Abstract
Pharmaceutical products continue to be developed for improved patient compliance, but the issue of hydrophobicity, low permeability, short half-life, and stability of various drugs always has been a challenging task for formulation development. Conventional formulations lack the features of prolong release of drug over an extended period of time and site-specific action. To resolve these issues, cyclodextrin has emerged as an exceptional pharmaceutical excipient with the features of bioavailability, nontoxicity, inclusion ability, and water solubility. Additionally, nanoparticles overcome the drawbacks associated with conventional formulations such as prolong release and site-specific action of active molecules. Cyclodextrins and chemically altered cyclodextrins in combination with certain polymers have sparked various researchers for exceptional advancements in the field of novel drug delivery. More recently, these substances have been incorporated in polymer systems to develop nanoparticles. Cyclodextrin conjugated nanoparticles offer synergistic advantages such as enhanced drug solubility, served as drug carriers to a specific target site such as cancer cells with minimum toxicity to normal cells, greater surface area over microparticles, and higher stability over liposomes. Accordingly, nanoparticles based on cyclodextrins have gained notable interest, and a variety of nanoparticles have been developed since the last two decades.
This chapter review major critical research on cyclodextrin-based nanoparticles to explain their versatility and high potential for advanced drug delivery, protein and peptide drug delivery, and gene delivery. This chapter also highlights the role of cyclodextrins in specific types of nanoparticles such as gold, silver, and magnetic, polymeric, and lipid-based nanoparticles. Finally, pharmaceutical applications of amphiphilic cyclodextrin nanoparticles and miscellaneous administration routes of cyclodextrin-based nanoparticles are also discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
7.1 Introduction
The presence of the hydrophobic cavity renders cyclodextrins able to form inclusion complexes. These molecules contribute distinguished advantages due to their novel architectural features to form inclusion complexes with several kinds of molecules like ions, protein, and oligonucleotides (Lysik and Wu-Pong 2003). Inclusion complexes are formed when the “guest” molecule usually a drug is partially or fully included inside the “host’s cavity” (Szente and Szejtli 1999). Owing to the hydrophobic cavity, cyclodextrins as ghosts offer the guest a suitable environment for interaction (Fig. 7.1). The outer hydrophilic surface of cyclodextrins is compatible with water, which allows hydrogen bonding cohesive interactions (Challa et al. 2005).
In 1891, Villiers first discovered the cyclodextrins. He determined a crystal structure called “cellulosine” from the isolation of starch after the digestion by Bacillus amylobacter. Then, at the beginning of the nineteenth century, Schardinger described two crystalline dextrins (α-cyclodextrin and β-cyclodextrin), which were separated from bacterial digestion of potato starch (Schardinger 1903; Crini 2014). Furthermore, Schardinger performed many experiments with cyclodextrins and was acknowledged as an originator of cyclodextrin chemistry by experts. In the decade of 1930, Freudenberg discovered γ-cyclodextrin, and he earned the credit of the first patent publication, including the first fundamental article on the role of cyclodextrins in drug formulations (Loftsson and Duchêne 2007). Till 1970 to present, cyclodextrin has been recognized as a valuable pharmaceutical excipient in the formulation development of numerous active pharmaceutical excipients, including novel drug delivery systems.
Therefore, to fulfill the need of the pharmaceutical industry for purified cyclodextrin, researchers have demonstrated several biotechnological techniques to obtain purified cyclodextrin products (Kurkov and Loftsson 2013). Literature reveals that cyclodextrins and their derivatives have been used in conventional and novel drug delivery systems such as emulsions, microspheres, liposomes, and nanoparticles. Following this, to overcome the issue of poor water solubility of natural cyclodextrins, several cyclodextrin derivatives were synthesized with the substitution of hydroxyl groups. Otherwise, cyclodextrins have been used to enhance the drug loading capacity of polymeric microspheres. Additionally, novel surface-active cyclodextrin derivatives have also been developed to impart a cutting edge in formulation development (Bilensoy et al. 2005).
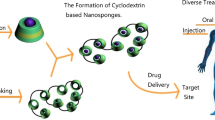
Cyclodextrins and their derivatives have been successfully employed to create novel nanomaterials. More recently, these products have been incorporated in polymer systems to develop nanoparticles that are suitable in the solubility of the hydrophobic drug. Cyclodextrin conjugated nanoparticles offer numerous advantages such as enhanced drug solubility, serve as drug carriers to a specific target site such as cancer cells with minimum toxicity to normal cells, greater surface area over microparticles, and higher stability over liposomes.
This chapter discusses on cyclodextrin-based nanoparticles to explain their versatility and high potential for advanced drug delivery, protein and peptide delivery, and gene delivery. It also highlights the role of cyclodextrins in specific types of nanoparticles such as gold, silver, and magnetic, polymeric, and lipid-based nanoparticles. Additionally, pharmaceutical applications of amphiphilic cyclodextrin nanoparticles and miscellaneous administration routes of cyclodextrin-based nanoparticles are also discussed.
7.2 Cyclodextrin Nanoparticles as Drug Delivery System
Since the nineteenth century, extraordinary scientific work has been done to explore the pharmaceutical potential of cyclodextrins, and they have been the subject of numerous scientific literature; cyclodextrins have been used in the pharmaceutical industry since the 1970s with the first product prostaglandin E2/β-cyclodextrins (Prostarmon ETM sublingual tablet) marketed in Japan in 1976. Cyclodextrins offer notable advantages due to their unique ability to form inclusion complexes with a variety of organic and inorganic lipophilic molecules. Cyclodextrins act as true carriers by dissolving and delivering hydrophobic drug molecules through the aqueous exterior of lipophilic biological membrane barriers, e.g., mucosa. In general, only dissolved drug molecules can partition into the barrier and then penetrate through it. In addition, cyclodextrins are known to self-assemble to form nanosized aggregates in aqueous solutions and thus have the potential of being developed into novel drug delivery systems (Messner et al. 2010). This characteristic is promising for a broad range of nanotechnology domains such as drug delivery, cancer therapy, gene delivery, and biosensing.
Cyclodextrins are advantageous in drug delivery because the bucket-shaped cavity shields the drug from degradation and irritation is lessened at the administration site. Cyclodextrin-based nanoparticles facilitate a novel drug delivery system with the advantages of both components: the cyclodextrin molecules offer enhanced water solubility and drug loading, while the nanoparticles afford targeted drug delivery. Additionally, cyclodextrins can surmount the constraints of nanoparticles such as low encapsulation efficiency and drug loading. Cyclodextrin-based nanoparticles yield a novel drug delivery system with the advantages of both components: the cyclodextrin molecules offer enhanced water solubility and drug loading, while the nanoparticles afford targeted drug delivery.
7.2.1 Anticancer Drugs
Oral administration of paclitaxel is still considered one of the most suitable and safe modes of delivery. Hamada et al. (2006) studied the aqueous solubility behavior of anticancer agent paclitaxel employing 11 kinds of cyclodextrins and the bioactivity of the paclitaxel-cyclodextrin inclusion complex. They have reported that 2,6-dimethyl-β-cyclodextrin was most effective, and paclitaxel showed significant solubility in 2,6-dimethyl-β-cyclodextrin aqueous solution. Moreover, this inclusion complex revealed a 1.23-fold polymerization activity as paclitaxel in a tubulin assay. Cancer therapy is currently changing from a standardized systemic strategy to more personalized and precisely customized disease therapy. Such personalized treatments use multifunctional drugs in association with drug carrier systems to target the molecular level at a specific site.
Nanotechnology-based drug delivery system provides an exceptional platform for the delivery of anticancer agents in order to enhance their targeting ability and bioavailability. One of the main advantages of loading anticancer drugs into nanoparticles is to enhance their cellular uptakes by bypassing the different multidrug-resistant mechanisms. Nanoparticle-based targeting of tumor cells has emerged as a potential therapeutic arsenal against cancer. For example, paclitaxel isolated from Taxus brevifolia is a potent anticancer agent approved for the treatment of a large number of solid tumors. But its hydrophobic nature results in low bioavailability.
Therefore, to overcome the issue of hydrophobicity, Bilensoy and co-workers have developed amphiphilic cyclodextrin as a nanoparticulate carrier system for paclitaxel drug delivery. This yielded nanospheres via nanoprecipitation technique with good cytotoxicity against L929 cells and was well studied using atomic force microscopy micrographs (Bilensoy et al. 2008a, b). Further studies demonstrated high encapsulation efficiency and a threefold increase in the loading capacity of nanoparticles when formed directly from the inclusion complex. Prolonged release rates for the drug were obtained: 12 h for nanospheres and 24 h for nanocapsules. In another approach Agüeros et al. (2009) investigated the concept of utilizing cyclodextrin-polyanhydride nanoparticles for oral delivery of paclitaxel. The addition of cyclodextrin increases the solubility by developing an inclusion complex, and the use of polyanhydride enhances the intestinal permeability. Additionally, they have also performed oral bioavailability studies in rodents by producing a synergistic effect using three distinct cyclodextrins, particularly β-cyclodextrin, 2-hydroxypropyl-β-cyclodextrin, and 6-mono-deoxy-6-monoamino-β-cyclodextrin in the combination of polyanhydride nanoparticles.
7.2.2 Proteins and Peptides
Various problems associated with the practical use of therapeutic peptides and proteins are their chemical and enzymatic instability, poor absorption through biological membranes, rapid plasma clearance, peculiar dose-response curves, and immunogenicity. Cyclodextrins, because of their adaptability in pharmaceutical use and ability to interact with cellular membranes, can act as potential carriers for the delivery of proteins, peptides, and oligonucleotide drugs (Irie and Uekama 1997). The combination between cyclodextrins and nanoparticles can also be of interest for the delivery of proteins and peptides. Cyclodextrin complexation represents an effective strategy for improving protein therapy by stabilizing them against aggregation, thermal denaturation, and degradation. Proteins are mostly hydrophilic and too bulky to be wholly included in the cavity of cyclodextrins. Nevertheless, the hydrophobic side chains in the peptides may penetrate into the cavity of the oligosaccharide, leading to the formation of non-covalent inclusion complexes, which improves the stability of proteins.
Da Silveira et al. (1998) have prepared and evaluated nanoparticulate systems of progesterone composed of poly(isobutyl cyanoacrylate) and cyclodextrins for enhancing the loading of the particles with substances. The nanoparticles were synthesized by polymerization of isobutyl cyanoacrylate in the presence of cyclodextrins or progesterone/hydroxypropyl-β-cyclodextrin complex. The particle size, zeta potential, and loading capacity of the particles were determined. The authors have demonstrated that nanoparticles could be efficiently prepared in the presence of cyclodextrins. Additionally, an increase in hydroxypropyl-β-cyclodextrin concentration resulted in small nanoparticles of size less than 50 nm. It was also observed that the addition of a higher concentration of cyclodextrins yields a 50-fold increase in progesterone loading compared to nanoparticles prepared without cyclodextrins. The poly(isobutyl cyanoacrylate)-cyclodextrin nanoparticles were delineated by the presence of many lipophilic sites belonging to the cyclodextrins which were tightly attached to the structure of the particles.
Sajeesh and Sharma (2006) reported the formulation development and evaluation of an oral insulin delivery system based on hydroxypropyl-β-cyclodextrin insulin complex encapsulated in polymethacrylic acid-chitosan-polyether (polyethylene glycol-polypropylene glycol copolymer). They have formulated the nanoparticles by the free radical polymerization of methacrylic acid in the presence of polymer chitosan and polyether in a solvent-/surfactant-free medium. Hydroxypropyl-β-cyclodextrin was incorporated to prepare a non-covalent inclusion complex with insulin. Furthermore, hydroxypropyl-β-cyclodextrin complexed insulin was entrapped into nanoparticles by diffusion filling technique, and their in vitro release profile was assessed at different pH. They found that nanoparticles exhibited satisfactory insulin encapsulation efficiency and pH-dependent release profile. An enzyme-linked immunosorbent assay study illustrated that insulin encapsulated inside the particles was biologically active. The mucoadhesive study of prepared nanoparticles was conducted using freshly extracted rat intestinal mucosa, and the nanoparticles were observed fairly adhesive to the mucosal membrane.
Recently, researchers have been focused on the nasal mucosa as an alternative route to the oral and parenteral routes, because it offers advantages such as large absorptive surface area and the high vascularity of the nasal mucosa, which facilitate direct passage of drug into the systemic circulation, thereby bypassing first-pass liver metabolism. However, the bioavailability of intranasally administered peptide and protein drugs may be low due to their high molecular weight and hydrophilicity (Merkus et al. 1991). Cyclodextrin has the ability to form non-covalent inclusion complexes with a large variety of drugs/proteins. Moreover, cyclodextrins are believed to enhance nasal absorption of peptides by opening tight junctions and/or solubilizing membrane components (Merkus et al. 1999).
Hyperbranched polyglycerols are readily available, highly water soluble, biocompatible, and stable polymer. Therefore, to explore these properties of hyperbranched polyglycerols, Zhang et al. (2011) fabricated a cyclodextrin and hyperbranched polyglycerols coupling based novel nanoparticle system to enhance the nasal transport of insulin. Insulin-loaded hyperbranched polyglycerol-β-cyclodextrin nanoparticles of size ranging from 198 to 340 nm with a positive charge were prepared. The nanoparticles showed a prominent capacity of incorporating insulin. In vitro release study showed significant release rate of insulin under acidic conditions than physiological conditions. In vitro cytotoxic evaluation against Caco-2 cells exhibited that hyperbranched polyglycerol-β-cyclodextrin had significant biocompatibility. Moreover, the capacity of hyperbranched polyglycerol-β-cyclodextrin nanoparticles to penetrate the nasal mucosal epithelia was proved by confocal laser scanning microscopy.
