraus 2019a; Otter et al. this volume). Finally, Schultz and colleagues included contaminants in their review of environmental alterations propagating from aquatic to terrestrial ecosystems (Schultz et al. 2015). In their conceptual model, they highlighted the potential for cascading food web effects of insect-mediated contaminant flux as well as loss of aquatic insect prey because of aquatic contaminants (Schultz et al. 2015).
2.2 Ecotoxicology Predicts Ecology: Exposure Drives Subsidy
Contaminants affect production of adult aquatic insects and processing of terrestrial detritus in aquatic ecosystems. These impacts ripple through linked aquatic-terrestrial ecosystems, altering subsidies dynamics in food webs and ecosystems. For example, trace metal inputs resulting from widespread hard rock mining can be ubiquitous limiters of aquatic to terrestrial subsidies. Burdon (this volume) describes the role of acid mine drainage (mixtures of elevated concentrations of trace metals, major ions, and low pH) and agricultural land use in driving a productivity gradient in adult aquatic insect emergence from New Zealand streams. Burdon found that emergence was lowest from mining-impacted streams and that the shift in emergence production due to adult aquatic insect biomass explained >20% of the variance in riparian spider densities. Kraus and Pomeranz (this volume) similarly summarize findings from the United States showing a 97% reduction in adult aquatic insect emergence from mountain streams in the Colorado Mineral Belt (USA) resulting from highly elevated concentrations of bioavailable trace metals. This loss of insect prey coincided with a 75% reduction in riparian spider populations (also explaining ~20% of the variation in densities) observed across the landscape. Additionally, Kraus and Pomeranz (this volume) demonstrate how the diet of insectivorous stream trout shifts over this same stream-metal gradient, with consumption of terrestrial arthropod prey increasing 82% in the most contaminated streams (Kraus et al. 2016). Loss of resource subsidy exchange in linked aquatic-terrestrial food webs can lead to reduced frequency of trophic interactions, fewer interacting taxa, and altered food web stability (Sullivan and Manning 2019).
Insecticides and elevated concentrations of nutrients and major ions from salt pollution can also reduce quantity of resource subsidies to land and water. For example, Morrissey and colleagues found that adult aquatic insect emergence was greatly reduced by a commonly used class of agricultural insecticide, neonicotinoids (Morrissey et al. 2015; Cavallaro et al. 2018; Stanton et al. 2018). In a laboratory study of 6 neonicotinoid compounds, Raby et al. (2018) found that percentage of insects to complete adult emergence was the most sensitive life history endpoint for midges and mayfly species (based on EC50 concentrations) for 5 of the 6 compounds. Furthermore, emergence biomass was reduced by >90% at concentrations that killed only 50% of pre-emergent larvae. In a study of contaminant effects on detrital subsidies to streams, Evans-White et al. (this volume) concluded that elevated concentrations of salts and nutrients altered decomposition of terrestrial plant detritus. This change reduced local detrital subsidization of stream life and led to a shift in the community composition of microbial and insect detritivores (Evans-White et al. this volume).
Because of the negative effects of some contaminants on subsidy production, these contaminants can also reduce contaminant flux (the product of contaminant concentration in subsidy and subsidy biomass or production) across ecosystem boundaries (Kraus 2019a; Kraus and Pomeranz this volume). For example, the reduction in aquatic insect emergence caused by trace metal toxicity to larvae, combined with excretion of metals by emerged adults during metamorphosis, leads to lower insect-mediated metal flux from high metal streams (Kraus et al. 2014a). This phenomenon could have important, albeit untested, effects on contaminant flux in ecosystems containing contaminant mixtures whereby biomagnifying contaminants are also present. For example, in the Prairie Pothole Region of North America, wetlands accumulate atmospherically derived methyl mercury (Hall et al. 2009), and large fluxes of methylmercury (mass of methylmercury per surface area of water per day) can be exported from wetlands by adult aquatic insects (Chumchal and Drenner 2015). However, agricultural runoff to these wetlands, specifically inputs of insecticides, is known to directly reduce adult aquatic insect emergence (Raby et al. 2018). As a result, the effects of toxic insecticides on production of adult aquatic insect subsidies (i.e., direct mortality of larvae) could reduce methylmercury flux from these wetlands to linked terrestrial food webs.
Variation in species sensitivity to contaminants leads to changes in relative species abundance and diversity, as well as interactions among aquatic organisms (Clements et al. 2000; Clements and Newman 2002; Clements et al. 2010; Clements et al. 2016). Thus, species sensitivity can alter the effects of aquatic contaminants given the respective characteristics and consumption of resource subsidies in linked aquatic-terrestrial ecosystems (Kraus et al. 2014a; Sullivan and Manning 2019; Kraus and Pomeranz this volume). For example, mixtures of trace metals including Cu, Cd, Zn, and Pb structure stream communities in areas of historic mining such as the Colorado Mineral Belt (Clements et al. 2000). Some species of mayfly are more sensitive to the effects of dissolved metals, and concomitant low pH, compared to other taxa such as dipterans (Clements et al. 2000). As a result, emergence of mayflies drops more precipitously than emergence of dipterans over the same metals gradient (Kraus et al. 2014a). Moreover, riparian spiders that eat adult aquatic insects at these sites decline with availability of dipteran, but not mayfly prey (Kraus et al. 2014a). Thus, relative sensitivity of taxa, as well as their decline over the contamination gradient, affects the response of riparian predators to subsidies.
Sensitivity to contaminants also varies with life stage, which has strong implications for predicting effects of aquatic contaminants on adult aquatic insect emergence. For example, Wesner and colleagues as well as others have shown that effects of contaminants on adult aquatic insect emergence can be stronger than predicted by effects on larvae (Schmidt et al. 2013; Wesner et al. 2014; Henry and Wesner 2018; Wesner 2019; Raby et al. 2018; Kotalik et al. 2019). One potential mechanism responsible for this relationship appears to be enhanced mortality during metamorphosis, in which death occurs disproportionately in the penultimate developmental stage (e.g., pupal stage in holometabolous insects or wingpad stage in hemimetabolous insects) (Wesner et al. 2014; Raby et al. 2018; Wesner et al. this volume). This presents a challenge in using bioassessments based on larval insects alone to infer patterns of emergence, since larval populations may show only subtle changes in response to contaminants even as adult populations show precipitous declines (Schmidt et al. 2013; Raby et al. 2018; Wesner 2019). However, declines in adult abundances do not necessarily lead to declines in larval populations due to high fecundity and immigration of aquatic insects. For example, genetic analyses suggest that only a small number of successful adult matings are needed to sustain robust larval populations (Bunn and Hughes 1997). Future work modeling the stage-specific responses of insects to contaminants, perhaps using recently developed theory in population and community ecology on stage-structured dynamics (e.g., de Roos and Persson 2013), would improve our understanding of ontogenetic changes in contaminant sensitivity.
Several approaches have been developed that could be used to predict the effects of contaminants on resource subsidies. For example, Rohr and colleagues use community ecology theory to derive predictions regarding effects of chemical contaminants on different aquatic ecosystems (Rohr et al. 2006; Clements and Rohr 2009). They predict that in some cases, contaminants act like predators, depressing prey availability and potentially leading to indirect effects such as trophic cascades and apparent competition. An experimental test of these hypotheses uncovered that response of aquatic ecosystems to a mixture of agrochemicals was best predicted by direct effects of contaminant(s), reproductive rates, interaction strengths with other taxa, and links to ecosystem function of each functional group of species included in the experiment (Halstead et al. 2014). Other researchers similarly showed that community level effects of a widely used insecticide (bifenthrin) cascaded through the food web to alter adult aquatic insect emergence, reducing aquatic subsidies available to terrestrial consumers (Rogers et al. 2016).
