Abstract
Monolayer molybdenum disulfide (MoS2), a 2D semiconducting dichalcogenide material with a bandgap of 1.8–1.9 eV, has demonstrated promise for future use in field-effect transistors and optoelectronics. Various approaches have been used for MoS2 processing, the most common being chemical vapor deposition. During chemical vapor deposition, precursors such as Mo, MoO3, and MoCl5 have been used to form a vapor reaction with sulfur, resulting in thin films of MoS2. Currently, MoO3 ribbons and powder and MoCl5 powder have been used. In addition, sputtering of Mo produces continuous MoS2 films as well. Here we compare the structural properties of MoS2 grown by sulfurization of pulse vapor deposited MoO3 and Mo precursor films. Transmission electron microscopy and atomic force microscopy results demonstrate uniform and continuous film growth for the MoS2 films produced from Mo when compared to the films produced from MoO3. X-ray photoelectron spectroscopy results show that both precursors produce MoS2 films that were stoichiometric and had ~7–8 layers in thickness. We also found that, like other reports, infiltrating reduced graphene oxide during the sulfurization process increases MoS2 grain growth. Correlations between Mo and MoO3 layers and resulting 2D MoS2 film chemistry and structure are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
4.1 Introduction
Two-dimensional (2D) materials such as graphene, molybdenum disulfide (MoS2) , tungsten diselenide (WSe2) , black phosphorus, and boron nitride (BN) have attracted much attention due to their extraordinary electronic and optical properties, making them ideal candidates for next-generation electronic and optoelectronic devices [1,2,3,4]. In particular, a monolayer of MoS2 has a direct bandgap of 1.8–1.9 eV [5, 6], making it an ideal candidate for the mentioned applications [1, 2]. Growth processes of 2D MoS2 include mechanical exfoliation [7,8,9], chemical vapor deposition (CVD) [5, 6], intercalation-assisted exfoliation [10,11,12,13], physical vapor deposition [14, 15], metal organic chemical vapor deposition [16], and a wet chemistry approach involving thermal decomposition of a precursor containing Mo and S [17]. An advantage of CVD growth of MoS2 is the ability to grow large area films for device fabrication. Molybdenum disulfide films grown using CVD have demonstrated promising results for semiconductor grade material properties, with observed field-effect mobilities around 500 cm2/Vs [18]. During CVD growth, sulfurization of molybdenum-containing precursors such as Mo, MoO3 , and MoCl5 is usually performed. In the case of MoO3 [6] and MoCl5 [19], the precursors have been powders or ribbons, whereas Mo has been prepared by e-beam evaporation [5] or sputtering [20]. At the Air Force Research Laboratory, we observed the structure properties of MoS2 films grown by sulfurization of DC magnetron sputtered MoO3 and Mo precursor films at room temperature. In addition, reduced graphene oxide (rGO), known for increasing layer and domain size of MoS2 [21, 22], was incorporated in our growth process of MoS2. This chapter will be focused on our reported data related to this work.
4.2 MoS2 Research
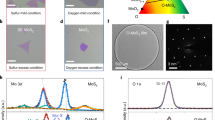
Thin films of metallic Mo and MoO3 were sputtered on c-face sapphire substrates (diameter of 25.4 mm) using a DC magnetron sputtering system (500 V at 100 mA) at room temperature, with an argon pressure of 0.92 Pa. The thickness of the precursor (3 nm for both Mo and MoO3) was controlled by manipulating the sputtering so that there were equal amounts of Mo sputtered in the MoO3 and Mo films. Table 4.1 lists the precursors and sample names. The substrates were ultrasonically cleaned in acetone for 5 minutes prior to deposition of Mo and MoO3. Following sputtering of Mo and MoO3 onto the substrates, the precursors were separately placed in the center of the quartz tube (Fig. 4.1). Sulfur powder (2 g) was placed in a ceramic boat, upstream from the Mo and MoO3 films. Reduced graphene oxide (Sigma-Aldrich) was dispersed in isopropyl alcohol and drop cast on separate sapphire substrates. The rGO samples were air dried before they were placed in the furnace next to the sputtered precursor films of Mo and MoO3 (with a distance of 5 mm between the precursor and rGO samples). After pumping the furnace down to a vacuum pressure of 667 Pa, the samples were heated to 300 °C at 20 °C/min and held there for 15 minutes. This enabled the removal of any residual water molecules. Next, the precursors were heated to 850 °C at a rate of 20 °C/min. As the temperature of the furnace approached 850 °C (around 830 °C), the boat with sulfur was heated to 125 °C using a heating tape. Both the precursors and the sulfur were held at their temperatures for 1 hour, followed by cooling to room temperature. All experiments were performed in an Ar/H2 environment, with a flow rate of 75 sccm.