Three alkyl carbonates of γ-cyclodextrin (hexyl-,octyl-, and dodecyl-carbonate) have also been examined as a formulation additive to prepare solid nanoparticles for the effective delivery of progesterone. They have developed spherical nanoparticles in the size range of 80–200 nm. The prepared nanoparticles when subjected to evaluation showed negative surface charges and extended release rate of progesterone (Cavalli et al. 2007). Glutathione is the main thiolated small peptide in mammalian cells. It possesses reducing and nucleophilic properties which enable glutathione a key redox buffer to inhibit the oxidative damage caused by free radicals. Glutathione is used for the treatment of drug poisoning and protection against cytotoxic chemotherapy and radiation trauma. However, its clinical use encounters many hurdles associated with its low and variable oral bioavailability, non-enzymatic pH-dependent oxidation, and chemical and enzymatic degradation of the peptide in the jejunum. Hence, inclusion and preservation of glutathione into conventional pharmaceutical dosage forms are challenging tasks Currently, glutathione is only available on the market as parenteral dosage forms (Gluthion) (Langie et al. 2007). Therefore, to resolve these issues, Trapani et al. (2010) have developed new nanoparticles containing chitosan or cyclodextrin and evaluate their potential for oral delivery of the peptide glutathione. More precisely, nanoparticle formulations composed of chitosan, chitosan/alpha-cyclodextrin, and chitosan/sulphobutylether-β-cyclodextrin were investigated for this application. They have demonstrated that by selecting the most suitable cyclodextrin, physicochemical characteristics of the nanoparticles and their ability to load glutathione can be altered. Chitosan nanoparticles containing the anionic cyclodextrin sulfobutylether 7 m-β-cyclodextrin seem to be significant potential oral glutathione carriers, as they combine enhanced glutathione loading along with the ability to improve glutathione permeabilization through the intestine, as observed in a frog intestinal sac model.
Zhang et al. (2009) have demonstrated the feasibility of chitosan bearing β-cyclodextrin nanocomplexes for controlled protein release. Briefly, chitosan bearing β-cyclodextrin nanocomplexes was fabricated by a single-step scheme with N-succinylated chitosan and mono(6-(2-aminoethyl)amino-6-deoxy)-β-cyclodextrin along with water-soluble carbodiimide. The results of in vitro cytotoxicity against NIH 3 T3 cells confirmed that prepared nanocomplexes have not cytotoxic effect. In conclusion, these results proved chitosan bearing β-cyclodextrin copolymer as a novel carrier for controlled protein release.
More recently, He et al. (2019) reported a novel oral protein delivery system of ovalbumin with improved intestinal permeability and enhanced antigen stability. The delivery system based on the combination of chitosan nanoparticles and antigen-cyclodextrin inclusion complex was prepared by a precipitation/coacervation method. Results of the in vivo study of nanoparticles revealed that ovalbumin-loaded cyclodextrin/chitosan nanoparticles possess the capacity to induce an intestinal mucosal immune response.
7.3 Cyclodextrin Nanoparticles as Gene Delivery Systems
Gene therapy as an option to conventional drug therapy paves the new way for the treatment of both genetic and inherited diseases, such as hemophilia, cystic fibrosis, and cancers. The efficacy of a viral or non-viral vector directly determines the benefit of gene therapy. Gene therapy offer advantages over conventional protein therapy such as improved bioavailability and reduced systemic toxicity. Therefore, to avoid the toxicity issue of viral vectors, researchers have developed cyclodextrin-based nanoparticles as non-viral vectors. The unique characteristic of the presence of two well-differentiated faces in the cyclodextrins, specifically the narrower rim owning the primary hydroxyl groups and the wider rim positioning two chemically distinct sets of secondary hydroxyls, is an intrinsic characteristic of this family of cyclo-oligosaccharides that classifies them as genuine Janus (the Roman god of doorways and passages, depicted with two faces on opposite sides of his head) molecular nanoparticles.
In 1999, Gonzalez et al. (1999) published the first report of the synthesis of the cationic cyclodextrin polymers and the characterization of the cationic cyclodextrin nanoparticles loaded with plasmid DNA including their in vitro transfection efficiency. Teijeiro-Osorio et al. (2009) first reported the potential of a new generation of hybrid polysaccharide nanocarriers composed of chitosan and anionic cyclodextrins for gene delivery to the airway epithelium. They investigated hybrid polysaccharide nanocarriers to evaluate their ability to penetrate epithelial cells and improve gene expression in the Calu-3 cell culture model. Furthermore, hybrid chitosan and anionic cyclodextrin nanoparticles were developed and loaded with plasmid DNA model that encodes the expression of secreted alkaline phosphatase. Results of cellular uptake studies revealed that the nanoparticles were efficiently internalized by the cells and confirm their potential as gene vectors. The transfection ability of the various nanoparticle formulations showed a significantly higher response than the naked DNA (control). This resulted in enhanced delivery of DNA and reduces the otherwise high levels of protein expression in the respiratory mucosa.
Similarly, cyclodextrin-based polycations have also been studied for nucleic acid delivery. Park et al. (2006) have demonstrated a new strategy for surface-mediated gene delivery based on inclusion complex formation between the solid surface and delivery carrier. The complexation ability of β-cyclodextrin molecule was utilized to especially immobilize cyclodextrin-modified polyethylenimine nanoparticles, on adamantane-altered self-assembled monolayers. Cyclodextrin-modified polyethylenimine nanoparticles are particularly immobilized on the chip surface by cyclodextrin-adamantane inclusion complex formation. The results of competition studies with free cyclodextrins showed multivalent interactions between cyclodextrin-modified polyethylenimine nanoparticles and the adamantane-modified surface results in tremendous binding affinity. Therefore, the ability of cyclodextrin-modified polyethylenimine nanoparticles to form inclusion complexes can be employed to achieve specific, high-affinity loading of delivery carriers onto solid surfaces.
Huntington’s disease is an uncommon autosomal neurodegenerative disease provoked by the expression of a toxic huntingtin protein. The huntingtin gene renders instructions for producing a protein called huntingtin. This protein plays a vital role in neurons in the brain and is essential for healthy growth before birth. The application of short interfering RNAs (siRNAs) is a promising approach to restrict the mutation of protein. The main hindrance in siRNA-based strategies is the lack of efficient and nontoxic transportation vectors to ensure target delivery to the nervous system. This stimulated Godinho et al. (2013) to develop modified amphiphilic β-cyclodextrins as novel siRNA neuronal carriers. The results showed that the cyclodextrin formed nanosized particles were stable in artificial cerebrospinal fluid. Furthermore, these complexes significantly reduced the expression of the huntingtin gene in rat striatal cells and human Huntington’s disease primary fibroblasts. These findings firmly support the utility of modified β-cyclodextrins as safe and effective siRNA delivery vectors (Godinho et al. 2013).
Since the past few years, several strategies of low and high molecular weight polyethylenimines have been attempted for safe and effective DNA and siRNA delivery but were limited by transfection and cytotoxic activity. Therefore a copolymer-based delivery system has been developed by researchers. Ping et al. (2011) synthesized chitosan-graft-polyethylenimine-β-cyclodextrin copolymers for DNA and siRNA delivery. These two hydrophilic cationic copolymers were fabricated by a reductive amination reaction between oxidized chitosan and low molecular weight polyethylenimine-modified β-cyclodextrin. Furthermore, the characterization of nanoparticles was carried out by proton nuclear magnetic resonance spectroscopy and gel permeation chromatography. These polycations displayed significant ability to condense both plasmid DNA and small interfering RNA into compact and spherical nanoparticles. Additionally, both polymers delineate improved gene transfection activity performance in comparison with native CTS in HEK293, L929, and COS7 cell lines.
In the past decade, low and high molecular weight polyethylenimines have been extensively studied for DNA and siRNA delivery but have faced challenges of transfection and cytotoxic activity. A new strategy was suggested where polyplexes formed between polymers and DNA were further enhanced by the addition of β-cyclodextrin. In another laboratory work to facilitate the delivery of siRNA, cationic cyclodextrin conjugated with poly(ethylene glycol) (PEG) chain to expedite the attachment of targeting group anisamide. Parenteral administration of anisamide-tagged PEGylated cyclodextrin nanoparticles presented notable tumor inactivation with diminished toxicity when investigated preclinically in a rodent prostate tumor model. Hence, serving as an excellent drug delivery system of siRNA delivery for prostate cancer therapy (Guo et al. 2012). The siRNAs generally exhibit weak cell penetration with limited stability; the inclusion of cyclodextrins as a key excipient can aid in the delivery of oligonucleotides.
Accordingly, in light of these facts, Méndez-Ardoy et al. (2011) developed and evaluated polycationic amphiphilic cyclodextrins along with nucleic acids as therapeutic gene vectors for in vivo purpose. Briefly, a tetradecacationic structure consisting of 14 primary amino groups and 7 thioureido groups in the primary face of the cyclodextrin core and 14 hexanoyl chains in the second face was assessed for therapeutic gene delivery. The results obtained delineate that polycationic amphiphilic cyclodextrins having the capacities of self-assembling as a nanosystem for gene delivery, and when evaluated on HeLa and HepG2 cells, exhibited significant cell death with reduced toxicity. Furthermore, in vivo experiments performed on rats correlated with previous results that revealed high transfection levels in the liver with reduced toxicity. Usually, acquired resistance to a cytotoxic agent is mediated by overexpression of a membrane-associated protein that encodes via multidrug resistance gene-1.
Zokaei et al. (2019) recently developed chitosan β-cyclodextrin complexes as a tropical agent. These polymer cyclodextrin complexes loaded with the mRNA-cleaving DNAzyme that targets the mRNA of the multidrug resistance protein 1 (MDR1) gene in the doxorubicin-resistant breast cancer cell line. Results proved the downregulation of MDR1 mRNAs in MCF-7/DR/DNZ by a real-time polymer chain reaction, compared to the MCF-7/DR as control. Additionally, the results of the water-soluble tetrazolium salt 1 (WST1) assay displayed a significant decrease in drug resistance in mRNA-cleaving DNAzyme-treated cells 24 h after transfection. To sum up, results substantiate chitosan β-cyclodextrin complexes in association with chemotherapy drug for cancer therapy as well as notably valuable at the delivery of DNAzyme in reviving chemosensitivity.
7.4 Role of Cyclodextrin in Magnetic Nanoparticles
The most common class of substances used in the fabrication of theranostic nanostructures is carbon nanotubes, graphene, up-converting nanoparticles, and gold nanostructures. Nevertheless, the issue of nanotoxicity, complicated and time-consuming synthetic procedures, and poor stability represents the main obstacles for the therapeutic application of these nanomaterials. Therefore, magnetic nanoparticles offer several advantages over other types of nanomaterials, such as narrow size distribution, high colloidal stability, low toxicity, and high specific surface area to render them suitable for biomedical applications (Ahmed et al. 2014). Additionally, magnetic nanoparticles displayed the phenomenon of superparamagnetism: they are promptly magnetized under the influence of the external magnetic field and vice versa. This unique characteristic facilitates easy magnetic separation, removal, and recovery of magnetic nanoparticles. The before-mentioned magnetic property allows the nanoparticles to localize at the targeted site in vivo in response to the externally applied magnetic field. Silica is generally added to the surface of the nanoparticles to prevent their oxidation which leads to demagnetization which subsequently maintains the stability of magnetic nanoparticles.
Furthermore, to explore the versatility of magnetic nanoparticles, cyclodextrins are added into the silica-coated surface of the magnetic nanoparticles via linkers such as 3-aminopropyltriethoxysilane. Thus, allowing cyclodextrin to function as a carrier for numerous active pharmaceutical ingredients, proteins, and cell-targeting ligands on the surface of the magnetic nanoparticles. Moreover, it also prevents premature drug release into non-target regions. Wang et al. (2003) first proved the role of cyclodextrin to enhance the stability of magnetic nanoparticles in an aqueous medium. They have modified the surface properties of these nanoparticles through the formation of an inclusion complex between surface-bound surfactant molecules and α-cyclodextrin, thus improving its dispersion for a longer period in water. Furthermore, Xia et al. (2007) reported a simple pathway to synthesize water-soluble Fe3O4 nanoparticles with a surface-surrounded layer by incorporating cyclodextrin, polyethylene glycol, and nonylphenol ether in water. Additionally, a method for fabricating cyclodextrin concentration-dependent nanostructured spherical aggregates from individual magnetic nanoparticles was also described. This method is very significant because it instantly produces water-soluble magnetic nanoparticles and is useful for biomedical applications of magnetic nanoparticles.
To investigate the efficiency of cyclodextrin-based magnetic nanoparticles as a carrier for hydrophobic drug delivery, Banerjee and Chen (2007) have developed cyclodextrin-citrate-gum Arabic modified magnetic nanoparticles. Briefly, magnetic nanoparticles were synthesized by grafting the citrate-modified cyclodextrin along with gum Arabic modified magnetic nanoparticles through carbodiimide activation. The cyclodextrin grafting was validated by Fourier transform infrared spectroscopy. The practicability of using this novel formulation as a vehicle for hydrophobic drug delivery was described by examining the formation of the inclusion complex and the in vitro release profile using ketoprofen as standard hydrophobic drug delivery. The results of the experimental study showed that cyclodextrin-citrate-gum Arabic modified magnetic nanoparticles exhibited a considerable adsorption capability for ketoprofen as compared to gum Arabic modified magnetic nanoparticles. Therefore, this system seems to be a very promising vehicle for the administration of hydrophobic drugs.
