Abstract
In the search of the ideal dacryocystorhinostomy (DCR) that avoids a surgical scar in the medial canthus, transcanalicular laser-assisted dacryocystorhinostomy (TCL-DCR) is considered a fast and relatively simple alternative procedure for the management of nasolacrimal duct obstruction. The success rates of TCL-DCR are between 34% and 95.2%, and anatomical success rates up to 100% have been published. The functional failure rate of TCL-DCR is higher, often related to a sump syndrome in the majority of patients, when compared to non-laser-assisted DCR methods. Complications, such as tissue necrosis and proximal lacrimal drainage system trauma, have been reported. Surgical modifications to decrease the amount of laser energy delivered have resulted in better anatomical and functional outcomes.
In this chapter, transcanalicular laser-assisted dacryocystorhinostomy is presented, including its historical evolution, types of lasers used, indications, and surgical techniques. Advantages and disadvantages of this approach, techniques for optimizing outcomes, and complication avoidance and management are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Dacryocystorhinostomy
- Endocanalicular
- Lacrimal surgery
- Laser dacryocystorhinostomy
- Nasolacrimal duct obstruction
- Scarless lacrimal surgery
- Diode laser
- Holmium YAG laser
- Laser complications
- Functional epiphora
Introduction
Epiphora , due to primary and secondary nasolacrimal duct obstructions (NLDO), is generally managed by dacryocystorhinostomy (DCR) techniques aiming to reconstruct the lacrimal drainage system. The obstructed outflow pathway is redirected by connecting the lacrimal sac to the middle nasal cavity to relieve symptoms. External dacryocystorhinostomy (EX-DCR) is the standard surgical technique for the treatment of NLDO, described first by Toti in 1904 [1]. Dupuy-Dutemps and Bourguet modified it in 1921 with the introduction of the anastomosis of the lacrimal sac and nasal mucosal linings [2]. Despite the published high success rates of EX-DCR exceeding 90% [3,4,5], the search for a scarless DCR technique that can be performed quickly and with high success rates has continued [6,7,8,9,10,11,12].
Development of high-resolution fiber-optic endoscopes and fine endonasal surgical tools in the late 1980s intensified the interest in endoscopic management of lacrimal obstructions, where laser energy could be used to rapidly create a precise and bloodless rhinostomy opening during a scarless technique [13, 14]. Laser energy can be delivered either nasally, as in endonasal laser-assisted DCR (ENL-DCR) , or via a probe passed through the canaliculus, referred to as transcanalicular laser-assisted DCR (TCL-DCR). Both techniques allow the surgeon to create the alternative route for lacrimal drainage; however, TCL-DCR is likely safer than ENL-DCR, as the laser beam is directed toward the nose and away from the orbit. In Fig. 46.1a, TCL-DCR is schematically presented.
(a) Schematic illustration of the transcanalicular laser DCR. The laser fiber-optic probe is introduced through the inferior punctum and canaliculus until a hard bony stop is felt. The aiming beam is turned on and visualized transmucosally in the nasal cavity with the endoscope, typically at the level anterior to the middle turbinate. (b) Performing TCL-DCR surgery with the Multidiode S30 OFT (INTERMEDIC Inc., Spain). (c) Multidiode S30 OFT (INTERMEDIC Inc., Spain)
Endoscopic DCR procedures avoid the external scar as well but necessitate additional surgical equipment and visualization systems. Laser delivery systems facilitate the precise removal of bone and soft tissues and minimize bleeding while creating the rhinostomy during endoscopic lacrimal techniques. However, the cost and maintenance of these devices are high. Laser safety measures also deserve maximum attention in surgical rooms.
Historical Perspective
Jack published the first transcanalicular DCR in 1963 [15]. In 1992, Levin and Silkiss studied laser endocanalicular DCRs on cadavers [16, 17], while Christenbury performed the first TCL-DCR with argon laser assistance on patients with NLDO [18]. Various laser wavelengths have been utilized to create the rhinostomy in lacrimal surgery , such as neodymium:YAG (Nd:YAG) laser [19,20,21,22,23,24], holmium:yttrium-aluminum-garnet (Ho:YAG) laser [25,26,27], potassium-tytanyl-phosphate (KTP) laser [28, 29], and erbium: YAG (Er:YAG) laser [30].
The use of solid-state diode laser for lacrimal surgery was first reported in 1994 by Mchugh, and an interventional case series of TCL-DCR with high success was reported by Eloy in 2000 [31, 32]. The diode laser subsequently gained popularity among other laser delivery devices, possibly due to relatively lower costs of buying and maintenance. TCL-DCR continues to remain the subject of many ongoing studies [26, 32,33,34,35,36,37,38,39].
Management of Epiphora via TCL-DCR
Transcanalicular laser-assisted DCR is performed with the assistance of nasal endoscopy to visualize the aiming beam and laser probe in the middle meatus during lacrimal surgery. The laser delivers focal energy to open the rhinostomy site, for the management of NLDO as explained below. The major advantages of TCL-DCR are briefly listed in Table 46.1.