Also applying community ecology theory to predict effects of chemical contaminants, Clements and colleagues developed hypotheses of how environmental gradients, such as previous chemical exposure, upstream-downstream community variation and disturbances, could shape community responses to contamination (i.e., context-dependency, Clements et al. 2012; Clements et al. 2016). Specifically, they suggested that disturbance would vary longitudinally (although not necessarily in a linear manner) based on the Intermediate Disturbance Hypothesis (Menge and Sutherland 1987). They expected contaminants to have the least effects (greater resilience) on communities within the watershed experiencing intermediate levels of disturbance (Kiffney and Clements 1996; Clements et al. 2016). In support of this hypothesis, they found early successional diatom communities in headwater streams in Colorado to be less sensitive to the direct effects of metal pollution than late successional diatoms in higher-order streams (Medley and Clements 1998). Although implications for aquatic-terrestrial linkages were not discussed, these patterns suggest that position within the watershed could partially determine the effects of contaminants on the production and use of resource subsidies (see Sect. 2.3 Other Considerations below for more on this point).
In contrast to the previous approaches, Kraus and colleagues use physicochemical properties of contaminants to predict the effects of chemical contaminants on aquatic-terrestrial linkages (Kraus et al. 2014b; Kraus 2019a, b). For example, trace metals, PAHs, and current-use insecticides that stress larval aquatic insects during their development drastically reduce emergence of adult aquatic insects (via larval mortality), thereby reducing the quantity of aquatic prey subsidies available to terrestrial consumers (Kraus et al. 2014a; Wesner et al. 2014; Kraus 2019a). Across contaminant classes, except for insecticides, they also suggest that classes more toxic to insects are less likely to persist in insect bodies across metamorphosis (Kraus 2019b). This relationship between direct toxicity and persistence of contaminant classes across metamorphosis drives the effects of aquatic contaminants on subsidized insectivores (Kraus 2019b). For directly toxic contaminants, Kraus’ framework suggests that reduction of emergence production, rather than increased exposure, will be the main mechanism by which insectivores of adult aquatic insects are affected by aquatic contamination.
2.3 Other Considerations
Resource exchange between aquatic and terrestrial ecosystems is a well-studied phenomenon in ecology (Minshall 1967; Polis et al. 1997; Baxter et al. 2005; Gratton and Vander Zanden 2009; Muehlbauer et al. 2014, see Collins and Baxter this volume for a brief overview). Theory describing animals with complex life histories and factors affecting timing and success of ontogenetic switch points (i.e., metamorphosis) is also well developed (Wilbur and Collins 1973; Wilbur 1980; Wilbur 1997; Schreiber and Rudolf 2008; de Roos and Persson 2013). Combined, these bodies of research may be usefully applied to predict the effects of aquatic contaminants on aquatic-terrestrial linkages.
As previously discussed, ecological drivers of subsidy production and use by consumers can govern contaminant effects on aquatic-terrestrial linkages. Polis and colleagues introduced the concept that productivity of the ecosystem producing the subsidy (donor) and ecosystem receiving the subsidy (recipient) would drive the direction and effects of resources subsidies across ecosystem boundaries (Polis et al. 1997, 2004). Specifically, they hypothesized that the effects of subsidies on consumers would increase with the ratio of donor to recipient production. In a meta-analysis, Marczak et al. (2007) found empirical evidence that consumer response to subsidies increased with the ratio of resource subsidies to alternative in situ prey. This concept suggests that contaminant flux in resource subsidies may have larger effects on consumers when resource subsidies are relatively more abundant than in situ resources.
Also building on the idea that relative productivity would influence consumer use and response to subsidies, Wipfli and Baxter (2010) presented a framework based on Vannote et al.’s (1980) River Continuum Concept. They predicted that the role of upstream, terrestrial, and marine allochthonous production in salmonid resource use would vary longitudinally within a watershed. They suggested that terrestrial resources would be more likely to support salmonid production in low-order streams, and that marine inputs become more important closer to the ocean. In line with this prediction, we would expect that the effects of aquatic contamination on the use of terrestrial inputs by salmonids would be more important in headwater streams. Several studies have found that reduction of aquatic prey by aquatic pollution can lead to increased reliance by fish on terrestrial insect subsidies in low-order streams (Kraus et al. 2016; Burdon et al. 2019). Thus, longitudinal position in the watershed could influence reliance of consumers on subsidies and increase the propensity of subsidies to mitigate effects of aquatic contamination.
Habitat shape or dimensions also affect subsidy fluxes across the aquatic-terrestrial boundary. For example, secondary production of macroinvertebrates is >5 times higher in streams than in lakes. However, an average-sized lake exports ~10 times higher insect emergence biomass than an average-sized stream in part, because an average stream width is smaller than an average lake radius (Gratton and Vander Zanden 2009). At the landscape scale, stream network geometry generates hotspots of adult aquatic insect flux, particularly at stream confluences (Sabo and Hagen 2012; Kopp and Allen 2019). Integrating the disciplines of freshwater geomorphology and subsidy production at the landscape scale (e.g., Schmidt et al. 2018) represents an exciting opportunity to predict cross-ecosystem contaminant flux.
Finally, for animals with complex life histories, larval conditions and environmental predictability can alter optimal timing and size at metamorphosis (Wilbur and Collins 1973). These factors can affect success of metamorphosis, future fitness, and subsidies to terrestrial consumers (Kotalik et al. this volume; Schreiber and Rudolf 2008; de Roos and Persson 2013). Contaminants can increase timing to metamorphosis if larval conditions are so stressful that animals cannot survive the total reorganization of body tissues (Clements et al. 2000; Kotalik and Clements 2019). Alternatively, if contaminants are pulsed, then naïve larvae that are developmentally mature enough to emerge, emerge earlier than noncontaminated larvae (Lee et al. 2016; Richmond et al. 2016; Rogers et al. 2016). Importantly, these physiological responses are predicated on the history of stress and previous exposure, as well as other interacting factors (including density and resource availability) that affect the timing, size, and ability of immature larvae or nymphs to metamorphose into an adult (Courtney and Clements 2000; Cain et al. 2006).
3 Synthesis Models
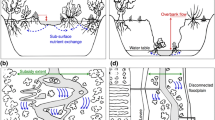
Multiple factors contribute to the biologically mediated flux of energy, nutrients, and contaminants across ecosystem boundaries (Fig. 1). To better understand the similarities and differences among these factors, we categorized them into four broad groups. Landscape factors are defined as those that shape the distribution of chemical contaminants and their bioavailability across the landscape. Ecotoxicological factors represent parameters associated with studies of the effects of chemical contaminants on organisms and environmental health including the chemical properties of contaminants and interaction of contaminants with the environment and living organisms. Ecological factors include measurements of organisms and their traits (e.g., both functional traits and physiological sensitivity) at different levels of organization. We group nutrients with ecological factors as they are a commonly measured parameter of ecological studies. To summarize the effects of these multiple factors on adult aquatic insect emergence, contaminant flux mediated by these insects, and impacts on terrestrial consumers, we employed a control model that expresses the directionality of control by “drivers” on ecosystem “endpoints” (Gentile et al. 2001).