Chemical vapor deposition setup for MoS2 growth on Al2O3 substrates. (Reproduced from Pacley et al. [3], with the permission of the American Vacuum Society)
Transmission electron microscopy (TEM) imaging of the MoS2 film cross sections for samples S1 (MoS2 grown from Mo precursor) and S1r (MoS2 film grown using Mo precursor with rGO seed) are shown in Fig. 4.2 [3]. The precursor films, MoO3 and Mo, are both shown in Fig. 4.2a, b [3]. Samples S1 and S1r (Fig. 4.2c, e) show uniform and continuous layer growth of MoS2 [3]. Both samples have a thickness of 7–8 nm, indicating the rGO used during the CVD growth of sample S1r had no effect on the film thickness. Atomic force microscopy (AFM) showed that samples S1 and S1r had an RMS of 0.360 nm and 2.43 nm (respectively), and the grain size increased from 4.5 nm to 17.7 nm, respectively (see Fig. 4.3a, b) [3]. This increase in the grain size indicated that the rGO played a role in grain growth of the MoS2. In contrast to the uniform and continuous film growth of samples S1 and S1r, samples S2 (MoO3 precursor) and S2r (MoO3 precursor with rGO seed) demonstrated a non-uniform, outward growth of MoS2 (Fig. 4.2d, f) [3]. It is reported that at 600 °C, MoO3 reduces to MoO2 under an H2 environment [23]. In this research, there was indication that MoO2 had formed after annealing MoO3 at 850 °C. X-ray photoelectron spectra (Fig. 4.4a) showed peaks at 229.57 and 232.7 for Mo(IV), which is typical of MoS2 and MoO2, and 232.19 and 235.32 for Mo(VI), which is typical of MoO3. AFM was performed on the same annealed sample (Fig. 4.3c) [3], and we noticed small islands across the substrate. The islands were formed when the sputtered MoO3 film reduced to MoO2 during annealing at 850 °C. Consequently, sulfurization of MoO2 islands caused MoS2 growth in a Volmer-Weber growth mechanism, which is a result of the film not wetting the substrate [24]. Moser and Levy reported similar growth patterns using sputtering technique to deposit thick MoS2 films [25].
TEM image of (a) MoO3 precursor film used for MoS2 growth; (b) Mo precursor film for MoS2 growth; (c) sample S1 (MoS2 on sapphire using Mo precursor) showing a layer thickness of 7 nm; (d) sample S2 (MoS2 on sapphire using a MoO3 precursor) showing an outward growth of MoS2, with a thickness of 15 nm; (e) sample S1r (Mo precursor) using rGO with a measured thickness of 7–8 nm; and (f) sample S2r (MoO3) using rGO with a thickness of 7 nm. (Reproduced from Pacley et al. [3], with the permission of the American Vacuum Society)
AFM surface topography (1 × 1 μm) for (a) MoO3 precursor that was heated to 850 °C forming MoO2 islands; (b) sample S1 (Mo precursor) showing a dense film of MoS2 with a grain size of 4.4 nm; (c) MoS2 sample S2 (MoO3 precursor) with a grain size of 7.9 nm; (d) MoS2 of MoS2, with a thickness of 15 nm; (e) sample S1r (Mo precursor) using rGO with a measured thickness of 7–8 nm; and (f) sample S2r (MoO3) using rGO with a thickness of 7 nm. (Reproduced from Pacley et al. [3], with the permission of the American Vacuum Society)
XPS spectra of annealed MoO3, MoS2 films S1(MoS2 using Mo precursor), S2 (MoS2 using MoO3 precursor), S1r (MoS2 using Mo precursor and rGO), and S2r (MoS2 using MoO3 precursor and rGO). The annealed MoO3 (a) shows Mo(IV) peaks which are indicative of MoO2 and Mo(VI) peaks that occur when MoO3 is present. Both (b) and (c) show spectra for samples S1 and S2, respectively, having a stoichiometric structure. In (d) and (e), the stoichiometry has increased for samples S1r and S2r, respectively, indicating the rGO played a role in increasing the stoichiometry. (Reproduced from Pacley et al. [3], with the permission of the American Vacuum Society)
Figure 4.3d, e shows the grain structures of MoS2 grown using the sputtered MoO3 films (S2 and S2r) [3]. The RMS values for these samples were 2.00 nm (S2) and 3.66 nm (S2r), and the grain size increased from 7.9 nm (S2) to 12.2 nm (S2r) when rGO was used during the growth. This correlates well with the data from samples S1 and S1r that suggest rGO promotes grain growth when using sputtered precursor films. There was also a decrease in the film thickness, going from 15 nm (S2) to 7 nm (S2r) when rGO was used during the sulfurization process (Fig. 4.2d, f). Ling et al. [22] report that organic seed promoters (such as PTAS) enable heterogeneous nucleation sites and that the size of the MoS2 domains is dependent upon the distance of the seed promoter from the precursor. We believe this is what occurred when rGO was used in our experiments involving sputtered Mo and MoO3 films. However, further investigation needs to be conducted to better understand the kinetics, and mechanism of increasing grain size, when using rGO during the sulfurization sputtered films.
