Abstract
Photocatalysis is one of the fastest growing technologies for the treatment of pollutants, utilizing the mechanism of reaction with the help of light (photo emissions). Photocatalysis has captured broad academic and research interest during the past three decades for its potential of controlling pollution in air and water. Its qualities, such as low cost and high efficiency, have caused researchers all over the world to focus on it and also promoted many industrial applications and much research. Photocatalysis has been used to remove major air pollutants, disinfect water, and oxidize various organic chemicals. In this connection, this chapter considers the properties of the ideal photocatalyst, available photocatalytic materials for air pollution control, common indoor air pollutants and their severe health effects, and purification techniques for indoor air pollution. Furthermore, photocatalytic oxidation techniques for the removal of volatile organic compounds are discussed in detail.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
9.1 Introduction
Today the entire world is struggling with the severe effects of environmental pollution and researchers all over the world are looking to provide solutions for this problem (Anpo 2000; Naushad 2014). In 2012, one in eight of all deaths, around 7 million people, died because of bad air quality, which has a vast negative impact around the worldwide (Naushad 2014; Lelieveld et al. 2015). To have more yield and more profits, chemical industries provide products with more favourable usage although they have hazardous impacts on the human health and environment. Compounds such as phenol, pesticides from wastewater sewage, polymeric surfactants, and herbicides release dangerous chemical substances that are not easily biodegradable (Hoffmann et al. 1995; Alshehri et al. 2014; Al-Othman et al. 2011; Al-Kahtani et al. 2018).
People think that pollutants occur more in the outdoor environment compared to indoors because of heavy traffic pollutes the air. But, although this may be shocking, an urban area with average traffic might be cleaner than indoor air. Some of the studies reported that the concentration of air pollution indoors is five times higher than in the outdoor environment (Zhang and Smith 2003; Bernstein et al. 2008; Kolarik et al. 2010). Factors such as smoke, electronic equipment, inadequate ventilation, mould damage, and furniture increase air pollution in schools, homes, and public places. Sick building syndrome is a disorder found to occur in a group of people who spent time in a certain building with indoor air pollution (Wolkoff and Nielsen 2001; Crook and Burton 2010; Norhidayah et al. 2013). Some pollutants may cause diseases such as respiratory infections or cancer that are diagnosed later (Zhang and Smith 2003). From the foregoing facts and the literature, it is evident that the people spend 90% of their time in an indoor environment, which is a key factor to be considered for human health and comfort (Li 2016). Also, some of the studies state that compared to the outdoor environment there is a lag in public health awareness in indoor air pollution (Challoner and Gill 2014; Rohra and Taneja 2016).
The quality of the indoor air can be improved by eliminating or reducing contaminants by ventilation, by controlling indoor source pollutant emission, and by using low emission materials. Adequate ventilation and effective infiltration are the traditional methods to improve indoor air quality (IAQ) in which indoor air pollution can be reduced by introducing outside air into the building. As ventilation is inadequate (Bennett 2009) in modernized buildings, the only way to achieve air quality is to purify the air.
Photocatalysis is one of the fastest growing technologies for the treatment of air pollutants, utilizing the mechanism of reaction with the help of light (photo emissions). Fujishima and Honda 1972, while working on the photolysis of water, initially recognized this as a heterogeneous photocatalyst. Much research was put forward in their work towards the development and growth of photocatalysis techniques, and in recent years researchers have explored and reported the use of semiconductor materials as photocatalysts for elimination of organic and inorganic species. It has been considered an effective tool for the protection of the environment because of its large oxidation capacity (Robert and Malato 2002). Still, applications towards the improvement of indoor air by reducing the contaminants are open to wide usages. Hence, this chapter discusses photocatalysis for efficient indoor air pollution remediation, techniques and methods available for the elimination of pollutants, and the perspectives.
9.2 Ideal Catalyst
Fujishima et al. have reported that a photocatalyst to be considered as an ideal candidate should possess such characteristics as these (Fujishima et al. 2000):
-
1.
Photo-activity, nontoxic nature, low cost
-
2.
Inert to chemical and biological activity
-
3.
Photo-corrosion stability
-
4.
Active towards visible light
The various steps involved in photocatalysis activities (Shan et al. 2010) are diffusion, adsorption, reaction, desorption, and diffusion of the final products from the surface of the catalyst (Serpone and Emeline 2012). Figure 9.1 represents the steps involved in photocatalytic activities.
Schematic representation of photocatalysis scheme (Demeestere et al. 2007)
9.3 Photocatalytic Materials for Air Pollution
Semiconductors such as TiO2 , Fe2O3, ZnO, CdS, WO3, and SnO2 (Vinu and Madras 2010) have major roles in photocatalytic activity. Compared to TiO2, all the other candidates have some limitations: for example, ZnO is unstable at low pH values, CdS, iron oxides, and ZnS suffer from corrosion, and WO3 is less active. Among these, TiO2 is considered to be a potential candidate for photocatalytic activities because of its numerous advantages: chemical stability, low cost, expected electronic and optical properties, and nontoxicity (Litter 1999). Figure 9.2 represents the energy bandgap diagram of various semiconductor oxides.
Energy bandgap diagram of various semiconductor oxides (Demeestere et al. 2007)
Generally, TiO2 is available in three forms: anatase, rutile, and brookite. Of these the third is an uncommon form and stable only at high temperatures. Figure 9.3 represents the structures of TiO2 in three different forms. Although TiO2 has several merits towards photocatalytic activities, it is also restricted because of the wide bandgap (3.2 eV), which requires an excitation source of ultraviolet (UV) wavelength at 387 nm. The availability of this excitation energy is only 5% (Fig. 9.4). The photocatalytic oxidation (PCO) of volatile organic compounds (VOC) and inorganic pollutants are mostly treated by using wide bandgap semiconductor materials. The surface reactants on the semiconductors are the main reason for the foundation of the PCO air purification process (Bickley and Stone 1973; Spanhel et al. 1987; Cant and Cole 1992; Linsebigler et al. 1996; Khan and Ghoshal 2000; Shen and Ku 2002). The properties such as catalyzing, adsorbing, deodorizing, and disinfection of TiO2 project it as a potential candidate for air pollution treatments.
Various TiO2 phases (Smyth 2010)
Electromagnetic spectrum representing the entire wavelength region (www.miniphysics.com)
Serious studies were put forward by researchers all over the world to improve the photocatalytic activity of existing materials or to find an alternate to enhance photocatalytic activity. Li et al. (2005) stated that shifting the optical absorption point of TiO2 near to the lower energy can improve photocatalytic activities in the visible wavelength. It is a renewable energy source and can be used for large-scale operations. Photocatalysis can be achieved by doping anions (such as sulfur, fluorine, nitrogen, boron, and carbon) in TiO2 to achieve high efficiency. Several modification techniques are used for producing highly active TiO2 for photocatalytic activities, such as doped TiO2 (e.g., self-doping, non-metal doping, co-doping, metal doping), mesoporous TiO2, shape-controlled TiO2, supported TiO2, surface-modified TiO2, and dye-sensitized TiO2 (Sun et al. 2010).
Nitrides, metal sulfides, and oxynitrides are also proposed as candidates for photocatalytic activity using visible light (Inoue 2009). Researchers have proposed a surface plasmonic effect-based plasmonic photocatalyst (Kale et al. 2014). Very recently, graphitic carbon nitride-based metal-free photocatalysts have also been used as a photocatalyst using visible light (Sun et al. 2014; Cao et al. 2015). Although the technique and the enactment of PCO for purification of air pollutants are customized, the difficult aspect is the advances in photocatalytic material and reactors for this application.
Machado et al. 2006 reported that photocatalytic activities can be enhanced by increasing the surface area of the catalyst. A high interaction of reactant with catalyst takes place when the surface area is large. Doping of materials helps reduce the energy bandgap between the valence and conduction bands, which in turn requires low energy (visible light source) for the excitation of electrons from the lower to the higher band (Juanru et al. 2007).
9.4 Biological and Chemical Contaminants
People spend most of their time in an indoor air environment, which determines the health and well-being of a person. Perilous substances emanating from construction materials and indoor tools cause a wide range of health problems and even may be fatal. Around 4.3 million people died in 2012 because of illnesses produced by household air pollution (WHO 2014). Indoor contaminants can be classified into two major categories: chemical and biological.