Conceptual control model of major factors governing effects of aquatic contaminants on resource subsidies and subsidized consumers with a focus on freshwater-terrestrial linkages. Factors are grouped by category including landscape, ecotoxicological, and ecological and variable type. For variable type, squares are landscape drivers, circles are chemical stressors, diamonds are pathways or proximate drivers, and hexagons are endpoints or measured outcomes. Bold hexagons highlight focal endpoints. Arrows represent directionality of control. (See Table 1 for further details)
In general, the factors influencing contaminant effects on aquatic-terrestrial linkages are dependent upon the identity of contaminants within the study systems. This pattern arises because contaminants vary in the control pathways that dominate their effects on contaminant flux, subsidies, and consumers (Fig. 2, Table 1). For example, methylmercury does not tend to drive subsidy production but does drive contaminant flux by biomagnifying within the food web. For example, Chumchal and Drenner (this volume) show that pond permanency, nutrients, and predators alter mercury flux to terrestrial systems by changing the community composition, emergence production, and methylmercury concentration of adult aquatic insects emerging from ponds. Specifically, fish reduce emergence of large predatory dragonflies and damselflies (Odonata) but not smaller bodied dipterans, while nutrients increase overall emergence biomass. More ephemeral ponds are less likely to have fish, and thus have higher exports of mercury-laden predatory odonates and odonate-mediated adult emergence mercury flux. Meanwhile, the concentration of methylmercury available for uptake into freshwater food webs is codetermined by atmospheric (Hammerschmidt and Fitzgerald 2006), land-use, and organic factors such as presence of organic matter (dissolved organic carbon, DOC; e.g., Chaves-Ulloa et al. 2016), and anaerobic/redox conditions commonly linked to mercury methylation in freshwater ecosystems (Ullrich et al. 2001). The concentrations of methylmercury within food webs is also strongly influenced by trophic biomagnification, which leads to exponentially higher concentrations as the number of trophic levels within a food web increases. Thus, understanding the effects of mercury on cross-ecosystem linkages requires knowledge of the ecological, chemical, and landscape factors affecting aquatic productivity, food web structure, and mercury methylation (Fig. 2a).
Conceptual control model highlighting major factors found to govern effects of various aquatic contaminants on resource subsidies and subsidized consumers. Bold arrows and symbols represent pathways shown to be important in driving effects for methylmercury (a) as summarized in Chumchal and Drenner (this volume), trace metals (b) as summarized in Kraus and Pomeranz (this volume), and nutrients (c) as summarized in Evans-White et al. (this volume). Arrows represent directionality of control. (See Fig. 1 and Table 1 for further details)
On the other hand, at toxic concentrations, trace metals drive subsidy production and thus contaminant flux in mineralized streams in areas mined for hard-rock minerals (Paetzold et al. 2011; Kraus et al. 2014a; Kraus and Pomeranz this volume). Mayfly emergence decreases more rapidly across a concentration gradient of trace metals than emergence of aquatic dipterans (Kraus et al. 2014a). Furthermore, this relationship has been validated using stream mesocosm exposures on representative montane stream benthic assemblages (Kotalik and Clements 2019; Kotalik et al. 2019). The distribution and concentration of trace metals in these streams is determined by rock type, mineralization, and mining history (Wanty et al. 2009; Schmidt et al. 2012). In industrial and urban scenarios, trace metals inputs to freshwaters might be more linked to land use, point sources, and atmospheric sources (Nriagu 1996). However, in all cases, the effects of trace metals on benthic insects are influenced by metal concentration, water chemistry (which affects bioavailability), and species traits (Clements et al. 2000; Buchwalter et al. 2008). Thus, understanding the effects of trace metals on cross-ecosystem linkages requires focusing on lithology, land use, and water chemistry parameters affecting their bioavailability as well as taxon-specific sensitivity (Fig. 2b).
Finally, nutrients can function as both a subsidy and a contaminant in aquatic ecosystems (Evans-White et al. this volume). As previously mentioned, nutrients can increase emergence of aquatic insects, and thus resource subsidies to terrestrial insectivores (Chumchal and Drenner this volume). They can also increase the nutritive value of terrestrial detrital subsidies to aquatic ecosystems by decreasing the carbon to nitrogen ratio (C:N) on biofilms that colonize detritus before and during decomposition (Evans-White et al. this volume). At sufficiently high concentrations, however, nutrients become chemical contaminants of aquatic ecosystems. For example, high nitrogen and phosphorous runoff from agricultural fertilizers increases algal blooms, resulting in anoxic conditions in lakes and estuaries (Dodds 2006). In terms of terrestrial detrital subsidies to aquatic ecosystems, high nutrient concentrations can lead to rapid decomposition of detritus and release of associated carbon into the water column (Benstead et al. 2009; Rosemond et al. 2015; Evans-White et al. this volume). In streams, this pattern leads to increased rates of downstream export of the energy derived from these detrital subsidies, resulting in reduced resources for local detritivores (Evans-White et al. this volume). These effects are especially marked for less labile detritus (Ferreira et al. 2015), which depends in part on the species of plant contributing terrestrial organic carbon to the aquatic ecosystem (Tank et al. 2010). Thus, understanding the effects of nutrients as a contaminant on cross-ecosystem linkages requires knowledge of the landscape factors affecting nutrient concentrations and ecological factors influencing terrestrial plant species composition (Fig. 2c; Cross et al. 2003).
For all contaminants, their influence on aquatic-terrestrial linkages is constrained by traits and behaviors of resource subsidies and their consumers. For example, subsidies that are comprised of living organisms “choosing” to move between aquatic and terrestrial ecosystems, either as part of their development or foraging (i.e., “active” subsidies, sensu Kraus et al. 2011), respond directly to the effects of contaminants. These responses vary among species and can take the form of habitat selection, changed foraging decisions, increased contaminant bioaccumulation, reduced reproduction, and reduced survival through metamorphosis (Custer et al. 2003; Vonesh and Kraus 2009; Schmidt et al. 2013; Wesner et al. 2014; Kraus et al. 2014b, 2016). The effects of aquatic contaminants on active subsidies such as animals with complex life cycles are thus likely to be mediated by behavior, sensitivity to contaminants, and processing of contaminants by the subsidies. On the other hand, subsidies that are passively deposited or moved between ecosystems by factors external to themselves (i.e., salmon carcasses, leaf litter, or accidental input of terrestrial organisms into water) integrate the effects of contaminants on the living organisms that are their vectors of movement or responsible for their processing/consumption (Kraus et al. 2010). The effects of aquatic contaminants on passive subsidies are thus more likely to be mediated by contaminant effects on consumers of those resources. Therefore, predicting the response of subsidies and linked consumers to contaminants depends in part on understanding ecological responses of those organisms/resources.
To further help predict the effects of contaminant physicochemical properties in governing aquatic-terrestrial linkages, we built a food web model that can be applied to multiple contaminants and subsidy types (Fig. 3, Table 2). This model differs from our control model (Fig. 1) in that it shows energy and contaminant flow among focal taxa of the linked aquatic-terrestrial food web (Fig. 3a). Like the patterns illustrated in the control model, we suggest that the pathways of major energy and contaminant fluxes through the food web will be governed in part by contaminant identity and physiochemical properties (see Kraus 2019a, for more details). Specifically, we show how some contaminants can virtually eliminate aquatic prey for terrestrial consumers, leading to prey loss as the mechanism driving effects on terrestrial consumers (Fig. 3b). For detrital inputs, loss of aquatic prey for aquatic consumers can increase consumption and, reliance upon, terrestrial subsidies (Fig. 3b). Lastly, bioaccumulative aquatic contaminants can negatively impact terrestrial consumers of animals with complex life histories (e.g., insects) by exposing consumers to elevated concentrations of contaminants in prey (Fig. 3c). For terrestrial inputs to aquatic ecosystems, bioaccumulative contaminants can increase detrital contaminant concentrations either through adsorption or uptake by biofilms. However, alternative aquatic prey may also be exposed in these environments and accumulate contaminants, making aquatic food web exposure possible through either aquatic or terrestrial prey sources (Fig. 3c).