A decade later, Chen et al. (2017) have amalgamated double-layer polymer-coated magnetic targeted nanoparticles (coated with β-cyclodextrin and polymer chitosan) to ensure stability and biocompatibility of the nanoparticles and effective drug delivery of ibuprofen, a hydrophobic drug delivery. They noted that nanocarriers exhibited sufficient magnetic properties, high drug loading capacity, and significant in vitro drug release. In conclusion, the cyclodextrin chitosan-coated magnetic nanoparticles represent a promising nanocarrier for the delivery of hydrophobic drugs. Recently, the same authors have proposed a new approach for the production of β-cyclodextrin-based magnetic nanocarriers via a molecular docking technique. Fe3O4 magnetic nanoparticles surface-modified with β-cyclodextrin and its derivatives, carboxymethyl-β-cyclodextrin, and maleated-β-cyclodextrin, respectively, were manufactured via a layer-by-layer approach, incorporating carmofur, an antineoplastic agent, as a model drug. The results of loading and release studies were similar to the previously reported literature. Herein the introduction of the molecular docking technique establishes a method to fast select an effective β-cyclodextrin-based surface coating for the development of high-performance magnetic nanoparticles (Chen et al. 2019).
During the last decade, researchers published numerous designings and preparation techniques of cyclodextrin-functionalized superparamagnetic nanoparticles by conjugating cyclodextrin units onto the surface of magnetite nanoparticles. Similarly, in one of the notable contributions, Lu and co-workers have explained an innovative protocol for the development of cyclodextrin-based magnetic nanoparticles intending to establish their wide variety of biomedical applications as targeted drug delivery vehicles. Briefly, Li et al. (2011) fabricated superparamagnetic β-cyclodextrin-functionalized composite nanoparticles with core-shell compositions via cross-linking reaction (by using epichlorohydrin as cross-linking agent) on the surface of carboxymethyl β-cyclodextrin-modified magnetite nanoparticles. This strategy ensures that the innermost magnetite nanoparticles could sense and react to an externally applied magnetic field. Additionally, the outermost cyclodextrin moiety represents an inclusion site for drugs. Luo et al. (2012) developed a novel redox responsive controlled drug release system based on magnetic nanoparticles to provide efficient intracellular anticancer drug delivery. Disulfide bonds are employed as intermediate linkers to immobilize polyethylenimine/β-cyclodextrin molecules as nanoreservoirs for drug loading onto magnetic nanoparticles. The endocytotic pathway and endosomal escape of the smart controlled drug release system are proposed as an end result. Badruddoza et al. (2013) synthesized highly uniform silica-coated iron oxide (Fe3O4) magnetic nanoparticles and cross-linked them with carboxymethylated-β-cyclodextrin, intending to equip magnetic nanoparticles with drug storage and delivery carrier. These magnetic nanoparticles contributed quadruple features of magnetism, luminescence, cell targeting, and hydrophobic drug delivery. These magnetic nanoparticles included functionalities of fluorescence labeling by fluorescent dye-conjugated and a common cancer-targeting ligand, folic acid, that Banerjee’s magnetic nanoparticles lacked. Preliminary outcomes of this study recommend that such magnetic nanoparticles can be a smart theranostic candidate for an optically trackable, cancer cell-targeting, and delivery vehicle of the hydrophobic drug delivery (all-trans-retinoic acid), loaded in cyclodextrin moiety. Ding et al. (2015) developed a novel hydrogel of poorly soluble drug 5-fluorouracil, based on chitosan-cross-linked carboxymethyl-β-cyclodextrin polymer-modified Fe3O4 magnetic nanoparticles intending to explore the inclusion abilities of cyclodextrin hydrophobic cavity with this insoluble anticancer drug through host-guest interactions. Experimental results showed that the nanocarriers displayed a high loading efficiency and pH-dependent swelling and diffusion-controlled drug release. This report tentatively proposed the mechanism of 5-fluorouracil encapsulated into the magnetic chitosan nanoparticles.
The literature reveals that the Food and Drug Administration has approved more than 150 chemotherapeutic agents for cancer treatment. But, most of these drugs exert cytotoxic action on tumors as well as normal cells and cause severe adverse effects on the patients. Additionally, numerous anticancer agents have poor solubility and stability in the aqueous and biological system. Several researchers have developed some novel approaches to ensure the safe and effective delivery of such chemotherapeutic agents. Cyclodextrins can be associated with iron oxide magnetic nanoparticles to increase their stability and dispersion in water. Anirudhan et al. (2015) have developed magnetic nanoparticles of anticancer drug 5-fluorouracil, which is used to treat breast, stomach, and lung cancer and has detrimental effects on normal cells, as it is not targeted specifically for the tumor cells. The authors prepared 3-methacryloxypropyltrimethoxysilane-coated magnetic nanoparticles polymerized with glycidylmethacrylate-grafted-maleated cyclodextrin in the presence of ethylene glycol dimethacrylate as cross-linker and a′-azobisisobutyronitrile as initiator. The prepared nanoparticles exhibited significant antimicrobial activity against gram-positive and gram-negative bacteria. The cytotoxicity studies were also performed using MCF-7 (human breast carcinoma) cells and observed that prepared nanoparticles are biocompatible and provide sustained and controlled release of drugs to the targeted site. To overcome the hindrances of earlier reported methods of targeted drug delivery, Enoch et al. (2018) synthesized β-cyclodextrin-based magnetic nanoparticles. Briefly, manganese ferrite nanoparticles were synthesized by coating with β-cyclodextrin-modified polyethylene glycol as a vehicle for the anticancer drug camptothecin. The fabricated nanoparticles showed superparamagnetic behavior. Further research showed that coating the magnetic nanoparticles with the cyclodextrin-tethered polymer improves the drug loading capacity and sustained drug release. Additionally, cytotoxicity is also enhanced when the drug is incorporated in the polymer-coated magnetic nanoparticles.
Nowadays mild acid response nanodrug delivery system has been gaining attention in the research of drug delivery based on the responsive property to the abnormal physiological environment and to the normal physiological environment. However, it lacks the site-specific action. In light of these facts, Wang et al. (2018) fabricated a magnetic and pH-sensitive composite nanoparticulate system prepared by double emulsion technique (Fig. 7.2) and incorporating acetylated β-cyclodextrin as a key ingredient to recognize the pH response and Fe3O4 as a component to realize magnetic response. Results showed irreversible pH response property and reversible magnetic responsive properties at different pH environment for the composite nanoparticle. Mrówczyński et al. (2018) developed polydopamine-coated magnetic nanoparticles of doxorubicin combined with mono-6-thio-β-cyclodextrin for cancer therapy. The hepatic cancer cellular uptake of doxorubicin-loaded nanoparticles was investigated by confocal imaging microscopy, and the significant anticancer effect of magnetic nanoparticles was observed.
7.5 Role of Cyclodextrin in Polymeric Nanoparticles
Polymeric nanoparticles are both biocompatible and biodegradable, so the fate of the nanoparticles in biological system is not a concern. Examples of natural and synthetic polymers used in the nanoparticles are chitosan, polyethylene glycol, poly (lactic acid), and poly (lactic-co-glycolic acid) poly(cyanoacrylates). The inclusion property of cyclodextrin renders polymeric nanoparticles to conveniently deliver hydrophobic molecules to the targeted site by encapsulating the drugs in the hydrophobic cyclodextrin cavity. The polymeric nanoparticles have cyclodextrin casting outer shells, while the core of the polymeric nanoparticles is composed of natural or synthetic polymer. Thus, the drugs can be loaded in the core of the polymeric nanoparticles, or it can be conjugated with the cyclodextrin in the outer shell. Nanoparticulate systems can be prepared either by dispersion of preformed polymers or polymerization. Among the polymers used in the nanoparticle preparation is poly(cyanoacrylates) which are particularly interesting, because of their biodegradability and very simple polymerization process. One of the major drawbacks of this type of nanoparticle is related to the difficulty of entrapping in hydrophobic drugs.
Da Silveira et al. (1998) first proposed cyclodextrin to overcome this problem. The authors proposed possibility of preparing nanoparticles of poly(isobutyl cyanoacrylate) in the presence of hydroxypropyl-β-cyclodextrin by anionic polymerization of isobutyl cyanoacrylate. Later, Ren et al. (2009) dissolved adamantane-end-capped poly(ε-caprolactone) and poly(vinylpyrrolidone)-cyclodextrin in N-methyl-2-pyrrolidone, a common solvent for both polymers. Further addition of this mixed polymer solution in solvent results in self-assembled polymeric nanoparticles. The same year, Zhang and Ma (2009) reported synthesis of polymeric core-shell assemblies based on inclusion complexes of a wide variety of poorly soluble drugs such as indomethacin and coumarin as guest molecules with a polyaspartamide copolymer composed of a block carrying cyclodextrin units and a polyethylene glycol block (polyethylene glycol-β-cyclodextrin). Subsequently incubation of these assemblies in buffer medium provides a prolong release of these compounds. The chronological summary of various major cyclodextrin-based polymeric nanoparticles is illustrated in Table 7.1.
7.6 Cyclodextrin-Based Lipid Nanoparticles
Lipid-based cyclodextrin nanoparticles represent numerous examples such as liposomes, nanoemulsions, solid lipid nanoparticles, or nanostructured lipid carriers. The association of cyclodextrin into lipid nanoparticle formulations not only promotes the hydrophobic drug loading within the aqueous components of the lipid cyclodextrin nanoparticles but also maintains the targetability of nanoparticles. It has been confirmed from earlier investigations that highly lipophilic molecules included in the liposome membranes are released rapidly after in vivo administration (Maestrelli et al. 2005).
To ensure stable encapsulation, McCormack and Gregoriadis (1994a, b) suggested an approach wherein cyclodextrin/drug inclusion complexes are embedded into liposomes. This strategy identified a novel drug delivery system consisting of liposomes and cyclodextrin complexes of lipophilic drugs and designated as drug-in-cyclodextrin-in-liposome. In liposomes, cyclodextrin complexation competes with liposomal membrane binding, which strengthens the potential benefit of complexation in increasing hydrophobic drug retention. This concept was applied to dexamethasone retinol and retinoic acid included in hydroxypropyl-β-cyclodextrin (McCormack and Gregoriadis 1994a, b, 1996). Arima et al. (2006) developed PEGylated liposomes entrapping the doxorubicin complex with γ-cyclodextrin and evaluated the antitumor effect of doxorubicin in rodents bearing colon-26 tumor cells. The findings of the study displayed retardation in tumor growth and an increase in drug retention. From the historical point of view, a number of lipophilic drugs have been encapsulated in the drug-in-cyclodextrin-in-liposome system. They include Ca2+ channel blocker drug nifedipine (Škalko et al. 1996); anti-inflammatory drugs such as prednisolone (Fatouros et al. 2001), ketoprofen (Maestrelli et al. 2005), betamethasone (Piel et al. 2006), celecoxib (Jain et al. 2007), and indomethacin (Chen et al. 2007); anesthetic drugs such as benzocaine, butamben (Maestrelli et al. 2010), and prilocaine (Bragagni et al. 2010); and anticancer drugs such as ß-lapachone (Cavalcanti et al. 2011) and tretinoin (Ascenso et al. 2013).
Curcumin is well known for its therapeutic effects such as antibacterial, anti-inflammatory, antioxidant, and antitumor. But, it exhibits instability and poor solubility. Therefore, to improve the cytotoxic effect and to resolve the issue of instability and solubility, Dhule et al. (2012) fabricated curcumin-loaded cyclodextrin-based liposomal nanoparticles and studied to treat osteosarcoma. The resulting 2-hydroxypropyl-γ-cyclodextrin/curcumin-liposome complex exhibits promising cytotoxic potential and represents themselves as a potential delivery vehicle for the treatment of cancers of different tissue origin.
Ji et al. (2016) practiced the use of cyclodextrin to enhance the tumor-targeting ability of the lipid nanoparticles on the outside of the liposomal wall. The surface of the liposome consisted of pirfenidone-loaded β-cyclodextrin linked with a cleavable peptide, along with arginyl glycyl aspartic acid peptide (most common peptide motif responsible for cell adhesion to the extracellular matrix) to target pancreatic tumor cells, while the interior of the liposome carried the chemotherapeutic agent gemcitabine. Solid lipid nanoparticles represent an alternative carrier system to conventional colloidal carriers due to their specific features such as the use of natural fabrication components, size and related narrow distribution, enhanced stability, and increased permeation through biological barriers. Additionally, increased solubility, biocompatibility, ease of manufacture, and different possible administration routes enable solid lipid nanoparticles a frontline drug delivery system.
Skiba et al. (1993) first proposed the concept of employing cyclodextrin in the solid nanoparticulate system and filed a patent for this invention. This invention described the development and application of a novel cyclodextrin-based dispersible colloidal system in the form of spherical particles of matrix type with size ranging from 90 to 900 nm (nanospheres), which might contain an active pharmaceutical ingredient. This nanoparticulate system was used as a carrier for numerous pharmaceuticals and cosmetic agents.
