Abstract
Pediatric traumatic brain injury (TBI) is the most common cause of acquired brain injury in childhood and leads to neuropathological, neurocognitive, and neurobehavioral morbidities. Unfortunately, these morbidities frequently persist and impact day-to-day long-term outcomes including adaptive behavior, educational attainment, employment, and quality of life. Relative risk of poor outcome is influenced by a variety of individual (e.g., age at injury, cognitive reserve), socioeconomic, and injury-related (e.g., duration of posttraumatic amnesia) factors. Certain sequelae, particularly neurocognitive deficits, have been tied to neuroanatomical abnormalities, most often via volumetric and diffusion tensor magnetic resonance imaging. While application of cognitive rehabilitation approaches has lagged behind that found in adult TBI, recent research has targeted neurocognitive function after pediatric TBI, with modest success albeit limited evidence of far transfer of effects. Neuroimaging has been very seldom used to assess treatment-related changes in brain system integrity after pediatric TBI but provides a robust opportunity to elucidate the mechanisms underlying response to intervention in this population.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Pediatric TBI Background
Epidemiology of Pediatric TBI
Traumatic brain injury (TBI) occurs when the usual function of the brain is disrupted by a bump, blow, or jolt to the head or by a penetrating injury (Marr & Coronado, 2004). TBI is the most common cause of acquired brain injury in childhood and is a leading cause of death and disability in children and adolescents (Faul, Xu, Wald, & Coronado, 2010). Although prevalence and incidence rate reports vary across sources, recent estimates provided by the United States Centers for Disease Control and Prevention (CDC) indicate that an estimated 640,000 emergency department visits and 18,000 hospitalizations in 2013 were due to TBI (Taylor, Bell, Breiding, & Xu, 2017). Compared to estimates from a decade prior (i.e., 2002–2006), this is a slight increase in emergency department visits, but a significant decrease in hospitalizations, down from roughly 60,000 (Faul, Wald, Xu, & Coronado, 2010). This may not reflect a true increase in the incidence of TBI, but rather a growing public health concern about TBI (particularly mild TBI, or concussion) and corresponding state legislation dictating healthcare provider clearance to return to play for youth athletes.
Rates of pediatric TBI and associated emergency department visits and hospitalizations are not distributed randomly across the population or across injury mechanism; rather, they vary based on both demographic and injury-related factors. Boys are about twice as likely to sustain a TBI as girls across all ages. Incidence rates are particularly high among very young children (i.e., ≤4 years old; 1,035 per 100,000), largely due to injuries sustained in falls or as a consequence of inflicted, non-accidental head trauma. Non-accidental trauma (e.g., injury due to abuse or assault) also contributes to the relatively greater mortality rate in this age group. Among older children and adolescents, falls remain a common mechanism of injury, as do motor vehicle accidents and other transportation-related injuries (e.g., bicycle accidents), although sport- and recreation-related injuries are the single most common mechanism. Older adolescents (i.e., ages 15–19 years) have a higher rate of hospitalization and death from TBI, given the frequency of injuries sustained in motor vehicle collision, which tend to be disproportionately more severe than injury by other mechanisms.
Socioeconomic status and race are also linked to variability in incidence and prevalence rates (especially for more severe injuries), hospitalizations, and deaths from TBI arising from motor vehicle crashes; specifically, among young children (i.e., ages 0–9 years), those who are Black are more likely to be hospitalized or die from their injuries, as are those of lower socioeconomic status. Black, Hispanic, and Native American children are also more likely than White children to sustain a severe TBI and have a higher mortality rate (Centers for Disease Control and Prevention, 2011; Falcone Jr, Martin, Brown, & Garcia, 2008; Howard, Joseph, & Natale, 2005; Linton & Kim, 2014). Pre-injury emotional and behavioral functioning may also play a role in the incidence of TBI; children with pre-injury psychiatric disorders (e.g., ADHD, anxiety), for example, are more likely to sustain a TBI than are children with no psychiatric history (Bloom et al., 2001).
Rates of pediatric TBI also vary based on injury severity. Though imperfect, the most common method of classification of TBI severity is the Glasgow Coma Scale (Teasdale & Jennett, 1974), which integrates information about the extent of motor response, verbal response, and eye-opening to rate level of consciousness. Specific criteria are outlined in Table 11.1, with total scores ranging from 3 to 15; scores from 3 to 8 are traditionally classified as severe, scores from 9 to 12 as moderate, and scores from 13 to 15 as mild. Some injuries are described as “complicated mild,” which indicates GCS score of 13–15 accompanied by significant abnormalities on neuroimaging (typically computed tomography [CT] scans) conducted at the time of injury. Often additional criteria are considered when assessing TBI severity; for instance, assessment using GCS alone can lead to questionable classification when a child is particularly young, or when a child or adolescent requires medical sedation. In these cases, classification metrics such as the pediatric version of the GCS (pGCS), duration of loss of consciousness, or duration of posttraumatic amnesia can provide important additional information. Criteria for the pGCS are annotated(*) in Table 11.1, and a summary of classification criteria is provided in Table 11.2.
Regardless of method, the vast majority of TBIs in childhood are classified as mild. Though somewhat variable across studies, epidemiological estimates indicate that between 70 and 90% of TBI-related visits to the emergency department are for injuries eventually classified as mild TBI (Cassidy et al., 2004; Faul et al., 2010; Rivara et al., 2012). TBI places a significant financial burden on the public healthcare system. One recent study estimated that, within the first 3 months after an injury, related costs exceeded $1,000 for mild injuries, $4,300 for moderate injuries, and $7,200 for severe injuries. Though costs are clearly higher for individual severe TBI relative to mild TBI, population-level costs for mild TBI were estimated to approach $700 million within 3 months after injury because of the high frequency of mild TBI, while costs for moderate and severe TBI were estimated at $81 and $175 million, respectively (Graves, Rivara, & Vavilala, 2015).
Fortunately, overall mortality rates are on the decline, with roughly 1,500 deaths attributed to TBI in 2013 (Taylor et al., 2017) relative to an average of 6,100 between 2002 and 2006 (Faul et al., 2010). As mortality from TBI decreases, long-term morbidities are increasingly recognized and have been the focus of concerted research efforts. At least 145,000 children and adolescents are thought to be living with persistent and significant social, behavioral, physical, or cognitive sequelae from TBI (Zaloshnja, Miller, Langlois, & Selassie, 2008). These long-term morbidities vary widely by injury severity and associated neuropathological and pathophysiological characteristics of the injury.
Neuropathology and Pathophysiology of Pediatric TBI
Pediatric TBI can result in a variety of neuropathological changes. Some are a direct result of the impact on the brain itself, whereas others are a consequence of a cascade of neurochemical processes that follow the impact. Some changes occur immediately upon impact, while others emerge or evolve over the days, weeks, and months following the injury (Farkas & Povlishock, 2007; Giza & Hovda, 2001; Povlishock & Katz, 2005; Toledo et al., 2012). Structural neuropathological changes are often discussed as either primary or secondary consequences of TBI. Primary injuries occur as a direct result of the force to the head and include things like skull fracture, lacerations and contusions, and shear or strain injury to brain tissue or vasculature. In contrast, secondary injuries come about indirectly; some examples include brain swelling or edema and increased intracranial pressure. Swelling can also cause a reduction in blood flow, leading to hypoxic or ischemic injury.
Primary injuries reflect both the biomechanics and the physics of injury. In many cases, these injuries occur due to the properties of acceleration–deceleration. Upon impact, a child’s skull may abruptly stop, but the brain tissue does not. In addition to a possible skull fracture, this acceleration–deceleration can cause tearing of blood vessels and shear injury to nerve fibers. These injuries contribute to evolving hemorrhage and diffuse axonal injury, respectively (Yeates, 2010a). Focal lesions may also occur; they disproportionately occur in the anterior frontal and temporal brain regions (Wilde et al., 2005) partially because of the greater likelihood of sustaining a TBI to the front of the head, and partially due to the presence of bony prominences in the anterior and middle fossa of the skull (Bigler, 2007).
In many cases, secondary injuries that arise over time are the most life threatening, including brain swelling, cerebral edema, and associated increased intracranial pressure. When swelling occurs, the normal balance of blood volume, cortical tissue, and cerebrospinal fluid is disrupted. When cerebral blood flow is diminished, blood volume can increase, further contributing to elevated intracranial pressure. At their worst, swelling and elevated intracranial pressure can cause herniation of the brain and even death; at milder levels, hypoxic or ischemic injury can occur, exacerbating any associated excitotoxic damage (Toledo et al., 2012; Yeates, 2010a).
Other neuropathological changes occur due to neuroinflammatory and metabolic cascades during and after the acute injury period. After TBI, inflammation can lead to an excess of microglia and microphages, as well as reactive astrogliosis (Kelley, Lifshitz, & Povlishock, 2007; Shultz, Bao, et al., 2012, Shultz, MacFabe, Foley, Taylor, & Cain, 2012). Additionally, TBI can lead to the excessive production of free radicals and excitatory amino acids, which can disrupt cell metabolism and lead to cell death (Hunter, Wilde, Tong, & Holshouser, 2012). Several studies utilizing magnetic resonance spectroscopy have noted reduced N-acetylaspartate (NAA), even in the absence of visible injury on conventional neuroimaging. This likely reflects the neuronal dysfunction and neuronal loss that occurs acutely after TBI. Choline, on the other hand, is often elevated due to shear injury to myelin and cell membranes. Glutamate is another excitatory amino acid that can be released into extracellular space after TBI. This also contributes to neuronal death, particularly in regions with a large number of glutamate receptors such as the hippocampus (Wilde et al., 2007). TBI can also disrupt normal cellular calcium homeostasis; when elevated, circulating calcium can worsen the effects of hypoxic or ischemic injury.