9.4.1 Biological Contaminants
Biological contaminants are living entities that can be transported via air and are often invisible. Bacteria, viruses, animal dander, moulds, and pollen are some of the common biological impurities which can cause dangerous effects in humans (Nelson et al. 1988). Nutrients and poor moisture content are the two important reasons for the growth of these pollutants (US Department of Health and Human Services 2006). The amount of contaminant that is needed to cause infection in the human being is unknown but will vary from one person to other.
Among the contaminants, airborne bacteria and fungi are considered to be major pollutants that cause challenging health issues. These pollutants will grow under high humidity conditions in materials such as grease, soap scum, dirt, textiles, and paper. Once the mould colony has been formed, it will start floating on the air (Dallongeville et al. 2015) and fall on land or surfaces, which leads to symptoms such as eye and skin irritation, nasal stuffiness, and wheezing (Taskinen et al. 1997). Madureira et al. (2015) proposed a brief investigation of biological pollutants by accumulating air samples from homes, primary schools, childcare centres, and elderly care centres in the area of North Portugal, and reported that the pollutants are high in childcare centres and schools because of lack of ventilation and different density of occupants. Dust mines are another source causing biological pollutants (Calderón et al. 2015).
9.4.2 Chemical Contaminants
Among chemical contaminants, gases such as nitrogen dioxide (NO2), carbon monoxide (CO), sulfur dioxide (SO2), particulate matter (PM2.5 and PM10), and chemical and microbial volatile organic compounds (VOCs) are the most serious candidates causing air pollution (Bernstein et al. 2008). One second-most candidate is radon, a colourless radioactive gas that causes lung cancer in most countries (Environmental Protection Agency 2017).
Possible health problems caused by some of the most significant indoor air pollutants, their effects, and the indoor sources are listed in Table 9.1. The first step to reduce or avoid problems from these materials is to know their impact.
Stranger et al. (2009) reported a brief investigation on particulate matter in which the collected samples revealed that 90% of indoor and 85% of outdoor particles are smaller than 2.5 μg. The approximate indoor mass density of PM2.5 was found to be 46.6 μg/m3 and 29.5 μg/m3 in a smoker’s and non-smoker’s house, respectively, which are above the normal PM2.5 EU norm of 25 μg/m3. The indoor PM2.5 concentrations of ten various buildings in Dublin, Ireland, were analyzed and reported by Challoner and Gill 2014. The concentration of PM2.5 was found to be greater during working hours when compared to non-working hours, of which five locations exceeded the EU norm.
The concentration of PM10 is approximately equal to 49.4 μg/m3 in a smoker’s house and 33.8 μg/m3 in a non-smoker’s house (Stranger et al. 2009). The obtained PM10 concentration is found to be more or less equal with the WHO indoor air guidelines value and in good agreement with the mass concentration ranges published by the US Environmental Protection Agency (USEPA 2004a, b).
Concentrations of NO2 for personal exposure, indoor and outdoor, were 41.4, 32.6, and 38.9 μg/m3, respectively. The concentrations of NO2 were in the range 25–43 μg/m3 for personal exposure, 13–62 μg/m3 indoors, 24–61 μg/m3 outdoors, and 27–36 μg/m3 in the workplace, as reported in the INDEX report by the European Commission (EC). In European homes, the maximum associated levels for the use of gas appliances are in the range 180–2500 μg/m3 (Koistinen et al. 2008). Rivas et al. (2014) recently examined the concentrations of NO2 in Barcelona schools and reported that the mean concentration of NO2 (30 μg/m3) is below the guideline level. The concentration of SO2 both indoors and outdoors was investigated by Stranger et al. (2009), who reported that the levels found were much lower than the guidelines level.
The indoor air quality in ten energy-efficient French schools was examined for 4.5 days (Verriele et al. 2016), and about 150 VOC species were characterized. Measured VOC level varied from 100 μg/m3 to 680 μg/m3, depending on occupancy level. At most, 40–85% of ketones and aldehydes are detected from the overall VOC. Acetone, formaldehyde, pentanal, acetaldehyde, 2-butanone, heptane, hexaldehyde, and toluene are present in major concentrations. Aromatic compounds and formaldehyde have concentrations of 8–50 μg/m3 and 14.8 μg/m3, respectively, and terpenes are always a minor compound (Verriele et al. 2016). VOC concentration varies sharply in various indoor environments.
9.5 Methods to Control the Mobilization of TiO2
For the development of photo-reactors for the purification of air contaminants, the controlling of TiO2 is a tedious process. To control the mobilization of TiO2, techniques such as fabrication of TiO2 as a film and depositing TiO2 on the supported materials are followed. Thevenet et al. (2014) reported that the mobilization of TiO2 can be controlled by depositing P25-loaded TiO2 in glass fibers and using these for PCO removal of acetylene. Colloidal silica at 50% and 50% TiO2 by weight are suspended in water for coating on silica glass fibers. Materials such as PVC sheets (Tejasvi et al. 2015), monoliths (Monteiro et al. 2015), optical fibers (Hou and Ku 2013), and graphene oxides (Andryushina and Stroyuk 2014) were also used for controlling the mobilization of photocatalytic materials.
Compared to the deposition of TiO2 on supported materials, the synthesis of TiO2 film is the most powerful method for controlling the mobilization of the photocatalyst materials. Antonello et al. (2014) proposed a novel method for the fabrication of transparent TiO2 with high mechanical strength using an electrochemical technique. The fabricated film was then used for the PCO removal of volatile oxidation compounds as well as a self-cleaning material. TiO2 thin films for application to glass windows were suggested by Xie et al. (2016), using a polyvinylpyrrolidone (PVP) modified sol-gel route.
Chemical vapour deposition (CVD), electrophoretic deposition, sol-gel, layer-by-layer method, and thermal methods are important methods proposed for controlling TiO2 mobilization. In CVD techniques, at high temperature the supporting material is exposed to the gaseous form of the catalyst, which decomposes at the support surface (Shan et al. 2010; Robert et al. 2013). These methods can be used for the synthesis of N-doped TiO2 from various mixtures (Dunnill et al. 2009), in situ crystallization of TiO2 (Nizard et al. 2008; Sun et al. 2008), and for controlling film thickness (Nolan et al. 2006).
The sol-gel is a very simple method, in which the support materials are dipped into a solution of TiO2 mixture heated at high temperature above 450 °C at a controlled rate. Flexibility, simplicity, and low cost make this method the one most used (Shan et al. 2010; Lopez et al. 2013; Sampaio et al. 2013). For substrates such as stainless steel, Al2O3 monoliths, and aluminum and glass fibers, a low-temperature sol-gel method is proposed to fabricate thick, robust, and stable photocatalytic layers for various commercial applications (Kete et al. 2014).
In thermal techniques, the catalyst mixture is directly interspersed on the supporting substrate and then thermally treated to form desired characteristics (Tennakone et al. 1995; Shan et al. 2010). However, performance and characteristics of the deposited catalyst hinge on the calcination temperature (Yu et al. 2000). In the layer-by-layer method, the support material that is to be coated is dipped or sprayed using a catalyst solution for the required thickness. The thickness of the catalyst can be easily controlled and hence a large surface area can be obtained (Yu et al. 2000; Nolan et al. 2006; Krogman et al. 2008; Nizard et al. 2008; Sun et al. 2008; Nakajima et al. 2009; Priya et al. 2009). In the electrophoretic deposition method, under the influence of an electric field, the charge particles are moved in a stable support material. This method is found to be most environmentally safe for the complex shapes and provides uniform film thickness. It can be used to deposit TiO2 composites on the metal (Raddaha et al. 2014) or plastic (Chen et al. 2011) substrates, TiO2 nanotube arrays (Bavykin et al. 2013), and perhaps used for coating biological molecules in future (Chávez-Valdez et al. 2012).
9.6 Purification Techniques for Indoor Air Pollution
Various measures should be taken to provide a healthy and comfortable indoor environment by limiting the concentration levels of pollutants and improving indoor air quality (IAQ). Various methods for prevention of air pollution are indoor source control, ventilation, and indoor air purification. Both destructive and nondestructive methods are explained briefly in the following sections.