Diagnostic Evaluation and Patient Selection for TCL-DCR
Patients with nasolacrimal drainage system obstructions typically present to clinic with epiphora and/or dacryocystitis. A large tear lake with mucoid or purulent discharge is a common sign of nasolacrimal duct obstruction. All patients with a history of tearing and/or dacryocystitis must be evaluated via a clinical work-up and ophthalmic examination to rule out other causes of epiphora, such as blepharitis, conjunctivitis, keratitis, eyelid malpositions or laxity, and orbicularis oculi muscle weakness. The lacrimal sac area should be palpated for a mass and/or copious mucopurulent reflux, indicating a dacryocele (dacryocystocele). If a dacryocele is evident, it would be anticipated that the enlarged lacrimal sac flaps would need to be trimmed down for a successful DCR. Therefore, such patients may not be appropriate candidates for minimally invasive techniques, or the sac flaps would require adequate shortening transnasally during the endoscopic surgery.
The puncta and lacrimal canaliculi must be evaluated if canaliculitis is suspected. Definitive treatment of this condition typically requires canaliculotomy for drainage and curettage.
When considering the transcanalicular approach, office lacrimal irrigation to evaluate its patency and to determine the level of any obstruction in the proximal drainage system is mandatory.
Nasal cavity examination by the surgeon may identify intranasal pathologies such as hypertrophic or bullous middle turbinate, nasal polyps, and septal deviation. In patients with narrow nasal cavities, endoscopic surgery may be more challenging to perform.
A 99mTc-DTPA (diethylenetriaminepentaacetic acid) dacryoscintigraphy scan can be considered for patients with a history of tearing despite patent lacrimal irrigation, as this may detect functional epiphora in addition to a prolonged dye disappearance test. In contrast, dacryocystography (DCG) can be helpful to diagnose an anatomic etiology for outflow obstruction, such as a filling defect from a lacrimal sac mass, foreign bodies, or dacryolith. Dilated or undersized fibrotic sacs can be detected by DCG and be helpful in the differential diagnosis.
Punctal and/or canalicular stenosis , history of nasal and/or lacrimal surgery , naso-orbital trauma, congenital nasolacrimal duct occlusion, and previous transconjunctival blepharoplasty are important risk factors to elicit before TCL-DCR.
In some instances, TCL-DCR may not be easily performed in patients with prominent frontal processes and wide dorsum nasi, as the manipulation of the laser probe is limited. The use of pediatric TCL-DCR surgery is controversial due to the risk of significant fibrosis caused by heat convection. Table 46.2 summarizes the contraindications for TCL-DCR.
Surgery
TCL-DCR surgery with diode laser assistance has become increasingly popular worldwide (Fig. 46.1a–c). This technique can be performed under local anesthesia, with or without sedation, or under general anesthesia.
Preoperative Preparation
Premedication to aid in hemostasis and analgesia is performed with 0.05% xylometazoline +1% lidocaine nasal spray and nasal cavity packing using absorbent sponges soaked in xylometazoline 0.05% with 1:200,000 epinephrine. The uncinate process mucosa and medial canthal area are infiltrated with approximately 1–4 ml of 1:1 volume 1% lidocaine HCl with 0.0125 mg/ml adrenaline and 0.05% bupivacaine HCl. An acrylic corneal protector is placed to protect cornea. Laser safety rules must be strictly applied to prevent operating room hazards and patient or staff injuries.
Surgical Steps
Surgery is performed with a clean setup and sterile draping. The 980 nanometer (nm) wavelength diode laser is delivered via a silica-silica polyamide laser fiber optic. The Multidiode S30 OFT laser delivery device (INTERMEDIC Inc., Spain), equipped with a 600 μm silica-silica polyamide laser fiber optic is displayed in Fig. 46.1c.
The preferred total caliper of the probe with the sleeve is between 300 and 600 microns. Laser settings are adjusted for each patient, ranging between 8 and 12 W power range, 350–500 milliseconds pulse time, and 350–500 milliseconds pause duration between pulses to use the lowest amount of energy sufficient to create an osteotomy.
The diode laser fiber optic is passed through the canaliculus into the lacrimal sac, until the bright transillumination of the aiming beam can be seen via a nasal endoscope with a 0° or 30° angle (Fig. 46.2a). An anteriorly located ethmoidal cell may prevent direct access to the nasal cavity, or ethmoidal cells may be entered instead. The aiming beam is dull in these cases (Fig. 46.2b).
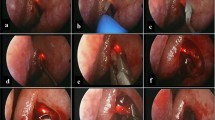
The surgical steps of TCL-DCR are demonstrated in Fig. 46.3a–d. The upper canaliculus is typically the preferred entrance as long as the aiming beam is clearly visible across the middle turbinate. Infracture or medialization of the middle turbinate may be necessary in some cases for better view of the lateral nasal wall (Fig. 46.4). A bullous or large middle concha may be partially resected to debulk its volume to prevent functional blockage of the ostium (Fig. 46.5).
Surgical steps of TCL-DCR: (a) rhinostomy, initial opening; (b) aiming beam in the lacrimal sac; (c) rhinostomy, mid-surgery; (d) probe tip in the lacrimal sac entering the nasal cavity next to the middle turbinate; (e) antifibrotic agent application to the rhinostomy site; (f) rhinostomy site and silicone tube
Laser energy is the delivered until the large bony opening is created, at least 5 mm in diameter. The ostium can then be further enlarged with rongeurs through the nasal cavity to decrease the amount of laser energy delivered. A can opener type of laser application (Fig. 46.6) may also decrease the total laser energy. The rhinostomy is opened by the removal of the sac mucosa-lacrimal bone-nasal mucosa button created.