Although some of the acute neuropathological changes that occur after TBI are expected to abate over time, others are more lasting. Specifically, while metabolic and neurochemical changes are temporary, cortical thinning, cerebral atrophy, and associated ventricular enlargement can be persistent. Diffuse axonal injury and white matter degeneration persist, especially in cases of severe TBI. Recent longitudinal research has provided a clearer appreciation of the temporal dynamics of white matter pathology after TBI (e.g., Dennis, Babikian, Giza, Thompson, & Asarnow, 2017; Wilde, Ayoub, Bigler, Chu, Hunter, Wu, et al., 2012). White matter injury is most often assessed using diffusion tensor imaging (DTI), which quantifies the extent of the restriction of diffusion of water along axons. During DTI, information about water diffusion is collected across a variety of spatial directions and modeled as an ellipsoid. The most common DTI metric used to convey white matter organization is fractional anisotropy (FA), which is the ratio of the extent of diffusion along the three main axes. Ranging from 0 to 1, an FA value of 0 indicates entirely isotropic diffusion, while an FA value of 1 indicates entirely anisotropic diffusion (i.e., perfect diffusion along the white matter tract). Thus, lower FA values generally suggest compromised white matter integrity. Close to the time of injury (i.e., within 3 months), however, FA tends to increase due to the impact of brain swelling on the restriction of water diffusivity. In contrast, farther from the time of injury, FA tends to decrease as swelling recedes and damage to white matter can be detected more clearly.
Typical Cognitive Profile and Impact on Everyday Functioning
The typical neurocognitive and neurobehavioral consequences of TBI involve domains that correspond to the brain systems most vulnerable to traumatic injury, particularly the anterior frontal and temporal cortical regions and white matter pathways. Subcortical structures can also be affected, leading to difficulties with autonomic regulation and motor coordination. The assessment of the neurocognitive and behavioral sequelae of TBI demands an appreciation for normal developmental processes and of how recovery or the emergence of deficits occurs in both a developmental and environmental context that has implications for both prognosis and intervention.
After mild TBI (especially a single, uncomplicated mild TBI), the development of long-term cognitive deficits is highly unlikely (Babikian et al., 2011; Babikian & Asarnow, 2009; Bijur, Haslum, & Golding, 1990; Brosseau-Lachaine, Gagnon, Forget, & Faubert, 2008; Carroll et al., 2004; Hung et al., 2014; Yeates, 2010b; Yeates et al., 1999, 2009). More likely, falling outside of the realm of cognitive deficits, nearly a third of children and adolescents who sustain a mild TBI endorse behavioral or psychological symptoms that persist beyond 4 weeks. In some cases, these symptoms reflect pre-injury health or psychosocial factors rather than the injury itself, perhaps exacerbated by inaccurate expectations a child or family may have about the consequences of mild TBI (Covassin, Moran, & Wilhelm, 2013; Eisenberg, Andrea, Meehan, & Mannix, 2013).
Overall, neurocognitive and behavioral sequelae of TBI are far more common in moderate to severe TBI. The typical pattern of deficits following moderate to severe injury is outlined below.
Neurocognitive Functioning
A wealth of literature outlines associations between TBI and outcomes in various neurocognitive domains. Early research efforts relied on global assessment of intellectual functioning and found that children with TBI performed more poorly on IQ measures relative to either healthy control or orthopedic injury control groups. Global measures have been criticized, however, as they can obscure more nuanced strengths and weaknesses. For instance, children with TBI often perform more poorly on nonverbal intelligence tasks, or those with a speed-based component, relative to verbal intelligence measures (Babikian & Asarnow, 2009). Thus, more recent investigations have focused on more specific aspects of neurocognitive functioning.
The most common and specific neurocognitive changes following TBI include deficits in nonverbal skills, attention, memory , processing speed, and executive functions (e.g., cognitive flexibility, working memory, planning, organization, self-regulation; Babikian & Asarnow, 2009; Babikian, Merkley, Savage, Giza, & Levin, 2015). Although overt aphasias are rare following the acute injury period, subtler language deficits can persist, particularly in pragmatic domains (Yeates, 2010a).
Executive dysfunction is particularly common, perhaps due to the contribution of prefrontal and cingulate regions to these skills and the vulnerability of such regions to injury. These deficits can persist, even up to 10 years post-injury in cases of severe TBI (Beauchamp et al., 2011; Mangeot, Armstrong, Colvin, Yeates, & Taylor, 2002). In terms of prevalence, one study found that roughly 20% of children with severe TBI displayed symptoms consistent with attention-deficit/hyperactivity disorder (ADHD) , compared to only 4% of children with orthopedic injury. When premorbid history of attention problems was taken into account, rates of post-injury clinically significant symptoms of attention problems on the CBCL increased to 82% for children with severe TBI, relative to 42% of children with orthopedic injury (Yeates et al., 2005).
Psychosocial Adjustment/Psychiatric Functioning
Emotional and behavioral disorders can also emerge following TBI and can also contribute to overall impairment, decreased quality of life, and poor functional independence. Behavior problems and mood disorders such as depression or anxiety are common, more so in children and adolescents with severe injuries (Schwartz et al., 2003), affecting up to nearly two-thirds of those with severe TBI (Bloom et al., 2001). Children and adolescents with TBI are more likely than their healthy peers to display disruptive behaviors, conduct problems, and emotional distress (Anderson, Catroppa, Haritou, Morse, & Rosenfeld, 2005) and are at increased risk of engaging in risky lifestyle behaviors such as substance use and self-harm (Babikian et al., 2015). Unfortunately, although many of the cognitive deficits that occur after TBI subside over time, emotional and behavioral symptoms are more persistent, and may even worsen (Fay et al., 2009), especially in the context of poor family functioning or discord (Yeates et al., 2001).
Psychosocial morbidities are also common after TBI. Children and adolescents may become socially withdrawn from peer groups or become reluctant to participate in previously enjoyed activities (Bedell & Dumas, 2004). They may display poor social skills, lower self-esteem about social skills, reduced social participation and peer interaction, and greater loneliness and isolation. Their poor social outcomes may be related to impairment in basic social skills, poor self-regulation during social interactions, and difficulties with social problem-solving. Studies examining predictors of social adjustment have noted that the developmental timing of the injury, injury severity and location, and the impact of the TBI on basic cognitive and social-cognitive skills are important considerations (Rosema, Crowe, & Anderson, 2012; Yeates et al., 2007). Furthermore, the quality and pervasiveness of social difficulties can change over time. In a sense, children can “grow into” social deficits as they transition to adolescence, likely due to the increasing complexity of social relationships (Tonks et al., 2009; Turkstra, 2000; Turkstra, McDonald, & DePompei, 2001). Although neurocognitive deficits may play a role in difficulties navigating the complex social world of adolescence, other aspects of social information processing contribute more to the variability in social adjustment observed after TBI. Theory of mind, or the ability to make inferences about the internal or mental states of others, can be particularly challenging for children following severe TBI and accounts for significant variance in social adjustment, above and beyond performance on measures of executive function or processing speed (Robinson, Fountain-Zaragoza, et al., 2014).
Adaptive Functioning
Traumatic brain injury can also impact day-to-day adaptive functioning. Following TBI, particularly severe injuries, children and adolescents are at increased risk of deficits in adaptive functioning, including reduced social participation, leisure skills, and social adjustment (Shultz et al., 2016). Deficits in communication, daily living skills, and other functional behaviors have also been documented (Catroppa, Anderson, Morse, Haritou, & Rosenfeld, 2008; Fay et al., 2009; Stancin et al., 2002; Taylor et al., 1999, 2002), particularly when family functioning is poor (Taylor et al., 1999). Unfortunately, these types of deficits also tend to persist and contribute to worse long-term outcomes (Cameto, Levine, & Wagner, 2004; Koskiniemi, Kyykkä, Nybo, & Jarho, 1995; Nybo, Sainio, & Müller, 2004; Sariaslan, Sharp, D’Onofrio, Larsson, & Fazel, 2016; Todis & Glang, 2008; Todis, Glang, Bullis, Ettel, & Hood, 2011). In the long term, young adults who sustain a severe TBI during childhood have fewer close friendships, experience poorer educational outcomes, have higher rates of unemployment or underemployment, and endorse lower quality of life, compared to healthy peers (Anderson, Brown, Newitt, & Hoile, 2009; Cattelani, Lombardi, Brianti, & Mazzucchi, 1998; Todis & Glang, 2008).
Factors that Influence Outcomes
One intriguing facet of pediatric TBI is the heterogeneity in long-term cognitive, emotional, and behavioral outcomes. As noted above in the discussion of TBI incidence and prevalence rates, a number of factors are linked with outcomes, including characteristics of the injured child, demographic and family factors, characteristics of the injury itself, and associated neuroanatomical abnormalities.
Individual Characteristics
The types and severity of long-term consequences of pediatric TBI vary based on characteristics of the individual child or adolescent. One important factor is the age at injury. The impact of acceleration–deceleration on diffuse brain systems (particularly white matter) and the diffuse consequences of secondary injuries contribute to poorer outcomes among those who sustain injuries at a younger age (Anderson, Bechara, Damasio, Tranel, & Damasio, 1999; Barnes, Dennis, & Wilkinson, 1999; Dennis, Wilkinson, Koski, & Humphreys, 1995; Ewing-Cobbs et al., 1997; Taylor & Alden, 1997; Verger et al., 2001). Early injury is also linked with poorer distal outcomes like poor employment outcomes (Anderson, Catroppa, Morse, Haritou, & Rosenfeld, 2005; Todis et al., 2011).