9.6.1 Indoor Source Control
As said by the modern hygiene and preventive medicine pioneer Max Von Pettenkoffer (1958), “If there is a pile of manure in a space, do not try to remove the odor by ventilation. Remove the pile of manure.” (Paunović et al. 2005). This quote explains the significance of controlling the source in an indoor environment as a primary approach. To improve IAQ, the most inexpensive operative method is to eradicate or decrease the indoor air pollution sources (Guo et al. 2003).
Source control includes different principles and applications based on the properties of the particular contaminants, preventing contaminants from entering the buildings, and exchanging emitting sources with low-emitting or non-emitting sources (Nazaroff 2013). Also, indoor environment factors such as climate and humidity can be controlled to decrease the emission rate or to avoid conditions that can lead to the generation of contaminants (Clements-Croome et al. 2008).
Several combinations are proposed to improve IAQ. Hult et al. (2015) have carried out examinations of new US residences to determine the extent to which formaldehyde exposure can be reduced by controlling indoor sources (building materials). Homes built with low-VOC materials were compared for formaldehyde concentration with buildings with conventional materials. At an air exchange rate of 0.35 per hour, the formaldehyde concentration in conventionally built homes (46 μg/m3) was found to be higher than in homes with low-emitting materials (34 μg/m3), that is, 27% reduction was found in buildings with low-emitting materials. This investigation proves the merits and importance of source control.
9.6.2 Ventilation
The second most useful approach for maintaining good IAQ is by providing ventilation by either natural or mechanical methods (Technical 2008). By ventilation, indoor pollutants can be reduced by mixing with conditioned outdoor air, dispensing this conditioned air throughout the building, and exhausting indoor air to the outside environment.
The difference in air pressure of indoor and outdoor environments provides ventilation by passing the air through leaks in the building shell. However, this is not possible in recently built new buildings, which are constructed to be more airtight and use insulation to reduce loss of energy. Fresh air is reduced in mechanical ventilation systems to exchange indoor air with a supply of relatively clean outdoor air (Yu et al. 2009).
Lyng et al. (2015) has studied the effect of concentrations of polychlorinated biphenyl (PCB) in air by employing mechanically balanced ventilation units. Air exchange rates were increased from 0.2 to 5.5 per hour with mechanical ventilation, and PCB concentrations were decreased to 30%. Hence, it is evident that indoor air pollution can be significantly reduced by installing ventilation in buildings.
9.6.3 Filtration
Filtering is one of the most important techniques in which contaminants responsible for air pollution can be removed and contaminants in the environment can be reduced significantly. Mechanical and electronic air filters are the two basic types of filters employed to remove pollutants and airborne microbes. Mechanical filters simply capture the pollutant particles passing through them. High-efficiency particulate air (HEPA) filters are examples of such mechanical filters, with the filtering efficiency of 99.97% for larger particles (0.3 μm airborne particles). In spite of its advantages, HEPA has disadvantages such as low filtering efficiency for smaller particles: these may be a source of infection from microorganisms harmful to human health and act as a source of pollution when they have not been used properly. Deposition of organic and inorganic matter on the filter leads to microbial growth that decreases filter efficiency and finally causes filter deterioration (Yu et al. 2009). Also, and at the same time, the reaction of deposited particles in the filter with ozone results in formation of by-products such as formic acid, formaldehyde, ultra-fine particles, and carbonyls (Hyttinen et al. 2006; Waring et al. 2008; Yu et al. 2009). Hence, use of mechanical filters for prevention of air pollution requires constant monitoring, cleaning, and replacing of filters (Zhang et al. 2011).
Electronic air filters such as electrostatic precipitators (ESP), which follow the principle of electrostatic attraction to catch air particles, can be used to remove contaminants from the air. The air is passed through an ionization section of the filter where the particles are charged. The ionized particles then pass through the collector plates of opposite charge where they are attracted and trapped by the plates. For the efficient use of filters, the particles must be cleaned frequently from the collector plates. In wet ESP, water is used to clean off the particles whereas in dry ESP rappers are used. A demerit of the ESP is the rapping re-entrainment. While cleaning the dust from the filters, some dust must remain in the filter itself, which causes filter efficiency to decrease. In another method to remove particles from the air, a large quantity of charged particles is passed through a room using an ionizer. Instead of collecting the charged particles as in ESP, here the airborne particles mingle with the ions and seem to be become charged particles. These particles in turn become attached to the nearby surface or attract each other and go out of the air (Environmental Protection Agency 2014b).
Compared to the filters (HEPA or ESP), ionizers seem to have less efficiency in filtering particles such as pollen, tobacco, fungal spores, smoke, and dust (Shaughnessy et al. 1994; Pierce et al. 1996). Regrettably, a few of the electronic filters emit ozone as a secondary product of their operation (Poppendieck et al. 2014). In a comparative study of different ionizers the ozone generation rates range from 56 μg/h to 2757 μg/h (Niu et al. 2001). Berry et al. (2007) examined the consequences of indoor ozone level with a commercially available ionic air cleaner. The measured ozone level was found to be very high in front of the ionic cleaner (77 ppb) when compared to the same room without an ionic cleaner (13–19 ppb). The obtained ozone level was found to be close to EC guidelines.
9.6.4 Adsorption
Electronic and mechanical air filters remove only particulate matter, whereas adsorption units can remove VOC. In the adsorption process, the gasses are attracted towards the surface of a solid such as zeolite, activated carbon, or activated alumina and subsequently removed from the air stream (Yu et al. 2009). Adsorption can occur as physical adsorption or chemical adsorption. If adsorption is induced by Van der Waals forces (intermolecular attractive force), it is then called physical adsorption in which there is no modification in the properties of either the adsorbent or the gas. Chemical adsorption, also called chemisorption, includes the formation of new chemical bonds (Zhong and Haghighat 2015; WHO 2000). It is a vital reaction in which electron or ion exchange occurs among gas and adsorbent. Particles that have a greater attraction towards the adsorbent will be adsorbed and the remaining molecules will be retained in air. Hence, this method is only efficient for a limited quantity of pollutants. It is also reported that the accretion of pollutants will provide a path for the growth of bacteria and moulds (Van Durme et al. 2008). Schleibinger and Rüden 1999 reported growth of VOC before and after the use of filter pieces. They also proved the presence of VOCs such as esters, alcohols, aldehydes, and ketones in the used filter. Another major demerit is that adsorption will be easily carried out at humidity and lower temperatures, which has a negative impact on targeted indoor pollutants. Similar to filters, absorbents also have limited capacity in reducing air pollution. Hence, these should be frequently maintained for better results (Ge et al. 2015).
9.6.5 Ultraviolet Germicidal Irradiation
The aforementioned techniques will change the phase of the molecules from one form to another instead of destroying them. To destroy pollutants in the air, numerous innovative methods have been proposed, named advanced oxidation processes (AOP). These methods eliminate a broad range of VOCs by producing such agents as atomic oxygen and ozone. Ozone generators, photocatalytic oxidation, plasma reactors, and UV lamps are some examples of AOP. Ultraviolet germicidal irradiation (UVGI) kills biological pollutants by flouting the molecular bonds in their DNA using ultraviolet radiation (Zhang et al. 2015). The replication of the pollutants can be completely limited by breaking their DNA, which in turn causes cell death (Bennett 2009). The UVGI cleaners can be used for the timely purpose but not as an effective replacement technique for filtration systems (Zhang et al. 2011). The efficiency of the UVGI has been limited because of the devastation of moulds and bacteria requires high radiation, which in turn became harmful (Environmental Protection Agency 2009; Wang et al. 2009).
9.6.6 Ozonation
Ozonisers as mostly used for indoor air purification purposefully emit ozone by utilizing electrical discharge or UV lamps to clean the environment. The ozone generators are considered to be an effective purification tool that is highly oxidative and destroys most of the unsaturated VOCs in the indoor air environment. The ozone vendors generally state that ozone generators are effective and safe devices for reducing indoor air pollution. They also claim that the ozone, also called energized oxygen, activated oxygen, or super oxygen, can kill all the contaminants and provide a pollutant-free indoor air environment by destroying all airborne particles, viruses, moulds, chemicals, odors, and bacteria and produce secondary products such as water, carbon dioxide, and oxygen. In spite of all the advantages just mentioned, the ozone purifiers have many demerits (Environmental Protection Agency 2009, 2014b).