Simplified food web model of freshwater-terrestrial linkages in reference conditions (a) and affected by bioaccumulative (b) and toxic contaminants (c). Arrows represent directional flux of energy (black) and contaminants (red). Bioaccumulative contaminants are more likely to lead to consumer exposure, while toxic contaminants reduce availability of aquatic resources and may cause consumers to shift diets toward terrestrial prey or suffer population declines. Variation in weights of arrows represents general differences in relative magnitude of fluxes. (See Table 2 for more details)
4 Data Gaps
Mapping these control and mechanistic pathways reveals crucial management foci as well as data gaps in predicting the effects of aquatic contaminants on aquatic-terrestrial linkages. For example, further linking of information on transport of aquatic mercury by adult aquatic insects with information on the distribution and bioavailability of methylmercury in freshwaters would allow derivation of spatial predictions of biologically mediated mercury flux to terrestrial consumers across the landscape. Shape and hydrology of aquatic ecosystems would also influence these predictions. For lotic systems, researchers and managers might need to focus on downstream effects of mercury contamination (Otter et al. this volume), especially since streams and rivers tend to become wider downstream and may export more adult aquatic insects compared to lower-order streams (Gratton and Vander Zanden 2009). For lentic systems, surface area as well as the depth profile could affect flux of aquatic insects, and therefore mercury to land (Gratton and Vander Zanden 2009; Chumchal and Drenner 2015). Most information regarding the effects of trace metals on aquatic-terrestrial linkages comes from field and mesocosm studies of high elevation montane stream communities with adult aquatic insects and spiders as the focal subsidy/consumer (Kraus et al. 2016; Kotalik and Clements 2019; Kotalik et al. 2019). To understand how these patterns play out in different ecosystems, other ecological drivers such as subsidy production, community composition, and subsidy/consumer traits need to be considered. For example, environmental gradients such as disturbance (e.g., drought and flooding frequency) or longitudinal variation (e.g., low to high stream order) within a watershed can change the relative magnitude of resource subsidies and contaminant flux, as well as their impacts on consumers (Greenwood and McIntosh 2008; Wipfli and Baxter 2010; Schultz et al. 2015; Burdon this volume; Chumchal and Drenner this volume). Finally, while the effects of nutrient enrichment on aquatic ecosystems have generally been well studied, the effects of nutrients on cross-ecosystem linkages are less well known. For example, harmful algal blooms can create anoxic conditions that might lead to reduced emergence as well as increased export of algal toxins in adult aquatic insects (Moy et al. 2016).
The effects of other contaminants such as current-use pesticides, pharmaceuticals, and biotoxins (as well as contaminant mixtures) on aquatic-terrestrial linkages are poorly understood, and there are many gaps in the mapping of their pathways of effects. For example, in the case of current-use pesticides, Morrissey and colleagues have published field and laboratory studies on effects of neonicotinoids on emerging aquatic insects and insectivorous birds in the heavily agricultural Prairie Pothole region of Canada (Morrissey et al. 2015; Cavallaro et al. 2018; Stanton et al. 2018). However, no data have yet been published for other current-use pesticides or on current-use pesticide accumulation and transfer across metamorphosis for these insects. For pharmaceuticals and personal care products (POPs), as well as algal toxins, insect-mediated transfer of these compounds to aquatic and riparian birds and riparian spiders has only recently been documented (Moy et al. 2016; Richmond et al. 2018). However, effects of these transfers on insect consumers or on patterns of adult aquatic insect emergence are not well understood. Finally, Kraus and colleagues synthesized the current knowledge on transfer of contaminants across metamorphosis (Kraus et al. 2014b), but there are many contaminants that were either missing from this synthesis or were incompletely studied. Even metamorphic transfer of mercury, which is a particularly well-studied contaminant, was only measured in a few studies. In fact, transfer of methylmercury, which is the most common organic form of mercury found in animals, was only reported in one study.
In addition to contaminant-specific data gaps, we also lack knowledge about certain aspects of how aquatic contaminants alter aquatic-terrestrial linkages more broadly. In particular, the specific roles of multiple chemical contaminants in driving patterns observed in the environment require further study. For example, agricultural systems and wastewater effluent may contain multiple pesticides, nutrients, trace metals, and other contaminants. We expect that these contaminants will have interactive effects on aquatic-terrestrial linkages. Study of the role of aquatic insects in transferring these contaminants and contaminant mixtures across metamorphosis is in its infancy, as are the effects on detrital processing. But given their near-ubiquitous distribution and their known effects on aquatic ecosystems, greater understanding of their effect pathways on aquatic-terrestrial linkages will be crucial for researchers formulating questions and for resource practitioners managing risks in the environment. Mapping of multiple pathways of exposure and toxicity using our synthetic models could help with generating predictions under these complex scenarios. Other gaps in our knowledge include information on how disrupted aquatic-terrestrial linkages affect a broad suite of consumers. Specifically, what are the effects of shifting diets, contaminated prey, loss of prey, and change in prey taxonomic composition, and are there any generalizations to be made across ecosystems (as in Wipfli and Baxter 2010)? Finally, we lack knowledge about how regional/continental variation in the distribution of contaminants on the landscape impacts aquatic-terrestrial linkages and recipient consumers. Unanswered questions include: what are the effects of different contaminants across the landscape? How do regional patterns in contaminant distribution and bioavailability impact cross-ecosystem linkages? How do the effects of contaminants interact with other global stressors? Ultimately, including aquatic-terrestrial linkages and resource subsidy metrics (such as detrital endpoints and adult aquatic insect emergence) in regulatory and management decisions will be necessary to estimate “safe” contaminant concentrations for linked aquatic and riparian life.
5 Applications and Next Steps
One of the central themes that emerges from our conceptual control model is that understanding contaminant effects across ecosystem boundaries requires expertise from a wide range of scientific disciplines including geology, analytical chemistry, biogeochemistry, hydrology, ecology, physiology, statistical modeling, and landscape ecology, among others. As those of us who have tried to assemble these teams can attest, the most challenging aspect of building interdisciplinary teams is in finding experts who can communicate across disciplinary boundaries (Fiore 2008). We hope that our conceptual control model (Fig. 1) will improve communication by explicitly demonstrating how knowledge, for example, of lithography can help to predict the risk of metal contamination in riparian birds.
We could also view the conceptual model (Fig. 1) as a guide to risk assessment in the face of incomplete knowledge. For example, Dixon (2012, p. 191) used models like ours to derive a risk assessment of agriculturally applied chlorpyrifos, an organophosphate insecticide, to four species of birds without collecting new empirical data. The assessment used conceptual and mechanistic models to build a roadmap of how chlorpyrifos could move from seed coatings in corn fields to tissue concentrations in birds with different foraging traits. Dixon then quantified the risk to birds by assigning literature-derived values to each step along the roadmap, including studies that measured diet composition, consumption rate and foraging time of birds, dissipation rates of chlorpyrifos from plant surfaces, population size of seeds, and concentration-dependent mortality rates of birds to contaminants.