An individual’s pre-injury functioning can also predict long-term outcomes after TBI. Children with better cognitive functioning prior to an injury preserve more functional capacity afterward (Dennis, 2000; Dennis, Yeates, Taylor, & Fletcher, 2007; Kesler, Adams, Blasey, & Bigler, 2003), consistent with theories of “cognitive reserve.” The cognitive reserve hypothesis (Kesler et al., 2003; Stern, 2003) proposes that those with higher pre-morbid functioning have preserved functional capacity after CNS injury due to the brain’s ability to make efficient use of existing networks, which has been supported in few available studies of pediatric TBI (Farmer et al., 2002; Fay et al., 2010). Pre-injury behavioral and mental health histories are also important to consider. Children with a pre-injury history of ADHD, learning disability, or mood disorder are at higher risk for long-term morbidities following TBI (Biederman et al., 2015; Haarbauer-Krupa et al., 2017; Kania, Shaikh, White, & Ackerman, 2016; Yeates et al., 2005).
Socioeconomic and Family Characteristics
Socioeconomic status and a family’s overall functioning also play a role in the emergence and persistence of long-term morbidities after TBI (Taylor et al., 2002, 2010; Taylor, Swartwout, et al., 2008; Yeates et al., 2012; Yeates & Taylor, 1997). Some of this influence is likely due to increased support during the acute recovery and rehabilitation process, which can increase the effectiveness of these interventions (Sander, Maestas, Sherer, Malec, & Nakase-Richardson, 2012). These family-level factors and social-environmental influences continue to play a role in influencing outcomes over the long run as well (Gerring & Wade, 2012). Socioeconomically, children who live in households with fewer resources or economic disadvantage tend to have worse cognitive and academic outcomes, particularly after severe injury (Taylor et al., 2002; Yeates, Taylor, Walz, Stancin, & Wade, 2010). Low economic resources can also impact social and behavioral outcomes. Children may have less access to or ability to consistently seek out specialized care. They may be less able to manage appointments, have limited access to transportation, or have struggle with the addition of a significant health event to an already chaotic or stressed home environment (Johnstone, Nossaman, Schopp, Holmquist, & Rupright, 2002; Lishner, Levine, & Patrick, 1996; Marcin et al., 2004; Mayer, Slifkin, & Skinner, 2005). Family socioeconomic disadvantage, when combined with severe TBI, is associated with the poorest outcomes for children and adolescents.
Also within the home, the level of parental responsiveness and warmth and the types of discipline used predict a child’s recovery, especially behavioral outcomes (Wade, Cassedy, Walz, Taylor, Stancin, & Yeates, 2011; Wade et al., 2002). As mentioned above, a TBI is an unexpected and significant health event that can significantly alter typical family functioning, roles, and responsibilities. A parent’s absence from work may introduce additional financial strain, although it is sometimes necessary to provide the level of supervision required after a significant injury. Parents may change their parenting or disciplinary approach after TBI (Root et al., 2016), and this may or may not extend to other siblings, possibly introducing an additional basis for family conflict. Overall, pediatric TBI introduces numerous new sources of stress to a family system that can disrupt marital relationships (Wade, Taylor, Drotar, Stancin, & Yeates, 1996), intensify parental or sibling distress (Jaffe et al., 1992; Rivara et al., 1996; Wade, Taylor, Drotar, Stancin, & Yeates, 1998), and contribute to maladaptive or insufficient coping responses (Wade et al., 2001, 2002; Yeates et al., 2002). These factors are strongly related to a child’s recovery after TBI, especially emotional and behavioral outcomes (Rivara et al., 1996; Taylor et al., 1999, 2002; Wade, Carey, & Wolfe, 2006; Yeates et al., 2010).
Injury Characteristics
Injury severity is one of the most commonly cited predictors of long-term outcomes after TBI. Overall, the more severe an injury, the worse the neuropsychological and behavioral outcomes (Anderson et al., 2006; Fletcher, Ewing-Cobbs, Francis, & Levin, 1995; Schwartz et al., 2003; Taylor, Yeates, et al., 2008; Taylor et al., 2001). Individual studies vary in how injury severity is assessed. As mentioned previously, the GCS score is one common measure that reflects a child or adolescent’s level of consciousness and responsiveness after TBI. It has been linked with both cognitive and functional outcome, as well as the extent of long-term cerebral atrophy (Ghosh et al., 2009). Duration of loss of consciousness and duration of posttraumatic amnesia are other metrics, and while the GCS provides a snapshot of a child’s consciousness at a single time point, duration of loss of consciousness, and posttraumatic amnesia reflect rate of recovery over time. These measures generally perform better than static measures (e.g., GCS) in predicting distal outcomes.
Children who sustain non-accidental or abusive head trauma, especially at a younger age, typically demonstrate worse outcomes because of the severity of such injuries. Non-accidental head trauma is more common in infants and very young children. They suffer more extensive secondary injuries, particularly hypoxic injury due to brain swelling, which causes diminished blood circulation in the brain (Ewing-Cobbs, Prasad, Kramer, & Landry, 1999; Hymel, Makoroff, Laskey, Conaway, & Blackman, 2007; Ichord et al., 2007). Unfortunately, children who sustain non-accidental head trauma are also more likely to come from families with higher stress and fewer resources. This confluence of factors contributes to especially high risk for poor outcomes and death in this young population.
Neuroanatomical Abnormalities
As neuroimaging methods have improved, so too has research on the neuroanatomical abnormalities that predict long-term cognitive and behavioral deficits after pediatric TBI. After injury, CT is typically the modality of choice for initial assessment, as it is readily available, fast, and effective in detecting abnormalities that require emergent intervention (e.g., mass lesions). That said, CT is not especially sensitive to subtler neuroanatomical abnormalities that are common after TBI; magnetic resonance imaging (MRI) has eclipsed CT as the method of choice for detecting smaller lesions, microstructural changes, and cortical atrophy and is often conducted when neurological findings are present but are unexplained by CT (Duckworth & Stevens, 2010).
Individual studies have linked diminished functional connectivity in the corticostriatal motor network and poor response inhibition (Stephens et al., 2017), as well as relatively greater activation in the right frontal and parietal regions during the completion of an inhibitory control paradigm during functional MRI (Tlustos et al., 2011). Others have focused on the assessment of volumetric changes (e.g., lesion volume), using a variety of neuroimaging techniques . For instance, in one longitudinal study of changes in cortical thickness following moderate to severe TBI, Wilde and colleagues found decreased cortical thickness bilaterally within several frontal regions and the anterior cingulate when compared to children who had sustained an orthopedic injury, at 3 months post-injury (Wilde, Merkley, Bigler, Max, Schmidt, Ayoub, et al., 2012). Fifteen months later, persistent decreased cortical thickness was observed in the bilateral frontal, fusiform, and lingual regions, and progressive cortical thinning was apparent in other bilateral frontal, fusiform, and left parietal regions. Longitudinal changes in cortical thickness within the left frontal, right medial frontal, and anterior cingulate regions were significantly correlated with the aspects of day-to-day executive function (Wilde, Merkley, et al., 2012). Similarly, children with increased intracranial pressure after TBI displayed reduced white and gray matter volumes in the frontal, callosal, and subcortical regions that corresponded with poorer performance on measures of working memory, decision-making, and impulsivity (Slawik et al., 2009).
Much existing research in pediatric TBI focuses on the assessment of white matter pathways, with the goal of quantifying the degree of diffuse traumatic or axonal injury and its association with cognitive outcomes. Diffuse damage to white matter pathways is generally considered to be a major contributor to the cognitive impairments faced by children after TBI (Arfanakis et al., 2002). As noted above, DTI is the most common sequence used to assess white matter integrity. A recent meta-analysis synthesized the findings of 14 individual studies that used DTI to examine the link between white matter integrity and cognitive sequelae of pediatric TBI (Roberts, Mathias, & Rose, 2016). Shortly after injury, increases in FA and decreases in apparent diffusion coefficient (ADC; reflects the rate of diffusion without respect to direction) were associated with poorer performance on the measures of executive function and memory; associations with other cognitive domains (e.g., attention, information processing, construction, motor function) were consistently in the expected direction with moderate to large effect sizes, but statistical significance was sometimes precluded by small samples. At longer intervals post-injury, as expected, effects were in the opposite direction; in other words, decreases in FA were related to poorer performance on measures of academic achievement, attention, information processing, construction, overall cognition, memory, and motor function, with effects in the moderate to large range (Roberts et al., 2016). The change in direction of association likely reflects the early effects of brain swelling and edema, in contrast to the later effects of white matter damage.
Neuroanatomical abnormalities have also been linked to social impairments, particularly theory of mind and other aspects of social cognition. Lesion burden in the mirror neuron empathy network (i.e., inferior frontal, inferior parietal, and premotor regions) and diminished volume in the posterior cingulate/retrosplenial cortex and hippocampal formation were associated with poorer performance on a conative theory of mind task that required appreciation of complex social overtures like irony and empathy (Dennis et al., 2013). Susceptibility weighted imaging, which is particularly sensitive to the microhemorrhagic lesions that accompany diffuse axonal injury, has also been associated with poorer performance on a cognitive theory of mind task (Ryan et al., 2015a, 2015b). In contrast, others have found inconsistent relationships among lesion burden or focal pathology and social deficits (Bigler et al., 2013), illustrating the heterogeneity and complexity of TBI in children. Little research has examined the link between neuroanatomical abnormalities and emotional and behavioral outcomes, although isolated studies have linked white matter integrity within the uncinate fasciculus to emotional and behavioral regulation (Johnson et al., 2011) and white matter integrity within the cingulum, genu of the corpus callosum, inferior longitudinal fasciculus, and inferior fronto-occipital fasciculus to emotional processing (Schmidt et al., 2013).
Cognitive Rehabilitation Techniques and Findings
As appreciation of the long-term morbidities of pediatric TBI has increased, so too have efforts to develop and implement interventions to address them. The amount of empirical evidence supporting interventions for neurocognitive deficits after pediatric TBI continues to lag substantially behind that for adult TBI, but efforts have increased over the past two decades. Existing interventions largely fall under the umbrella of “cognitive rehabilitation,” primarily targeting deficits in neurocognitive functions such as memory, attention, and executive functions. Many of these studies assess the impact of interventions using behavioral or cognitive outcome measures, and very few examine changes in aspects of structural or functional brain integrity post-intervention.
