Abstract
Aneurysm treatment with clipping or coiling is indicated to prevent aneurysm re-rupture after aneurysmal subarachnoid hemorrhage (SAH). Any form of benefit of treatment requires active treatment to improve on natural history of the aneurysm. Primarily, morbidity and mortality of aneurysm treatment need to be lower than morbidity from re-rupture. For ruptured aneurysms, the risk of re-rupture within 6 months was reported at 40% for patients of all ages with a mortality of 78% [1]; and not less for elderly patients. Next to effects of initial bleeding, historical materials cite re-bleeding and surgical complications as main determinants of a bad outcome [2]. For subarachnoid hemorrhage, high age appears to increase treatment risk [3]. The highest mortality was seen in poor-grade patients over 75 who, however, were treated conservatively [4]. In addition, the expected remaining life-time for the age must be considered to assess potential benefit of preventing re-rupture, since competing risks are higher at a higher age. In contrast, several studies report favorable outcome after more aggressive management of aneurysms in an elderly population [5, 6].
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Aneurysm treatment with clipping or coiling is indicated to prevent aneurysm re-rupture after aneurysmal subarachnoid hemorrhage (SAH). Any form of benefit of treatment requires active treatment to improve on natural history of the aneurysm. Primarily, morbidity and mortality of aneurysm treatment need to be lower than morbidity from re-rupture. For ruptured aneurysms, the risk of re-rupture within 6 months was reported at 40% for patients of all ages with a mortality of 78% [1]; and not less for elderly patients. Next to effects of initial bleeding, historical materials cite re-bleeding and surgical complications as main determinants of a bad outcome [2]. For subarachnoid hemorrhage, high age appears to increase treatment risk [3]. The highest mortality was seen in poor-grade patients over 75 who, however, were treated conservatively [4]. In addition, the expected remaining life-time for the age must be considered to assess potential benefit of preventing re-rupture, since competing risks are higher at a higher age. In contrast, several studies report favorable outcome after more aggressive management of aneurysms in an elderly population [5, 6].
Hence, precise information of how age affects outcome of aneurysm treatment would be necessary for practical decision-making. The rationale for treatment is unclear in advanced age, since a limited expected life-span affects long-term benefit of treatment and advanced age with potential health problems increase complications. This review was made to investigate published information on outcomes of treatment in elderly patients.
We were seeking information on outcomes of treatment in elderly patients with the primary intent to find data to support microsurgical or endovascular strategies in elderly patients.
Methods
A Pub Med Search was made on 15th September, 2018 with the search terms” elderly patients”, “age”, “Intracranial aneurysm”, “clipping”, “coiling”. English literature was searched and reference lists of selected papers were reviewed for additional articles. We retrieved 286 abstracts, that were screened for contents. In total, 31 articles were selected and after reading, 17 articles were deemed relevant and used for further analyses. The articles were used to extract data for meta-analytical investigation.
A random-effects model, which acknowledges the existence of different effects sizes underlying different studies, was used in this analysis. We adopted the “restricted maximum likelihood”-estimator in the random-effects model based on meta-analytic studies comparing bias and efficiency of meta- analytic variance estimators in random-effects models [7, 8].
I2 quantifies the proportion of variance in study effect estimates, which is attributable to heterogeneity rather than chance. Thus, an I2-value of 0% correlates with no inconsistency between studies.
Heterogeneity was quantified accordingly to Higgins et al., with “low”, “moderate” and “high” corresponding to I2-values of 25%, 50% and 75% [9]. The p-value for χ2-test was computed to determine whether significant heterogeneity existed.
Statistical analyses were performed in R-Studio. This meta-analysis and its graphical content were made by using the “metafor”-package [10].
The primary outcome was: first, to establish the 1-year survival after treatment with either endovascular treatment or microsurgical clipping; and second, to quantify the proportion of patients achieving a favorable outcome after treated with either endovascular coiling or microsurgical clipping. A favorable outcome was defined in alignment with the vast majority of definitions applied within the individual studies, which comprised either a Glasgow Outcome Scale (GOS) equal to “good recovery (GR): none or minor physical or mental deficits that affects daily life” or “moderate disability (MD): independent, but cannot resume work/school or all previous activities” or a modified Rankin Scale (mRS) equal to “0: no symptoms”, “1: no significant disability, despite symptoms; able to perform all usual duties and activities” and “2: slight disability; unable to perform all previous activities but able to look after own affairs without assistance”.