Similarly, Cavalli et al. (1999) developed inclusion complexes of hydrocortisone and progesterone with β-cyclodextrin or 2-hydroxypropyl-β-cyclodextrin to modulate the release kinetics. The inclusion complexes were incorporated into two types of solid lipid nanoparticles. The results showed that using the β-cyclodextrin complexes, the incorporation of the more hydrophilic drug, hydrocortisone, was higher than that of progesterone. The release of hydrocortisone and progesterone from solid lipid nanoparticles was lower when they were incorporated as inclusion complexes than as free molecules. To study the morphology of cyclodextrin-based solid lipid nanoparticles, Dubes et al. (2003) have performed scanning electron microscopy and atomic force microscopy for the imagery of solid lipid nanoparticles developed from an amphiphilic cyclodextrin, 2,3-di-o-alkanoyl-β-cyclodextrin, β-cyclodextrin 21C6. The results of the study reveal that the vacuum drying technique used in sample preparation for scanning electron microscopy causes shrinkage in the size of the solid lipid nanoparticles, whereas the deposition method used for atomic force microscopy produces small clusters of solid lipid nanoparticles.
Chirio et al. (2011) formulated solid lipid nanoparticles of curcumin, composed of different triglycerides with or without various modified α- and γ-cyclodextrins. The synthesized nanoparticles showed significant entrapment efficiency, improved hydrolytic stability, and a notable reduction in curcumin photodegradation when the drug was incorporated in tristearin solid lipid nanoparticles. Moreover, the results of the skin uptake study displayed an improvement in curcumin skin accumulation when curcumin was incorporated in solid lipid nanoparticles obtained with all cyclodextrin derivatives, particularly with most lipophilic ones. Solid lipid nanoparticles act as a protection to bioactive molecules.
To explore this unique feature of solid lipid nanoparticles, Carlotti et al. (2012) evaluated the potential of solid lipid nanoparticles as a protective carrier of resveratrol, against photosensitivity. A notable decline in photodegradation was recorded in drug-loaded solid lipid nanoparticles in comparison to tetradecyl-γ-cyclodextrin. Moreover, significant in vitro skin accumulation and an enhanced antioxidative efficacy were also observed in resveratrol-loaded nanoparticles.
Spada et al. (2012) formed diclofenac sodium-loaded solid lipid nanoparticles composed of different ratios of Compritol AT088 and 2-hydroxypropyl-β-cyclodextrin via an oil/water hot homogenization method for colon-specific drug delivery. In vitro and ex vivo investigations confirmed solid lipid nanoparticles as a prominent carrier for colonic drug delivery. Additionally, in vitro toxicity studies showed that the solid lipid nanoparticles are well tolerated. Furthermore, Baek and Cho (2013) developed the surface-modified paclitaxel-filled solid lipid nanoparticles with hydroxypropyl-β-cyclodextrin. This cyclodextrin has been recognized to solubilize the drugs and inhibit the oxidation of lipids (Zafar et al. 2013). The formulated solid lipid nanoparticles showed a paclitaxel encapsulation percentage of 71% with a mean size of 251 nm. The Caco-2 cell uptake of paclitaxel from solid lipid nanoparticles was 5.3-fold greater than the Taxol formulation.
Nanostructured lipid carriers represent an upgraded generation of lipid nanoparticle, which overcome the major drawback of solid lipid nanoparticles, particularly the tendency of discharge of the drug during storage as an outcome of their highly ordered crystalline composition. Nanostructured lipid carriers composed by a solid lipid matrix are developed from a mixture of biocompatible solid and liquid lipids and an aqueous phase containing a surfactant or a blend of surfactants. Recently, several reports have been published demonstrating the strategy of simultaneously utilizing the advantages of both cyclodextrin and nanolipid carriers by locking them in a novel drug delivery system, by incorporating the drug-cyclodextrin inclusion complex into the lipid nanoparticles. A brief summary of recently developed cyclodextrin-based solid lipid nanoparticles and lipid nanoparticles loaded with various drugs used as anticancer, antihypertensive, diuretic, and anti-inflammatory is illustrated in Table 7.2.
7.7 Role of Cyclodextrin in Gold and Silver Nanoparticles
In recent years, gold and silver nanoparticles have been widely investigated for nanomedicine due to their superior optical, chemical, and biological properties which make them ideal for molecular imaging, drug/gene delivery, biosensors, and therapeutic agents. Furthermore, silver nanoparticles have antibacterial properties that make them desirable for drug delivery systems as they provide additional antimicrobial action. When combined with cyclodextrins, these metallic nanoparticles can become more targeted. Gold and silver cyclodextrin nanoparticles are commonly produced by connecting cyclodextrin to the metallic core using a linker, such as adamantane, which forms a strong stable complex with the cyclodextrins (Fig. 7.2). Sometimes the cyclodextrins are added by using cyclodextrin-loaded macromolecules (cyclodextrin-modified hyaluronic acid).
Liu et al. (1998) first developed a novel technique for the surface derivatization of gold colloidal particles to prepare gold colloidal particles of diameter > 10 nm. They demonstrated aqueous solubilization of aliphatic thiols by α-cyclodextrin, which effectively binds to the aliphatic chains and carries the hydrophobic thiol molecules to the surface of the gold particles. Two years later, the same authors developed cyclodextrin-modified gold nanospheres by reduction of AuCl4 in the presence of perthiolated cyclodextrin receptors resulting in gold nanospheres modified with attached cyclodextrins. It is proposed that cyclodextrin-modified gold nanospheres represent multisite hosts in aqueous media. The most common application of cyclodextrin-gold nanoparticles is in biosensor technology.
Holzinger et al. (2009) developed a single-walled carbon nanotube with β-cyclodextrin-functionalized gold nanoparticles attached to the surface, loaded with polymerized adamantane. The association between the polymerized adamantane and the β-cyclodextrin-functionalized gold nanoparticles ideally simulates the biological interactions of biotin and avidin. To explore the potential of gold nanoparticles for photothermal release of drug, Sierpe et al. (2015) carried out the synthesis of a 1:1 β-cyclodextrin-phenylethylamine inclusion complex and the adhesion of gold nanoparticles onto microcrystals of this complex. Wang et al. (2016a, b) described an easy method to produce the host-guest assembly of gold nanoparticles induced by intracellular glutathione. Briefly, gold nanoparticle aggregates were developed from dispersive gold nanoparticles by host-guest interactions between ferrocene and β-cyclodextrin and triggered by intracellular glutathione. It is well known that ferrocene develops an inclusion complex with β-cyclodextrin and detaches upon ionization (Fc+). Results showed that the synthesized aggregates are retained for a long time in cancer cells and provoke apoptosis of cells when exposed to near-infrared irradiation. It has been proven from several scientific investigations that β-cyclodextrin-functionalized gold nanoparticles are more efficient in anticancer therapy when incorporated with anticancer agents. For example, Ha et al. (2013) formulated gold nanoparticles decorated with polyethylene glycol and poly (N-isopropyl acrylamide) linked via complexation between β-cyclodextrin and adamantane groups. It has been observed that exposure of these gold nanoparticles to low temperature facilitates the release of the doxorubicin as well as transports it directly to the cancer cell nucleus by dismantling.
Furthermore, Bakar et al. (2015) reported decreased breast cancer cell (MCF-7) proliferation by complexing various ligands (pinoresinol, lariciresinol, and secoisolariciresinol) with thiolated-β-cyclodextrin and decorating them on the exterior of gold nanoparticles. Conventional anticancer molecules such as doxorubicin, paclitaxel, and docetaxel were incorporated into the β-cyclodextrin-functionalized gold nanoparticles and targeted to cancer cells. Findings of cell line studies showed that the doxorubicin-loaded β-cyclodextrin gold nanoparticles enhanced the cellular uptake and exerted a significant antiproliferative effect. Similarly, Wang et al. (2016a, b) constructed a twofold nanoparticulate delivery system based on host-guest nanoplatforms loaded with anticancer agent docetaxel and genetic material siRNA using gold nanorods coated with polyethylenimine-grafted β-cyclodextrin. The developed gold nanoparticles, upon exposure to near-infrared laser irradiation, generate a significant hyperthermia effect to trigger siRNA and docetaxel release from the cyclodextrin, and it has been observed that developed gold nanoparticles in combination with laser irradiation remarkably inhibit lung metastasis of 4T1 breast tumors.
A general method to improve the dispersity of oleic acid-stabilized silver nanoparticles was proposed by Wang et al. (2016a, b). The authors have modified the surface properties of the nanoparticles by the configuration of an inclusion complex between surface-bound surfactant particles and α-cyclodextrin. Results showed that initial cyclodextrin concentration is a key factor responsible for the phase transfer of nanoparticles to the aqueous solutions (Wang et al. 2016a, b). To explore this concept of phase transfer of silver nanoparticles in aqueous solvents, George et al. (2011) synthesized antibacterial water-soluble, silver nanoparticles encapsulated in β-cyclodextrin. The β-cyclodextrin-encapsulated silver nanoparticle revealed remarkable stability and antimicrobial effect when tested in vitro against Pseudomonas aeruginosa and Staphylococcus aureus. One year later, the same researcher’s group synthesized silver nanoparticles by reducing silver acetate with a long-chain aliphatic amine stabilized with β-cyclodextrin. This formulation revealed significant results when evaluated for antifungal activity against various human pathogens such as Aspergillus fumigatus, Mucor, and Chrysosporium species.
Gannimani et al. (2016) coupled the antibacterial properties of silver nanoparticles and hydrophobic drug carrier characteristic of cyclodextrin to fabricate supramolecules to provide cutting edge for antibacterial efficacy of chloramphenicol. Likewise, Gaurav et al. (2015) exploited an interesting mechanistic feature of metal nanoparticles for multiple therapeutic targets. They utilized β-cyclodextrin to solubilize clotrimazole, an antifungal agent, and then attach to albumin-stabilized silver nanoparticles; here albumin facilitates to reduce the interaction between the silver nanoparticles and the cyclodextrin-clotrimazole complex. These hybrid nanoparticles exerted a synergistic effect when evaluated for antifungal activity against candida yeast cells. β-Cyclodextrin-capped silver nanoparticles also have a notable contribution to cytotoxic therapeutic applications. Jaiswal et al. (2015) proved the ability of β-cyclodextrin silver nanoparticles to regulate biofilm growth and lessen the cytotoxic effect. The cytotoxicity of silver nanoparticles in human HaCat skin cells was eliminated due to the shielding capping of β-cyclodextrin. These nanoparticles particularly target cancerous cells and bypass uptake in normal cells. Zhai et al. (2017) reported the formulation and therapeutic application of biocompatible nanoparticles for the examination of the uptake of nanoparticles into viable cells in a microfluidic chip by utilizing surface-enhanced Raman spectroscopy, which modified the surface of β-cyclodextrin-capped silver nanoparticles using para-amino thiophenol and folic acid. The para-amino thiophenol molecules serve as the Raman reporter, while the folic acid fragments have a high proclivity for folate receptors that are overexpressed on the surface cancerous cells so that the nanoparticles can penetrate the cells and be observed by the Raman reporter.
Like gold nanoparticles, silver nanoparticles represent valuable attributes such as high functionalization and good catalytic activity, which make them suitable for biosensor applications. Therefore, with an aim to explore this characteristic of silver nanoparticles, Gao et al. (2015) developed a novel sandwich-type electrochemical immunosensor based on host-guest interaction for the detection of alpha-fetoprotein (a protein made in the liver of a developing baby). The developed immunosensor displays a wide linear calibration range from 0.001 to 5.0 ng/mL with a low detection limit (0.2 pg/mL) for the detection of alpha-fetoprotein. This method has a large-scale application promise in the clinical domain. Hui et al. (2015) explored β-cyclodextrin as a reducing agent between silver nitrate and graphene oxide in a guanine and adenine biosensor. In this study, β-cyclodextrin acts as a stabilizer as well as a dispersant for the silver nanoparticles and graphene oxide. This property of β-cyclodextrin offered a microenvironment that produced a rapid absorption of guanine and adenine leading to faster electrocatalysis.
7.8 Pharmaceutical Applications of Amphiphilic Cyclodextrin Nanoparticles
The potential use of cyclodextrin in a biological system needs amphiphilic properties because natural cyclodextrin has relatively low solubility both in water and organic solvents, thus limiting their uses in pharmaceutical formulations. Amphiphilic or ionizable cyclodextrins can modify the rate or time of drug release and bind to the surface membrane of cells that may be used for the enhancement of drug absorption across biological barriers. Improvement of the interaction of cyclodextrins with hydrophobic drugs renders self-assembly capacity in aqueous solutions (Bilensoy and Hincal 2009). Amphiphilic cyclodextrins are obtained by grafting of 6-C aliphatic chains linked with ester or/and amide groups of different length on the primary/secondary face of the glucopyranose units (Duchêne et al. 1999a). The major advantage of amphiphilic cyclodextrins is their self-assembly properties which are sufficient to form different nanosized carriers spontaneously without the presence of surfactants. This superiority gives them amphiphilic properties resulting in the formation of supramolecular aggregates in the nanometer range (Duchêne et al. 1999b).