Cognitive and Behavioral Outcomes
Typical approaches to cognitive rehabilitation involve intensive, hands-on, or computerized training in specific skill areas. For example, Galbiati et al. (2009) assessed the benefits of an attention-training program for children who were being discharged from inpatient rehabilitation 6–10 months after sustaining a severe TBI. Children participated in intensive training with a therapist four times per week for 45 min per session, over 6 months. Individual sessions included 15 min of tabletop tasks that targeted selective, focused, sustained, and divided attention. For example, during one task, a participant might be presented with a vignette or picture depicting a situation where attention would be challenging; the participant then responded to open- or closed-ended questions to generate options or suggestions to manage attention in that context. The remaining 30 min was spent on computer-based activities (i.e., RehaCom (Schuhfried, 1996) or Attenzione e Concentrazione (Di Nuovo, 2001), based on participant age) that targeted vigilance, attention/concentration, and response behavior. Post-intervention, the children who received the intervention improved significantly on a computerized attention task (CPT-II), as well as on ratings of adaptive behavior provided by parents, and showed significantly more improvement on these measures than did a control group (Galbiati et al., 2009). While this certainly suggests some promise for a combination of massed practice and teaching of metacognitive skills, the intervention was very time-intensive, and the extent to which the testing and parent-reported improvements would transfer to observable behavior is unknown.
A similar pattern of results was observed in a pilot case series involving another computerized cognitive training program, Brain Games (Apple, SpriteKit). After 8 weeks of training, five participants with moderate to severe TBI improved on a flanker and computerized performance test, as well as on parent-reported BRIEF measures; however, no evidence for far transfer of treatment effects was provided (Verhelst, Vander Linden, Vingerhoets, & Caeyenberghs, 2017). Another pilot study assessed the effectiveness of the Attention Improvement Management (AIM) program, which purports to improve attention and executive function skills (Treble-Barna, Sohlberg, Harn, & Wade, 2016). Thirteen children with complicated mild to severe TBI and a comparison group of 11 healthy children participated in the intervention, which included 10 weeks of face-to-face sessions with a clinician. During sessions, attention-training tasks were selected through a computerized portal, and clinicians coached participants on how different metacognitive strategies could be applied in real-life situations. Between sessions, the participants were instructed to practice the computerized tasks at home 2–4 times per week. Children with TBI improved more than healthy children on a measure of sustained attention after the intervention, as well as on parent-reported executive function. That said, several measures of attention and executive function remained unchanged post-intervention, and the program had considerable attrition and low adherence to the ideal schedule of home-based practice.
Other intervention approaches first trialed in other pediatric populations have been more recently used with children following TBI. Cogmed™ (Pearson Education, Inc.), for instance, is an adaptive working memory training program that has been used in a variety of populations of children with neurodevelopmental (e.g., learning disabilities, ADHD) and acquired neurocognitive deficits. Recently, children with moderate to severe TBI were invited to participate in a Cogmed™ intervention trial that involved 30–40 min sessions, 5 days per week, for 5 weeks (Phillips et al., 2016). As mentioned above, this program is adaptive, so individual trials increase or decrease in difficulty to match a child’s performance. Though this intervention was completed at home, children were supervised by parents and had a weekly check-in with a certified training coach. As a control arm, children in an active placebo group also completed the Cogmed™ program, but used a version that was not adaptive and remained at a low difficulty level across the entire intervention. Overall, children in the adaptive arm showed significantly greater gains than children in the control arm on working memory tasks and in some reading-based academic domains, suggesting some possible transfer to daily academic functioning.
Finally, some investigators have attempted to intervene to address behavior problems that occur after TBI. The most prominent series of studies has examined the impact of several different modalities of family-based problem-solving interventions, which have generally proven efficacious in reducing both behavior problems and deficits in executive function relative to internet-based resources (Kurowski et al., 2013; Wade et al., 2006; Wade, Walz, Carey, McMullen, Cass, Mahone, & Yeates, 2011; Wade et al., 2010; Wade, Stancin, Kirkwood, & Brown, 2014; Wade et al., 2015). These findings were most impressive for older youth and those from lower-income families (Kurowski et al., 2013; Wade et al., 2006). Continuing efforts from this research group includes assessment of the extent to which telehealth provision of these interventions is feasible, acceptable, and effective in addressing neurocognitive and behavioral deficits after TBI when compared with internet-based resources or traditional face-to-face intervention.
In a recent review of interventions targeting attention problems, Backeljauw and Kurowski (2014) outlined a number of issues that limit the impact of this growing body of research. In particular, they noted a lack of consistent outcome measures across studies, as well as a lack of measures that assess changes in day-to-day functioning, which are needed to assess the broader effectiveness of interventions. Furthermore, existing research is so limited that it precludes efforts to prescribe individualized or tailored empirically based interventions. Many individual studies have small samples, occur at a single center, and are unable to delineate findings based on individual (e.g., gender) or injury-related factors (e.g., severity, time since injury). Others pool samples of children with “acquired brain injury” (ABI), obscuring specific effects for children with TBI relative to children with other forms of ABI.
These challenges affect both qualitative reviews and quantitative meta-analysis (Robinson, Kaizar, Catroppa, Godfrey, & Yeates, 2014). For instance, in a recent meta-analysis of randomized controlled trials of cognitive interventions for central nervous system and neurodevelopmental disorders, large, significant, and positive treatment effects were found for attention, working memory, and memory tasks, while small but significant effects were found for academic achievement and behavioral rating scales. However, effects across domains displayed notable heterogeneity, including across diagnostic groups, although the number of available studies and the small sample sizes in those studies did not allow for more nuanced analysis of subgroups (Robinson, Kaizar, et al., 2014). In general, less evidence was found for the generalization of intervention to everyday behavior than for immediate transfer to cognitive tasks. Heterogeneity in intervention methods, outcome measures, and participant groups diminishes the likelihood that existing research can meaningfully inform targeted or precision-based implementation, despite a strong desire to be able to do so within the broader field. Overall, while this is an important area of research that has the potential to positively impact children and their families, substantial work remains to be done.
Neuroimaging Outcomes
Surprisingly few intervention studies have assessed treatment-related changes in structural or functional brain integrity after pediatric TBI, and most of the existing studies do not focus on cognitive rehabilitation programs. For instance, structural connectivity analysis has been used to assess changes in response to an aerobic training program for adolescents with persistent post-concussive symptoms, with some notable increases in global brain efficiency and decreases in normalized characteristic path length following the intervention (Yuan, Wade, Quatman-Yates, Hugentobler, Gubanich, & Kurowski, 2017). DTI has also been used to assess changes in white matter integrity in the cerebellum in response to postural control/balance training following pediatric TBI. Participants who completed 8 weeks of a balance training using PC-based portable balancers showed increased mean diffusivity in the inferior cerebellar peduncle; increases in FA in the inferior cerebellar peduncle were also associated with improvement on balance testing (Drijkoningen et al., 2015).
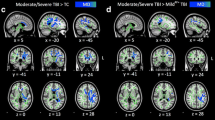
We could identify only one study that has assessed structural and functional changes in brain integrity following cognitive rehabilitation. Yuan and colleagues (2017) evaluated the impact of participation in the AIM program described above (Treble-Barna et al., 2016) using graph theory analysis to analyze global and regional brain networks. They found significantly higher small-worldness in the TBI group relative to the healthy control group at baseline, which indicates that the networks in the TBI group have more closely connected, short-distance connections, rather than long-distance connections, possibly reflecting greater reliance on segregated versus integrated brain networks. Following the AIM intervention, children in the TBI group showed reduced small-worldness and normalized clustering coefficient at follow-up. Improved network characteristics, including mean local efficiency and normalized characteristic path length, were associated with improvements in verbal inhibition, parent- and child-reported executive function, and sustained attention (Yuan, Treble-Barna, Sohlberg, Harn, & Wade, 2017).
Practice Considerations
Though currently quite limited, the existing body of research should begin to inform clinical practice and research, given the public health significance of pediatric TBI. Furthermore, ongoing and future research should continue to strengthen and refine our approaches to managing the neurocognitive and behavioral sequelae of these injuries.
Considerations for Assessment
One key role for practicing clinicians, particularly neuropsychologists, is providing rigorous assessment to help develop educational accommodations and behavioral and emotional supports for children who have sustained TBI. When approaching assessment, a variety of risk factors for poor outcome should be considered, including demographic and socioeconomic disadvantage, worse pre-injury functioning, and more severe injury. These should be weighed against factors that might promote more resilience, such as greater cognitive reserve or a high degree of family support. Assessments should target the domains most vulnerable to TBI (e.g., executive functioning, memory, processing speed), as well as skills that are less vulnerable (e.g., basic reading skills) to identify areas of strength and weakness.
Using what is known about the evolution of deficits across time after injury, clinicians can help families and educators anticipate potential future challenges as a child continues their recovery, and as he or she approaches major developmental milestones. While some challenges may be cognitive, the social, emotional, and behavioral morbidities that occur after pediatric TBI also must be remembered, and clinicians must provide guidance and recommendations that address each unique aspect of a child’s overall functioning.
Considerations for Treatment
As discussed above, strong empirical support for any single type of treatment (whether cognitive or behavioral) for the outcomes of childhood TBI is lacking. Across different specific intervention programs, some support can be found for at least some potential benefit of cognitive interventions that target skills like working memory and attention, at least in terms of near transfer, and additional research is underway. Support for transfer to more distal skills and assessment of whether benefits are sustained over time is even more limited. Given the demanding schedule of cognitive interventions, the benefits of these treatments should be weighed against other competing demands, particularly during acute recovery.