We sought to include a set of covariates for a meta-regression analysis to explore this heterogenous group of patients and how these may have affected outcome. However, the only consistent covariates were mean age and the proportion of “poor prognosis” patients—although, different assessment schemes were used for determining poor prognosis; in alignment with the majority of the included studies, we defined “poor prognosis” as a NIS-SAH Severity Score greater than 7, a World Federation of Neurosurgical Societies (WFNS)-score of 4–5 or, a Hunt-Hess grade equal to 4 or 5. We calculated the fraction of “poor prognosis” patients per total cohort as surrogate marker for the baseline severity of the included patients.
We grouped data in decades based on the age mean for the specific cohort. Age groups were compared corresponding to a mean age between 60 and 69 year, 70 and 79 year, 80 and 89 year or older than 90 year and across all age groups.
Results
A total of 31 articles were read of which 17 were eligible for quantitative synthesis, comprising 3998 patients treated with endovascular coiling whereas 2461 patients underwent microsurgical clipping—see Table 5.1 for further information; the age distribution in studies addressing endovascular coiling (n = 15), two studies were allocated in the age group between 60 and 69 year [11, 26], 11 studies were allocated in the age group between 70 and 79 year [12,13,14,15,16,17,18,19,20,21,22], and one study was allocated in the age group between 80 and 89 year [25]. One study did not report the mean age [27]; whereas the age distribution in studies addressing microsurgical clipping (n = 8), four studies were allocated in the age group between 70 and 79 year [17,18,19, 23] and three studies were allocated in the age group between 80 and 89 year [23,24,25]. The last study did not report a mean age but comprised a range between 70 and 82 year [16].
Favorable Outcome
A random-effects model was used to produce a weighted proportion of patients achieving favorable outcome in each treatment (Fig. 5.1a, b). In total, 55% (95% CI: 45%; 65%) across all age groups achieved a favorable outcome after treatment with endovascular coiling (Fig. 5.1a); similarly, 56% (95% CI: 52%; 59%) of patients achieved favorable outcome after treatment with microsurgical clipping (Fig. 5.1b). Notably, the overall I2-percentage was 87.2% and considered high. The χ2-p-value for all studies combined was 0, indicating that highly significant heterogeneity was observed in the analysis of endovascular treatment. In quite contrast, the I2-percentage and χ2-p-value was 12.2% and 0.38, respectively, indicating low, non-significant heterogeneity, hence between-study consistency.
Figure 5.1c depicts a Funnel Plot (the proportion of patients achieving a favorable outcome in each individual study plotted against the standard error (an index of precision). The white funnel illustrate 95% confidence band corresponding to each standard error) of the random-effects model used for analysis of favorable outcome after endovascular coiling. The studies are spread out an only poorly contained within the funnel—which give arise to the large heterogeneity observed. It demonstrates the complexity and difficulty in encapsulating this patient group due to considerable differences in e.g. baseline patient characteristics or selective cohorts used for different studies.
One-Year Mortality
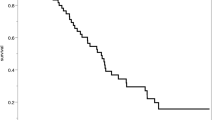
Similarly, we used a random-effects model to determine the weighted proportion of patients being alive 1-year after treatment (Fig. 5.2a–d). Both models were associated with very high and significant heterogeneity at I2-percentages of 98.8% and 95.7% for the endovascular coiling and microsurgical clipping, respectively, and χ2-p-values of 0.
(a) A Forest Plot illustrating 1-year survival after endovascular coiling. (b) A Forest Plot illustrating 1-year survival after microsurgical clipping. (c) A Funnel Plot computed based on the random-effects model used in (a); determining the proportion of patients being alive 1-year after endovascular treated patients. (d) A funnel plot computed based on the random-effects model used in (a); determining the proportion of patients being alive 1-year after microsurgical treated patients
The proportion being alive after 1-year was 67% and 59% for endovascular (Fig. 5.2a) and microsurgical (Fig. 5.2b) treatment, respectively. Surprisingly, in both models the one study that comprised the (80–89 year)-age group suggest a better 1-year prognosis in this group compared to the (70–79 year)-age group [25]. Funnel Plots were computed for both models, and greatly visualizes the how scattered the studies are due to heterogeneous study groups.
A meta-regression including the proportion of baseline “poor grade” per total cohort did not significantly intercept, meaning that baseline surrogate marker for “poor grade” could not be demonstrated to alter the 1-year survival outcome.
Level of Evidence
All studies represent retrospective reviews of cohorts, where patients were offered treatment at the discretion of physicians in charge or one represent a selected subgroup of patients at equipoise regarding better benefit of clipping or coiling. The quality per GRADE was considered very low, since selection bias regarding expected benefit was the basis of treatment allocation.