According to the chemical structure of the amphiphilic cyclodextrin, different carrier systems could be obtained such as solid lipid nanoparticles, bilayer vesicles, liposomes, and nanoparticles (Donohue et al. 2002; Dubes et al. 2003). According to the groups conjugated onto cyclodextrins, amphiphilic cyclodextrin-based systems can be classified as nonionic, cationic, and anionic materials (Zhang and Ma 2013). Amphiphilic cyclodextrins have been synthesized to overcome the drawbacks of natural cyclodextrins. In addition, they have the ability to form self-assembled nanoparticulate systems. Their unique properties can improve the drug loading capacity, cellular interaction and tumoral penetration, drug release profiles, and cytotoxicity of drug delivery systems. Skiba et al. (1996) synthesized an amphiphilic β-cyclodextrin-based nanosphere drug delivery system prepared by uniform dispersion of an organic solution of modified β-cyclodextrin in the hydrophilic phase with or without surfactant. A preliminary study for the effect of surfactant on the size, physicochemical properties, and stability of the nanospheres was carried out. Furthermore, in a similar study, the authors reported the synthesis and characterization of amphiphilic cyclodextrin-based new nanosphere drug delivery system consisted of fatty acid chain of either 6 or 12 or 14 carbon atoms assembled at the 02 and 03 positions of the β-cyclodextrin molecule. Table 7.3 summarized the various potential pharmaceutical applications of amphiphilic cyclodextrin-based nanoparticles such as anticancer, cholesterol-targeted, folate-targeted, and amphiphilic cyclodextrin nanoparticles for gene delivery.
7.9 Miscellaneous
Nowadays, numerous types of neutral, amphiphilic, modified cyclodextrins are incorporated in the formulation development of novel drug delivery systems that can enable effective delivery of drugs across mucosal or dermal membranes. As per the biopharmaceutical classification system of drugs, poor drug solubility or poor mucosa permeability attributes of drugs limit their pharmaceutical applications. These cyclodextrin-based polymeric nanoparticles represent a more reliable drug delivery system when compared with control nanoparticles; they displayed homogeneous bioadhesive interactions with the gastrointestinal mucosa due to the presence of several hydroxyl groups in cyclodextrin nanoparticles, which would promote hydrogen bonding with the gut, subsequently enhancing the bioadhesive potential (Agüeros et al. 2011). Furthermore, Luppi et al. (2011) examined the potential of different cyclodextrins in nasal drug delivery using albumin nanoparticles for the treatment of the most common neurodegenerative disorder Alzheimer’s disease to validate their effect on the drug release, mucoadhesiveness of nanoparticles, and permeability of model drug tacrine. In another approach, β-cyclodextrin was conjugated with chitosan and further quaternized with glycidyltrimethylammonium chloride for mucoadhesive drug delivery system.
Transdermal delivery of drugs has always been a challenging task due to barrier property of skin; due to unique properties of cyclodextrin-based nanoparticles, they represent an alternative choice for effective transdermal delivery of drugs. Maestrelli et al. (2006) synthesized chitosan nanoparticles in the presence of cyclodextrin as a nanocarrier system for the triclosan (an antifungal agent) and furosemide (a diuretic). This nanocarrier system showed immediate drug release, followed by a delayed-release of drug. It confirms the inclusion of the drug inside the cyclodextrin cavity and later encapsulation inside the chitosan polymer. Thus, representing an ideal nanocarrier system for transdermal drug delivery. Similarly, Khalil et al. (2012) formulated nanoparticles of warfarin, an anticoagulant drug, by loading it in chitosan-cyclodextrin-complexed nanoparticle systems for transdermal drug delivery. The results of in vitro release studies and ex vivo permeation studies of warfarin-cyclodextrin-complexed nanoparticles paved the new way for the delivery of hydrophobic drugs. To improve the percutaneous absorption, Kwon and Kim (2010) fabricated mono-olein cubic phase nanoparticles comprising of 2-hydroxypropyl-beta-cyclodextrin, complexed with minoxidil (a vasodilator drug), which were prepared by hydrating molten minoxidil with the inclusion complex solution. The cubic phase transparent gels were obtained in the presence of pluronic F127 as a dispersant that showed significant in vitro skin permeability when compared with native delivery carriers. Econazole nitrate, a poorly soluble antifungal agent for ocular drug delivery, was developed as the chitosan nanoparticles in the presence of sulfobutyl-ether-beta-cyclodextrin. Mucoadhesiveness property of chitosan enabled nanoparticles to interrelate with the ocular mucosa; besides the use of an anionic, sulfobutyl-ether-beta-cyclodextrin magnified the solubility and delivery of econazole to the cornea (Mahmoud et al. 2011).
Gil et al. (2009) synthesized the novel quaternary ammonium β-cyclodextrin nanoparticles as drug delivery carriers for doxorubicin to improve the permeability of doxorubicin, a hydrophobic anticancer drug across the blood-brain barrier. When quaternary ammonium β-cyclodextrin was experimented in different in vitro studies, it was found to be nontoxic at a concentration of up to 500 mg/ml in bovine brain microvessel endothelial cells and showed great potential for safe and effective delivery of doxorubicin and other therapeutic agents across the most complexed blood-brain barrier. Nanoparticle-based biomedicine has gained enormous acceptance for theranostic applications, due to their advantage features over conventional therapy. Therefore, in search of a novel theranostic material, Datz et al. (2018) have synthesized a new β-cyclodextrin-based biocompatible and multifunctional substance that cross-linked with rigid organic linker molecules to yield thermostable, readily water-dispersible particles having a nanosize range (∼150 nm). In the next step, these nanoparticles covalently linked with dye molecules to enable effective tracking of them during in vitro cell experiments. Results showed the successful nuclei staining with Hoechst 33342 dye, including effective cell killing with the doxorubicin cargo molecules. Concludingly, these findings represent a promising approach for the development of novel theranostic systems.
7.10 Conclusion
The application of cyclodextrin-based nanomaterials is beginning to confirm an effective degree of control in drug delivery and other therapeutic applications, although their safe, effective, and economical delivery is yet not significantly demonstrated. The chemical modification of cyclodextrin polymers is a promising strategy to extend their pharmaceutical applications. Some nanoparticles, such as CRLX101 and CALAA-01, are tumor-targeted and siRNA delivery nanopharmaceuticals respectively, they contain a cyclodextrin-containing polymer. These nanoparticles formulated with a polyethylene glycol steric stabilization agent, are among the most appropriate nanotherapeutics in clinical phase II trials for cancer diseases (Weiss et al. 2011; Zuckerman et al. 2014).
There is a flourishing discussion about the inherent advantages, characteristic features, and potential therapeutic applications of cyclodextrin-based nanoparticles reported by the scientist fraternity in the past decades to date. These cyclodextrin-based nanoparticles are enriched with various properties of an ideal novel drug delivery system. Moreover, they overcome the limitations of other carrier systems as well. Cyclodextrin-based nanoparticles have emerged as a very promising carrier system for high molecular weight compounds such as peptides, proteins, DNA, and other genetic materials. Nanoparticles containing cyclodextrins have shown their potential to improve the loading capacity of carriers such as liposomes, nanoparticles, and solid lipid nanoparticles. This review also highlights the new-generation cyclodextrin-based nanocarrier for the delivery of associated DNA or proteins with remarkable cytotoxicity against various cancer cell lines. Amphiphilic cyclodextrins represent unique properties of ideal pharmaceutical excipients, such as the ability to form nanoparticles spontaneously. Moreover, they possess the advantages of natural cyclodextrins suitable for drug delivery, together with the advantages of delivery of drugs through various administration routes such as oral, transdermal, nasal, ocular, and across the biological membranes including the blood-brain barrier.
Abbreviations
- DNA:
-
Deoxyribonucleic acid
- MCF-7:
-
Michigan Cancer Foundation-7
- MDR1:
-
Multidrug resistance protein 1
- mRNA:
-
Messenger ribonucleic acid
- RNA:
-
Ribonucleic acid
- siRNA:
-
Small interfering ribonucleic acid
References
Agüeros M, Ruiz-Gatón L, Vauthier C, Bouchemal K, Espuelas S, Ponchel G, Irache JM (2009) Combined hydroxypropyl-β-cyclodextrin and poly(anhydride) nanoparticles improve the oral permeability of paclitaxel. Eur J Pharm Sci 38:405–413. https://doi.org/10.1016/j.ejps.2009.09.010
Agüeros M, Espuelas S, Esparza I, Calleja P, Peñuelas I, Ponchel G, Irache JM (2011) Cyclodextrin-poly(anhydride) nanoparticles as new vehicles for oral drug delivery. Expert Opin Drug Deliv 8:721–734. https://doi.org/10.1517/17425247.2011.572069
Ahmed GHG, Laíño RB, Calzón JAG, García MED (2014) Magnetic nanoparticles grafted with β-cyclodextrin for solid-phase extraction of 5-hydroxy-3-indole acetic acid. Microchim Acta 181:941–948. https://doi.org/10.1007/s00604-014-1192-y
Ammar H, El-Nahhas S, Ghorab M, Salama A (2012) Chitosan/Cyclodextrin nanoparticles as drug delivery system. J Incl Phenom Macrocycl Chem 72:9950–9955. https://doi.org/10.1007/s10847-011
Anirudhan TS, Dilu D, Sandeep S (2013) Synthesis and characterisation of chitosan crosslinked-β-cyclodextrin grafted silylated magnetic nanoparticles for controlled release of Indomethacin. J Magn Magn Mater 343:149–156. https://doi.org/10.1016/j.jmmm.2013.04.007
Anirudhan TS, Divya PL, Nima J (2015) Synthesis and characterization of silane coated magnetic nanoparticles/glycidylmethacrylate-grafted-maleated cyclodextrin composite hydrogel as a drug carrier for the controlled delivery of 5-fluorouracil. Mater Sci Eng C 55:471–481. https://doi.org/10.1016/j.msec.2015.05.068
Aranda C, Urbiola K, Méndez Ardoy A, García Fernández JM, Ortiz Mellet C, de Ilarduya CT (2013) Targeted gene delivery by new folate-polycationic amphiphilic cyclodextrin-DNA nanocomplexes in vitro and in vivo. Eur J Pharm Biopharm 85:390–397. https://doi.org/10.1016/j.ejpb.2013.06.011
Arima H, Hagiwara Y, Hirayama F, Uekama K (2006) Enhancement of antitumor effect of doxorubicin by its complexation with γ-cyclodextrin in pegylated liposomes. J Drug Target 14:225–232. https://doi.org/10.1080/10611860600711136
Ascenso A, Cruz M, Euletério C, Carvalho FA, Santos NC, Marques HC, Simões S (2013) Novel tretinoin formulations: a drug-in-cyclodextrin-in-liposome approach. J Liposome Res 23:211–219. https://doi.org/10.3109/08982104.2013.788026
Badruddoza AZM, Rahman MT, Ghosh S, Hossain MZ, Shi J, Hidajat K, Uddin MS (2013) β-Cyclodextrin conjugated magnetic, fluorescent silica core-shell nanoparticles for biomedical applications. Carbohydr Polym 95:449–457. https://doi.org/10.1016/j.carbpol.2013.02.046
Baek JS, Cho CW (2013) 2-Hydroxypropyl-β-cyclodextrin-modified SLN of paclitaxel for overcoming p-glycoprotein function in multidrug-resistant breast cancer cells. J Pharm Pharmacol 65:72–78. https://doi.org/10.1111/j.2042-7158.2012.01578.x