Even less research has focused on the treatment programs for the emotional and behavioral sequelae of pediatric TBI. However, this is an area of significant interest and growth, including outside of the realm of “cognitive rehabilitation.” For instance, a recent study of children and adolescents with mild TBI and prolonged post-concussive symptoms found significant symptom improvement and increased quality of life ratings in response to participation in a cognitive behavioral intervention (McNally et al., 2018). The intervention focused on psychoeducation about mild TBI and typical outcomes, activity and sleep scheduling, relaxation and biofeedback training, and cognitive restructuring. Though quite preliminary, the development of these types of program, and “manualization” and dissemination, may prove highly beneficial for youth with TBI.
Considerations for Research
Finally, considerable room for growth exists in how researchers develop, implement, and assess cognitive rehabilitation programs. As mentioned above, many existing studies are limited by small and heterogeneous samples that limit the ability to examine mediators or moderators of treatment effects. In a broader healthcare system that is moving toward precision medicine, this gap is highly problematic. Increasing multicenter collaborations may help address this issue. When conducting pilot or single-center studies, moving toward consistency in outcome measures would at least allow for side-by-side or meta-analytic review of findings, perhaps guided by the NIH Common Data Elements Project (McCauley et al., 2012). Though challenging, randomized and controlled trials are especially useful in determining the efficacy, as well as the effectiveness, of interventions.
As interventions continue to be developed and tested, researchers should also incorporate neuroimaging methods when assessing their efficacy. While we believe this is true across different types of brain injury, use of neuroimaging markers in pediatrics can elucidate how TBI operates within the context of neurodevelopment, and how these dynamic processes can validate treatment, inform risk stratification, and foster and monitor recovery. Mounting evidence supports these methods, particularly DTI and measures of network connectivity, as sensitive to the structural and functional consequences of TBI on the brain. If these methods prove useful in predicting or assessing response to treatment, our understanding of the mechanisms supporting functional improvements in this population would be greatly increased. Furthermore, though neuroimaging is costly, and some methods are not readily available at some institutions, the discovery that a particular sequence or set of sequences is predictive of or sensitive to recovery could inform the clinical management of acute injury, monitoring over time, and the implementation of individualized treatment for children and adolescents with TBI.
References
Anderson, S. W., Bechara, A., Damasio, H., Tranel, D., & Damasio, A. R. (1999). Impairment of social and moral behavior related to early damage in human prefrontal cortex. Nature Neuroscience, 2(11), 1032–1037.
Anderson, V., Brown, S., Newitt, H., & Hoile, H. (2009). Educational, vocational, psychosocial, and quality-of-life outcomes for adult survivors of childhood traumatic brain injury. The Journal of Head Trauma Rehabilitation, 24(5), 303–312.
Anderson, V., Catroppa, C., Morse, S., Haritou, F., & Rosenfeld, J. (2005). Attentional and processing skills following traumatic brain injury in early childhood. Brain Injury, 19(9), 699–710.
Anderson, V. A., Catroppa, C., Dudgeon, P., Morse, S. A., Haritou, F., & Rosenfeld, J. V. (2006). Understanding predictors of functional recovery and outcome 30 months following early childhood head injury. Neuropsychology, 20(1), 42.
Anderson, V. A., Catroppa, C., Haritou, F., Morse, S., & Rosenfeld, J. V. (2005). Identifying factors contributing to child and family outcome 30 months after traumatic brain injury in children. Journal of Neurology, Neurosurgery & Psychiatry, 76(3), 401–408.
Arfanakis, K., Haughton, V. M., Carew, J. D., Rogers, B. P., Dempsey, R. J., & Meyerand, M. E. (2002). Diffusion tensor MR imaging in diffuse axonal injury. American Journal of Neuroradiology, 23(5), 794–802.
Babikian, T., & Asarnow, R. (2009). Neurocognitive outcomes and recovery after pediatric TBI: Meta-analytic review of the literature. Neuropsychology, 23(3), 283.
Babikian, T., Merkley, T., Savage, R. C., Giza, C. C., & Levin, H. (2015). Chronic aspects of pediatric traumatic brain injury: Review of the literature. Journal of Neurotrauma, 32(23), 1849–1860.
Babikian, T., Satz, P., Zaucha, K., Light, R., Lewis, R. S., & Asarnow, R. F. (2011). The UCLA longitudinal study of neurocognitive outcomes following mild pediatric traumatic brain injury. Journal of the International Neuropsychological Society, 17(5), 886–895.
Backeljauw, B., & Kurowski, B. G. (2014). Interventions for attention problems after pediatric traumatic brain injury: What is the evidence? PM&R, 6(9), 814–824.
Barnes, M. A., Dennis, M., & Wilkinson, M. (1999). Reading after closed head injury in childhood: Effects on accuracy, fluency, and comprehension. Developmental Neuropsychology, 15(1), 1–24.
Beauchamp, M., Catroppa, C., Godfrey, C., Morse, S., Rosenfeld, J. V., & Anderson, V. (2011). Selective changes in executive functioning ten years after severe childhood traumatic brain injury. Developmental Neuropsychology, 36(5), 578–595.
Bedell, G. M., & Dumas, H. M. (2004). Social participation of children and youth with acquired brain injuries discharged from inpatient rehabilitation: A follow-up study. Brain Injury, 18(1), 65–82.
Biederman, J., Feinberg, L., Chan, J., Adeyemo, B. O., Woodworth, K. Y., Panis, W., … Kenworthy, T. (2015). Mild traumatic brain injury and attention-deficit hyperactivity disorder in young student athletes. The Journal of Nervous and Mental Disease, 203(11), 813.
Bigler, E. D. (2007). Anterior and middle cranial fossa in traumatic brain injury: Relevant neuroanatomy and neuropathology in the study of neuropsychological outcome. Neuropsychology, 21(5), 515–531.
Bigler, E. D., Yeates, K. O., Dennis, M., Gerhardt, C. A., Rubin, K. H., Stancin, T., … Vannatta, K. (2013). Neuroimaging and social behavior in children after traumatic brain injury: Findings from the Social Outcomes of Brain Injury in Kids (SOBIK) study. NeuroRehabilitation, 32(4), 707–720.
Bijur, P. E., Haslum, M., & Golding, J. (1990). Cognitive and behavioral sequelae of mild head injury in children. Pediatrics, 86(3), 337–344.
Bloom, D. R., Levin, H. S., Ewing-Cobbs, L., Saunders, A. E., Song, J., Fletcher, J. M., & Kowatch, R. A. (2001). Lifetime and novel psychiatric disorders after pediatric traumatic brain injury. Journal of the American Academy of Child & Adolescent Psychiatry, 40(5), 572–579.
Brosseau-Lachaine, O., Gagnon, I., Forget, R., & Faubert, J. (2008). Mild traumatic brain injury induces prolonged visual processing deficits in children. Brain Injury, 22(9), 657–668.
Cameto, R., Levine, P., & Wagner, M. (2004). Transition planning for students with disabilities: A special topic report of findings from the National Longitudinal Transition Study-2 (NLTS2). Report from the National Center for Special Education Research, Washington DC.
Carroll, L., Cassidy, J. D., Peloso, P., Borg, J., Von Holst, H., Holm, L., … Pépin, M. (2004). Prognosis for mild traumatic brain injury: Results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. Journal of Rehabilitation Medicine, 43, 84–105.
Cassidy, J. D., Carroll, L., Peloso, P., Borg, J., Von Holst, H., Holm, L., … Coronado, V. (2004). Incidence, risk factors and prevention of mild traumatic brain injury: Results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. Journal of Rehabilitation Medicine, 36(0), 28–60.
Catroppa, C., Anderson, V. A., Morse, S. A., Haritou, F., & Rosenfeld, J. V. (2008). Outcome and predictors of functional recovery 5 years following pediatric traumatic brain injury (TBI). Journal of Pediatric Psychology, 33(7), 707–718.
Cattelani, R., Lombardi, F., Brianti, R., & Mazzucchi, A. (1998). Traumatic brain injury in childhood: Intellectual, behavioural and social outcome into adulthood. Brain Injury, 12(4), 283–296.
Centers for Disease Control and Prevention (2011). Surveillance Report of Traumatic Brain Injury-related Deaths - United States, 1997-2007. Centers for Disease Control and Prevention, U.S. Department of Health and Human Services.
Covassin, T., Moran, R., & Wilhelm, K. (2013). Concussion symptoms and neurocognitive performance of high school and college athletes who incur multiple concussions. The American Journal of Sports Medicine, 41(12), 2885–2889.
Dennis, E. L., Babikian, T., Giza, C. C., Thompson, P. M., & Asarnow, R. F. (2017). Diffusion MRI in pediatric brain injury. Child’s Nervous System, 33(10), 1683–1692.
Dennis, M. (2000). Childhood medical disorders and cognitive impairment: Biological risk, time, development, and reserve. In K. O. Yeates, D. M. Ris, & H. G. Taylor (Eds.), Pediatric neuropsychology: Research, theory, and practice (pp. 3–22). New York, NY: Guilford Press.
Dennis, M., Simic, N., Bigler, E. D., Abildskov, T., Agostino, A., Taylor, H. G., … Yeates, K. O. (2013). Cognitive, affective, and conative theory of mind (ToM) in children with traumatic brain injury. Developmental Cognitive Neuroscience, 5, 25–39.
Dennis, M., Wilkinson, M., Koski, L., & Humphreys, R. P. (1995). Attention deficits in the long term after childhood head injury. In S. H. Broman & M. E. Michel (Eds.), Traumatic head injury in children (pp. 165–187). New York, NY: Oxford University Press.
Dennis, M., Yeates, K. O., Taylor, H. G., & Fletcher, J. M. (2007). Brain reserve capacity, cognitive reserve capacity, and age-based functional plasticity after congenital and acquired brain injury in children. In Cognitive reserve (pp. 53–83). New York, NY: Psychology Press.
Di Nuovo, S. (2001). Attenzione e concentrazione. Trento, Italy: Erickson.