In principle, ozone generators are not an efficient method for destroying pollutants at low ozone levels, being an effective approach only at unsafe, high ozone levels. The emitted levels of ozone generators are approximately 500 ppb (Hubbard et al. 2005), which is five times higher than the maximum ozone exposure reported by the World Health Organization (WHO). Boeniger 1995 also reported that the reaction time for ozone with air pollutants is very high and may last for several months or even years. Contrary to claims by ozone vendors, ozone generators are not efficient in removing formaldehyde (Esswein and Boeniger 1994) or carbon monoxide (Shaughnessy et al. 1994) from the air. Some researchers also report that the reaction of ozone with pollutants may result in harmful secondary products (Weschler et al. 1992a, b; Weschler and Shields 1996).
9.7 Photocatalytic Oxidation
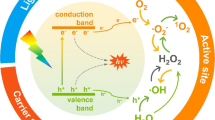
Photocatalytic oxidation (PCO) is an inventive and encouraging technique for removing contaminants from indoor air. With the help of light energy, enactment of the semiconductor is improved and thus can eradicate a wide range of indoor pollutants into CO2 and H2O (Demeestere et al. 2007; Nakata and Fujishima 2012; Chen et al. 2015a). Based on band structure, the semiconductors are characterized and the energy bandgap is defined as the bandgap (Eg) between the valence band and conduction band. The semiconductor material is activated by UV light that has energy equal to or greater than the bandgap. The electron in the valence band absorbs the energy from a photon and shifts to the higher valence band (conduction band), creating a hole in the conduction band. This pair is called an electron–hole pair.
9.7.1 Photocatalyst Oxidation Removal of Volatile Organic Compounds
Compared to outdoor air quality, indoor air quality is a cause of major anxiety and many problems to humans. VOCs, important contaminants in indoor air pollution, have become the most widespread PCO targets (Zhao and Yang 2003; Wang et al. 2007). Some studies related to TiO2-based PCO for VOCs are tabulated in Table 9.2. The PCO confiscation of VOCs together with toluene, acetone, benzene, acetaldehyde, etc. are removed using continuous reactors rather than aqueous degradation. The reproducibility of pure TiO2 depends on parameters such as shape control (Le Bechec et al. 2015), size dependence (Bianchi et al. 2014), preferentially exposed facets (Ren et al. 2016), and tailored porous structures (He et al. 2014). The larger Brunauer–Emmett–Teller (BET) surface, a more active surface, and pre-adsorption would provide good PCO performance. Figures 9.5 and 9.6 represent the mechanism proposed for the photocatalytic oxidation degradation of benzene and trichloroethylene, respectively.
Photocatalytic oxidation of benzene (Ma et al. 2007)
Mechanism for the degradation of trichloroethylene (TCE) (Ou and Lo 2007)
In PCO air purification, better stability and higher activity can be achieved by the modification of TiO2 methods of transition metal doping (Park et al. 2004; Huang et al. 2015), non-metal doping (Chen et al. 2013), semiconductor coupling (Liu et al. 2013), and noble metal doping (Murcia et al. 2013). The immobilization technique and material development can be assimilated in practical applications to obtain better outcomes.
Pham and Lee 2015 reported that the V-doped TiO improved photocatalytic activity. The prepared V-doped TiO was then loaded onto polyurethane and then allowed to remove the toluene in a continuous reactor. It is evident from Table 9.2 that the TiO-based photocatalysts are efficient candidates to decompose a vast range of organic contaminants.
9.7.2 Removal of Inorganic Gas Pollutants Using PCO
Inorganic gas pollutants such as NOx, CO, H2S, and SOx are very hazardous in both the outdoor and indoor environment. In contrast to VOC removal, research on the removal of inorganic gas pollutants is very minor. Continuous reactors are considered to be a best choice for the removal of organic compounds in practice. High mechanical strength can be provided by the TiO2 film rather than powders. It is also reported that the functionalized polystyrene or polyamide six fibers can be used for the conversion of NO to NO2 and HNO3 (Szatmáry et al. 2014). Hence, the TiO2 (anatase) are coated on the electron-spun polymeric nanofibers. The TiO2 (pristine) are found to be active only to UV; to make that active in the visible region, oxygen-deficient TiO2 was prepared for the oxidation of NO in the visible region (Wang et al. 2015). The reduction in the bandgap, increased absorption threshold to visible region, and improved electron–hole pair separation rates are studied and confirmed by the first principle density functional theory calculations.
Metal doping can be used in designing enhanced TiO2 for photocatalyst oxidation of inorganic gas contaminants. Hu et al. (2015) reported the removal of NOx can be improved by the synergistic effect of two Pt species together in the catalyst by doping the Pt4+ ions in TiO2 as Pt/TiO2. Ma et al. (2015) has reported that the UV-visible absorption spectra and TiO2 photoluminescence activity can be improved by doping the Fe ions. They have used this method in the visible light photocatalytic oxidation of NOx.
Light absorbance and improved photocatalysis can also be achieved by coupling the TiO2 to another semiconductor. Alonso-Tellez et al. (2014) reported the new method of coupling in the photocatalytic oxidation of H2S. The photocatalyst can also be obtained by applying the surface modification method. Jiao et al. reported the preparation of RuO2/TiO2/Pt (ternary photocatalyst) with the precise particle size of RuO2 at about 2 nm, which can be used to remove CO under UV. They have also reported that the finest RuO2 altered TiO2 has 20 times greater improved activity than P25, 15 times more than anatase, and 8 times than rutile TiO2, correspondingly. For the improvement in PCO removal of CO, the noteworthy exposure of RuO2 (1 1 0) facets are supposed to be the key factor.
9.7.3 Non-Titanium-Based Photocatalyst for Air Purification
Researchers are focusing towards the development of photocatalysts based on TiO2 rather than other novel photocatalytic materials. Great amounts of other novel photocatalysts are also available for the photocatalytic activities of air purification. Zinc oxide is the next best semiconductor photocatalytic candidate. The photocatalytic activity of ZnO is much higher when compared to TiO2 in aqueous phase, but the stability of ZnO might be a problem. It has poor stability, but the stability of ZnO can be improved only at high temperatures. Liao et al. (2013) has examined the characteristics of the modified morphology (six prisms of both short and long pyramid shape) and defect-induced ZnO. The modified ZnO has much higher degradation efficiency than the P25 for formaldehyde removal because the modified ZnO polar planes possess higher surface energy, which will result in increased photocatalytic activity.
Y.C. Chen et al. proposed a ZnO/graphene composite by a green one-pot hydrothermal method for the removal of gaseous acetaldehyde (Liao et al. 2013). The photocatalytic activity can be improved by increasing the transfer rate of electrons by introducing rGO (reduced graphene oxide). The degradation rate and CO2 generation can be drastically improved by controlling the proper rGO content. Sugranez et al. reported that the Fe2O3 can be a perfect candidate for the PCO removal of NOx by retaining HNO2/NO3. The Fe2O3 is a very stable iron oxide that holds n-type semiconductor properties and an energy bandgap of 1.9–2.2 eV.
Bimetal oxides rather than a single metal oxide also can be used as photocatalyst. Huang et al. reported a ZrxTi1xO2 photocatalyst material which can be used for the decomposition of gaseous HCHO (Chen et al. 2015b). For the removal of formaldehyde using PCO compared to the single Bi2SiO5, improved results were observed by using coupled Bi2SiO4 and AgI prepared by an in situ preparation method (Wan and Zhang 2015).
Graphitic carbon nitride has considered as a much desired metal-free photocatalyst candidate for air pollution (Sun and Wang 2014; Sun et al. 2014). Sun et al. (2015) reported that the Ag nanoparticle-deposited g-C3N4 nano sheets could be used for the PCO removal of NOx under visible light radiation. Ag nanoparticles not only improve photocatalytic activity but also lead to the selectivity of final products. Katsumata et al. (2013) proposed g-C3N4/WO3 composites for the photo-degradation of acetaldehyde pollution.