X-ray photoelectron spectroscopy (XPS) was performed for composition and chemistry analysis of the films that were grown in this research. The survey spectra (not shown) from sulfurized thin films of samples S1/S1r and S2/S2r showed peaks from Mo and S, as expected. As mentioned previously, Fig. 4.4a shows the spectra for annealed MoO3 [3]. The influence of the rGO on MoS2 stoichiometry was analyzed by comparing the S:Mo ratios obtained from the XPS spectra. The MoS2 films grown from samples S1 and S1r had S:Mo ratios of 1.9 and 2.1, respectively (see Fig. 4.4b, d). Samples S2 and S2r demonstrated the same respective S:Mo ratios of 1.9 and 2.1 (see Fig. 4.4c, e). The apparent improvement in the film stoichiometry is most likely a result of the Mo:S averaging over large spot size analysis area in XPS, which is orders of magnitude larger when compared to the average grain sizes of synthesized MoS2 films. The presence of the rGO helped to increase the MoS2 grain size areas and correspondingly decrease contributions of photoelectrons escaped from the grain boundary areas.
Curve fits to the Mo 3d doublets for all of the samples revealed two populations of Mo atoms. The Mo 3d5/2 peak at 229.8 eV and Mo 3d3/2 peak at 232.9 eV reveal the presence of Mo(IV), with a binding energy typical of MoS2 or MoO2 [26]. The Mo 3d5/2 peak at 232.7 eV and Mo 3d3/2 peak at 235.8 eV indicate the presence of Mo(VI), with a binding energy typical of MoO3 [27]. This suggests that there is likely some MoO2 present at the surface or at grain boundaries within the films. However, the intensity for both the Mo(IV) and Mo(VI) peaks are so low, that the presence of MoO2 and MoO3 is negligible.
4.3 Conclusion
The influence of metallic Mo and MoO3 thin-film precursors on the structure of MoS2 films grown by CVD was investigated. TEM established that rGO did not have an impact on the MoS2 film thickness for sputtered Mo but that it was responsible for the increase in the grain size. We also observed an increase in the grain size when rGO was used during sulfurization of sputtered MoO3. Reports demonstrate that seed promoters diffuse onto growth substrates, acting as nucleation sites for MoS2 growth. In addition, the size of the MoS2 domains is dependent upon the distance between the seed promoter and the growth substrate. In conclusion, sputtered Mo precursor films produce better uniformity and continuous MoS2 films, making these nanocrystalline films potentially applicable for electronic and optoelectronic devices.
References
B. Radisavljevic, A. Radenovic, J. Brivio, et al., Single-layer MoS2 transistors. Nat. Nanotechnol. 6, 147 (2011)
Z. Yin, H. Li, H. Li, et al., Single-layer MoS2 phototransistors. ACS Nano 6(1), 74 (2012)
S. Pacley, J. Hu, M. Jespersen, et al., Impact of reduced graphene oxide on MoS2 grown by sulfurization of sputtered MoO3 and Mo precursor films. J. Vac. Sci. Technol. A 34(4), 041505–041501 (2016)
D.Y. Zemlyanov, M. Jespersen, D.N. Zakharov, et al., Versatile technique for assessing thickness of 2D layered materials by XPS. Nanotechnology 29(115705), 1 (2018)
Y. Zhan, Z. Liu, S. Najmaei, et al., Large-area vapor-phase growth and characterization of MoS2 atomic layers on a SiO2 substrate. Small 8(7), 966 (2012)
S. Najmaei, Z. Liu, X. Zou, et al., Vapour phase growth and grain boundary structure of molybdenum disulphide atomic layers. Nat. Mater. 12(8), 754 (2013)
B. Radisavljevic, M.B. Whitwick, A. Kis, Integrated circuits and logic operations based on single-layer MoS2. ACS Nano 5(12), 9934 (2011)
K.F. Mak, C. Lee, J. Hone, et al., Atomically thin MoS2: A new direct-gap semiconductor. Phys. Rev. Lett. 105, 136805–136801 (2010)
J. Brivio, D.T.L. Alexander, A. Kis, Ripples and layers in ultrathin MoS2 membranes. Nano Lett. 11(12), 5148 (2011)