Drijkoningen, D., Caeyenberghs, K., Leunissen, I., Vander Linden, C., Leemans, A., Sunaert, S., … Swinnen, S. P. (2015). Training-induced improvements in postural control are accompanied by alterations in cerebellar white matter in brain injured patients. NeuroImage: Clinical, 7, 240–251.
Duckworth, J. L., & Stevens, R. D. (2010). Imaging brain trauma. Current Opinion in Critical Care, 16(2), 92–97.
Eisenberg, M. A., Andrea, J., Meehan, W., & Mannix, R. (2013). Time interval between concussions and symptom duration. Pediatrics, 132(1), 8–17.
Ewing-Cobbs, L., Fletcher, J. M., Levin, H. S., Francis, D. J., Davidson, K., & Miner, M. E. (1997). Longitudinal neuropsychological outcome in infants and preschoolers with traumatic brain injury. Journal of the International Neuropsychological Society, 3(6), 581–591.
Ewing-Cobbs, L., Prasad, M., Kramer, L., & Landry, S. (1999). Inflicted traumatic brain injury: Relationship of developmental outcome to severity of injury. Pediatric Neurosurgery, 31(5), 251–258.
Falcone, R. A., Jr., Martin, C., Brown, R. L., & Garcia, V. F. (2008). Despite overall low pediatric head injury mortality, disparities exist between races. Journal of Pediatric Surgery, 43(10), 1858–1864.
Farkas, O., & Povlishock, J. T. (2007). Cellular and subcellular change evoked by diffuse traumatic brain injury: A complex web of change extending far beyond focal damage. Progress in Brain Research, 161, 43–59.
Farmer, J. E., Kanne, S. M., Haut, J. S., Williams, J., Johnstone, B., & Kirk, K. (2002). Memory functioning following traumatic brain injury in children with premorbid learning problems. Developmental Neuropsychology, 22(2), 455–469.
Faul, M., Xu, L., Wald, M. M., & Coronado, V. G. (2010). Traumatic brain injury in the United States: Emergency department visits, hospitalizations, and deaths, 2002–2006. Atlanta, GA: Centers for Disease Control and Prevention, National Center for Injury Prevention and Control.
Fay, T. B., Yeates, K. O., Taylor, H. G., Bangert, B., Dietrich, A., Nuss, K. E., … Wright, M. (2010). Cognitive reserve as a moderator of postconcussive symptoms in children with complicated and uncomplicated mild traumatic brain injury. Journal of the International Neuropsychological Society, 16(1), 94–105.
Fay, T. B., Yeates, K. O., Wade, S. L., Drotar, D., Stancin, T., & Taylor, H. G. (2009). Predicting longitudinal patterns of functional deficits in children with traumatic brain injury. Neuropsychology, 23(3), 271–282.
Fletcher, J. M., Ewing-Cobbs, L., Francis, D. J., & Levin, H. S. (1995). Variability in outcomes after traumatic brain injury in children: A developmental perspective. In S. H. Broman & M. E. Michel (Eds.), Traumatic head injury in children (p. 3). New York, NY: Oxford University Press.
Galbiati, S., Recla, M., Pastore, V., Liscio, M., Bardoni, A., Castelli, E., & Strazzer, S. (2009). Attention remediation following traumatic brain injury in childhood and adolescence. Neuropsychology, 23(1), 40.
Gerring, J. P., & Wade, S. (2012). The essential role of psychosocial risk and protective factors in pediatric traumatic brain injury research. Journal of Neurotrauma, 29(4), 621–628.
Ghosh, A., Wilde, E. A., Hunter, J. V., Bigler, E. D., Chu, Z., Li, X., … Levin, H. S. (2009). The relation between Glasgow coma scale score and later cerebral atrophy in paediatric traumatic brain injury. Brain Injury, 23(3), 228–233.
Giza, C. C., & Hovda, D. A. (2001). The neurometabolic cascade of concussion. Journal of Athletic Training, 36(3), 228.
Graves, J. M., Rivara, F. P., & Vavilala, M. S. (2015). Health care costs 1 year after pediatric traumatic brain injury. American Journal of Public Health, 105(10), e35–e41.
Haarbauer-Krupa, J., Ciccia, A., Dodd, J., Ettel, D., Kurowski, B., Lumba-Brown, A., & Suskauer, S. (2017). Service delivery in the healthcare and educational systems for children following traumatic brain injury: Gaps in care. The Journal of Head Trauma Rehabilitation, 32(6), 367.
Howard, I., Joseph, J. G., & Natale, J. E. (2005). Pediatric traumatic brain injury: Do racial/ethnic disparities exist in brain injury severity, mortality, or medical disposition? Ethnicity and Disease, 15(4), S5.
Hung, R., Carroll, L. J., Cancelliere, C., Côté, P., Rumney, P., Keightley, M., … Cassidy, J. D. (2014). Systematic review of the clinical course, natural history, and prognosis for pediatric mild traumatic brain injury: Results of the International Collaboration on Mild Traumatic Brain Injury Prognosis. Archives of Physical Medicine and Rehabilitation, 95(3), S174–S191.
Hunter, J. V., Wilde, E. A., Tong, K. A., & Holshouser, B. A. (2012). Emerging imaging tools for use with traumatic brain injury research. Journal of Neurotrauma, 29(4), 654–671.
Hymel, K. P., Makoroff, K. L., Laskey, A. L., Conaway, M. R., & Blackman, J. A. (2007). Mechanisms, clinical presentations, injuries, and outcomes from inflicted versus noninflicted head trauma during infancy: Results of a prospective, multicentered, comparative study. Pediatrics, 119(5), 922–929.
Ichord, R. N., Naim, M., Pollock, A. N., Nance, M. L., Margulies, S. S., & Christian, C. W. (2007). Hypoxic-ischemic injury complicates inflicted and accidental traumatic brain injury in young children: The role of diffusion-weighted imaging. Journal of Neurotrauma, 24(1), 106–118.
Jaffe, K. M., Fay, G. C., Polissar, N. L., Martin, K. M., Shurtleff, H., J’May, B. R., & Winn, H. R. (1992). Severity of pediatric traumatic brain injury and early neurobehavioral outcome: A cohort study. Archives of Physical Medicine and Rehabilitation, 73(6), 540–547.
Johnson, C. P., Juranek, J., Kramer, L. A., Prasad, M. R., Swank, P. R., & Ewing-Cobbs, L. (2011). Predicting behavioral deficits in pediatric traumatic brain injury through uncinate fasciculus integrity. Journal of the International Neuropsychological Society, 17(4), 663–673.
Johnstone, B., Nossaman, L. D., Schopp, L. H., Holmquist, L., & Rupright, S. J. (2002). Distribution of services and supports for people with traumatic brain injury in ruraland urban Missouri. The Journal of Rural Health, 18(1), 109–117.
Kania, K., Shaikh, K. A., White, I. K., & Ackerman, L. L. (2016). Follow-up issues in children with mild traumatic brain injuries. Journal of Neurosurgery: Pediatrics, 18(2), 224–230.
Kelley, B. J., Lifshitz, J., & Povlishock, J. T. (2007). Neuroinflammatory responses after experimental diffuse traumatic brain injury. Journal of Neuropathology & Experimental Neurology, 66(11), 989–1001.
Kesler, S. R., Adams, H. F., Blasey, C. M., & Bigler, E. D. (2003). Premorbid intellectual functioning, education, and brain size in traumatic brain injury: An investigation of the cognitive reserve hypothesis. Applied Neuropsychology, 10(3), 153–162.
Koskiniemi, M., Kyykkä, T., Nybo, T., & Jarho, L. (1995). Long-term outcome after severe brain injury in preschoolers is worse than expected. Archives of Pediatrics & Adolescent Medicine, 149(3), 249–254.
Kurowski, B. G., Wade, S. L., Kirkwood, M. W., Brown, T. M., Stancin, T., Cassedy, A., & Taylor, H. G. (2013). Association of parent ratings of executive function with global-and setting-specific behavioral impairment after adolescent traumatic brain injury. Archives of Physical Medicine and Rehabilitation, 94(3), 543–550.
Linton, K. F., & Kim, B. J. (2014). Traumatic brain injury as a result of violence in Native American and Black communities spanning from childhood to older adulthood. Brain Injury, 28(8), 1076–1081.
Lishner, D. M., Levine, P., & Patrick, D. (1996). Access to primary health care among persons with disabilities in rural areas: A summary of the literature. The Journal of Rural Health, 12(1), 45–53.
Mangeot, S., Armstrong, K., Colvin, A. N., Yeates, K. O., & Taylor, H. G. (2002). Long-term executive function deficits in children with traumatic brain injuries: Assessment using the Behavior Rating Inventory of Executive Function (BRIEF). Child Neuropsychology, 8(4), 271–284.
Marcin, J. P., Schepps, D. E., Page, K. A., Struve, S. N., Nagrampa, E., & Dimand, R. J. (2004). The use of telemedicine to provide pediatric critical care consultations to pediatric trauma patients admitted to a remote trauma intensive care unit: A preliminary report. Pediatric Critical Care Medicine, 5(3), 251–256.
Marr, A. L., & Coronado, V. G. (2004). Central nervous system injury surveillance data submission standards—2002. Atlanta, GA: US Department of Health and Human Services, CDC.
Mayer, M. L., Slifkin, R. T., & Skinner, A. C. (2005). The effects of rural residence and other social vulnerabilities on subjective measures of unmet need. Medical Care Research and Review, 62(5), 617–628.
McCauley, S. R., Wilde, E. A., Anderson, V. A., Bedell, G., Beers, S. R., Campbell, T. F., … Levin, H. S. (2012). Recommendations for the use of common outcome measures in pediatric traumatic brain injury research. Journal of Neurotrauma, 29(4), 678–705.