9.7.4 Photocatalysis with Ozone
Photocatalysis in combination with ozone (O3-PCO) has been found to have better efficiency in the destruction of pollutants such as toluene and formaldehyde. To make this method marketable, the stability and efficiency of the same were analyzed and reported by researchers. Haibao Huang in 2010 investigated and reported the mechanism to improve the stability and performance of the O3-PCO process under various operating circumstances. The outcome of the process shows that it has admirable stability during the change of initial O3 concentration and humidity. Compared to the PCO, the O3-PCO has excellent characteristics for eliminating pollutants. Efficiency in removing toluene and formaldehyde in the ozone-PCO process seemed to be increased from 12% to 96% and from 64% to 97.5%, respectively, when compared to that of PCO. Secondary products for this toluene oxidation were also significantly reduced. It is evident from the foregoing discussion that O3-PCO is an effective method to reduce the air pollutants compared to the PCO method.
9.8 Summary
In this chapter, we concluded that the basic properties of a photocatalyst, the photocatalyst materials, and their application for the removal of indoor air pollutants clearly indicate that the photocatalyst is an effective method for environmental cleanliness. It is a potential tool for numerous industrial applications such as wastewater treatment, air pollution, and hydrogen generation to maintain a hygienic environment. Although photocatalysts are considered to be suitable for environment pollution remediation, many improvements are still needed to provide a cheap, highly efficient, stable, nontoxic, and highly active photocatalyst in visible regions.
References
Al-Kahtani AA, Almuqati T, Alhokbany N, Ahamad T, Naushad M, Alshehri SM (2018) A clean approach for the reduction of hazardous 4-nitrophenol using gold nanoparticles decorated multiwalled carbon nanotubes. J Clean Prod 191:429–435
Alonso-Tellez A, Robert D, Keller V, Keller N (2014) H2S photocatalytic oxidation over WO3/TiO2 Hombikat UV100. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-013-2329-y
Al-Othman ZA, Inamuddin, Naushad M (2011) Adsorption thermodynamics of trichloroacetic acid herbicide on polypyrrole Th(IV) phosphate composite cation-exchanger. Chem Eng J 169:38–42. https://doi.org/10.1016/j.cej.2011.02.046
Alshehri SM, Naushad M, Ahamad T et al (2014) Synthesis, characterization of curcumin based ecofriendly antimicrobial bio-adsorbent for the removal of phenol from aqueous medium. Chem Eng J 254:181–189. https://doi.org/10.1016/j.cej.2014.05.100
Andryushina NS, Stroyuk OL (2014) Influence of colloidal graphene oxide on photocatalytic activity of nanocrystalline TiO2 in gas-phase ethanol and benzene oxidation. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2013.11.044
Anpo M (2000) Applications of titanium oxide photocatalysts and unique second-generation TiO2 photocatalysts able to operate under visible light irradiation for the reduction of environmental toxins on a global scale. Stud Surf Sci Catal. https://doi.org/10.1016/S0167-2991(00)80952-0
Antonello A, Soliveri G, Meroni D et al (2014) Photocatalytic remediation of indoor pollution by transparent TiO2 films. Catal Today. https://doi.org/10.1016/j.cattod.2013.12.033
Bahri M, Haghighat F (2014) Plasma-based indoor air cleaning technologies: the state of the art-review. Clean (Weinh) 42(12):1667–1680
Banisharif A, Khodadadi AA, Mortazavi Y et al (2015) Highly active Fe2O3-doped TiO2 photocatalyst for degradation of trichloroethylene in air under UV and visible light irradiation: experimental and computational studies. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2014.10.023
Bavykin DV, Passoni L, Walsh FC (2013) Hierarchical tube-in-tube structures prepared by electrophoretic deposition of nanostructured titanates into a TiO2 nanotube array. Chem Commun. https://doi.org/10.1039/c3cc43264e
Bennett A (2009) Strategies and technologies: controlling indoor air quality. Filtr Sep. https://doi.org/10.1016/S0015-1882(09)70155-7
Bernstein JA, Alexis N, Bacchus H et al (2008) The health effects of non-industrial indoor air pollution. J Allergy Clin Immunol. https://doi.org/10.1016/j.jaci.2007.10.045
Berry D, Mainelis G, Fennell D (2007) Effect of an ionic air cleaner on indoor/outdoor particle ratios in a residential environment. Aerosol Sci Technol 41:315–328
Bianchi CL, Gatto S, Pirola C et al (2014) Photocatalytic degradation of acetone, acetaldehyde and toluene in gas-phase: comparison between nano and micro-sized TiO2. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2013.02.047
Bickley RI, Stone FS (1973) Photoadsorption and photocatalysis at rutile surfaces. I. Photoadsorption of oxygen. J Catal. https://doi.org/10.1016/0021-9517(73)90310-2
Boeniger MF (1995) Use of ozone generating devices to improve indoor air quality. Am Ind Hyg Assoc J. https://doi.org/10.1080/15428119591016827
Calderón MA, Linneberg A, Kleine-Tebbe J et al (2015) Respiratory allergy caused by house dust mites: what do we really know? J Allergy Clin Immunol 136(1):38–48
Cant NW, Cole JR (1992) Photocatalysis of the reaction between ammonia and nitric oxide on TiO2 surfaces. J Catal. https://doi.org/10.1016/0021-9517(92)90231-6
Cao S, Low J, Yu J, Jaroniec M (2015) Polymeric photocatalysts based on graphitic carbon nitride. Adv Mater. https://doi.org/10.1002/adma.201500033
Challoner A, Gill L (2014) Indoor/outdoor air pollution relationships in ten commercial buildings: PM 2.5 and NO2. Build Environ. https://doi.org/10.1016/j.buildenv.2014.05.032
Chávez-Valdez A, Herrmann M, Boccaccini AR (2012) Alternating current electrophoretic deposition (EPD) of TiO2 nanoparticles in aqueous suspensions. J Colloid Interface Sci. https://doi.org/10.1016/j.jcis.2012.02.054
Chen HW, Liang CP, Huang HS et al (2011) Electrophoretic deposition of mesoporous TiO2 nanoparticles consisting of primary anatase nanocrystallites on a plastic substrate for flexible dye-sensitized solar cells. Chem Commun. https://doi.org/10.1039/c1cc12514a
Chen Y, Cao X, Gao B, Lin B (2013) A facile approach to synthesize N-doped and oxygen-deficient TiO2 with high visible-light activity for benzene decomposition. Mater Lett. https://doi.org/10.1016/j.matlet.2012.12.010
Chen K, Zhu L, Yang K (2015a) Tricrystalline TiO2 with enhanced photocatalytic activity and durability for removing volatile organic compounds from indoor air. J Environ Sci (China). https://doi.org/10.1016/j.jes.2014.10.023
Chen YC, Katsumata KI, Chiu YH et al (2015b) ZnO-graphene composites as practical photocatalysts for gaseous acetaldehyde degradation and electrolytic water oxidation. Appl Catal A Gen. https://doi.org/10.1016/j.apcata.2014.10.055
Clements-Croome DJ, Awbi HB, Bakó-Biró Z et al (2008) Ventilation rates in schools. Build Environ. https://doi.org/10.1016/j.buildenv.2006.03.018
Crook B, Burton NC (2010) Indoor moulds, sick building syndrome and building related illness. Fungal Biol Rev 24(3-4):106–113
Dallongeville A, Le Cann P, Zmirou-Navier D et al (2015) Concentration and determinants of molds and allergens in indoor air and house dust of French dwellings. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2015.06.039
Demeestere K, Dewulf J, Van Langenhove H (2007) Heterogeneous photocatalysis as an advanced oxidation process for the abatement of chlorinated, monocyclic aromatic and sulfurous volatile organic compounds in air: state of the art. Crit Rev Environ Sci Technol 37(6):489–538