Similarly, we envision scientists using our conceptual and mechanistic models (Figs. 1, 2, and 3) as a roadmap to model cross-ecosystem contaminant flux. By assigning numerical information and associated uncertainty to factors in Figs. 1, 2, and 3, perhaps with a Monte Carlo analysis, teams could prioritize data collection to areas of high uncertainty, thereby maximizing impact and limiting cost. For example, if a monitoring survey revealed that a wetland contained 7 μg/l of total dissolved Se, we would be concerned, because that value is >4 times higher than the US EPA aquatic life criterion. Yet, there is considerable mechanistic uncertainty in the concentrations of Se that will move through food webs or across ecosystems to affect birds receiving subsidies from wetlands. A first step in resolving this uncertainty might be to derive literature estimates of the variation in the fraction of total dissolved Se that becomes bioavailable, Se retention through food webs, and the consumption rates of local birds on adult aquatic insects. In doing so, we might discover large uncertainties in the fraction of bioavailable Se, but lower uncertainties in the fraction retained through metamorphosis (presumably most of it is retained, Kraus et al. 2014b). This would allow researchers to focus sampling on factors that affect bioavailability, such as the fraction of Se that is organic versus inorganic (Wang and Lovell 1997).
Aquatic contaminants affect aquatic-terrestrial linkages in cryptic and nonintuitive ways. Understanding how these contaminants affect aquatic-terrestrial linkages will require the continued use of standard ecotoxicological methods, such as laboratory-based concentration-response testing, community mesocosm exposures, and field surveys. Yet, these approaches are unlikely to keep pace with the volume of new contaminants. As a solution, systematic approaches that combine fundamental principles from multiple disciplines into contextual and predictive models will be essential. Our hope is that the conceptual frameworks we present here, as well as this book, will help researchers and practitioners formulate questions and conduct studies that increase understanding of our complex and increasingly battered natural world (Paine 1996) and aid in efficient and effective management decisions protective of linked aquatic-terrestrial ecosystems.
References
Alberts JM, Sullivan SM (2016) Factors influencing aquatic-to-terrestrial contaminant transport to terrestrial arthropod consumers in a multiuse river system. Environ Pollut 213:53–62
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Evol Syst 35:257–284
Baxter CV, Fausch KD, Carl Saunders W (2005) Tangled webs: reciprocal flows of invertebrate prey link streams and riparian zones. Freshw Biol 50:201–220
Beketov MA, Kefford BJ, Schafer RB, Liess M (2013) Pesticides reduce regional biodiversity of stream invertebrates. Proc Natl Acad Sci U S A 110:11039–11043
Benstead JP, Rosemond AD, Cross WF et al (2009) Nutrient enrichment alters storage and fluxes of detritus in a headwater stream ecosystem. Ecology 90:2556–2566
Bernhardt ES, Rosi EJ, Gessner MO (2017) Synthetic chemicals as agents of global change. Front Ecol Environ 15:84–90
Bluth GJS, Kump LR (1994) Lithologic and climatologic controls of river chemistry. Geochim Cosmochim Acta 58:2341–2359
Buchwalter DB, Cain DJ, Martin CA, Xie L, Luoma SN, Garland T (2008) Aquatic insect ecophysiological traits reveal phylogenetically based differences in dissolved cadmium susceptibility. Proc Natl Acad Sci 105:8321–8326
Bunn SE, Hughes JM (1997) Dispersal and recruitment in streams: evidence from genetic studies. J N Am Benthol Soc 16:338–346
Burdon FJ (this volume) Agriculture and mining contamination contribute to a productivity gradient driving cross-ecosystem associations between stream insects and riparian arachnids. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Burdon FJ, McIntosh AR, Harding JS (2019) Mechanisms of trophic niche compression: evidence from landscape disturbance. J Anim Ecol 00:1–15
Cain DJ, Buchwalter DB, Luoma SN (2006) Influence of metal exposure history on the bioaccumulation and subcellular distribution of aqueous cadmium in the insect Hydropsyche californica. Environ Toxicol Chem Int J 25:1042–1049
Carson R (1962) Silent spring. Houghton Mifflin Company
Cavallaro MC, Liber K, Headley JV, Peru KM, Morrissey CA (2018) Community-level and phenological responses of emerging aquatic insects exposed to 3 neonicotinoid insecticides: an in situ wetland limnocorral approach. Environ Toxicol Chem 37:2401–2412
Chaves-Ulloa R, Taylor BW, Broadley HJ, Cottingham KL, Baer NA, Weathers KC, Ewing HA, Chen CY (2016) Dissolved organic carbon modulates mercury concentrations in insect subsidies from streams to terrestrial consumers. Ecol Appl 26:1771–1784
Chumchal MM, Drenner RW (2015) An environmental problem hidden in plain sight? Small human-made ponds, emergent insects, and mercury contamination of biota in the Great Plains. Environ Toxicol Chem 34:1197–1205
Chumchal MM, Drenner RW (this volume) Ecological factors controlling insect-mediated methylmercury flux from aquatic to terrestrial ecosystems: lessons learned from mesocosm and pond experiments. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Clements WH and Newman MC (2002) Community Ecotoxicology. John Wiley and Sons Ltd
Clements WH, Newman MC (2003) Community ecotoxicology. Wiley, Chichester
Clements WH, Rohr JR (2009) Community responses to contaminants: using basic ecological principles to predict ecotoxicological effects. Environ Toxicol Chem 28:1789–1800
Clements WH, Carlisle DM, Lazorchak JM, Johnson PC (2000) Heavy metals structure benthic communities in Colorado mountain streams. Ecol Appl 10:626–638
Clements WH, Vieira NK, Church SE (2010) Quantifying restoration success and recovery in a metal-polluted stream: a 17-year assessment of physicochemical and biological responses. J Appl Ecol 47:899–910
Clements WH, Hickey CW, Kidd KA (2012) How do aquatic communities respond to contaminants? It depends on the ecological context. Environ Toxicol Chem 31:1932–1940
Clements WH, Kashian DR, Kiffney PM, Zuellig RE (2016) Perspectives on the context-dependency of stream community responses to contaminants. Freshw Biol 61:2162–2170
Collins SF, Baxter CV (this volume) Beyond “donors and recipients”: impacts of species gains and losses reverberate among ecosystems due to changes in resource subsidies. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Courtney LA, Clements WH (2000) Sensitivity to acidic pH in benthic invertebrate assemblages with different histories of exposure to metals. J N Am Benthol Soc 19(1):112–127
Cross WF, Benstead JP, Rosemond AD, Wallace JB (2003) Consumer-resource stoichiometry in detritus-based streams. Ecol Lett 6(8):721–732
Custer CM, Custer TW, Allen PD, Stromborg KL, Melancon MJ (1998) Reproduction and environmental contamination in tree swallows nesting in the Fox River drainage and Green Bay, Wisconsin, USA. Environ Toxicol Chem 17:1786–1798