Discussion
Favorable outcomes were reported after clipping in 56% (95% CI: 52–59%) and after coiling in 55% (95% CI: 45–65%) after aneurysmal subarachnoid hemorrhages. In total, 1-year survival after clipping was 59% (95% CI: 44–75%) and after coiling 67% (95% CI: 52–82%). One study indicated higher mortality in patients aged 80–89, but a very high heterogeneity did not allow identification of a coherent pattern; we found a low heterogeneity only in the analysis of favorable outcome after clipping. The studies showed no difference between different age cohorts. Taken together, the studies indicate that patients offered treatments with either clip or coil for aneurysmal SAH were more likely to experience a favorable outcome than the opposite, although morbidity was high and can be expected to increase with age, although available studies do not allow a direct comparison of the results of the different treatments and conservative management. The patients that are already offered treatment will probably continue to be treated, since meta-analyses did not indicate any unexpectedly bad results from active treatment. The results can only be understood to show that personal knowledge and individual decision making was used for management of the patients and the coarse comparisons of quantitatively synthesized data on the meta-level failed to provide new insights because the included studies reflected treatment of heterogenous, highly selected patients. It is probable that the decision- making physicians had knowledge to offer clipping or coiling to patients they expected to benefit from active treatment from previous experience; these explicit parameters were not revealed in the analyzed studies. Hence, an algorithmic approach that is a prerequisite for meaningful meta-analyses was not identified in any included study and, subsequently, the results provided no information for an age- related algorithm for aneurysm-management.
We could thus summarize outcomes in the treated cohorts where treatment was offered per best knowledge and experience; whether the outcomes are desirable is a matter of values and evaluation from experience of what an expected alternative outcome with different or conservative treatment would have been. Neither mortality nor favorable outcome was compared to valid controls.
There is no obvious reason why age groups should be divided into decades. This was chosen in alignment with the included studies to apply some categorization suitable for statistical analysis. Age as a solitary criterion for treatment allocation seem arbitrary as a 60-year old with a high comorbidity index and poor performance status may have a significant shorter life expectancy contrarily to a healthy 90-year old with a good performance status. Further, we categorized the vast majority of studies based on the reported mean age, e.g. a mean of 75 year to the (70–79 year)-age group, although the age ranged between the seventh and ninth decade of life; the use of a mean age without supporting standard deviations is a major flaw, which was not possible to implement.
We believe that our finding of an absent age-relation reflects strict selection of patients with favorable prognoses; probably more so in the oldest patients. Other observations suggest that age is a relevant prognostic factor. One study on unruptured aneurysms [28] and two registry studies on hospital discharge cohorts [29, 30] indicated a higher mortality by a factor of 1.4 in patient over 65 compared to those younger, and morbidity appears to increase with age [3]. Not surprisingly, we conclude that age is probably related to worse outcomes for clipping and coiling after aSAH. Still, the available literature showed that selected patients appear to do well with the treatments offered. Age in relation to management of intracranial aneurysms is complex, and individual decisions cannot be determined by findings in larger groups unless findings are unequivocal and can be known to apply to the individual patient. The available articles fail to provide such information. The articles either compare outcomes of older to younger patients and conclude that outcomes in older patients are worse than in younger.
However, there is no comparison to natural history, hence we rely on historical knowledge of natural history of ruptured and unruptured aneurysms. For the former, expected risk of death within a year of hemorrhage is sufficiently high to warrant coiling or clipping if this can be achieved at surgical mortality below 10–20%.
Conclusion
Today, the individual decisions to offer treatment reflect individual experience and expertise. Treatments will need to continue to be based on individual decision-making by experts and it is probably more worthwhile to collect treatment data in registries to analyze treatments to improve gradually, than to expect “high quality” information from prospective randomized trials: surgical decision-making handles a multitude of parameters other than age and it is not probable than a RCT can meaningfully control for these sufficiently to tailor individual algorithms for therapy.
Outcomes reflect populations with treatment selected already based on practical knowledge of individual risk and benefit. Hence, comparison between clipping and coiling was not relevant, while all studies showed that a substantial proportion of patients can be treated with limited morbidity and that morbidity appears to be lower than would be expected without treatment.
Box
What is known?
More than 40% of aSAH patients will suffer a re-hemorrhage within 6 months and up to 80% of them will die. Re-bleeding is an important potential cause of death in aSAH patients whose aneurysms remain unsecured which may be a proportionally higher risk for elderly patients.
What is new?