Dunnill CWH, Aiken ZA, Pratten J et al (2009) Enhanced photocatalytic activity under visible light in N-doped TiO2 thin films produced by APCVD preparations using t-butylamine as a nitrogen source and their potential for antibacterial films. J Photochem Photobiol A Chem. https://doi.org/10.1016/j.jphotochem.2009.07.024
Environmental Protection Agency (2009) Residential air cleaners. A summary of available information, 2nd edn. United States Environmental Protection Agency, Washington, DC. doi:EPA 402-F-09-002
Environmental Protection Agency (2014a) Ground level ozone – health effects. EPA, Washington, DC
Environmental Protection Agency (2014b) Ozone generators that are sold as air cleaners. www.epa.gov
Environmental Protection Agency (2017) Health risk of radon. US EPA, Washington, DC
Esswein EJ, Boeniger MF (1994) Effect of an ozone-generating air-purifying device on reducing concentrations of formaldehyde in air. Appl Occup Environ Hyg. https://doi.org/10.1080/1047322X.1994.10388285
Fujishima A, Honda K (1972) Electrochemical photolysis of water at a semiconductor electrode. Nature (Lond) doi: https://doi.org/10.1038/238037a0
Fujishima A, Rao TN, Tryk DA (2000) TiO2 photocatalysts and diamond electrodes. Electrochim Acta. https://doi.org/10.1016/S0013-4686(00)00620-4
Ge H, Hu D, Li X et al (2015) Removal of low-concentration benzene in indoor air with plasma-MnO2 catalysis system. J Electrost. https://doi.org/10.1016/j.elstat.2015.06.003
Guo H, Murray F, Lee SC (2003) The development of low volatile organic compound emission house: a case study. Build Environ. https://doi.org/10.1016/S0360-1323(03)00156-2
Han Z, Chang VW, Wang X et al (2013) Experimental study on visible-light induced photocatalytic oxidation of gaseous formaldehyde by polyester fiber supported photocatalysts. Chem Eng J. https://doi.org/10.1016/j.cej.2012.12.025
He F, Li J, Li T, Li G (2014) Solvothermal synthesis of mesoporous TiO2: the effect of morphology, size and calcination progress on photocatalytic activity in the degradation of gaseous benzene. Chem Eng J. https://doi.org/10.1016/j.cej.2013.10.028
Hoffmann MR, Martin ST, Choi W, Bahnemann DW (1995) Environmental applications of semiconductor photocatalysis. Chem Rev. https://doi.org/10.1021/cr00033a004
Hou WM, Ku Y (2013) Photocatalytic decomposition of gaseous isopropanol in a tubular optical fiber reactor under periodic UV-LED illumination. J Mol Catal A Chem. https://doi.org/10.1016/j.molcata.2013.03.016
Hu Y, Song X, Jiang S, Wei C (2015) Enhanced photocatalytic activity of Pt-doped TiO2 for NOx oxidation both under UV and visible light irradiation: a synergistic effect of lattice Pt4+ and surface PtO. Chem Eng J. https://doi.org/10.1016/j.cej.2015.03.135
Huang H, Huang H, Zhang L et al (2015) Enhanced degradation of gaseous benzene under vacuum ultraviolet (VUV) irradiation over TiO2 modified by transition metals. Chem Eng J. https://doi.org/10.1016/j.cej.2014.08.057
Hubbard HF, Coleman BK, Sarwar G, Corsi RL (2005) Effects of an ozone-generating air purifier on indoor secondary particles in three residential dwellings. Indoor Air. https://doi.org/10.1111/j.1600-0668.2005.00388.x
Hult EL, Willem H, Price PN et al (2015) Formaldehyde and acetaldehyde exposure mitigation in US residences: in-home measurements of ventilation control and source control. Indoor Air. https://doi.org/10.1111/ina.12160
Hyttinen M, Pasanen P, Kalliokoski P (2006) Removal of ozone on clean, dusty and sooty supply air filters. Atmos Environ. https://doi.org/10.1016/j.atmosenv.2005.09.040
Inoue Y (2009) Photocatalytic water splitting by RuO2-loaded metal oxides and nitrides with d0- and d10-related electronic configurations. Energy Environ Sci. https://doi.org/10.1039/b816677n
Juanru H, Mingwei L, Zhong C (2007) Advances in doping of titania photocatalytic catalysts. Ind Catal 15:1–4
Kale MJ, Avanesian T, Christopher P (2014) Direct photocatalysis by plasmonic nanostructures. ACS Catal 4(1):116–128
Katsumata KI, Motoyoshi R, Matsushita N, Okada K (2013) Preparation of graphitic carbon nitride (g-C3N4)/WO3 composites and enhanced visible-light-driven photodegradation of acetaldehyde gas. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2013.05.058
Kete M, Pavlica E, Fresno F et al (2014) Highly active photocatalytic coatings prepared by a low-temperature method. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-014-3077-3
Khan FI, Ghoshal AK (2000) Removal of volatile organic compounds from polluted air. J Loss Prev Process Ind. https://doi.org/10.1016/S0950-4230(00)00007-3
Koistinen K, Kotzias D, Kephalopoulos S et al (2008) The INDEX project: executive summary of a European Union project on indoor air pollutants. Allergy 63(7):810–819
Kolarik B, Wargocki P, Skorek-Osikowska A, Wisthaler A (2010) The effect of a photocatalytic air purifier on indoor air quality quantified using different measuring methods. Build Environ. https://doi.org/10.1016/j.buildenv.2009.12.006
Krogman KC, Zacharia NS, Grillo DM, Hammond PT (2008) Photocatalytic layer-by-layer coatings for degradation of acutely toxic agents. Chem Mater. https://doi.org/10.1021/cm703096w
Le Bechec M, Kinadjian N, Ollis D et al (2015) Comparison of kinetics of acetone, heptane and toluene photocatalytic mineralization over TiO2 microfibers and Quartzel® mats. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2015.05.015
Lelieveld J, Evans JS, Fnais M et al (2015) The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature. https://doi.org/10.1038/nature15371
Leung DYC (2015) Outdoor-indoor air pollution in urban environment: challenges and opportunity. Front Environ Sci. https://doi.org/10.3389/fenvs.2014.00069
Li Y (2016) The “impurity” of indoor air. Indoor Air 26(1):3–5
Li Y, Hwang DS, Lee NH, Kim SJ (2005) Synthesis and characterization of carbon-doped titania as an artificial solar light sensitive photocatalyst. Chem Phys Lett. https://doi.org/10.1016/j.cplett.2005.01.062
Liao Y, Xie C, Liu Y, Huang Q (2013) Enhancement of photocatalytic property of ZnO for gaseous formaldehyde degradation by modifying morphology and crystal defect. J Alloys Compd. https://doi.org/10.1016/j.jallcom.2012.09.109
Linsebigler AL, Lu GQ, Yates JT (1996) CO photooxidation on TiO2. J Phys Chem. https://doi.org/10.1021/jp952018f
Litter MI (1999) Heterogeneous photocatalysis: transition metal ions in photocatalytic systems. Appl Catal B Environ 23(2-3):89–114
Liu Z, Chen F, Fang P et al (2013) Study of adsorption-assisted photocatalytic oxidation of benzene with TiO2/SiO2 nanocomposites. Appl Catal A Gen. https://doi.org/10.1016/j.apcata.2012.11.020
Lopez L, Daoud WA, Dutta D et al (2013) Effect of substrate on surface morphology and photocatalysis of large-scale TiO2 films. Appl Surf Sci. https://doi.org/10.1016/j.apsusc.2012.10.156
Lyng NL, Gunnarsen L, Andersen HV (2015) The effect of ventilation on the indoor air concentration of PCB: an intervention study. Build Environ. https://doi.org/10.1016/j.buildenv.2015.08.019
Ma CM, Wang W, Ku Y, Jeng FT (2007) Photocatalytic degradation of benzene in air streams in an optical fiber photoreactor. Chem Eng Technol. https://doi.org/10.1002/ceat.200700138
Ma J, He H, Liu F (2015) Effect of Fe on the photocatalytic removal of NOx over visible light responsive Fe/TiO2 catalysts. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2015.05.003