Baek JS, Kim JH, Park JS, Cho CW (2015) Modification of paclitaxel-loaded solid lipid nanoparticles with 2-hydroxypropyl-β-cyclodextrin enhances absorption and reduces nephrotoxicity associated with intravenous injection. Int J Nanomedicine 10:5397–5405. https://doi.org/10.2147/IJN.S86474
Bakar F, Caglayan M, Onur F, Nebioglu S, Palabiyik I (2015) Gold nanoparticle-lignan complexes inhibited MCF-7 cell proliferation in vitro: a novel conjugation for cancer therapy. Anticancer Agents Med Chem 15:336–344. https://doi.org/10.2174/1871520614666141202144152
Banerjee SS, Chen DH (2007) Magnetic nanoparticles grafted with cyclodextrin for hydrophobic drug delivery. Chem Mater 19:6345–6349. https://doi.org/10.1021/cm702278u
Bilensoy E, Hincal AA (2009) Recent advances and future directions in amphiphilic cyclodextrin nanoparticles. Expert Opin Drug Deliv 6:1161–1173. https://doi.org/10.1517/17425240903222218
Bilensoy E, Gürkaynak O, Doǧan AL, Hincal AA (2008a) Safety and efficacy of amphiphilic ß-cyclodextrin nanoparticles for paclitaxel delivery. Int J Pharm 347:163–170. https://doi.org/10.1016/j.ijpharm.2007.06.051
Bilensoy E, Gürkaynak O, Ertan M, Şen M, Hincal AA (2008b) Development of nonsurfactant cyclodextrin nanoparticles loaded with anticancer drug paclitaxel. J Pharm Sci 97:1519–1529. https://doi.org/10.1002/jps.21111
Bragagni M, Maestrelli F, Mura P (2010) Physical chemical characterization of binary systems of prilocaine hydrochloride with triacetyl-β-cyclodextrin. J Incl Phenom Macrocycl Chem 68:437–445. https://doi.org/10.1007/s10847-010-9807-3
Bu X, Zhu T, Ma Y, Shen Q (2015) Co-administration with cell penetrating peptide enhances the oral bioavailability of docetaxel-loaded nanoparticles. Drug Dev Ind Pharm 41:764–771. https://doi.org/10.3109/03639045.2014.902465
Campos EVR, Proença PLF, Oliveira JL, Melville CC, Vechia JFD, De Andrade DJ, Fraceto LF (2018) Chitosan nanoparticles functionalized with β-cyclodextrin: a promising carrier for botanical pesticides. Sci Rep 8. https://doi.org/10.1038/s41598-018-20602-y
Carlotti M, Sapino S, Ugazio E, Gallarate M, Morel S (2012) Resveratrol in solid lipid nanoparticles. J Dispers Sci Technol 33:465–471. https://doi.org/10.1080/01932691.2010.548274
Cavalcanti IMF, Mendona EAM, Lira MCB, Honrato SB, Camara CA, Amorim RVS, Filho JM, Rabello MM, Hernandes MZ, Ayala AP, Santos-Magalhães NS (2011) The encapsulation of β-lapachone in 2-hydroxypropyl-β-cyclodextrin inclusion complex into liposomes: a physicochemical evaluation and molecular modeling approach. Eur J Pharm Sci 44:332–340. https://doi.org/10.1016/j.ejps.2011.08.011
Cavalli R, Peira E, Caputo O, Gasco MR (1999) Solid lipid nanoparticles as carriers of hydrocortisone and progesterone complexes with β-cyclodextrins. Int J Pharm 182:59–69. https://doi.org/10.1016/S0378-5173(99)00066-6
Cavalli R, Trotta F, Carlotti ME, Possetti B, Trotta M (2007) Nanoparticles derived from amphiphilic-γ-cyclodextrins. J Incl Phenom Macrocycl Chem 57:657–661. https://doi.org/10.1007/s10847-006-9269-9
Challa R, Ahuja A, Ali J, Khar RK (2005) Cyclodextrins in drug delivery: an updated review. AAPS Pharm SciTech 6:2. https://doi.org/10.1208/pt060243
Chen H, Gao J, Wang F, Wenquan L (2007) Preparation, characterization and pharmacokinetics of liposomes-encapsulated cyclodextrins inclusion complexes for hydrophobic drugs. Drug Deliv 14:201–208. https://doi.org/10.1080/10717540601036880
Chen P, Song H, Yao S, Tu X, Su M, Zhou L (2017) Magnetic targeted nanoparticles based on β-cyclodextrin and chitosan for hydrophobic drug delivery and a study of their mechanism. RSC Adv 7:29025–29034. https://doi.org/10.1039/C7RA02398G
Chen P, Yao S, Chen X, Huang Y, Song H (2019) A new strategy for the construction of β-cyclodextrin-based magnetic nanocarriers: a molecular docking technique. New J Chem 43:4282–4290. https://doi.org/10.1039/c8nj06131a
Chirio D, Gallarate M, Peira E, Battaglia L, Serpe L, Trotta M (2011) Formulation of curcumin-loaded solid lipid nanoparticles produced by fatty acids coacervation technique. J Microencapsul 28:537–548. https://doi.org/10.3109/02652048.2011.590615
Çirpanli Y, Bilensoy E, Lale Doǧan A, Çaliş S (2009) Comparative evaluation of polymeric and amphiphilic cyclodextrin nanoparticles for effective camptothecin delivery. Eur J Pharm Biopharm 73:82–89. https://doi.org/10.1016/j.ejpb.2009.04.013
Çirpanli Y, Allard E, Passirani C, Bilensoy E, Lemaire L, Caliş S, Benoit JP (2011) Antitumoral activity of camptothecin-loaded nanoparticles in 9L rat glioma model. Int J Pharm 17:201–206. https://doi.org/10.1016/j.ijpharm.2010.10.015
Cirri M, Bragagni M, Mennini N, Mura P (2012) Development of a new delivery system consisting in “drug – In cyclodextrin – In nanostructured lipid carriers” for ketoprofen topical delivery. Eur J Pharm Biopharm 80:46–53. https://doi.org/10.1016/j.ejpb.2011.07.015
Cirri M, Mennini N, Maestrelli F, Mura P, Ghelardini Cyclodextrini Cesare Mannelli L (2017) Development and in vivo evaluation of an innovative “hydrochlorothiazide-in cyclodextrins-in solid lipid nanoparticles” formulation with sustained release and enhanced oral bioavailability for potential hypertension treatment in pediatrics. Int J Pharm 521:73–83. https://doi.org/10.1016/j.ijpharm.2017.02.022
Cirri M, Maestrelli F, Mura P, Ghelardini C, Mannelli LDC (2018) Combined approach of cyclodextrin complexation and nanostructured lipid carriers for the development of a pediatric liquid oral dosage form of hydrochlorothiazide. Pharmaceutics 10:287. https://doi.org/10.3390/pharmaceutics10040287
Conte C, Costabile G, d’Angelo I, Pannico M, Musto P, Grassia G, Ialenti A, Tirino P, Miro A, Ungaro F, Quaglia F (2015) Skin transport of PEGylated poly(ε-caprolactone) nanoparticles assisted by (2-hydroxypropyl)-β-cyclodextrin. J Colloid Interface Sci 454:112–120. https://doi.org/10.1016/j.jcis.2015.05.010
Crini G (2014) Review: a history of cyclodextrins. Chem Rev 114:10940–10975. https://doi.org/10.1021/cr500081p
Da Silveira AM, Ponchel G, Puisieux F, Duchêne D (1998) Combined poly(isobutylcyanoacrylate) and cyclodextrins nanoparticles for enhancing the encapsulation of lipophilic drugs. Pharm Res 15:1051–1055. https://doi.org/10.1023/A:1011982211632
Datz S, Illes B, Gößl D, Schirnding CV, Engelke H, Bein T (2018) Biocompatible crosslinked β-cyclodextrin nanoparticles as multifunctional carriers for cellular delivery. Nanoscale 10:16284–16292. https://doi.org/10.1039/c8nr02462f
Dhule SS, Penfornis P, Frazier T, Walker R, Feldman J, Tan G, He J, Alb A, John V, Pochampally R (2012) Curcumin-loaded γ-cyclodextrin liposomal nanoparticles as delivery vehicles for osteosarcoma. Nanomedicine 8:440–451. https://doi.org/10.1016/j.nano.2011.07.011
Díaz-Moscoso A, Guilloteau N, Bienvenu C, Méndez-Ardoy A, Jiménez Blanco J, Benito J, Gourriérec L, Di Giorgio C, Vierling P, Defaye J, Mellet C, García Fernández J (2011) Mannosyl-coated nanocomplexes from amphiphilic cyclodextrins and pDNA for site-specific gene delivery. Biomaterials 32:7263–7273. https://doi.org/10.1016/j.biomaterials.2011.06.025
Ding Y, Shen SZ, Sun H, Sun K, Liu F, Qi Y, Yan J (2015) Design and construction of polymerized-chitosan coated Fe3O4 magnetic nanoparticles and its application for hydrophobic drug delivery. Mater Sci Eng C 48:487–498. https://doi.org/10.1016/j.msec.2014.12.036
Donohue R, Mazzaglia A, Ravoo BJ, Darcy R (2002) Cationic β-cyclodextrin bilayer vesicles. Chem Commun 2:2864–2865. https://doi.org/10.1039/b207238f
Dubes A, Parrot-Lopez H, Abdelwahed W, Degobert G, Fessi H, Shahgaldian P, Coleman AW (2003) Scanning electron microscopy and atomic force microscopy imaging of solid lipid nanoparticles derived from amphiphilic cyclodextrins. Eur J Pharm Biopharm 55:279–282. https://doi.org/10.1016/S0939-6411(03)00020-1
Duchêne D, Ponchel G, Wouessidjewe D (1999a) Cyclodextrins in targeting application to nanoparticles. Adv Drug Deliv Rev 36:29–40. https://doi.org/10.1016/S0169-409X(98)00053-2
Duchêne D, Wouessidjewe D, Ponchel G (1999b) Cyclodextrins and carrier systems. J Control Release 62:263–268. https://doi.org/10.1016/S0168-3659(99)00046-2
El Fagui A, Dalmas F, Lorthioir C, Wintgens V, Volet G, Amiel C (2011) Well-defined core-shell nanoparticles containing cyclodextrin in the shell: a comprehensive study. Polymer 52:3752–3761. https://doi.org/10.1016/j.polymer.2011.06.043
Enoch IVMV, Ramasamy S, Mohiyuddin S, Gopinath P (2018) Cyclodextrin-PEG conjugate-wrapped magnetic ferrite nanoparticles for enhanced drug loading and release. Appl Nanosci 8:273–284. https://doi.org/10.1007/s13204-018-0798-5
Erdogar N, Varan G, Bilensoy E (2017) Amphiphilic cyclodextrin derivatives for targeted drug delivery to tumors. Curr Top Med Chem 17:1521–1528. https://doi.org/10.2174/1568026616666161222101104
Evans J, Malhotra M, Fitzgerald K, Guo J, Cronin M, Curtin C, O’Brien F, Darcy R, O’Driscoll C (2017) Formulation and evaluation of anisamide-targeted amphiphilic cyclodextrin nanoparticles to promote therapeutic gene silencing in a 3d prostate cancer bone metastases model. Mol Pharm 3:42–52. https://doi.org/10.1021/acs.molpharmaceut.6b00646
Fatouros DG, Hatzidimitriou K, Antimisiaris SG (2001) Liposomes encapsulating prednisolone and prednisolone-cyclodextrin complexes: comparison of membrane integrity and drug release. Eur J Pharm Sci 13:287–296. https://doi.org/10.1016/S0928-0987(01)00114-2
Gannimani R, Ramesh M, Mtambo S, Pillay K, Soliman ME, Govender P (2016) γ-Cyclodextrin capped silver nanoparticles for molecular recognition and enhancement of antibacterial activity of chloramphenicol. J Inorg Biochem 157:15–24. https://doi.org/10.1016/j.jinorgbio.2016.01.008
Gao H, Wang YN, Fan YG, Ma JB (2005) Synthesis of a biodegradable tadpole-shaped polymer via the coupling reaction of polylactide onto mono(6-(2-aminoethyl)amino-6-deoxy)-β- cyclodextrin and its properties as the new carrier of protein delivery system. J Control Release 107:158–173. https://doi.org/10.1016/j.jconrel.2005.06.010
Gao J, Ma H, Lv X, Yan T, Li N, Cao W, Wei Q (2015) A novel electrochemical immunosensor using β-cyclodextrins functionalized silver supported adamantine-modified glucose oxidase as labels for ultrasensitive detection of alpha-fetoprotein. Anal Chim Acta 893:49–56. https://doi.org/10.1016/j.aca.2015.08.052
García-González L, Yépez-Mulía L, Ganem A (2016) Effect of β-cyclodextrin on the internalization of nanoparticles into intestine epithelial cells. Eur J Pharm Sci 81:113–118. https://doi.org/10.1016/j.ejps.2015.10.012
Gaurav C, Nikhil G, Deepti S, Kalra S, Goutam R, Amit GK (2015) Albumin stabilized silver nanoparticles-clotrimazole β-cyclodextrin hybrid nanocomposite for enriched anti-fungal activity in normal and drug resistant Candida cells. RSC Adv 5:71190–71202. https://doi.org/10.1039/c5ra08274a
George C, Kuriakose S, George S, Mathew T (2011) Antifungal activity of silver nanoparticle-encapsulated β-cyclodextrin against human opportunistic pathogens. Supramol Chem 23:593–597. https://doi.org/10.1080/10610278.2011.575471
Gèze A, Chau LT, Choisnard L, Mathieu JP, Marti-Batlle D, Riou L, Putaux JL, Wouessidjewe D (2007) Biodistribution of intravenously administered amphiphilic β-cyclodextrin nanospheres. Int J Pharm 344:135–142. https://doi.org/10.1016/j.ijpharm.2007.06.050
Ghera BB, Perret F, Chevalier Y, Parrot-Lopez H (2009) Novel nanoparticles made from amphiphilic perfluoroalkyl alpha-cyclodextrin derivatives: preparation, characterization and application to the transport of acyclovir. Int J Pharm 22:155–162. https://doi.org/10.1016/j.ijpharm.2009.04.004
Gil ES, Li J, Xiao H, Lowe TL (2009) Quaternary ammonium β-cyclodextrin nanoparticles for enhancing doxorubicin permeability across the in vitro blood-brain barrier. Biomacromolecules 10:505–516. https://doi.org/10.1021/bm801026k