Custer CM, Custer TW, Dummer PM, Munney KL (2003) Exposure and effects of chemical contaminants on tree swallows nesting along the Housatonic River, Berkshire County, Massachusetts, USA, 1998–2000. Environ Toxicol Chem 22:1605–1621
Custer CM, Custer TW, Rosiu CJ, Melancon MJ, Bickham JW, Matson CW (2005) Exposure and effects of 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin in tree swallows (Tachycineta bicolor) nesting along the Woonasquatucket River, Rhode Island, USA. Environ Toxicol Chem 24:93–109
de Roos AM, Persson L (2013) Population and community ecology of ontogenetic development. Princeton University Press, Princeton
Debecker S, Dinh KV, Stoks R (2017) Strong delayed interactive effects of metal exposure and warming: latitude-dependent synergisms persist across metamorphosis. Environ Sci Technol 51:2409–2417
DeForest DK, Brix KV, Adams WJ (2007) Assessing metal bioaccumulation in aquatic environments: the inverse relationship between bioaccumulation factors, trophic transfer factors and exposure concentration. Aquat Toxicol 84:236–246
Dixon KR (2012) Modeling and simulation in ecotoxicology with applications in matlab and simulink. CRC Press, Boca Raton
Dodds WK (2006) Nutrients and the “dead zone”: the link between nutrient ratios and dissolved oxygen in the northern Gulf of Mexico. Front Ecol Environ 4:211–217
Eng ML, Stutchbury BJ, Morrissey CA (2019) A neonicotinoid insecticide reduces fueling and delays migration in songbirds. Science 365:1177–1180
Evans-White M, Entrekin S, Bauer C (this volume) When nutrients become contaminants in aquatic systems: identifying responses to guide terrestrial-derived detrital endpoint development for managers. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Ferreira V, Castagneyrol B, Koricheva J, Gulis V, Chauvet E, Graça MA (2015) A meta-analysis of the effects of nutrient enrichment on litter decomposition in streams. Biol Rev 90:669–688
Fiore SM (2008) Interdisciplinarity as teamwork: how the science of teams can inform team science. Small Group Res 39:251–277
Fornaroli R, Ippolito A, Tolkkinen MJ, Mykrä H, Muotka T, Balistrieri LS, Schmidt TS (2018) Disentangling the effects of low pH and metal mixture toxicity on macroinvertebrate diversity. Environ Pollut 235:889–898
Gentile JH, Harwell MA, Cropper Jr. W, Harwell CC, DeAngelis D, Davis S, Ogden JC, Lirman D (2001) Ecological conceptual models: a framework and case study on ecosystem management for South Florida sustainability. STOTEN 274:231–253
Graf N, Battes KP, Cimpean M, Dittrich P, Entling MH, Link M, Scharmüller A, Schreiner VC, Szöcs E, Schäfer RB (2019) Do agricultural pesticides in streams influence riparian spiders? Sci Total Environ 660:126–135
Graf N, Battes KP, Cimpean M, Entling MH, Frisch K, Link M, Scharmüller A, Schreiner VC, Szöcs E, Zubrod JP, Schäfer RB (2020) Relationship between agricultural pesticides and the diet of riparian spiders in the field. Environ Sci Eur 32:1–12
Gratton C, Vander Zanden MJ (2009) Flux of aquatic insect productivity to land: comparison of lentic and lotic ecosystems. Ecology 90:2689–2699
Green RE, Newton IA, Shultz S, Cunningham AA, Gilbert M, Pain DJ, Prakash V (2004) Diclofenac poisoning as a cause of vulture population declines across the Indian subcontinent. J Appl Ecol 41:793–800
Greenwood MJ, McIntosh AR (2008) Flooding impacts on responses of a riparian consumer to cross-ecosystem subsidies. Ecology 89:1489–1496
Grier JW (1982) Ban of DDT and subsequent recovery of reproduction in bald eagles. Science 218:1232–1235
Gurnell A, Lee M, Souch C (2007) Urban rivers: hydrology, geomorphology, ecology and opportunities for change. Geogr Compass 1:1118–1137
Hairston NG, Smith FE, Slobodkin LB (1960) Community structure, population control, and competition. Am Nat 94:421–425
Hall BD, Baron LA, Somers CM (2009) Mercury concentrations in surface water and harvested waterfowl from the prairie pothole region of Saskatchewan. Environ Sci Technol 43:8759–8766
Hallmann CA, Foppen RP, van Turnhout CA, de Kroon H, Jongejans E (2014) Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511:341–343
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, Stenmans W, Müller A, Sumser H, Hörren T, Goulson D (2017) More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS One 12:e0185809
Halstead NT, McMahon TA, Johnson SA, Raffel TR, Romansic JM, Crumrine PW, Rohr JR (2014) Community ecology theory predicts the effects of agrochemical mixtures on aquatic biodiversity and ecosystem properties. Ecol Lett 17:932–941
Hamelink J, Landrum PF, Bergman H, Benson WH (1994) Bioavailability: physical, chemical, and biological interactions. CRC Press Inc., Boca Raton
Hammerschmidt CR, Fitzgerald WF (2006) Methylmercury in freshwater fish linked to atmospheric mercury deposition. Environ Sci Technol 40:7764–7770
Harada M (1995) Minamata disease: methylmercury poisoning in Japan caused by environmental pollution. Crit Rev Toxicol 25:1–24
Hare L (1992) Aquatic insects and trace metals: bioavailability, bioaccumulation, and toxicity. Crit Rev Toxicol 22:327–369
Henry BL, Wesner JS (2018) Severing ties: different responses of larval and adult aquatic insects to atrazine and selenium. Environ Sci Technol 52:8848–8857
Iwata T (2007) Linking stream habitats and spider distribution: spatial variations in trophic transfer across a forest–stream boundary. Ecol Res 22:619–628
Jansson M, Hickler T, Jonsson A, Karlsson J (2008) Links between terrestrial primary production and bacterial production and respiration in lakes in a climate gradient in subarctic Sweden. Ecosystems 11:367–376
Jones TA, Chumchal MM, Drenner RW, Timmins GN, Nowlin WH (2013) Bottom-up nutrient and top-down fish impacts on insect-mediated mercury flux from aquatic ecosystems. Environ Tox Chem 32:612–618
Kaushal SS, Mayer PM, Vidon PG, Smith RM, Pennino MJ, Newcomer TA, Duan S, Welty C, Belt KT (2014) Land use and climate variability amplify carbon, nutrient, and contaminant pulses: a review with management implications. JAWRA J Am Water Resour Assoc 50:585–614
Kiffney PM, Clements WH (1996) Effects of metals on stream macroinvertebrate assemblages from different altitudes. Ecol Appl 6:472–481
Kopp DA, Allen DC (2019) Stream network geometry and the spatial influence of aquatic insect subsidies across the contiguous United States. Ecosphere 10:e02926
Kotalik CJ, Clements WH (2019) Stream mesocosm experiments show significant differences in sensitivity of larval and emerging adults to metals. Environ Sci Technol 53:8362–8370
Kotalik CJ, Cadmus P, Clements WH (2019) Indirect effects of iron oxide on stream benthic communities: capturing ecological complexity with controlled mesocosm experiments. Environ Sci Technol 53:11532–11540
Kotalik CJ, Schmidt TS, Mebane CA (this volume) Mesocosms to evaluate aquatic-terrestrial contaminant linkages using aquatic insect emergence: utility for aquatic life criteria development. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Kraus JM (2019a) Contaminants in linked aquatic–terrestrial ecosystems: predicting effects of aquatic pollution on adult aquatic insects and terrestrial insectivores. Freshwater Sci 38:919–927