In a patient with a wide dorsum nasi, the frontal process of the maxillary bone is generally thick. In these patients, it should be kept in mind that the total laser energy delivered to open a large enough rhinostomy would be quite high and could cause extensive thermal damage to the neighboring tissues.
It is also recommended to minimize the in-out maneuvers with the laser probe through the canaliculi in order to avoid trauma to the delicate endothelial lining of the proximal drainage system. The debris around the osteotomy is preferably removed transnasally with a Takahashi endo-forceps and backbiting rongeur. Once the carbonized debris (Fig. 46.7) is cleared around the ostium, bicanalicular silicone intubation is performed to facilitate healthy endocanalicular epithelialization.
Adjunctive antifibrotic agents applied during TCL-DCR should be applied precisely to the ostium via an irrigation cannula, followed by copious irrigation of the ocular surface. A dry surgical sponge is placed in the nasal ostium during this procedure to minimize contact of the healthy nasal mucosa from the antifibrotic agent. The lacrimal tract can then be irrigated with 0.2% dexamethasone and 2% gentamycin mixture or 0.9% NaCl solution.
Occasionally, the surgeon may choose to convert to a conventional external DCR with skin approach if an adequately sized bony ostium could not be created via TCL-DCR. The common technical and intraoperative difficulties in performing TCL-DCR are listed in Table 46.3.
Postoperative Care
After surgery, eye patching for 4 hours with sterile antibiotic and corticosteroid drops is often advised. Although rarely necessary, nasal packing can be removed later that day or the following day if used. Cold compresses used frequently in the first 24 hours after surgery help decrease pain and edema. Topical, and occasionally systemic, antibiotics are prescribed for 1 week. Nasal and ocular steroids and nasal saline spray are continued for 3 weeks postoperatively. Follow-up visits are recommended at postoperative day 1, day 3, week 1, month 1, and every 3 months until the end of the first postoperative year to check for recurrence of symptoms or infection. The ostium is examined, and any debris clogging the ostium removed if still present after the first postoperative week. The patency of the new rhinostomy is assessed by dye disappearance test (DDT), or lacrimal irrigation if the DDT is fluorescein-negative intranasally. Bicanalicular stents are typically removed between 1 and 3 months postoperatively.
Tissue Response to Laser in TCL-DCR
Laser wavelength, laser power and energy, method of application (contact or noncontact), application mode (continuous, burst, or pulsed), and tissue properties play important roles in the tissue response to laser [39]. Several studies have assessed the histological effects of different wavelength lasers on various tissues [40,41,42,43,44]. Scanning electron microscopy studies revealed large coagulation zones underneath the ablated hypertrophic inferior nasal turbinates following Nd:YAG and diode lasers. The coagulation effect was noted to be minimal with CO2 lasers, whereas the ablative effect of CO2 was the most precise [45].
In vitro thermal tissue effects induced by contact application of fiber-guided lasers, such as Ho:YAG, Nd:YAG, and diode lasers (830, 940 nm), which are similar to those used for endocanalicular DCR approaches, revealed that the 830 nm diode laser created the widest ablation zones.
Although the 810–980 nm diode laser, operating in wavelengths near the infrared part of the light spectrum, is highly absorbed by melanin and hemoglobin to provide good hemostasis, the coagulative effect of the contact mode was less than the noncontact mode of the 830 nm diode laser and contact mode of the Ho:YAG laser [41, 44].
Histopathologically, inflammation and reepithelialization are delayed when lasers are used, as compared to the steel scalpel, as shown on guinea pig oral mucosa. Complete reepithelialization occurred by the end of the first week with the scalpel, compared to the fourth week with CO2 laser. Resolution of inflammation was also delayed up to 4 weeks with lasers and bipolar cautery, compared to resolution of inflammation by the second week when using the scalpel [42, 43, 46]. The most extensive coagulation damage with delayed reepithelialization was noted with Nd:YAG applications, and more prominent inflammatory infiltration was seen in Nd:YAG and combined CO2-Nd:YAG wounds compared to CO2 and non-laser wounds [43, 47].
Briefly, the 2940 nm Er:YAG and 2140 nm Ho:YAG lasers have good bone ablation properties with limited coagulative effects [48]. The 532 nm KTP laser has both good cutting and coagulating properties. The 1064 nm Nd:YAG laser can lead to significant collateral damage in spite of its good ablative effect. Diode lasers in the 830–980 nm wavelength segment have demonstrated both good cutting and coagulating effects with less collateral damage and are more effective in the noncontact mode. Due to these characteristics, combined with its applicability via fiber probes ranging between 0.4 and 1.0 mm and fiber-optic tips that confine the laser energy to the tip, the diode laser has gained popularity in endocanalicular nasolacrimal surgery [49]. Newer compact and more easily operated diode laser delivery systems are also available, which enable the surgeon to mobilize the device if needed (Fig. 46.1c).
However, based on the current data available in peer-reviewed literature, it is unclear whether a particular laser source provides a significant advantage over the others with respect to clinical outcomes [24, 39, 49]. Practically speaking, laser assistance in lacrimal surgery enables precise, bloodless, easy, and fast ablation of the mucosa-bone- mucosa tissues at the rhinostomy site with minimal trauma to adjacent tissues. Adequate coagulation is achieved in the well-vascularized nasal mucosa; however, some degree of heat is conveyed to the vicinity of the surgical site.
The advantages of diode lasers are therefore reported as its ability to deliver a sufficiently powerful laser beam via a relatively narrow optical fiber with less collateral heat spread and less residual thermal damage to tissue.