The literature search and analyses of articles did not reveal any relevant novel information, apart from an indication that a meta-analysis for the research-question may be futile. It was due to the complexity—manifested as large analytical heterogeneity—derived from competing risks such as comorbidities and other severe illness, different and inconsistent usage of assessment schemes, improved and advancements within the applied treatment techniques throughout the different study periods and patient inclusion and different primary study objectives yielding subsets of selective cohorts that may not be comparable.
What are the consequences for clinical practice?
Age should not be a solitary determinant of treatment allocation. Clinical practice should continue to comprise surgery or endovascular treatment of aneurysms in selected patients based on expert knowledge, multidisciplinary interaction and specialized patient assessment while long-term data can be gathered with use of registries.
References
Nishioka H, Torner JC, Graf CJ, Kassell NF, Sahs AL, Goettler LC. Cooperative study of intracranial aneurysms and subarachnoid hemorrhage: a long-term prognostic study. II. Ruptured intracranial aneurysms managed conservatively. Arch Neurol. 1984;41:1142–6.
Saveland H, Brandt L. Which are the major determinants for outcome in aneurysmal subarachnoid hemorrhage? A prospective total management study from a strictly unselected series. Acta Neurol Scand. 1994;90:245–50.
Goldberg J, Schoeni D, Mordasini P, Z’Graggen W, Gralla J, Raabe A, et al. Survival and outcome after poor-grade aneurysmal subarachnoid hemorrhage in elderly patients. Stroke. 2018;49:2883–9. https://doi.org/10.1161/STROKEAHA.118.022869.
Nieuwkamp DJ, Rinkel GJE, Silva R, Greebe P, Schokking DA, Ferro JM. Subarachnoid haemorrhage in patients > or = 75 years: clinical course, treatment and outcome. J Neurol Neurosurg Psychiatry. 2006;77:933–7. https://doi.org/10.1136/jnnp.2005.084350.
Johansson M, Cesarini KG, Contant CF, Persson L, Enblad P. Changes in intervention and outcome in elderly patients with subarachnoid hemorrhage. Stroke. 2001;32:2845–949.
Suzuki Y, Watanabe A, Wakui K, Horiuchi T, Hongo K. Results of clipping surgery for aneurysmal subarachnoid hemorrhage in elderly patients aged 90 or older. Acta Neurochir Suppl. 2016;123:13–6. https://doi.org/10.1007/978-3-319-29887-0_2.
Thompson SG, Sharp SJ. Explaining heterogeneity in meta-analysis: a comparison of methods. Stat Med. 1999;18:2693–708.
Viechtbauer W. Bias and efficiency of meta-analytic variance estimators in the random- effects model. J Educ Behav Stat. 2005;30:261–93. https://doi.org/10.3102/10769986030003261.
Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60. https://doi.org/10.1136/bmj.327.7414.557.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36:1–48.
Duan G, Yang P, Li Q, Zuo Q, Zhang L, Hong B, et al. Prognosis predicting score for endovascular treatment of aneurysmal subarachnoid hemorrhage: a risk modeling study for individual elderly patients. Medicine (Baltimore). 2016;95:e2686. https://doi.org/10.1097/MD.0000000000002686.
Sedat J, Dib M, Lonjon M, Litrico S, Von Langsdorf D, Fontaine D, et al. Endovascular treatment of ruptured intracranial aneurysms in patients aged 65 years and older: follow-up of 52 patients after 1 year. Stroke. 2002;33:2620–5.
Johansson M, Norback O, Gal G, Cesarini KG, Tovi M, Solander S, et al. Clinical outcome after endovascular coil embolization in elderly patients with subarachnoid hemorrhage. Neuroradiology. 2004;46:385–91. https://doi.org/10.1007/s00234-004-1170-x.
Watanabe D, Hashimoto T, Koyama S, Ohashi HT, Okada H, Ichimasu N, et al. Endovascular treatment of ruptured intracranial aneurysms in patients 70 years of age and older. Surg Neurol Int. 2014;5:104. https://doi.org/10.4103/2152-7806.136090.
Gu D-Q, Zhang X, Luo B, Long X-A, Duan C-Z. Impact of ultra-early coiling on clinical outcome after aneurysmal subarachnoid hemorrhage in elderly patients. Acad Radiol. 2012;19:3–7. https://doi.org/10.1016/j.acra.2011.09.012.
Karamanakos PN, Koivisto T, Vanninen R, Khallaf M, Ronkainen A, Parviainen I, et al. The impact of endovascular management on the outcome of aneurysmal subarachnoid hemorrhage in the elderly in eastern Finland. Acta Neurochir. 2010;152:1493–502. https://doi.org/10.1007/s00701-010-0714-6.