Kraus JM (2019b) Applying the ecology of aquatic–terrestrial linkages to freshwater and riparian management. Freshwater Sci 38:917–918
Kraus JM, Pomeranz JPF (this volume) Cross-ecosystem linkages and trace metals at the land-water interface. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Kraus JM, Pletcher LT, Vonesh JR (2011) Variation in active and passive resource inputs to experimental pools: mechanisms and possible consequences for food webs. Freshwater Biol 56:491–502
Kraus JM, Schmidt TS, Walters DM, Wanty RB, Zuellig RE, Wolf RE (2014a) Cross-ecosystem impacts of stream pollution reduce resource and contaminant flux to riparian food webs. Ecol Appl 24:235–243
Kraus JM, Walters DM, Wesner JS, Stricker CA, Schmidt TS, Zuellig RE (2014b) Metamorphosis alters contaminants and chemical tracers in insects: implications for food webs. Environ Sci Technol 48:10957–10965
Kraus JM, Pomeranz JF, Todd AS, Walters DM, Schmidt TS, Wanty RB (2016) Aquatic pollution increases use of terrestrial prey subsidies by stream fish. J Appl Ecol 53:44–53
Kraus JM, Gibson PP, Walters DM, Mills MA (2017) Riparian spiders as sentinels of polychlorinated biphenyl contamination across heterogeneous aquatic ecosystems. Environ Toxicol Chem 36:1278–1286
Lamouroux N, Poff NL, Angermeier PL (2002) Intercontinental convergence of stream fish community traits along geomorphic and hydraulic gradients. Ecology 83:1792–1807
Lavoie RA, Hebert CE, Rail JF, Braune BM, Yumvihoze E, Hill LG, Lean DR (2010) Trophic structure and mercury distribution in a Gulf of St. Lawrence (Canada) food web using stable isotope analysis. Sci Total Environ 408:5529–5539
Lee SS, Paspalof AM, Snow DD, Richmond EK, Rosi-Marshall EJ, Kelly JJ (2016) Occurrence and potential biological effects of amphetamine on stream communities. Environ Sci Technol 50(17):9727–9735
Marczak LB, Thompson RM, Richardson JS (2007) Meta-analysis: trophic level, habitat, and productivity shape the food web effects of resource subsidies. Ecology 88:140–148
Medley CN, Clements WH (1998) Responses of diatom communities to heavy metals in streams: the influence of longitudinal variation. Ecol Appl 8:631–644
Menge BA, Sutherland JP (1987) Community regulation: variation in disturbance, competition, and predation in relation to environmental stress and recruitment. Am Nat 130:730–757
Miller DM, Finn SP, Woodward A, Torregrosa A, Miller ME, Bedford DR, Brasher AM (2010) Conceptual ecological models to guide integrated landscape monitoring of the Great Basin. US Geol Surv Sci Investig Rep 5133:134
Minshall GW (1967) Role of allochthonous detritus in the trophic structure of a woodland springbrook community. Ecology 48:139–149
Morin PJ (2009) Community ecology. Wiley, New Brunswick
Morrissey CA, Mineau P, Devries JH, Sanchez-Bayo F, Liess M, Cavallaro MC, Liber K (2015) Neonicotinoid contamination of global surface waters and associated risk to aquatic invertebrates: a review. Environ Int 74:291–303
Moy NJ, Dodson J, Tassone SJ, Bukaveckas PA, Bulluck LP (2016) Biotransport of algal toxins to riparian food webs. Environ Sci Technol 50(18):10007–10014
Muehlbauer JD, Collins SF, Doyle MW, Tockner K (2014) How wide is a stream? Spatial extent of the potential “stream signature” in terrestrial food webs using meta-analysis. Ecology 95(1):44–55. https://doi.org/10.1890/12-1628.1
Muehlbauer JD, Larsen S, Jonsson M, Emilson EJS (this volume) Variables affecting resource subsidies from streams and rivers to land and their susceptibility to global change stressors. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Nakano S, Murakami M (2001) Reciprocal subsidies: dynamic interdependence between terrestrial and aquatic food webs. Proc Natl Acad Sci 98:166–170
Newman MC, Jagoe CH (1996) Ecotoxicology: a hierarchical treatment. CRC Press, Boca Raton
Nriagu JO (1996) A history of global metal pollution. Science 272:223–224
Nriagu JO, Pacyna JM (1988) Quantitative assessment of worldwide contamination of the air, water, and soils with trace metals. Nature 333:134–139
Oaks JL, Gilbert M, Virani MZ, Watson RT, Meteyer CU, Rideout BA, Shivaprasad HL, Ahmed S, Chaudhry MJ, Arshad M, Mahmood S (2004) Diclofenac residues as the cause of vulture population decline in Pakistan. Nature 427:630–633
Otter R, Beaubien GB, Olson CI, Walters DM, Mills MA (this volume) Practical considerations for the incorporation of insect-mediated contaminant flux into ecological risk assessments. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Paetzold A, Smith M, Warren PH, Maltby L (2011) Environmental impact propagated by cross-system subsidy: chronic stream pollution controls riparian spider populations. Ecology 92:1711–1716
Paine RT (1996) Preface. In: Polis GA, Winemiller KO (eds) Food webs: integration of patterns and dynamics. Springer Science, Berlin
Poff NL, Bledsoe BP, Cuhaciyan CO (2006) Hydrologic variation with land use across the contiguous United States: geomorphic and ecological consequences for stream ecosystems. Geomorphology 79:264–285
Polis GA, Anderson WB, Holt RD (1997) Toward an integration of landscape and food web ecology: the dynamics of spatially subsidized food webs. Annu Rev Ecol Syst 28:289–316
Polis GA, Power ME, Huxel GR (2004) Food webs at the landscape level. University of Chicago Press, Chicago
Raby M, Zhao X, Hao C, Poirier DG, Sibley PK (2018) Chronic toxicity of 6 neonicotinoid insecticides to Chironomus dilutus and Neocloeon triangulifer. Environ Toxicol Chem 37:2727–2739
Raikow DF, Walters DM, Fritz KM, Mills MA (2011) The distance that contaminated aquatic subsidies extend into lake riparian zones. Ecol Appl 21:983–990
Richmond EK, Rosi-Marshall EJ, Lee SS, Thompson RM, Grace MR (2016) Antidepressants in stream ecosystems: influence of selective serotonin reuptake inhibitors (SSRIs) on algal production and insect emergence. Freshwater Sci 35(3):845–855
Richmond EK, Rosi EJ, Walters DM, Fick J, Hamilton SK, Brodin T, Sundelin A, Grace MR (2018) A diverse suite of pharmaceuticals contaminates stream and riparian food webs. Nat Commun 9:4491. https://doi.org/10.1038/s41467-018-06822-w
Rockström J, Steffen W, Noone K, Persson Å, Chapin FS III, Lambin EF, Lenton TM, Scheffer M, Folke C, Schellnhuber HJ, Nykvist B (2009) A safe operating space for humanity. Nature 461:472
Rogers HA, Schmidt TS, Dabney BL, Hladik ML, Mahler BJ, Van Metre PC (2016) Bifenthrin causes trophic cascade and altered insect emergence in mesocosms: implications for small streams. Environ Sci Technol 50(21):11974–11983
Rohr JR, Kerby JL, Sih A (2006) Community ecology as a framework for predicting contaminant effects. Trends Ecol Evol 21:606–613
Rosemond AD, Benstead JP, Bumpers PM, Gulis V, Kominoski JS, Manning DW, Suberkropp K, Wallace JB (2015) Experimental nutrient additions accelerate terrestrial carbon loss from stream ecosystems. Science 347(6226):1142–1145
Rosi-Marshall EJ, Royer TV (2012) Pharmaceutical compounds and ecosystem function: an emerging research challenge for aquatic ecologists. Ecosystems 15:867–880
Sabo JL, Hagen EM (2012) A network theory for resource exchange between rivers and their watersheds. Water Resour Res 48:1–17