H. Ramakrishna Matte, A. Gomathi, A. Manna, et al., MoS2 and WS2 analogues of graphene. Angew. Chem. Int. Ed. 49(24), 4059–4062 (2010). https://doi.org/10.1002/anie.201000009
Z. Zeng, Z. Yin, X. Huang, et al., Single-layer semiconducting nanosheets: High-yield preparation and device fabrication. Angew. Chem. Int. Ed. 50(47), 11093–11097 (2011). https://doi.org/10.1002/anie.201106004
V. Nicolosi, M. Chhowalla, M.G. Kanatzidis, et al., Liquid exfoliation of layered materials. Science 340(6139), 1226419 (2013). https://doi.org/10.1126/science.1226419
G. Eda, H. Yamaguchi, D. Voiry, et al., Photoluminescence from chemically exfoliated MoS2. Nano Lett. 11(12), 5111–5116 (2011). https://doi.org/10.1021/nl201874w
C. Muratore, A.A. Voevodin, Control of molybdenum disulfide basal plane orientation during coating growth in pulsed magnetron sputtering discharges. Thin Solid Films 517(19), 5605–5610 (2009). https://doi.org/10.1016/j.tsf.2009.01.190
C. Muratore, J.J. Hu, B. Wang, et al., Continuous ultra-thin MoS2 films grown by low-temperature physical vapor deposition. Appl. Phys. Lett. 104, 261604 (2014). https://doi.org/10.1063/1.4885391
K. Kang, S. Xie, L. Huang, et al., High-mobility three-atom-thick semiconducting films with wafer-scale homogeneity. Nature 520, 656 (2015)
C. Altavilla, M. Sarno, P. Ciambelli, A novel wet chemistry approach for the synthesis of hybrid 2D free-floating single or multilayer Nanosheets of MS2@oleylamine (M═Mo, W). Chem. Mater. 23(17), 3879–3885 (2011). https://doi.org/10.1021/cm200837g
H. Schmidt, S. Wang, L. Chu, et al., Transport properties of monolayer MoS2 grown by chemical vapor deposition. Nano Lett. 14(4), 1909–1913 (2014). https://doi.org/10.1021/nl4046922
Y. Yu, C. Li, Y. Liu, et al., Controlled scalable synthesis of uniform, high-quality monolayer and few-layer MoS2 films. Sci. Rep. 3, 1866 (2013)
N. Choudhary, J. Park, J.Y. Hwang, et al., Growth of large-scale and thickness-modulated MoS2 nanosheets. ACS Appl. Mater. Interfaces 6(23), 21215–21222 (2014). https://doi.org/10.1021/am506198b
Y. Lee, X. Zhang, W. Zhang, et al., Synthesis of large-area MoS2 atomic layers with chemical vapor deposition. Adv. Mater. 24(17), 2320–2325 (2012). https://doi.org/10.1002/adma.201104798
X. Ling, Y. Lee, Y. Lin, et al., Role of the seeding promoter in MoS2 growth by chemical vapor deposition. Nano Lett. 14(2), 464–472 (2014). https://doi.org/10.1021/nl4033704
E. Lalik, W.I.F. David, P. Barnes, et al., Mechanisms of reduction of MoO3 to MoO2 reconciled. J. Phys. Chem. B 105(38), 9153–9156 (2001). https://doi.org/10.1021/jp011622p
D.L. Smith, Thin Film Deposition: Principles and Practice, 1st edn. (McGraw-Hill Education, New York, 1995)
J. Moser, F. Levy, Growth mechanisms and near-interface structure in relation to orientation of MoS2 sputtered thin films. J. Mater. Res. 7(3), 734–740 (1992). https://doi.org/10.1557/JMR.1992.0734
G. Seifert, J. Finster, H. Müller, SW Xα calculations and x-ray photoelectron spectra of molybdenum(II) chloride cluster compounds. Chem. Phys. Lett. 75(2), 373–377 (1980). https://doi.org/10.1016/0009-2614(80)80534-3
P.A. Spevack, N.S. McIntyre, Thermal reduction of molybdenum trioxide. J. Phys. Chem. 96(22), 9029–9035 (1992). https://doi.org/10.1021/j100201a062
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pacley, S. (2020). 2D Materials: Molybdenum Disulfide for Electronic and Optoelectronic Devices. In: Kinsella, M. (eds) Women in Aerospace Materials. Women in Engineering and Science. Springer, Cham. https://doi.org/10.1007/978-3-030-40779-7_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-40779-7_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-40778-0
Online ISBN: 978-3-030-40779-7
eBook Packages: EngineeringEngineering (R0)