Godinho BMDC, Ogier JR, Darcy R, O’Driscoll CM, Cryan JF (2013) Self-assembling modified β-cyclodextrin nanoparticles as neuronal siRNA delivery vectors: focus on Huntington’s disease. Mol Pharm 10:640–649. https://doi.org/10.1021/mp3003946
Godinho BMDC, Ogier JR, Quinlan A, Darcy R, Griffin BT, Cryan JF, Caitriona MOD (2014) PEGylated cyclodextrins as novel siRNA nanosystems: correlations between polyethylene glycol length and nanoparticle stability. Int J Pharm 473:105–112. https://doi.org/10.1016/j.ijpharm.2014.06.054
Gonzalez H, Hwang SJ, Davis ME (1999) New class of polymers for the delivery of macromolecular therapeutics. Bioconjug Chem 10:1068–1074. https://doi.org/10.1021/bc990072j
Gooding M, Malhotra M, McCarthy DJ, Godinho BMDC, Cryan JF, Darcy R, O’Driscoll CM (2015) Synthesis and characterization of rabies virus glycoprotein-tagged amphiphilic cyclodextrins for siRNA delivery in human glioblastoma cells: in vitro analysis. Eur J Pharm Sci 71:80–92. https://doi.org/10.1016/j.ejps.2015.02.007
Gorjikhah F, Azizi Jalalian F, Salehi R, Panahi Y, Hasanzadeh A, Alizadeh E, Akbarzadeh A, Davaran S (2017) Preparation and characterization of PLGA-β-cyclodextrin polymeric nanoparticles containing methotrexate and evaluation of their effects on T47D cell line. Artif Cells Nanomed Biotechnol 45:432–440. https://doi.org/10.3109/21691401.2016.1160915
Guo J, Ogier JR, Desgranges S, Darcy R, ODriscoll C (2012) Anisamide-targeted cyclodextrin nanoparticles for siRNA delivery to prostate tumours in mice. Biomaterials 33:7775–7784. https://doi.org/10.1016/j.biomaterials.2012.07.012
Guo B, Xu D, Liu X, Liao C, Li S, Huang Z, Li X, Yi J (2017) Characterization and cytotoxicity of PLGA nanoparticles loaded with formononetin cyclodextrin complex. J Drug Delivery Sci Technol 41:375–383. https://doi.org/10.1016/j.jddst.2017.08.010
Ha W, Kang Y, Peng SL, Ding LS, Zhang S, Li BJ (2013) Vesicular gold assemblies based on host-guest inclusion and its controllable release of doxorubicin. Nanotechnology 24. https://doi.org/10.1088/0957-4484/24/49/495103
Hamada H, Ishihara K, Masuoka N, Mikuni K, Nakajima N (2006) Enhancement of water-solubility and bioactivity of paclitaxel using modified cyclodextrins. J Biosci Bioeng 102:369–371. https://doi.org/10.1263/jbb.102.369
He M, Zhong C, Hu H, Jin Y, Chen Y, Lou K, Gao F (2019) Cyclodextrin/chitosan nanoparticles for oral ovalbumin delivery: preparation, characterization and intestinal mucosal immunity in mice. Asian J Pharm Sci 14:193–203. https://doi.org/10.1016/j.ajps.2018.04.001
Hippalgaonkar K, Adelli GR, Hippalgaonkar K, Repka MA, Majumdar S (2013) Indomethacin-loaded solid lipid nanoparticles for ocular delivery: development, characterization, and in vitro evaluation. J Ocul Pharmacol Ther 29:216–228. https://doi.org/10.1089/jop.2012.006
Holzinger M, Bouffier L, Villalonga R, Cosnier S (2009) Adamantane/β-cyclodextrin affinity biosensors based on single-walled carbon nanotubes. Biosens Bioelectron 24:1128–1134. https://doi.org/10.1016/j.bios.2008.06.029
Hou X, Zhang W, He M, Lu Y, Lou K, Gao F (2017) Preparation and characterization of β-cyclodextrin grafted N-maleoyl chitosan nanoparticles for drug delivery. Asian J Pharm Sci 12:558–568. https://doi.org/10.1016/j.ajps.2017.07.007
Hui Y, Ma X, Hou X, Chen F, Yu J (2015) Silver nanoparticles-β-cyclodextrin-graphene nanocomposites based biosensor for guanine and adenine sensing. Ionics (Kiel) 21:1751–1759. https://doi.org/10.1007/s11581-014-1343-5
Irie T, Uekama K (1997) Pharmaceutical applications of cyclodextrins. III. Toxicological issues and safety evaluation. J Pharm Sci 86:147–162. https://doi.org/10.1021/js960213f
Jain SK, Gupta Y, Jain A, Bhola M (2007) Multivesicular liposomes bearing celecoxib-β-cyclodextrin complex for transdermal delivery. Drug Deliv 14:327–335. https://doi.org/10.1080/10717540601098740
Jaiswal S, Bhattacharya K, McHale P, Duffy B (2015) Dual effects of β-cyclodextrin-stabilised silver nanoparticles: enhanced biofilm inhibition and reduced cytotoxicity. J Mater Sci Mater Med 26. https://doi.org/10.1007/s10856-014-5367-1
Ji T, Li S, Zhang Y, Lang J, Ding Y, Zhao X, Zhao R, Li Y, Shi J, Hao J, Zhao Y, Nie G (2016) An MMP-2 responsive liposome integrating antifibrosis and chemotherapeutic cyclodextrin drugs for enhanced drug perfusion and efficacy in pancreatic cancer. ACS Appl Mater Interfaces 8:3438–3445. https://doi.org/10.1021/acsami.5b11619
Kadari A, Gudem S, Kulhari H, Bhandi MM, Borkar RM, Kolapalli VRM, Sistla R (2017) Enhanced oral bioavailability and anticancer efficacy of fisetin by encapsulating as inclusion complex with HPβCD in polymeric nanoparticles. Drug Deliv 24:224–232. https://doi.org/10.1080/10717544.2016.1245366
Khalil SK, El-Feky GS, El-Banna ST, Khalil WA (2012) Preparation and evaluation of warfarin-β-cyclodextrin loaded chitosan nanoparticles for transdermal delivery. Carbohydr Polym 15:1244–1253. https://doi.org/10.1016/j.carbpol.2012.06.056
Krauland AH, Alonso MJ (2007) Chitosan/cyclodextrin nanoparticles as macromolecular drug delivery system. Int J Pharm 340:134–142. https://doi.org/10.1016/j.ijpharm.2007.03.005
Kurkov SV, Loftsson T (2013) Cyclodextrins. Int J Pharm 30:167–180. https://doi.org/10.1016/j.ijpharm.2012.06.055
Kwon TK, Kim JC (2010) In vitro skin permeation of mono olein nanoparticles containing hydroxypropyl beta-cyclodextrin/minoxidil complex. Int J Pharm 392:268–273. https://doi.org/10.1016/j.ijpharm.2010.03.04
Langie SAS, Knaapen AM, Houben JMJ, van Kempen FC, de Hoon JPJ, Gottschalk RWH, Godschalk RWL, van Schooten FJ (2007) The role of glutathione in the regulation of nucleotide excision repair during oxidative stress. Toxicol Lett 168:302–309. https://doi.org/10.1016/j.toxlet.2006.10.027
Li R, Liu S, Zhao J, Otsuka H, Takahara A (2011) Preparation of superparamagnetic β-cyclodextrin-functionalized composite nanoparticles with core-shell structures. Polym Bull 66:1125–1136. https://doi.org/10.1007/s00289-010-0410-y
Lin C, Chen F, Ye T, Zhang L, Zhang W, Liu D, Xiong W, Yang X, Pan W (2014) A novel oral delivery system consisting in “drug-in cyclodextrin-in nanostructured lipid carriers” for poorly water-soluble drug: Vinpocetine. Int J Pharm 465:90–96. https://doi.org/10.1016/j.ijpharm.2014.02.013
Liu J, Xu R, Kaifer AE (1998) In situ modification of the surface of gold colloidal particles. Preparation of cyclodextrin-based rotaxanes supported on gold nanospheres. Langmuir 14:7337–7339. https://doi.org/10.1021/la981385q
Loftsson T, Duchêne D (2007) Cyclodextrins and their pharmaceutical applications. Int J Pharm 329:1–11. https://doi.org/10.1016/j.ijpharm.2006.10.044
Luo Z, Cai K, Hu Y, Li J, Ding X, Zhang B, Xu D, Yang W, Liu P (2012) Redox-responsive molecular nanoreservoirs for controlled intracellular anticancer drug delivery based on magnetic nanoparticles. Adv Mater 24:431–435. https://doi.org/10.1002/adma.201103458
Luppi B, Bigucci F, Corace G, Delucca A, Cerchiara T, Sorrenti M, Catenacci L, Di Pietra AM, Zecchi V (2011) Albumin nanoparticles carrying cyclodextrins for nasal delivery of the anti-Alzheimer drug tacrine. Eur J Pharm Sci 44:559–565. https://doi.org/10.1016/j.ejps.2011.10.002
Lysik MA, Wu-Pong S (2003) Innovations in oligonucleotide drug delivery. J Pharm Sci 92:1559–1573. https://doi.org/10.1002/jps.10399
Mady FM, Abou-Taleb AE, Khaled KA, Yamasaki K, Iohara D, Taguchi K, Anraku M, Hirayama F, Uekama K, Otagiri M (2010) Evaluation of carboxymethyl-β-cyclodextrin with acid function: improvement of chemical stability, oral bioavailability and bitter taste of famotidine. Int J Pharm 397:1–8. https://doi.org/10.1016/j.ijpharm.2010.06.018
Maestrelli F, González-Rodríguez ML, Rabasco AM, Mura P (2005) Preparation and characterisation of liposomes encapsulating ketoprofen-cyclodextrin complexes for transdermal drug delivery. Int J Pharm 298:55–67. https://doi.org/10.1016/j.ijpharm.2005.03.033
Maestrelli F, Garcia-Fuentes M, Mura P, Alonso MJ (2006) A new drug nanocarrier consisting of chitosan and hydroxyl propyl cyclodextrin. Eur J Pharm Biopharm 63:79–86. https://doi.org/10.1016/j.ejpb.2005.12.006
Maestrelli F, González-Rodríguez ML, Rabasco AM, Ghelardini C, Mura P (2010) New “drug-in cyclodextrin-in deformable liposomes” formulations to improve the therapeutic efficacy of local anaesthetics. Int J Pharm 395:222–231. https://doi.org/10.1016/j.ijpharm.2010.05.046
Mahmoud AA, El-Feky GS, Kamel R, Awad GEA (2011) Chitosan/sulfobutylether-β-cyclodextrin nanoparticles as a potential approach for ocular drug delivery. Int J Pharm 413:229–236. https://doi.org/10.1016/j.ijpharm.2011.04.031
McCormack B, Gregoriadis G (1994a) Drugs-in-cyclodextrins-in liposomes: a novel concept in drug delivery. Int J Pharm 112:249–258. https://doi.org/10.1016/0378-5173(94)90361-1
McCormack B, Gregoriadis G (1994b) Entrapment of cyclodextrin-drug complexes into liposomes: potential advantages in drug delivery. J Drug Target 2:449–454. https://doi.org/10.3109/10611869408996821
McCormack B, Gregoriadis G (1996) Comparative studies of the fate of free and liposome-entrapped hydroxypropyl-β-cyclodextrin/drug complexes after intravenous injection into rats: implications in drug delivery. Biochim Biophys Acta 1291:237–244. https://doi.org/10.1016/S0304-4165(96)00096-7
Memişoğlu E, Bochot A, Özalp M, Sen M, Duchêne D, Hincal A (2003) Direct formation of nanospheres from amphiphilic beta-cyclodextrin inclusion complexes. Pharm Res 20:117–125. https://doi.org/10.1023/A:1022263111961
Bilensoy E, Vural I, Bochot A, Renoir JM, Duchene D, Hincal AA (2005) Tamoxifen citrate loaded amphiphilic β-cyclodextrin nanoparticles: in vitro characterization and cytotoxicity. J Control Release 104:489–496. https://doi.org/10.1016/j.jconrel.2005.03.006
Méndez-Ardoy A, Guilloteau N, Di Giorgio C, Vierling P, Santoyo-González F, Ortiz Mellet C, García Fernández JM (2011) β-cyclodextrin-based polycationic amphiphilic “click” clusters: effect of structural modifications in their DNA complexing and delivery properties. J Org Chem 76:5882–5894. https://doi.org/10.1021/jo2007785
Mennini N, Cirri M, Maestrelli F, Mura P (2016) Comparison of liposomal and NLC (nanostructured lipid carrier) formulations for improving the transdermal delivery of oxaprozin: effect of cyclodextrin complexation. Int J Pharm 515:684–691. https://doi.org/10.1016/j.ijpharm.2016.11.013
Merkus FWHM, Verhoef JC, Romeijn SG, Schipper NGM (1991) Absorption enhancing effect of cyclodextrins on intranasally administered insulin in rats. Pharm Res 8:588–592. https://doi.org/10.1023/A:1015896405389
Merkus FWHM, Verhoef JC, Marttin E, Romeijn SG, Van Der Kuy PHM, Hermens WAJJ, Schipper NGM (1999) Cyclodextrins in nasal drug delivery. Adv Drug Deliv Rev 36:41–57. https://doi.org/10.1016/S0169-409X(98)00054-4
Messner M, Kurkov SV, Jansook P, Loftsson T (2010) Self-assembled cyclodextrin aggregates and nanoparticles. Int J Pharm 387:199–208. https://doi.org/10.1016/j.ijpharm.2009.11.035
Mrówczyński R, Jędrzak A, Szutkowski K, Grześkowiak BF, Coy E, Markiewicz R, Jesionowski T, Jurga S (2018) Cyclodextrin-based magnetic nanoparticles for cancer therapy. Nanomaterials (Basel) 8:170. https://doi.org/10.3390/nano8030170
Mura P, Maestrelli F, Cecchi M, Bragagni M, Almeida A (2010) Development of a new delivery system consisting in drug in cyclodextrin in PLGA nanoparticles. J Microencapsul 27:479–486. https://doi.org/10.3109/02652040903515508
Negi JS, Chattopadhyay P, Sharma AK, Ram V (2014) Preparation of gamma cyclodextrin stabilized solid lipid nanoparticles (SLNS) using stearic acid-γ-cyclodextrin inclusion complex. J Incl Phenom Macrocycl Chem 80:359–368. https://doi.org/10.1007/s10847-014-0415-5