Schiesari L, Leibold MA, Burton GA Jr (2018) Metacommunities, metaecosystems and the environmental fate of chemical contaminants. J Appl Ecol 55:1553–1563
Schindler DE, Smits AP (2017) Subsidies of aquatic resources in terrestrial ecosystems. Ecosystems 20(1):78–93. https://doi.org/10.1007/s10021-016-0050-7
Schmidt TS, Clements WH, Wanty RB, Verplanck PL, Church SE, San Juan CA, Fey DL, Rockwell BW, DeWitt EH, Klein TL (2012) Geologic processes influence the effects of mining on aquatic ecosystems. Ecol Appl 22:870–879
Schmidt TS, Kraus JM, Walters DM, Wanty RB (2013) Emergence flux declines disproportionately to larval density along a stream metals gradient. Environ Sci Technol 47:8784–8792
Schmidt TS, Van Metre PC, Carlisle DM (2018) Linking the agricultural landscape of the Midwest to stream health with tructural equation modeling. Environ Sci Technol 53:452–462
Schreiber S, Rudolf VH (2008) Crossing habitat boundaries: coupling dynamics of ecosystems through complex life cycles. Ecol Lett 11:576–587
Schultz R, Bundschuh M, Gergs R, Brühl CA, Diehl D, Entling MH, Fahse L, Froer O, Jungkunst HF, Lorke A, Schaefer RB (2015) Review on environmental alterations propagating from aquatic to terrestrial ecosystems. Sci Total Environ 538:246–261
Speir SL, Chumchal MM, Drenner RW, Cocke WG, Lewis ME, Whitt HJ (2014) Methyl mercury and stable isotopes of nitrogen reveal that a terrestrial spider has a diet of emergent aquatic insects. Environ Toxicol Chem 33:2506–2509
Stanton RL, Morrissey CA, Clark RG (2018) Analysis of trends and agricultural drivers of farmland bird declines in North America: a review. Agric Ecosyst Environ 254:244–254
Stehle S, Schulz R (2015) Agricultural insecticides threaten surface waters at the global scale. Proc Natl Acad Sci U S A 112:5750–5755
Sullivan SMP, Cristol DA (this volume) Ecological networks as a framework for understanding and predicting contaminant movement across the land-water interface. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Sullivan SM, Manning DW (2019) Aquatic–terrestrial linkages as complex systems: insights and advances from network models. Freshwater Sci 38:936–945
Sullivan SM, Rodewald AD (2012) In a state of flux: the energetic pathways that move contaminants from aquatic to terrestrial environments. Environ Toxicol Chem 31:1175–1183
Sullivan TJ, Webb JR, Snyder KU, Herlihy AT, Cosby BJ (2007) Spatial distribution of acid-sensitive and acid-impacted streams in relation to watershed features in the southern Appalachian Mountains. Water Air Soil Pollut 182:57–71
Tank JL, Rosi-Marshall EJ, Griffiths NA, Entrekin SA, Stephen ML (2010) A review of allochthonous organic matter dynamics and metabolism in streams. J N Am Benthol Soc 29(1):118–146
Townsend C, Doledec S, Scarsbrook M (1997) Species traits in relation to temporal and spatial heterogeneity in streams: a test of habitat templet theory. Freshw Biol 37:367–387
Tweedy BN, Drenner RW, Chumchal MM, Kennedy JH (2013) Effects of fish on emergent insect-mediated flux of methyl mercury across a gradient of contamination. Environ Sci Technol 47:1614–1619
Ullrich SM, Tanton TW, Abdrashitova SA (2001) Mercury in the aquatic environment: a review of factors affecting methylation. Crit Rev Environ Sci Technol 31:241–293
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137
Vonesh JR, Kraus JM (2009) Pesticide alters habitat selection and aquatic community composition. Oecologia 160:379–385
Walters DM, Fritz KM, Otter RR (2008) The dark side of subsidies: adult stream insects export organic contaminants to riparian predators. Ecol Appl 18:1835–1841
Walters DM, Mills MA, Fritz KM, Raikow DF (2010) Spider-mediated flux of PCBs from contaminated sediments to terrestrial ecosystems and potential risks to arachnivorous birds. Environ Sci Technol 44:2849–2856
Walters DM, Mills MA, Cade BS, Burkard LP (2011) Trophic magnification of PCBs and its relationship to the octanol−water partition coefficient. Environ Sci Technol 45:3917–3924
Walters DM, Jardine TD, Cade BS, Kidd KA, Muir DC, Leipzig-Scott P (2016) Trophic magnification of organic chemicals: a global synthesis. Environ Sci Technol 50:4650–4658
Wang C, Lovell RT (1997) Organic selenium sources, selenomethionine and selenoyeast, have higher bioavailability than an inorganic selenium source, sodium selenite, in diets for channel catfish (Ictalurus punctatus). Aquaculture 152:223–234
Wanty RB, Verplanck PL, San Juan CA, Church SE, Schmidt TS, Fey DL, DeWitt EH, Klein TL (2009) Geochemistry of surface water in alpine catchments in Central Colorado, USA: resolving host-rock effects at different spatial scales. Appl Geochem 24:600–610
Ward DM, Nislow KH, Folt CL (2012) Do low-mercury terrestrial resources subsidize low-mercury growth of stream fish? Differences between species along a productivity gradient. PLoS One 7:e49582
Wesner J (2019) Using stage-structured food webs to assess the effects of contaminants and predators on aquatic–terrestrial linkages. Freshwater Sci 38:928–935
Wesner JS, Kraus JM, Schmidt TS, Walters DM, Clements WH (2014) Metamorphosis enhances the effects of metal exposure on the mayfly, Centroptilum triangulifer. Environ Sci Technol 48:10415–10422
Wesner J, Kraus JM, Henry B, Kerby J (this volume) Metamorphosis and the impact of contaminants on ecological subsidies. In: Kraus JM, Walters DM, Mills MA (eds) Contaminants and ecological subsidies: the land-water interface. Springer, Cham
Wilbur HM (1980) Complex life cycles. Annu Rev Ecol Syst 11:67–93
Wilbur HM (1997) Experimental ecology of food webs: complex systems in temporary ponds. Ecology 78:2279–2302
Wilbur HM, Collins JP (1973) Ecological aspects of amphibian metamorphosis: non-normal distributions of competitive ability reflect selection for facultative metamorphosis. Science 182:1305–1314
Williams EB, Chumchal MM, Drenner RW, Kennedy JH (2017) Seasonality of odonate-mediated methylmercury flux from permanent and semipermanent ponds and potential risk to red-winged blackbirds (Agelaius phoeniceus). Environ Toxicol Chem 36:2833–2837
Wipfli MS, Baxter CV (2010) Linking ecosystems, food webs, and fish production: subsidies in salmonid watersheds. Fisheries 35:373–387
Wu F, Mu Y, Chang H, Zhao X, Giesy JP, Wu KB (2013) Predicting water quality criteria for protecting aquatic life from physicochemical properties of metals or metalloids. Environ Sci Technol 47:446–453
Acknowledgments
This work was funded by the USGS Contaminant Biology Program. This research was subjected to USGS review and approved for publication. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the US Government.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kraus, J.M., Wesner, J., Walters, D.M. (2020). Synthesis: A Framework for Predicting the Dark Side of Ecological Subsidies. In: Kraus, J.M., Walters, D.M., Mills, M.A. (eds) Contaminants and Ecological Subsidies. Springer, Cham. https://doi.org/10.1007/978-3-030-49480-3_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-49480-3_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-49479-7
Online ISBN: 978-3-030-49480-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)