Machado LCR, Torchia CB, Lago RM (2006) Floating photocatalysts based on TiO2 supported on high surface area exfoliated vermiculite for water decontamination. Catal Commun. https://doi.org/10.1016/j.catcom.2005.10.020
Madureira J, Paciência I, Rufo JC et al (2015) Assessment and determinants of airborne bacterial and fungal concentrations in different indoor environments: homes, child day-care centres, primary schools and elderly care centres. Atmos Environ. https://doi.org/10.1016/j.atmosenv.2015.03.026
Martínez Vargas DX, Rivera De la Rosa J, Lucio-Ortiz CJ et al (2015) Photocatalytic degradation of trichloroethylene in a continuous annular reactor using Cu-doped TiO2 catalysts by sol-gel synthesis. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2015.05.019
Monteiro RAR, Miranda SM, Rodrigues-Silva C et al (2015) Gas phase oxidation of n-decane and PCE by photocatalysis using an annular photoreactor packed with a monolithic catalytic bed coated with P25 and PC500. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2014.10.026
Murcia JJ, Hidalgo MC, Navío JA et al (2013) Cyclohexane photocatalytic oxidation on Pt/TiO2 catalysts. Catal Today. https://doi.org/10.1016/j.cattod.2012.11.018
Nakajima A, Akiyama Y, Yanagida S et al (2009) Preparation and properties of Cu-grafted transparent TiO2-nanosheet thin films. Mater Lett. https://doi.org/10.1016/j.matlet.2009.05.016
Nakata K, Fujishima A (2012) TiO2 photocatalysis: design and applications. J Photochem Photobiol C Photochem Rev. https://doi.org/10.1016/j.jphotochemrev.2012.06.001
Naushad M (2014) Surfactant assisted nano-composite cation exchanger: development, characterization and applications for the removal of toxic Pb2+ from aqueous medium. Chem Eng J 235:100–108. https://doi.org/10.1016/j.cej.2013.09.013
Nazaroff WW (2013) Four principles for achieving good indoor air quality. Indoor Air 23(5):353–356
Nelson HS, Hirsch SR, Ohman JL et al (1988) Recommendations for the use of residential air-cleaning devices in the treatment of allergic respiratory diseases. J Allergy Clin Immunol. https://doi.org/10.1016/0091-6749(88)90980-3
Niu JL, Tung TCW, Burnett J (2001) Quantification of dust removal and ozone emission of ionizer air-cleaners by chamber testing. J Electrost. https://doi.org/10.1016/S0304-3886(01)00118-8
Nizard H, Kosinova ML, Fainer NI et al (2008) Deposition of titanium dioxide from TTIP by plasma enhanced and remote plasma enhanced chemical vapor deposition. Surf Coat Technol. https://doi.org/10.1016/j.surfcoat.2008.02.023
Nolan MG, Pemble ME, Sheel DW, Yates HM (2006) One step process for chemical vapour deposition of titanium dioxide thin films incorporating controlled structure nanoparticles. Thin Solid Films. https://doi.org/10.1016/j.tsf.2006.07.182
Norhidayah A, Chia-Kuang L, Azhar MK, Nurulwahida S (2013) Indoor air quality and sick building syndrome in three selected buildings. Procedia Eng. https://doi.org/10.1016/j.proeng.2013.02.014
Ou HH, Lo SL (2007) Photocatalysis of gaseous trichloroethylene (TCE) over TiO2: the effect of oxygen and relative humidity on the generation of dichloroacetyl chloride (DCAC) and phosgene. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2006.12.039
Park SE, Joo H, Kang JW (2004) Effect of impurities in TiO2 thin films on trichloroethylene conversion. Sol Energy Mater Sol Cells. https://doi.org/10.1016/j.solmat.2004.02.012
Paunović K, Maksimović M, Davidović D et al (2005) Max Josef von Pettenkofer – founder of modern hygiene (1818–1901). Srp Arh Celok Lek 133(9-10):450–453
Pham TD, Lee BK (2015) Novel adsorption and photocatalytic oxidation for removal of gaseous toluene by V-doped TiO2/PU under visible light. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2015.07.048
Pierce WM, Janczewski JN, Roethlisberger B et al (1996) Effectiveness of auxiliary air cleaners in reducing ETS components in offices. ASHRAE J 38:51–57
Poppendieck DG, Rim D, Persily AK (2014) Ultrafine particle removal and ozone generation by in-duct electrostatic precipitators. Environ Sci Technol. https://doi.org/10.1021/es404884p
Priya DN, Modak JM, Raichur AM (2009) LbL fabricated poly(styrene sulfonate)/TiO2 multilayer thin films for environmental applications. ACS Appl Mater Interfaces. https://doi.org/10.1021/am900566n
Raddaha NS, Cordero-Arias L, Cabanas-Polo S et al (2014) Electrophoretic deposition of chitosan/h-BN and chitosan/h-BN/TiO2 composite coatings on stainless steel (316L) substrates. Materials (Basel). https://doi.org/10.3390/ma7031814
Ren L, Li Y, Hou J et al (2016) The pivotal effect of the interaction between reactant and anatase TiO2 nanosheets with exposed {001} facets on photocatalysis for the photocatalytic purification of VOCs. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2015.08.034
Rivas I, Viana M, Moreno T et al (2014) Child exposure to indoor and outdoor air pollutants in schools in Barcelona. Spain Environ Int. https://doi.org/10.1016/j.envint.2014.04.009
Robert D, Malato S (2002) Solar photocatalysis: a clean process for water detoxification. Sci Total Environ. https://doi.org/10.1016/S0048-9697(01)01094-4
Robert D, Keller V, Keller N (2013) Immobilization of a semiconductor photocatalyst on solid supports: methods, materials, and applications. In: Photocatalysis and water purification: from fundamentals to recent applications. Wiley-VCH, Weinheim
Rohra H, Taneja A (2016) Indoor air quality scenario in India—an outline of household fuel combustion. Atmos Environ 129:243–255
Sampaio MJ, Silva CG, Silva AMT et al (2013) Photocatalytic activity of TiO2-coated glass raschig rings on the degradation of phenolic derivatives under simulated solar light irradiation. Chem Eng J. https://doi.org/10.1016/j.cej.2012.11.027
Schleibinger H, Rüden H (1999) Air filters from HVAC systems as possible source of volatile organic compounds (VOC): laboratory and field assays. Atmos Environ. https://doi.org/10.1016/S1352-2310(99)00274-5
Serpone N, Emeline AV (2012) Semiconductor photocatalysis – past, present, and future outlook. J Phys Chem Lett 3(5):673–677
Shan AY, Ghazi TIM, Rashid SA (2010) Immobilisation of titanium dioxide onto supporting materials in heterogeneous photocatalysis: a review. Appl Catal A Gen. https://doi.org/10.1016/j.apcata.2010.08.053
Shaughnessy RJ, Levetin E, Blocker J, Sublette KL (1994) Effectiveness of portable indoor air cleaners: sensory testing results. Indoor Air. https://doi.org/10.1111/j.1600-0668.1994.t01-1-00006.x
Shen YS, Ku Y (2002) Decomposition of gas-phase trichloroethene by the UV/TiO2 process in the presence of ozone. Chemosphere. https://doi.org/10.1016/S0045-6535(00)00585-3
Smyth J (2010) The TiO2 Group. University of Colorado. USA
Spanhel L, Weller H, Henglein A (1987) Photochemistry of semiconductor colloids. 22. Electron ejection from illuminated cadmium sulfide into attached titanium and zinc oxide particles. J Am Chem Soc. https://doi.org/10.1021/ja00256a012
Stranger M, Potgieter-Vermaak SS, Van Grieken R (2009) Particulate matter and gaseous pollutants in residences in Antwerp, Belgium. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2008.10.019
Sun H, Wang S (2014) Research advances in the synthesis of nanocarbon-based photocatalysts and their applications for photocatalytic conversion of carbon dioxide to hydrocarbon fuels. Energy Fuel 28(1):22–36
Sun H, Wang C, Pang S et al (2008) Photocatalytic TiO2 films prepared by chemical vapor deposition at atmosphere pressure. J Non-Cryst Solids. https://doi.org/10.1016/j.jnoncrysol.2007.01.108
Sun H, Wang S, Ang HM et al (2010) Halogen element modified titanium dioxide for visible light photocatalysis. Chem Eng J 162(2):437–447