Surgical Outcome of TCL-DCR
Functional success after lacrimal drainage surgery is assessed by the recovery of patency by lacrimal irrigation and the resolution of epiphora and dacryocystitis. Anatomical success describes an unobstructed ostium and lacrimal pathway but may be paradoxically accompanied by ongoing symptoms of obstruction.
Success rates reported for TCL-DCR with erbium:YAG [30], neodymium:YAG, and holmium:YAG lasers are 75%, between 64% and 85%, and between 47% and 82.5%, respectively [19,20,21,22,23,24,25,26]. However, these studies are not comparable due to the variable demographics of the study groups, number of patients, and follow-up periods.
Despite the limited number of published articles involving laser-assisted lacrimal surgery , the increasing number of solid-state diode laser delivery devices in ophthalmology and otolaryngology worldwide has led to more studies of diode laser-assisted TCL-DCR since first published by Eloy in 2000 [33,34,35,36,37]. The success rates of diode laser-assisted TCL-DCR for treating primary NLDO in adults range from 34% [50] to 95.2% [51]. The majority of these interventional case series report the success rates of TCL-DCR higher than 80%, close to the success rates of EX-DCR of 90–98% [3, 52] and endonasal DCR, which are between 61% and 100% [53,54,55,56,57,58,59,60]. Several studies comparing the outcome of EX-DCR and TCL-DCR with diode laser assistance revealed functional success of 89–95.4% for EX- DCR versus 73.7–84% for TCL-DCR [61,62,63]. A similar study by Ajalloueyan reported 92.6% success for EX-DCR and 93.4% for TCL-DCR, with less hemorrhage and shorter surgical times (19 versus 67 minutes) noted in the diode laser TCL-DCR group [64]. The shorter operative time of diode laser-assisted TCL-DCR (approximately 20 minutes) versus EX- DCR (60–90 minutes) has also been confirmed by others [61, 62, 65]. Piedrola compared diode laser-assisted techniques for transcanalicular and transnasal DCRs with similar outcomes [66].
Piaton then introduced the use of mitomycin-C (MMC) as an adjunct in Ho:YAG and Nd:YAG laser-assisted TCL-DCR [19, 23]. Henson used 0.4 mg/ml MMC for 5 minutes during 40 diode laser TCL-DCR procedures and reported a success rate of 87.5% after 12 months [67]. Henson then stated that “the cicatricial closure of the ostium can still occur even with a single intraoperative application of MMC.” By applying multiple applications of MMC (0.4 mg/ml for 5 minutes) postoperatively after TCL-DCR, he then found higher success rates of 92.8% after 12 months in 125 diode laser-assisted TCL-DCR surgeries [68]. In general, although antifibrotic agents such as MMC and 5-FU have demonstrated potential benefits, the published data in laser-assisted DCR are not consistent, and high doses of adjunctive MMC applications during and/or after TCL-DCR may damage ocular and nasal surfaces [69,70,71,72].
Kaynak et al. reported a 60.3% functional success with diode laser-assisted TCL-DCR with intraoperative 0.2 mg/ml MMC applied at the rhinostomy site in a large series of patients with PANDO with 2-year follow-up. The relatively high failure rate was attributed to the overall greater total energy delivered, when compared to similar studies [38].
Low-level diode laser irradiation of cultured human oral mucosa fibroblasts in vitro has been found to lead to an increase in DNA synthesis [73]. In a similar study, laser-patterned microcoagulation using the 980 nm diode laser on the oral mucosa of healthy rabbits demonstrated stimulation of gingival and oral mucosal tissue regeneration. The study concluded that “laser-patterned microcoagulation treatment with the 980nm diode laser is a promising method for treating degenerative diseases of the oral soft tissues,” such as gingivitis and gingival recession [74]. With these studies in mind, it can be postulated that the high amount of laser energy used to create a large bony ostium in TCL-DCR may cause increased fibroblastic activity and wound healing, thus contributing to ostium fibrosis and surgical failure. Theoretically, laser delivery systems with irrigating ports may decrease the thermal effect of ablative lasers on the surgical site and surrounding tissues; however, published data is lacking in the ophthalmic literature. One study using a diode laser with an irrigation port for TCL-DCR yielded 83.3% functional success with an average of 245 joules of total energy delivered [37]. The higher success rates may be attributed to the lower heat delivered due to the irrigation system attached to the laser probe, which decreases thermal injury to the rhinostomy site and neighboring tissues.
In TCL-DCR, an inferiorly located rhinostomy of at least 7 mm diameter may sometimes be difficult to achieve by laser ablation only. Aggressive attempts may lead to higher energy delivery and subsequent thermal damage to both mucosal surfaces that may lead to fibrosis and failed TCL-DCR. To decrease the energy delivered and to place the rhinostomy more inferiorly, endonasal mechanical enlargement of the ostium have shown increased success rates. The mechanical removal of the composite mucosal tissue and bone along the rhinostomy circumference also removes the charred tissues as well, leaving raw rhinostomy edges for healing by secondary intention [75, 76].
Middle turbinectomy during TCL-DCR, if necessary, may also increase the success from 71% to 88% [77]. TCL-DCR can also be performed with anastomosed flaps with good success rates, although technically this is much more challenging [78].