McNally, K. A., Patrick, K. E., LaFleur, J. E., Dykstra, J. B., Monahan, K., & Hoskinson, K. R. (2018). Brief cognitive behavioral intervention for children and adolescents with persistent post-concussive symptoms: A pilot study. Child Neuropsychology, 24(3), 396–412.
Nybo, T., Sainio, M., & Müller, K. (2004). Stability of vocational outcome in adulthood after moderate to severe preschool brain injury. Journal of the International Neuropsychological Society, 10(5), 719–723.
Phillips, N. L., Mandalis, A., Benson, S., Parry, L., Epps, A., Morrow, A., & Lah, S. (2016). Computerized working memory training for children with moderate to severe traumatic brain injury: A double-blind, randomized, placebo-controlled trial. Journal of Neurotrauma, 33(23), 2097–2104.
Povlishock, J. T., & Katz, D. I. (2005). Update of neuropathology and neurological recovery after traumatic brain injury. The Journal of Head Trauma Rehabilitation, 20(1), 76–94.
Rivara, F. P., Koepsell, T. D., Wang, J., Temkin, N., Dorsch, A., Vavilala, M. S., … Jaffe, K. M. (2012). Incidence of disability among children 12 months after traumatic brain injury. American Journal of Public Health, 102(11), 2074–2079.
Rivara, J. B., Jaffe, K. M., Polissar, N. L., Fay, G. C., Liao, S., & Martin, K. M. (1996). Predictors of family functioning and change 3 years after traumatic brain injury in children. Archives of Physical Medicine and Rehabilitation, 77(8), 754–764.
Roberts, R. M., Mathias, J. L., & Rose, S. E. (2016). Relationship between diffusion tensor imaging (DTI) findings and cognition following pediatric TBI: A meta-analytic review. Developmental Neuropsychology, 41(3), 176–200.
Robinson, K. E., Fountain-Zaragoza, S., Dennis, M., Taylor, H. G., Bigler, E. D., Rubin, K., … Yeates, K. O. (2014). Executive functions and theory of mind as predictors of social adjustment in childhood traumatic brain injury. Journal of Neurotrauma, 31(22), 1835–1842.
Robinson, K. E., Kaizar, E., Catroppa, C., Godfrey, C., & Yeates, K. O. (2014). Systematic review and meta-analysis of cognitive interventions for children with central nervous system disorders and neurodevelopmental disorders. Journal of Pediatric Psychology, 39(8), 846–865.
Root, A. E., Wimsatt, M., Rubin, K. H., Bigler, E. D., Dennis, M., Gerhardt, C. A., … Yeates, K. O. (2016). Children with traumatic brain injury: Associations between parenting and social adjustment. Journal of Applied Developmental Psychology, 42, 1–7.
Rosema, S., Crowe, L., & Anderson, V. (2012). Social function in children and adolescents after traumatic brain injury: A systematic review 1989–2011. Journal of Neurotrauma, 29(7), 1277–1291.
Ryan, N. P., Catroppa, C., Cooper, J. M., Beare, R., Ditchfield, M., Coleman, L., … Anderson, V. A. (2015a). The emergence of age-dependent social cognitive deficits after generalized insult to the developing brain: A longitudinal prospective analysis using susceptibility-weighted imaging. Human Brain Mapping, 36(5), 1677–1691.
Ryan, N. P., Catroppa, C., Cooper, J. M., Beare, R., Ditchfield, M., Coleman, L., … Yeates, K. O. (2015b). Relationships between acute imaging biomarkers and theory of mind impairment in post-acute pediatric traumatic brain injury: A prospective analysis using susceptibility weighted imaging (SWI). Neuropsychologia, 66, 32–38.
Sander, A. M., Maestas, K. L., Sherer, M., Malec, J. F., & Nakase-Richardson, R. (2012). Relationship of caregiver and family functioning to participation outcomes after postacute rehabilitation for traumatic brain injury: A multicenter investigation. Archives of Physical Medicine and Rehabilitation, 93(5), 842–848.
Sariaslan, A., Sharp, D. J., D’Onofrio, B. M., Larsson, H., & Fazel, S. (2016). Long-term outcomes associated with traumatic brain injury in childhood and adolescence: A nationwide Swedish cohort study of a wide range of medical and social outcomes. PLoS Medicine, 13(8), e1002103.
Schmidt, A. T., Hanten, G., Li, X., Wilde, E. A., Ibarra, A. P., Chu, Z. D., … Levin, H. S. (2013). Emotional prosody and diffusion tensor imaging in children after traumatic brain injury. Brain Injury, 27(13–14), 1528–1535.
Schuhfried. (1996). RehaCom [Computer Software]. Bologna, Italy: Schuhfried.
Schwartz, L., Taylor, H. G., Drotar, D., Yeates, K. O., Wade, S. L., & Stancin, T. (2003). Long-term behavior problems following pediatric traumatic brain injury: Prevalence, predictors, and correlates. Journal of Pediatric Psychology, 28(4), 251–263.
Shultz, E. L., Hoskinson, K. R., Keim, M. C., Dennis, M., Taylor, H. G., Bigler, E. D., … Yeates, K. O. (2016). Adaptive functioning following pediatric traumatic brain injury: Relationship to executive function and processing speed. Neuropsychology, 30(7), 830–840.
Shultz, S. R., Bao, F., Omana, V., Chiu, C., Brown, A., & Cain, D. P. (2012). Repeated mild lateral fluid percussion brain injury in the rat causes cumulative long-term behavioral impairments, neuroinflammation, and cortical loss in an animal model of repeated concussion. Journal of Neurotrauma, 29(2), 281–294.
Shultz, S. R., MacFabe, D. F., Foley, K. A., Taylor, R., & Cain, D. P. (2012). Sub-concussive brain injury in the Long-Evans rat induces acute neuroinflammation in the absence of behavioral impairments. Behavioural Brain Research, 229(1), 145–152.
Slawik, H., Salmond, C. H., Taylor-Tavares, J. V., Williams, G. B., Sahakian, B. J., & Tasker, R. C. (2009). Frontal cerebral vulnerability and executive deficits from raised intracranial pressure in child traumatic brain injury. Journal of Neurotrauma, 26(11), 1891–1903.
Stancin, T., Drotar, D., Taylor, H. G., Yeates, K. O., Wade, S. L., & Minich, N. M. (2002). Health-related quality of life of children and adolescents after traumatic brain injury. Pediatrics, 109(2), e34–e34.
Stephens, J. A., Salorio, C. E., Gomes, J. P., Nebel, M. B., Mostofsky, S. H., & Suskauer, S. J. (2017). Response inhibition deficits and altered motor network connectivity in the chronic phase of pediatric traumatic brain injury. Journal of Neurotrauma, 34(22), 3117–3123.
Stern, Y. (2003). What is cognitive reserve? Theory and research application of the reserve concept. Journal of the International Neuropsychological Society, 8(3), 448–460.
Taylor, C. A., Bell, J. M., Breiding, M. J., & Xu, L. (2017). Traumatic brain injury–related emergency department visits, hospitalizations, and deaths—United States, 2007 and 2013. MMWR Surveillance Summaries, 66(9), 1.
Taylor, H. G., & Alden, J. (1997). Age-related differences in outcomes following childhood brain insults: An introduction and overview. Journal of the International Neuropsychological Society, 3(6), 555–567.
Taylor, H. G., Dietrich, A., Nuss, K., Wright, M., Rusin, J., Bangert, B., … Yeates, K. O. (2010). Post-concussive symptoms in children with mild traumatic brain injury. Neuropsychology, 24(2), 148.
Taylor, H. G., Swartwout, M. D., Yeates, K. O., Walz, N. C., Stancin, T., & Wade, S. L. (2008). Traumatic brain injury in young children: Postacute effects on cognitive and school readiness skills. Journal of the International Neuropsychological Society, 14(5), 734–745.
Taylor, H. G., Yeates, K. O., Rusin, J., Bangert, B., Dietrich, A., Nuss, K., … Minich, N. (2008). Post-concussive symptoms following mild head injury in children. The Clinical Neuropsychologist, 22(3), 451.
Taylor, H. G., Yeates, K. O., Wade, S. L., Drotar, D., Klein, S. K., & Stancin, T. (1999). Influences on first-year recovery from traumatic brain injury in children. Neuropsychology, 13(1), 76–89.
Taylor, H. G., Yeates, K. O., Wade, S. L., Drotar, D., Stancin, T., & Burant, C. (2001). Bidirectional child–family influences on outcomes of traumatic brain injury in children. Journal of the International Neuropsychological Society, 7(6), 755–767.
Taylor, H. G., Yeates, K. O., Wade, S. L., Drotar, D., Stancin, T., & Minich, N. (2002). A prospective study of short-and long-term outcomes after traumatic brain injury in children: Behavior and achievement. Neuropsychology, 16(1), 15–27.
Teasdale, G., & Jennett, B. (1974). Assessment of coma and impaired consciousness: A practical scale. The Lancet, 304(7872), 81–84.
Tlustos, S. J., Chiu, C. Y. P., Walz, N. C., Holland, S. K., Bernard, L., & Wade, S. L. (2011). Neural correlates of interference control in adolescents with traumatic brain injury: Functional magnetic resonance imaging study of the counting stroop task. Journal of the International Neuropsychological Society, 17(1), 181–189.
Todis, B., & Glang, A. (2008). Redefining success: Results of a qualitative study of postsecondary transition outcomes for youth with traumatic brain injury. The Journal of Head Trauma Rehabilitation, 23(4), 252–263.
Todis, B., Glang, A., Bullis, M., Ettel, D., & Hood, D. (2011). Longitudinal investigation of the post—High school transition experiences of adolescents with traumatic brain injury. The Journal of Head Trauma Rehabilitation, 26(2), 138–149.
Toledo, E., Lebel, A., Becerra, L., Minster, A., Linnman, C., Maleki, N., … Borsook, D. (2012). The young brain and concussion: Imaging as a biomarker for diagnosis and prognosis. Neuroscience & Biobehavioral Reviews, 36(6), 1510–1531.