Sun H, Zhou G, Wang Y et al (2014) A new metal-free carbon hybrid for enhanced photocatalysis. ACS Appl Mater Interfaces 6:16745–16754. https://doi.org/10.1021/am503820h
Sun Y, Xiong T, Ni Z et al (2015) Improving g-C3N4 photocatalysis for NOx removal by Ag nanoparticles decoration. Appl Surf Sci. https://doi.org/10.1016/j.apsusc.2015.07.071
Szatmáry L, Šubrt J, Kalousek V et al (2014) Low-temperature deposition of anatase on nanofiber materials for photocatalytic NOx removal. Catal Today. https://doi.org/10.1016/j.cattod.2013.09.023
Taskinen T, Meklin T, Nousiainen M et al (1997) Moisture and mould problems in schools and respiratory manifestations in schoolchildren: clinical and skin test findings. Acta Paediatr. https://doi.org/10.1111/j.1651-2227.1997.tb14841.x
Technical, Report Efficiency and REC House (EREC) (2008) Spot ventilation: Source control to improve indoor air quality
Tejasvi R, Sharma M, Upadhyay K (2015) Passive photo-catalytic destruction of air-borne VOCs in high traffic areas using TiO2-coated flexible PVC sheet. Chem Eng J. https://doi.org/10.1016/j.cej.2014.10.040
Tennakone K, Tilakaratne CTK, Kottegoda IRM (1995) Photocatalytic degradation of organic contaminants in water with TiO2 supported on polythene films. J Photochem Photobiol A Chem. https://doi.org/10.1016/1010-6030(94)03980-9
Thevenet F, Guillard C, Rousseau A (2014) Acetylene photocatalytic oxidation using continuous flow reactor: gas phase and adsorbed phase investigation, assessment of the photocatalyst deactivation. Chem Eng J. https://doi.org/10.1016/j.cej.2014.01.038
US Department of Health and Human Services (2006) Centers for Disease Control and Prevention. In: Healthy housing reference manual. US Department of Health and Human Services, Washington, DC
USEPA (2004a) Air quality criteria for particulate matter October 2004, volume 1. EPA, Washington, DC
USEPA (2004b) Air quality criteria for particulate matter October 2004, volume 2. EPA, Washington, DC
Van Durme J, Dewulf J, Leys C (2008) Development of heterogeneous plasma catalysis for the abatement of health damaging organic micropollutants in indoor environments, UGent. Faculty of Bioscience Engineering
Verriele M, Schoemaecker C, Hanoune B et al (2016) The MERMAID study: indoor and outdoor average pollutant concentrations in 10 low-energy school buildings in France. Indoor Air. https://doi.org/10.1111/ina.12258
Vinu R, Madras G (2010) Environmental remediation by photocatalysis. J Indian Inst Sci 90(2):189–230
Wan Z, Zhang G (2015) Synthesis and facet-dependent enhanced photocatalytic activity of Bi2 SiO5/AgI nanoplate photocatalysts. J Mater Chem A. https://doi.org/10.1039/C5TA03465E
Wang S, Ang HM, Tade MO (2007) Volatile organic compounds in indoor environment and photocatalytic oxidation: state of the art. Environ Int 33(5):694–705
Wang B, Mortazavi R, Haghighat F (2009) Evaluation of modeling and measurement techniques of ultraviolet germicidal irradiation effectiveness – towards the design of immune buildings. Indoor Built Environ. https://doi.org/10.1177/1420326X09103024
Wang C, Ma J, Liu F et al (2015) The effects of Mn2+ precursors on the structure and ozone decomposition activity of cryptomelane-type manganese oxide (OMS-2) catalysts. J Phys Chem C. https://doi.org/10.1021/acs.jpcc.5b08095
Waring MS, Siegel JA, Corsi RL (2008) Ultrafine particle removal and generation by portable air cleaners. Atmos Environ. https://doi.org/10.1016/j.atmosenv.2008.02.011
Weschler CJ, Shields HC (1996) Production of the hydroxyl radical in indoor air. Environ Sci Technol. https://doi.org/10.1021/es960032f
Weschler CJ, Hodgson AT, Wooley JD (1992a) Indoor chemistry: ozone, volatile organic compounds, and carpets. Environ Sci Technol. https://doi.org/10.1021/es00036a006
Weschler CJ, Michael B, Petros K (1992b) Indoor ozone and nitrogen dioxide: a potential pathway to the generation of nitrate radicals, dinitrogen pentoxide, and nitric acid indoors. Environ Sci Technol. https://doi.org/10.1021/es00025a022
WHO (2000) Air quality guidelines for Europe. Environ Sci Pollut Res. https://doi.org/10.1007/BF02986808
WHO (2014) Indoor air quality guidelines: household fuel combustion. World Health Organization. isbn:9789241548878
Wolkoff P, Nielsen GD (2001) Organic compounds in indoor air—their relevance for perceived indoor air quality? Atmos Environ 35(26):4407–4417
Xie H, Liu B, Zhao X (2016) Facile process to greatly improve the photocatalytic activity of the TiO2 thin film on window glass for the photodegradation of acetone and benzene. Chem Eng J. https://doi.org/10.1016/j.cej.2015.09.049
Yu JC, Lin J, Lo D, Lam SK (2000) Influence of thermal treatment on the adsorption of oxygen and photocatalytic activity of TiO2. Langmuir. https://doi.org/10.1021/la000309w
Yu BF, Hu ZB, Liu M et al (2009) Review of research on air-conditioning systems and indoor air quality control for human health. Int J Refrig 32(1):3–20
Zhang J, Smith KR (2003) Indoor air pollution: a global health concern. Br Med Bull 68:209–225
Zhang Y, Mo J, Li Y et al (2011) Can commonly-used fan-driven air cleaning technologies improve indoor air quality? A literature review. Atmos Environ 45(26):4329–4343
Zhang P, Liu L, He Y et al (2015) One-dimensional angular surface plasmon resonance imaging based array thermometer. Sens Actuators B Chem. https://doi.org/10.1016/j.snb.2014.10.055
Zhao J, Yang X (2003) Photocatalytic oxidation for indoor air purification: a literature review. Build Environ. https://doi.org/10.1016/S0360-1323(02)00212-3
Zhong L, Haghighat F (2015) Photocatalytic air cleaners and materials technologies – abilities and limitations. Build Environ. https://doi.org/10.1016/j.buildenv.2015.01.033
Zhuang H, Gu Q, Long J et al (2014) Visible light-driven decomposition of gaseous benzene on robust Sn2+-doped anatase TiO2 nanoparticles. RSC Adv. https://doi.org/10.1039/c4ra05904b
(2014) 7 million deaths annually linked to air pollution. Cent Eur J Public Health
(2005) WHO. Air quality guidelines for particulate matter, ozone, nitrogen dioxide and sulfur dioxide. Technical report, World Health Organization
(2014) US Environmental Protection Agency. National ambient air quality standards (NAAQS)
(2015) European Commission. Air quality standards
(2010) WHO Regional Office for Europe. Guidelines for indoor air quality. Technical report
(2016) The US Environmental Protection Agency Website. Exposure to radon causes lung cancer in non-smokers and smokers alike
(2007) United States Environmental Protection Agency. Benzo(a)pyrene (BaP). TEACH Chemical Summary 1:1–14
Acknowledgments
The authors (D. Shanmuga Sundar and Marcos Flores Carrasco) wish to thank the Conicyt FONDECYT (Fondo Nacional de Desarrollo Científico y Tecnológico) Project No. 3180089 and Millennium Nucleus MULTIMAT for funding and support.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Dhanabalan, S.S., Avaninathan, S.R., Rajendran, S., Carrasco, M.F. (2020). Photocatalysts for Indoor Air Pollution: A Brief Review. In: Rajendran, S., Naushad, M., Ponce, L., Lichtfouse, E. (eds) Green Photocatalysts for Energy and Environmental Process. Environmental Chemistry for a Sustainable World, vol 36. Springer, Cham. https://doi.org/10.1007/978-3-030-17638-9_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-17638-9_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-17637-2
Online ISBN: 978-3-030-17638-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)