The functional success rate of secondary diode laser TCL-DCR has been reported as 80% for treating failed external DCR (27.3-month follow-up). Similarly, patients who had failed TCL-DCR may be treated via subsequent external DCR with 80% success [79].
Uysal reported 100% anatomical success and 85% functional success at the end of 20-month follow-up in a study group of 18 children with NLDO between the ages of 4 and 10 years treated with TCL-DCR [80]. Çakmak reported an 87.5% success rate in eight children with a shorter follow-up of 9 months [81]. It may be that iatrogenic canalicular injury by the laser probe to the delicate proximal drainage system in children may lead to robust fibrosis and proximal lacrimal system obstruction. Generally, the success rate of TCL-DCR for the younger population is lower when compared with an elderly population [82]. TCL-DCR may therefore be considered as a relative contraindication in the pediatric population.
Table 46.4 summarizes the data from studies where various lasers are used in TCL-DCR.
Complications and Failure of TCL-DCR
Canalicular lacerations, punctal slitting, and punctal adhesions are not rare in TCL-DCR (Fig. 46.8a, b).
Due to the oval shape of the lacrimal sac, which is narrower inferiorly, the ability of the surgeon to move the laser probe anteriorly and posteriorly during TCL-DCR becomes more restricted as the probe is advanced more inferiorly, and punctal and canalicular lacerations can occur due to the forced manipulation. Therefore, the final ostium is usually wider superiorly and narrower inferiorly (Fig. 46.9a–f). The narrow inferior part can be easily occluded with mucosal proliferation forming a residual lacrimal sac leading to a sump syndrome. The patient may exhibit a patent ostium by irrigation (due to flow through the opening superiorly) but have functional epiphora. Closure of the ostium due to fibrosis and/or adhesions between the middle turbinate and the ostium are other causes of failure. Excessive thermal injury is a well-known risk factor for nasal mucosal and ostium adhesions.
Failure after TCL-DCR: (a) the typical upside-down pear-shaped ostium of laser-assisted TCL-DCR; (b) sump syndrome in TCL-DCR; (c) middle turbinate adhesion to the rhinostomy; (d) small pinhole ostium at the 3rd postoperative month; (e) false ostium in the anterior ethmoidal cell; (f) total closure of the ostium (in blue circle)
Lacrimal system tumors and dacryoliths may be missed using the TCL-DCR approach. Thus, a patient with a missed dacryolith during the TCL-DCR may postoperatively report persistent infection, regional pain, and functional epiphora , and therefore additional exploration is warranted. TCL-DCR would be contraindicated if these etiologies are suspected prior to surgery.
Although the transcanalicular route is a safer method of laser delivery in lacrimal surgery due to its direction away from the eye, there are risks of iatrogenic mechanical injury to the puncta, canaliculi, and/or the common canaliculus by the laser probe and sleeve. Dilating the puncta to enable the passage of the laser probe and/or maneuvering the probe to reach the maximum limits of the sac may cause small tears that subsequently enlarge to canalicular lacerations by the presence of the silicone tubes.
The lasers may also cause thermal canalicular burns and fibrosis that result in narrowing or occlusion of the upper lacrimal drainage system. Such complications may necessitate recanalization surgery with a conjunctivodacryocystorhinostomy with Jones tube.
Kaynak reported tube prolapse or stent loss in 17.7% of patients, which was speculated to be due to the absence of a visible wound that keeps the patient away from the medial canthal area (Fig. 46.10) [38].
Less commonly, tissue necrosis, nasocutaneous fistula at the nasojugal sulcus, and orbital infarction syndrome following diode laser-assisted TCL-DCR have been reported [83, 84].
The possible complications of TCL-DCR are listed in Table 46.5.
Conclusion
The relative technical ease, shorter surgery, and rapid recovery time associated with TCL-DCR compared to external DCR have attracted the attention of many surgeons in recent years. The published success rate of TCL-DCR varies widely from 34% to 95.2%. The differences in the number of cases, follow-up time, definition of success, and surgeon variations in the TDL-DCR technique make it difficult to compare these results. The use of silicone stents, endonasal instrumentation, newer lasers, and antifibrotic agents have been reported to increase the success rate of TCL-DCR for the management of NLDO. Failure of TCL-DCR is often due to adhesions between middle turbinate and the ostium, closure of the rhinostomy site due to significant heat spread with resultant fibrosis, or sump syndrome from occlusion of the inferior ostium in the majority of patients. Among the complications of this surgery, patients should also be informed of the possibility of thermal or mechanical injury to the proximal lacrimal drainage apparatus.
References
Toti A. Nuovo metodo conservatore di cura radicale delle suporazioni chroniche del sacco lacrimale. Clin Mod Firenze. 1904;10:385–9.
Dupuy-Dutemps L, Bourguet J. Procédé plastique de dacryocystorhinostomie et ses resultats. Ann Ocul J. 1921;158:241–61.
Tarbet KJ, Custer PL. External dacryocystorhinostomy. Surgical success, patient satisfaction, and economic cost. Ophthalmology. 1995;102:1065–70.
Warren JF, Seiff SR, Kavanagh MC. Long term results of external dacryocystorhinostomy. Ophthalmic Surg Lasers Imaging. 2005;36:446–50.
Fayers T, Laverde T, Tay E, Olver JM. Lacrimal surgery success after external dacryocystorhinostomy: functional and anatomical results using strict outcome criteria. Ophthal Plast Reconstr Surg. 2009;25:472–5.