Tonks, J., Slater, A., Frampton, I., Wall, S. E., Yates, P., & Williams, W. H. (2009). The development of emotion and empathy skills after childhood brain injury. Developmental Medicine & Child Neurology, 51(1), 8–16.
Treble-Barna, A., Sohlberg, M. M., Harn, B. E., & Wade, S. L. (2016). Cognitive intervention for attention and executive function impairments in children with traumatic brain injury. Journal of Head Trauma Rehabilitation, 31(6), 407–418.
Turkstra, L. S. (2000). Should my shirt be tucked in or left out? The communication context of adolescence. Aphasiology, 14(4), 349–364.
Turkstra, L. S., McDonald, S., & DePompei, R. (2001). Social information processing in adolescents: Data from normally developing adolescents and preliminary data from their peers with traumatic brain injury. The Journal of Head Trauma Rehabilitation, 16(5), 469–483.
Verger, K., Junque, C., Levin, H. S., Jurado, M. A., Perez-Gomez, M., Bartres-Faz, D., … Mercader, J. M. (2001). Correlation of atrophy measures on MRI with neuropsychological sequelae in children and adolescents with traumatic brain injury. Brain Injury, 15(3), 211–221.
Verhelst, H., Vander Linden, C., Vingerhoets, G., & Caeyenberghs, K. (2017). How to train an injured brain? A pilot feasibility study of home-based computerized cognitive training. Games for Health Journal, 6(1), 28–38.
Wade, S. L., Borawski, E. A., Taylor, H. G., Drotar, D., Yeates, K. O., & Stancin, T. (2001). The relationship of caregiver coping to family outcomes during the initial year following pediatric traumatic injury. Journal of Consulting and Clinical Psychology, 69(3), 406.
Wade, S. L., Carey, J., & Wolfe, C. R. (2006). The efficacy of an online cognitive-behavioral family intervention in improving child behavior and social competence following pediatric brain injury. Rehabilitation Psychology, 51(3), 179.
Wade, S. L., Cassedy, A., Walz, N. C., Taylor, H. G., Stancin, T., & Yeates, K. O. (2011). The relationship of parental warm responsiveness and negativity to emerging behavior problems following traumatic brain injury in young children. Developmental Psychology, 47(1), 119.
Wade, S. L., Kurowski, B. G., Kirkwood, M. W., Zhang, N., Cassedy, A., Brown, T. M., … Taylor, H. G. (2015). Online problem-solving therapy after traumatic brain injury: A randomized controlled trial. Pediatrics, 135(2), e487–e495.
Wade, S. L., Stancin, T., Kirkwood, M., & Brown, T. M. (2014). Counselor-assisted problem solving (CAPS) improves behavioral outcomes in older adolescents with complicated mild to severe TBI. The Journal of Head Trauma Rehabilitation, 29(3), 198.
Wade, S. L., Taylor, H. G., Drotar, D., Stancin, T., & Yeates, K. O. (1996). Childhood traumatic brain injury: Initial impact on the family. Journal of Learning Disabilities, 29(6), 652–661.
Wade, S. L., Taylor, H. G., Drotar, D., Stancin, T., & Yeates, K. O. (1998). Family burden and adaptation during the initial year after traumatic brain injury in children. Pediatrics, 102(1), 110–116.
Wade, S. L., Taylor, H. G., Drotar, D., Stancin, T., Yeates, K. O., & Minich, N. M. (2002). A prospective study of long-term caregiver and family adaptation following brain injury in children. The Journal of Head Trauma Rehabilitation, 17(2), 96–111.
Wade, S. L., Walz, N. C., Carey, J., McMullen, K. M., Cass, J., Mahone, E. M., & Yeates, K. O. (2011). Effect on behavior problems of teen online problem-solving for adolescent traumatic brain injury. Pediatrics, 128(4), e947–e953.
Wade, S. L., Walz, N. C., Carey, J., Williams, K. M., Cass, J., Herren, L., … Yeates, K. O. (2010). A randomized trial of teen online problem solving for improving executive function deficits following pediatric traumatic brain injury. The Journal of Head Trauma Rehabilitation, 25(6), 409–415.
Wilde, E. A., Ayoub, K. W., Bigler, E. D., Chu, Z. D., Hunter, J. V., Wu, T. C., … Levin, H. S. (2012). Diffusion tensor imaging in moderate-to-severe pediatric traumatic brain injury: Changes within an 18-month post-injury interval. Brain Imaging and Behavior, 6(3), 404–416.
Wilde, E. A., Bigler, E. D., Hunter, J. V., Fearing, M. A., Scheibel, R. S., Newsome, M. R., … Levin, H. S. (2007). Hippocampus, amygdala, and basal ganglia morphometrics in children after moderate-to-severe traumatic brain injury. Developmental Medicine & Child Neurology, 49(4), 294–299.
Wilde, E. A., Hunter, J. V., Newsome, M. R., Scheibel, R. S., Bigler, E. D., Johnson, J. L., … Pedroza, C. (2005). Frontal and temporal morphometric findings on MRI in children after moderate to severe traumatic brain injury. Journal of Neurotrauma, 22(3), 333–344.
Wilde, E. A., Merkley, T. L., Bigler, E. D., Max, J. E., Schmidt, A. T., Ayoub, K. W., … Chu, Z. D. (2012). Longitudinal changes in cortical thickness in children after traumatic brain injury and their relation to behavioral regulation and emotional control. International Journal of Developmental Neuroscience, 30(3), 267–276.
Yeates, K. O. (2010a). Traumatic brain injury. In K. O. Yeates, D. M. Ris, H. G. Taylor, & B. F. Pennington (Eds.), Pediatric neuropsychology: Research, theory, and practice (2nd ed., pp. 112–146). New York, NY: Guilford Press.
Yeates, K. O. (2010b). Mild traumatic brain injury and postconcussive symptoms in children and adolescents. Journal of the International Neuropsychological Society, 16(6), 953–960.
Yeates, K. O., Armstrong, K., Janusz, J., Taylor, H. G., Wade, S., Stancin, T., & Drotar, D. (2005). Long-term attention problems in children with traumatic brain injury. Journal of the American Academy of Child & Adolescent Psychiatry, 44(6), 574–584.
Yeates, K. O., Bigler, E. D., Dennis, M., Gerhardt, C. A., Rubin, K. H., Stancin, T., … Vannatta, K. (2007). Social outcomes in childhood brain disorder: A heuristic integration of social neuroscience and developmental psychology. Psychological Bulletin, 133(3), 535–556.
Yeates, K. O., Luria, J., Bartkowski, H., Rusin, J., Martin, L., & Bigler, E. D. (1999). Postconcussive symptoms in children with mild closed head injuries. The Journal of Head Trauma Rehabilitation, 14(4), 337–350.
Yeates, K. O., & Taylor, H. G. (1997). Predicting premorbid neuropsychological functioning following pediatric traumatic brain injury. Journal of Clinical and Experimental Neuropsychology, 19(6), 825–837.
Yeates, K. O., Taylor, H. G., Barry, C. T., Drotar, D., Wade, S. L., & Stancin, T. (2001). Neurobehavioral symptoms in childhood closed-head injuries: Changes in prevalence and correlates during the first year postinjury. Journal of Pediatric Psychology, 26(2), 79–91.
Yeates, K. O., Taylor, H. G., Rusin, J., Bangert, B., Dietrich, A., Nuss, K., … Jones, B. L. (2009). Longitudinal trajectories of postconcussive symptoms in children with mild traumatic brain injuries and their relationship to acute clinical status. Pediatrics, 123(3), 735–743.
Yeates, K. O., Taylor, H. G., Rusin, J., Bangert, B., Dietrich, A., Nuss, K., & Wright, M. (2012). Premorbid child and family functioning as predictors of post-concussive symptoms in children with mild traumatic brain injuries. International Journal of Developmental Neuroscience, 30(3), 231–237.
Yeates, K. O., Taylor, H. G., Wade, S. L., Drotar, D., Stancin, T., & Minich, N. (2002). A prospective study of short-and long-term neuropsychological outcomes after traumatic brain injury in children. Neuropsychology, 16(4), 514.
Yeates, K. O., Taylor, H. G., Walz, N. C., Stancin, T., & Wade, S. L. (2010). The family environment as a moderator of psychosocial outcomes following traumatic brain injury in young children. Neuropsychology, 24(3), 345.
Yuan, W., Treble-Barna, A., Sohlberg, M. M., Harn, B., & Wade, S. L. (2017). Changes in structural connectivity following a cognitive intervention in children with traumatic brain injury: A pilot study. Neurorehabilitation and Neural Repair, 31(2), 190–201.
Yuan, W., Wade, S. L., Quatman-Yates, C., Hugentobler, J. A., Gubanich, P. J., & Kurowski, B. G. (2017). Structural connectivity related to persistent symptoms after mild TBI in adolescents and response to aerobic training: Preliminary investigation. The Journal of Head Trauma Rehabilitation, 32(6), 378.
Zaloshnja, E., Miller, T., Langlois, J. A., & Selassie, A. W. (2008). Prevalence of long-term disability from traumatic brain injury in the civilian population of the United States, 2005. The Journal of Head Trauma Rehabilitation, 23(6), 394–400.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Hoskinson, K.R., Yeates, K.O. (2020). Pediatric TBI. In: DeLuca, J., Chiaravalloti, N.D., Weber, E. (eds) Cognitive Rehabilitation and Neuroimaging. Springer, Cham. https://doi.org/10.1007/978-3-030-48382-1_11
Download citation
DOI: https://doi.org/10.1007/978-3-030-48382-1_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-48381-4
Online ISBN: 978-3-030-48382-1
eBook Packages: Behavioral Science and PsychologyBehavioral Science and Psychology (R0)