Bekelis K, Gottlieb D, Su Y, O’Malley AJ, Labropoulos N, Goodney P, et al. Surgical clipping versus endovascular coiling for elderly patients presenting with subarachnoid hemorrhage. J Neurointerv Surg. 2016;8:913–8. https://doi.org/10.1136/neurintsurg-2015-011890.
Proust F, Gerardin E, Derrey S, Lesveque S, Ramos S, Langlois O, et al. Interdisciplinary treatment of ruptured cerebral aneurysms in elderly patients. J Neurosurg. 2010;112:1200–7. https://doi.org/10.3171/2009.10.JNS08754.
Park J, Woo H, Kang D-H, Kim Y. Critical age affecting 1-year functional outcome in elderly patients aged >/= 70 years with aneurysmal subarachnoid hemorrhage. Acta Neurochir. 2014;156:1655–61. https://doi.org/10.1007/s00701-014-2133-6.
Iosif C, Di Maria F, Sourour N, Degos V, Bonneville F, Biondi A, et al. Is a high initial World Federation of Neurosurgery (WFNS) grade really associated with a poor clinical outcome in elderly patients with ruptured intracranial aneurysms treated with coiling? J Neurointerv Surg. 2014;6:286–90. https://doi.org/10.1136/neurintsurg-2013-010711.
Jain R, Deveikis J, Thompson BG. Endovascular management of poor-grade aneurysmal subarachnoid hemorrhage in the geriatric population. AJNR Am J Neuroradiol. 2004;25:596–600.
Luo C-B, Teng MM-H, Chang F-C, Chang C-Y. Endovascular embolization of ruptured cerebral aneurysms in patients older than 70 years. J Clin Neurosci. 2007;14:127–32. https://doi.org/10.1016/j.jocn.2006.05.018.
Horiuchi T, Tanaka Y, Hongo K. Surgical treatment for aneurysmal subarachnoid hemorrhage in the 8th and 9th decades of life. Neurosurgery. 2005;56:469–75.
Horiuchi T, Tsutsumi K, Ito K, Hongo K. Results of clipping surgery for aneurysmal subarachnoid hemorrhage in the ninth and tenth decades of life. J Clin Neurosci. 2014;21:1567–9. https://doi.org/10.1016/j.jocn.2013.11.047.
Dasenbrock H, Gormley WB, Lee Y, Mor V, Mitchell SL, Fehnel CR. Long-term outcomes among octogenarians with aneurysmal subarachnoid hemorrhage. J Neurosurg. 2018;1:1–12. https://doi.org/10.3171/2018.3.JNS173057.
Zheng K, Zhao B, Tan X-X, Li Z-Q, Xiong Y, Zhong M, et al. Comparison of aggressive surgical treatment and palliative treatment in elderly patients with poor-grade intracranial aneurysmal subarachnoid hemorrhage. Biomed Res Int. 2018;2018:5818937. https://doi.org/10.1155/2018/5818937.
Wilson TJ, Davis MC, Stetler WR, Giles DA, Chaudhary N, Gemmete JJ, et al. Endovascular treatment for aneurysmal subarachnoid hemorrhage in the ninth decade of life and beyond. J Neurointerv Surg. 2014;6:175–7. https://doi.org/10.1136/neurintsurg-2013-010714.
Silva NA, Shao B, Sylvester MJ, Eloy JA, Gandhi CD. Unruptured aneurysms in the elderly: perioperative outcomes and cost analysis of endovascular coiling and surgical clipping. Neurosurg Focus. 2018;44:E4. https://doi.org/10.3171/2018.1.FOCUS17714.
Brinjikji W, Lanzino G, Rabinstein AA, Kallmes DF, Cloft HJ. Age-related trends in the treatment and outcomes of ruptured cerebral aneurysms: a study of the nationwide inpatient sample 2001-2009. AJNR Am J Neuroradiol. 2013;34:1022–7. https://doi.org/10.3174/ajnr.A3321.
Barker FG 2nd, Amin-Hanjani S, Butler WE, Hoh BL, Rabinov JD, Pryor JC, et al. Age- dependent differences in short-term outcome after surgical or endovascular treatment of unruptured intracranial aneurysms in the United States, 1996-2000. Neurosurgery. 2004;54:18–30.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Mirian, C., Mathiesen, T. (2019). A Restriction for the Surgical or Endovascular Treatment of a Ruptured Aneurysm in the Elderly?. In: Bartels, R., Rovers, M., Westert, G. (eds) Evidence for Neurosurgery. Springer, Cham. https://doi.org/10.1007/978-3-030-16323-5_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-16323-5_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-16322-8
Online ISBN: 978-3-030-16323-5
eBook Packages: MedicineMedicine (R0)