Caldwell GW. Two new operations for obstruction of the nasal duct with preservation of the canaliculi, and an incidental description of a new lachrymal probe. NY Med J. 1893;57:581.
Krasnov MM. Ultrasonic dacryocystorhinostomy. Am J Ophthalmol. 1971;72:200–1.
Javate RM, Campomanes BS, Co ND, et al. The endoscope and the radiofrequency unit in DCR surgery. Ophthal Plast Reconstr Surg. 1995;11:54–8.
Ibrahim HA, Noble JL, Batterbury M, Johnson CP, Williams R. Endoscopic-guided trephination dacryocystorhinostomy (Hesham DCR): technique and pilot trial. Ophthalmology. 2001;108:2337–45.
Wormald PJ. Powered endonasal DCR. Laryngoscope. 2002;112:69–71.
Adenis JP, Robert PY. Retrocaruncular approach to the medial orbit for dacryocystorhinostomy. Graefes Arch Clin Exp Ophthalmol. 2003;241:725–9.
Kaynak-Hekimhan P, Yilmaz OF. Transconjunctival dacryocystorhinostomy: scarless surgery without endoscope and laser assistance. Ophthal Plast Reconstr Surg. 2011;27:206–10.
Massaro BM, Gonnering RS, Harris GJ. Endonasal laser dacryocystorhinostomy. A new approach to nasolacrimal duct obstruction. Arch Ophthalmol. 1990;108:1172–6.
Gonnering RS, Lyon DB, Fisher JC. Endoscopic laser-assisted lacrimal surgery. Am J Ophthalmol. 1991;111:152–7.
Jack MK. Dacryocystorhinostomy: description of a transcanalicular method. Am J Ophthalmol. 1963;56:974–7.
Levin PS, Stormogipson DJ. Endocanalicular laser- assisted dacryocystorhinostomy. An anatomic study. Arch Ophthalmol. 1992;110(10):1488–90.
Silkiss RZ, Axelrod RN, Iwach AG, et al. Transcanalicular THC: YAG dacyrocystorhinostomy. Ophthalmic Surg. 1992;23:351–3.
Christenbury JD. Translacrimal laser dacrycystorhinostomy [letter]. Arch Opthalmol. 1992;110:170–1.
Piaton JM, Limon S, Ounnas N, et al. Transcanalicular endodacryocystorhinostomy using Neodynium:YAG laser. J Fr Ophthalmol. 1994;17(10):555–67.
Pearlman SJ, Michalos P, Leib ML, et al. Translacrimal transnasal laser-assisted dacryocystorhinostomy. Laryngoscope. 1997;107:1362–5.
Rosen N, Barak A, Rosner M. Transcanalicular laser-assisted dacryocystorhinostomy. Ophthalmic SurgLasers. 1997;28:723–6.
Fay AM, Michalos P, Rubin PA. Endocanalicular Nd: YAG laser dacryocystorhinostomy [review]. Int Ophthalmol Clin. 1999;39:177.
Piaton JM, Keller P, Limon S, et al. Holmium:YAG and neodymium:YAG laser assisted trans-canalicular Dacryocystorhinostomy. Results of 317 first procedures. J Fr Ophthalmol. 2001;24:253–64.
Hong JE, Hatton MP, Leib ML, et al. Endocanalicular laser dacryocystorhinostomy: an analysis of 118 consecutive surgeries. Ophthalmology. 2005;112(9):1629–33.
Dalez D, Lemagne JM. Transcanalicular dacryocystorhinostomy by pulse Holmium-YAG laser. Bull SocBelge Ophthalmol. 1996;263:139–40.
Kaynak P, Çelikoyar M, Göker S, Küçüksümer Y, Yılmaz ÖF. Endoskopik Ho:YAG laser dakriyosistorinostomi. Türk Oftalmoloji Gazetesi. 2002;32:332–6. (Article in Turkish).
Kaynak Hekimhan P, Yılmaz ÖF. Holmium:YAG LASER lacrimal by-pass surgery. Techin Ophthalmol. 2006;4:39–44.
Muellner K, Wolf G, Luxenberger W, et al. Laser-assisted transcanalicular dacryocystorhinostomy. Initial results Ophthalmologe. 2001;98:174–7.
Hofmann T, Lackner A, Muellner K, et al. Endolacrimal KTP laser-assisted dacryocystorhinostomy. Arch Otolaryngol Head Neck Surg. 2003;129(3):329–32.
Caversaccio M, Frenz M, Schar P, et al. Endonasal and transcanalicular Er:YAG laser dacryocystorhinostomy. Rhinology. 2001;39:28–32.
Mchugh JDA, Rose GE, Marshall J. The application of high-power diode lasers in ophthalmology. Laser Light Ophthalmol. 1994;6:229–38.
Eloy P, Trussart C, Jouzdani E, et al. Transcanalicular didoe laser assisted Dacryocystorhinostomy. Acta Otohinolaryngol Belg. 2000;54:157–63.
Alanon FFJ, Alanon FMA, Martinez FA, et al. Transcanalicular dacryocystorhinostomy technique using diode laser. Arch Soc Esp Oftalmol. 2004;79(7):325–30.
Plaza G, Betere F, Noquiera A. Transcanalicular dacryocystorhinostomy with diode laser: long-term results. Ophthal Plast Reconstr Surg. 2007;23(3):179–82.