Nguyen HT, Goycoolea FM (2017) Chitosan/Cyclodextrin/TPP nanoparticles loaded with quercetin as novel bacterial quorum sensing inhibitors. Molecules 22:1975. https://doi.org/10.3390/molecules22111975
Park IK, Von Recum HA, Jiang S, Pun SH (2006) Supramolecular assembly of cyclodextrin-based nanoparticles on solid surfaces for gene delivery. Langmuir 22:8478–8484. https://doi.org/10.1021/la061757s
Piel G, Piette M, Barillaro V, Castagne D, Evrard B, Delattre L (2006) Betamethasone-in-cyclodextrin-in-liposome: the effect of cyclodextrins on encapsulation efficiency and release kinetics. Int J Pharm 312:75–82. https://doi.org/10.1016/j.ijpharm.2005.12.044
Ping Y, Liu C, Zhang Z, Liu KL, Chen J, Li J (2011) Chitosan-graft-(PEI-β-cyclodextrin) copolymers and their supramolecular PEGylation for DNA and siRNA delivery. Biomaterials 32:8328–8341. https://doi.org/10.1016/j.biomaterials.2011.07.038
Pires FQ, da Silva JKR, Sa-Barreto LL, Gratieri T, Gelfuso GM, Cunha-Filho M (2019) Lipid nanoparticles as carriers of cyclodextrin inclusion complexes: a promising approach for cutaneous delivery of a volatile essential oil. Colloids Surf B Biointerfaces 182:110382. https://doi.org/10.1016/j.colsurfb.2019.110382
Prabha G, Raj V (2016) Formation and characterization of β-cyclodextrin (β-cyclodextrin) – polyethyleneglycol (PEG)-polyethyleneimine (PEI) coated Fe3O4 nanoparticles for loading and releasing 5-fluorouracil drug. Biomed Pharmacother 80:173–182. https://doi.org/10.1016/j.biopha.2016.03.015
Prandina A, Herfindal L, Radix S, Rongved P, Døskeland SO, Le Borgne M, Perret F (2018) Enhancement of iodinin solubility by encapsulation into cyclodextrin nanoparticles. J Enzyme Inhib Med Chem 33:370–375. https://doi.org/10.1080/14756366.2017.1421638
Quaglia F, Ostacolo L, Mazzaglia A, Villari V, Zaccaria D, Sciortino MT (2009) The intracellular effects of non-ionic amphiphilic cyclodextrin nanoparticles in the delivery of anticancer drugs. Biomaterials 30:374–382. https://doi.org/10.1016/j.biomaterials.2008.09.035
Ren S, Chen D, Jiang M (2009) Noncovalently connected micelles based on a β-cyclodextrin-containing polymer and adamantane end-capped poly(ε-caprolactone) via host-guest interactions. J Polym Sci Part A Polym Chem 47:4267–4278. https://doi.org/10.1002/pola.23479
Sajeesh S, Sharma CP (2006) Cyclodextrin-insulin complex encapsulated polymethacrylic acid based nanoparticles for oral insulin delivery. Int J Pharm 325:147–154. https://doi.org/10.1016/j.ijpharm.2006.06.019
Schardinger F (1903) Uber die Zulässigkeit des Warmhaltens von zum Gebuß bestimmten Nahrungsmittel mittelst Wärme speichernder Apparate, sog. Thermophore Wien K lin Wochenschr:468–474
Sierpe R, Lang E, Jara P, Guerrero AR, Chornik B, Kogan MJ, Yutronic N (2015) Gold nanoparticles interacting with β-cyclodextrin-phenylethylamine inclusion complex: a ternary system for photothermal drug release. ACS Appl Mater Interfaces 7:15177–15181. https://doi.org/10.1021/acsami.5b00186
Škalko N, Brandl M, Bećirević-Laćan M, Filipović-Grčić J, Jalšenjak I (1996) Liposomes with nifedipine and nifedipine-cyclodextrin complex: calorimetrical and plasma stability comparison. Eur J Pharm Sci 4:359–366. https://doi.org/10.1016/S0928-0987(96)00180-7
Skiba M, Wouessidjewe D, Coleman A, Fessi H, Devissaguet JP, Duchene D, Puisieux F (1993) Preparation and use of novel cyclodextrin-based dispersible colloidal systems in the form of nanospheres. PCT Int Appl 23 pp. PCT Applications FR 93/00594 (Publication No. WO 93/25195)
Skiba M, Duchêne D, Puisieux F, Wouessidjewe D (1996) Development of a new colloidal drug carrier from chemically modified cyclodextrins: nanospheres and influence of physicochemical and technological factors on particle size. Int J Pharm 129:113–121. https://doi.org/10.1016/0378-5173(95)04272-5
Spada G, Gavini E, Cossu M, Rassu G, Giunchedi P (2012) Solid lipid nanoparticles with and without hydroxypropyl β-cyclodextrin: a comparative study of nanoparticles designed for colonic drug delivery. Nanotechnology:23. https://doi.org/10.1088/0957-4484/23/9/095101
Szente L, Szejtli J (1999) Highly soluble cyclodextrin derivatives: chemistry, properties, and trends in development. Adv Drug Deliv Rev 36:17–28. https://doi.org/10.1016/S0169-409X(98)00092-1
Tao HQ, Meng Q, Li MH, Yu H, Liu MF, Du D, Sun SL, Yang HC, Wang YM, Ye W, Yang LZ, Zhu DL, Jiang CL, Peng HS (2013) HP-β-cyclodextrin-PLGA nanoparticles improve the penetration and bioavailability of puerarin and enhance the therapeutic effects on brain ischemia-reperfusion injury in rats. Naunyn Schmiedeberg’s Arch Pharmacol 386:61–70. https://doi.org/10.1007/s00210-012-0804-5
Teijeiro-Osorio D, Remuñán-López C, Alonso MJ (2009) Chitosan/cyclodextrin nanoparticles can efficiently transfect the airway epithelium in vitro. Eur J Pharm Biopharm 71:257–263. https://doi.org/10.1016/J.EJPB.2008.09.020
Trapani A, Garcia-Fuentes M, Alonso MJ (2008) Novel drug nanocarriers combining hydrophilic cyclodextrins and chitosan. Nanotechnology 19. https://doi.org/10.1088/0957-4484/19/18/185101
Trapani A, Sitterberg J, Bakowsky U, Kissel T (2009) The potential of glycol chitosan nanoparticles as carrier for low water soluble drugs. Int J Pharm 375:97–106. https://doi.org/10.1016/j.ijpharm.2009.03.041
Trapani A, Lopedota A, Franco M, Cioffi N, Ieva E, Garcia-Fuentes M, Alonso MJ (2010) A comparative study of chitosan and chitosan/cyclodextrin nanoparticles as potential carriers for the oral delivery of small peptides. Eur J Pharm Biopharm 75:26–32. https://doi.org/10.1016/j.ejpb.2010.01.010
Vaidya B, Parvathaneni V, Kulkarni NS, Shukla SK, Damon JK, Sarode A, Kanabar D, Garcia JV, Mitragotri S, Muth A, Gupta V (2019) Cyclodextrin modified erlotinib loaded PLGA nanoparticles for improved therapeutic efficacy against non-small cell lung cancer. Int J Biol Macromol 122:338–347. https://doi.org/10.1016/j.ijbiomac.2018.10.181
Vakhariya RR, Talokar SS, Salunkhe VR, Magdum CS (2017) Formulation development and optimization of simvastatin loaded solid lipid nanoparticles. Asian J Res Pharm Sci 7:49. https://doi.org/10.5958/2231-5659.2017.00008.x
Varan G, Öncül S, Ercan A, Benito JM, Ortiz Mellet C, Bilensoy E (2016) Cholesterol-targeted anticancer and apoptotic effects of anionic and polycationic amphiphilic cyclodextrin nanoparticles. J Pharm Sci 105:3172–3182. https://doi.org/10.1016/j.xphs.2016.06.021
Varan G, Patrulea V, Borchard G, Bilensoy E (2018) Cellular interaction and tumoral penetration properties of cyclodextrin nanoparticles on 3D breast tumor model. Nanomaterials 8:1–18. https://doi.org/10.3390/nano8020067
Wang Y, Wong JF, Teng X, Lin XZ, Yang H (2003) “Pulling” nanoparticles into water: phase transfer of oleic acid stabilized monodisperse nanoparticles into aqueous solutions of α-cyclodextrin. Nano Lett 3:1555–1559. https://doi.org/10.1021/nl034731j
Wang T, Zhang C, Liang XJ, Liang W, Wu Y (2011) Hydroxypropyl-β-cyclodextrin copolymers and their nanoparticles as doxorubicin delivery system. J Pharm Sci 100:1067–1079. https://doi.org/10.1002/jps.22352
Wang X, Chen C, Huo D, Qian H, Ding Y, Hu Y, Jiang X (2012) Synthesis of β-cyclodextrin modified chitosan-poly(acrylic acid) nanoparticles and use as drug carriers. Carbohydr Polym 90:361–369. https://doi.org/10.1016/j.carbpol.2012.05.052
Wang Y, Li H, Jin Q, Ji J (2016a) Intracellular host-guest assembly of gold nanoparticles triggered by glutathione. Chem Commun 52:582–585. https://doi.org/10.1039/c5cc07195j
Wang D, Wang T, Xu Z, Yu H, Feng B, Zhang J, Guo C, Yin Q, Zhang Z, Li Y (2016b) Cooperative treatment of metastatic breast cancer using host-guest nanoplatform coloaded with docetaxel and siRNA. Small 12:488–498. https://doi.org/10.1002/smll.201502913
Wang X, Gao Z, Zhang L, Wang H, Hu X (2018, 2018) A magnetic and pH-sensitive composite nanoparticle for drug delivery. J Nanomater. https://doi.org/10.1155/2018/1506342
Weiss G, Yen Y, Neidhart J, Young CS, Weiss S, Ryan J (2011) Abstract 1306: a phase 1b/2a study of CRLX101, a novel nanopharmaceutical of camptothecin, in solid tumor cancer patients who have failed standard therapy. Cancer Res 71:1306–1306. https://doi.org/10.1158/1538-7445.am2011-1306
Xia HB, Yi J, Foo PS, Liu B (2007) Facile fabrication of water-soluble magnetic nanoparticles and their spherical aggregates. Chem Mater 19:4087–4091. https://doi.org/10.1021/cm070918q
Xu Z, Liu S, Liu H, Yang C, Kang Y, Wang M (2015) Unimolecular micelles of amphiphilic cyclodextrin-core star-like block copolymers for anticancer drug delivery. Chem Commun 51:15768–15777. https://doi.org/10.1039/c5cc02743h
Ye Z, Zhang Q, Wang S, Bharate P, Varela-Aramburu S, Lu M, Seeberger PH, Yin J (2016) Tumor-targeted drug delivery with mannose-functionalized nanoparticles self-assembled from amphiphilic β-cyclodextrins. Chem Eur J 22:15216–15221. https://doi.org/10.1002/chem.201603294
Yuan Z, Ye Y, Gao F, Yuan HH, Lan M, Lou K, Wang W (2013) Chitosan-graft-β-cyclodextrin nanoparticles as a carrier for controlled drug release. Int J Pharm 446:191–198. https://doi.org/10.1016/j.ijpharm.2013.02.024
Zafar N, Fessi H, Elaissari A (2013) Cyclodextrin containing biodegradable particles: from preparation to drug delivery applications. Int J Pharm 461:351–366. https://doi.org/10.1016/j.ijpharm.2013.12.004
Zhai Z, Zhang F, Chen X, Zhong J, Liu G, Tian Y, Huang Q (2017) Uptake of silver nanoparticles by DHA-treated cancer cells examined by surface-enhanced Raman spectroscopy in a microfluidic chip. Lab Chip 17:1306–1313. https://doi.org/10.1039/c7lc00053g
Zhang J, Ma PX (2009) Polymeric core-shell assemblies mediated by host-guest interactions: versatile nanocarriers for drug delivery. Angew Chem Int Ed 48:964–968. https://doi.org/10.1002/anie.200804135
Zhang J, Ma PX (2013) Cyclodextrin-based supramolecular systems for drug delivery: recent progress and future perspective. Adv Drug Deliv Rev 65:1215–1233. https://doi.org/10.1016/j.addr.2013.05.001
Zhang X, Wu Z, Gao X, Shu S, Zhang H, Wang Z, Li C (2009) Chitosan bearing pendant cyclodextrin as a carrier for controlled protein release. Carbohydr Polym 77:394–401. https://doi.org/10.1016/j.carbpol.2009.01.018
Zhang N, Li JJ, Jiang W, Ren C, Li JJ, Xin J, Li K (2010) Effective protection and controlled release of insulin by cationic β-cyclodextrin polymers from alginate/chitosan nanoparticles. Int J Pharm 393:213–219. https://doi.org/10.1016/j.ijpharm.2010.04.006
Zhang X, Zheng X, Wu Z, Gao X, Shu S, Wang Z, Li C (2011) β-Cyclodextrin grafting hyperbranched polyglycerols as carriers for nasal insulin delivery. Carbohydr Polym 84:1419–1425. https://doi.org/10.1016/j.carbpol.2011.01.057
Zokaei E, Badoei-dalfrad A, Ansari M (2019) Therapeutic potential of DNAzyme loaded on chitosan/cyclodextrin nanoparticle to recovery of chemosensitivity in the MCF-7 cell line. Appl Biochem Biotechnol 187:708. https://doi.org/10.1007/s12010-018-2836-x
Zuckerman JE, Gritli I, Tolcher A, Heidel JD, Lim D, Morgan R, Chmielowski B, Ribas A, Davis ME, Yen Y (2014) Correlating animal and human phase Ia/Ib clinical data with CALAA-01, a targeted, polymer-based nanoparticle containing siRNA. Proc Natl Acad Sci U S A 111:11449–11454. https://doi.org/10.1073/pnas.1411393111
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 The Editor(s) (if applicable) and The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pandey, A. (2020). Role of Cyclodextrins in Nanoparticle-Based Systems for Drug Delivery. In: Crini, G., Fourmentin, S., Lichtfouse, E. (eds) The History of Cyclodextrins. Environmental Chemistry for a Sustainable World, vol 52. Springer, Cham. https://doi.org/10.1007/978-3-030-49308-0_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-49308-0_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-49307-3
Online ISBN: 978-3-030-49308-0
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)