Maeso RJ, Sellarès FMT. Trans-canalicular diode laser dacryocystorhinostomy: technical variations and results. Acta Otorrinolaringol Esp. 2007;58(1):10–5.
Cintra PPVC, Anselmo-Lima WT. Endocanalicular diode laser-assisted dacryocystorhinostomy. Otolaryngol Head Neck Surg. 2008;139:159–61.
Drnovšek-Olup B, Beltram M. Transcanalicular diode laser-assisted dacryocystorhinostomy. Indian J Ophthalmol. 2010;58:213–7.
Kaynak P, Ozturker C, Yazgan S, Ozturk Karabulut G, Akar S, Demirok A, Yilmaz OF. Transcanalicular diode laser assisted dacryocystorhinostomy ın primary acquired nasolacrimal duct obstruction: 2-year-follow up. Ophthal Plast Reconstr Surg. 2014;30:28–33.
Dowd T, Jones N, Eloy P, Oswal V. Laser assisted dacryocystorhinostomy. In: Oswal V, Remacle M, editors. Principles and practice of lasers in otorhinolaryngology and head and neck surgery. The Hague: Kugler Publishers; 2002. p. 187–219.
Janda P, Sroka R, Tauber S, et al. Diode laser treatment of hyperplastic inferior nasal turbinates. Lasers Surg Med. 2000;27:129–39.
Sroka R, Janda P, Killian T, et al. Comparison of long term results after Ho:YAG and diode laser treatment of hyperplastic inferior nasal turbinates. Lasers Surg Med. 2007;39:324–31.
Goharkhay K, Mortiz A, Wilder-Smith P, et al. Effects on oral soft tissue produced by diode laser in vitro. Lasers Surg Med. 1999;25:401–6.
Zborayova K, Ryska A, Lansky M, et al. Histomorphologic study of nasal turbinates after surgical treatment: a comparison of laser surgery and radiofrequency-induced thermotherapy effects in animals. Acta Otolaryngol. 2009;129:550–5.
Janda P, Sroka R, Mundweil B, et al. Comparison of thermal tissue effects induced by contact application of fiber guided laser systems. Lasers Surg Med. 2003;33:93–101.
Janda P, Sroka R, Betz CS, et al. Comparison of laser induced effects on hyperplastic inferior nasal turbinates by means of scanning electron microscopy. Lasers Surg Med. 2002;30:31–9.
Sinha UK, Gallagher LA. Effects of steel scalpel, ultrasonic scalpel, bipolar electrosurgery on wound healing in guinea pig oral mucosa. Laryngoscope. 2003;113:228–36.
Luomanen M, Rauhamaa-Mäkinen R, Meurman JH, et al. Healing of rat mucosa after irradiation with CO2, Nd:YAG, Co2-Nd:YAG combination lasers. Scand J Dent Res. 1994;102:223–8.
Kuchaer A, Novak P, et al. Endoscopic laser recanalisation of presaccal canalicular obstruction. Br J Ophthalmol. 1999;83(4):443–7.
Ali MJ. Primary endocanalicular laser dacryocystorhinostomy. In: Ali MJ, editor. Principles and practice of lacrimal surgery. New Delhi: Springer; 2015. p. 219–28.
Joshi RS. Conventional dacryocystorhinostomy in a failed transcanalicular laser-assisted dacryocystorhinostomy. Indian J Ophthalmol. 2011;59:383–5.
Nuhoglu F, Gurbuz B, Eltutar K. Long-term outcomes after trans- canalicular laser dacryocystorhinostomy. Acta Otorhinolaryngol Ital. 2012;32:258–62.
Welham RA, Henderson PH. Results of dacryocystorhinostomy analysis of causes for failure. Trans Ophthalmol Soc U K. 1973;93:601–9.
Ben Simon GJ, Joseph J, Lee S, et al. External versus endoscopic dacryocystorhinostomy for acquired nasolacrimal duct obstruction in a tertiary referral center. Ophthalmology. 2005;112:1463–8.
Woog JJ, Kennedy RH, Custer PL, et al. Endonasal dacryocysto- rhinostomy: a report by the American Academy of Ophthalmology. Ophthalmology. 2001;108:2369–77.
Hartikainen J, Grenman R, Puukka P, Seppa H. Prospective randomized comparison of external dacryocystorhinostomy and endonasal laser dacryocystorhinostomy. Ophthalmology. 1998;105:1106–13.
Moore WM, Bentley CR, Olver JM. Functional and anatomic results after two types of endoscopic endonasal dacryocystorhinostomy: surgical and holmium laser. Ophthalmology. 2002;109:1575–82.
Maini S, Raghava N, Youngs R, et al. Endoscopic endonasal laser versus endonasalsurgical dacryocystorhinostomy for epiphora due to nasolacrimal duct obstruction: prospective, randomized, controlled trial. J Laryngol Otol. 2007;121:1170–6.
Lester SE, Robson AK, Bearn M. Endoscopic ‘cold steel’ versus laser dacryocystorhinostomy: completing the audit cycle. J Laryngol Otol. 2008;122:924–7.
Mirza S, Al-Barmani A, Douglas SA, et al. A retrospective comparison of endonasal KTP laser dacryocystorhinostomy versus external dacryocystorhinostomy. Clin Otolaryngol Allied Sci. 2002;27:347–51.
Lee S, Yen MT. Laser-assisted dacryocystorhinostomy: a viable treatment option? Curr Opin Ophthalmol. 2011;22:413–8.
Taşkıran Çömez A, Karadağ O, Arıkan S, Gencer B, Kara S. Comparison of transcanalicular diode laser dacryocystorhinostomy and external dacryocystorhinostomy in patients with primary acquired nasolacrimal duct obstruction. Lasers Surg Med. 2014;4:275–80.
Mourya D, Rijal RK. Transcanalicular laser-assisted dacryocystorhinostomy with diode laser. Orbit. 2017;36:370–4.
Uludag G, Yeniad B, Ceylan E, Yildiz-Tas A, Kozer-Bilgin L. Outcome comparison between transcanalicular and external dacryocystorhinostomy. Int J Ophthalmol. 2015;18:353–7. https://doi.org/10.3980/j.issn.2222-3959.2015.02.25.
Ajalloueyan M, Fartookzadeh M, Parhizgar H. Use of laser for dacryocystorhinostomy. Arch Otolaryngol Head Neck Surg. 2007;133:340–3.
Derya K, Demirel S, Doganay S, Orman G, Cumurcu T, Gunduz A. Endoscopic transcanalicular diode laser dacryocystorhinostomy: Is it an alternative method to conventional external dacryocystorhinostomy? Ophthal Plast Reconstr Surg. 2013;29:15–7.
Piédrola Maroto D, Franco SJ. Endonasal versus transcanalicular endoscopic dacriocystorhinostomy using diode laser. Surgical techniques and outcomes. Acta Otorrinolaringolo Esp. 2008;59(6):283–7.
Henson RD, Henson RG Jr, Cruz HL Jr, et al. Use of the diode laser with intraoperative Mitomycin C in endocanalicular laser dacryocystorhinosotmy. Ophthal Plast Reconstr Surg. 2007;23:134–7.
Henson RD, Cruz HL, Henson RG Jr, et al. Postoperative application of mitomycin-C in endocanalicular laser dacryocystorhinostomy. Ophthal Plast Reconstr Surg. 2012;28:192–5.
Camara JG, Bengzon AU, Henson RD. The safety and efficacy of mitomycin C in endonasal endoscopic laser-assisted dacryocystorhinostomy. Ophthal Plast Reconstr Surg. 2000;16:114–8.
Alanon FMA, Alanon FFJ, Martinez FA, et al. Results of the application of Mitomycin-c during endonasal and endocanalicular dacryocystorhinostomy by diode laser. Acta Otorhinolaryngol Esp. 2006;57:355–8.
Ali MJ, Mariappan I, Maddileti S, et al. Mitomycin C in dacryocystorhinostomy: the search for the right concentration and duration – a fundamental study on human nasal mucosa fibroblasts. Ophthal Plast Reconstr Surg. 2013;29:469–74.
Kamal S, Ali MJ, Naik MN. Circumostial injection of Mitomycin C (COS-MMC) in external and endoscopic dacryocystorhinostomy: efficacy, safety profile and outcomes. Ophthal Plast Reconstr Surg. 2014;30:187–90.
Loevschall H, Arenholt-Bindslev D. Effect of low level diode laser irradiation of human oral mucosa fibroblasts in vitro. Lasers Surg Med. 1994;14:347–54.
Romanos GE, Gladkova ND, Feldchtein FI, et al. Oral mucosa response to laser patterned microcoagulation (LPM) treatment. An animal study. Lasers Med Sci. 2013;28:25–31.
Basmak H, Caklı H, Sahin A, Gursoy H, Ozer A, Altun E, Yildirim N. Comparison of endocanalicular laser dacryocystorhinostomy with and without endonasal procedures. Graefes Arch Clin Exp Ophthalmol. 2011;249(5):737–43.
Goel R, Nagpal S, Kumar S, Meher R, Kamal S, Garg S. Transcanalicular laser-assisted dacryocystorhinostomy with endonasal augmentation in primary nasolacrimal duct obstruction: Our experience. Ophthalmic Plast Reconstr Surg. 2017;33:408–12.
Basmak H, Cakli H, Sahin A, et al. What is the role of partial middle turbinectomy in endocanalicular laser- assisted endonasal dacryocystorhinostomy? Am J Rhinol Allergy. 2011;25:160–5.
Robert MC, Maleki B, Boulos P. Endocanalicular laser dacryocystorhinostomy with mucosal flaps. Ophthal Plast Reconstr Surg. 2013;29:294–7.
Narioka J, Ohashi Y. Transcanalicular-endonasal semiconductor diode laser-assisted revision surgery for failed external dacryocystorhinostomy. Am J Ophthalmol. 2008;146(1):60–8.
Cakmak SS, Yildirim M. Use of endocanalicular dacryocystorhinostomy with multidiode laser in children. Int J Pediatr Otorhinolaryngol. 2010;74:1320–2.
Uysal IO, Ozçimen M, Yener HI, Kal A. Pediatric endocanalicular diode laser dacryocystorhinostomy: results of a minimally invasive surgical technique. Eur Arch Otorhinolaryngol. 2011;268:1283–8. https://doi.org/10.1007/s00405-011-1585-z.
Ayintap E, Buttanri IB, Sadıgov F, Serin D, Ozsutcu M, Umurhan Akkan JC, Tuncer K. Analysis of age as a possible prognostic factor for transcanalicular multidiode laser dacryocystorhinostomy. J Ophthalmol. 2014;2014:913047. https://doi.org/10.1155/2014/913047.
Yeniad B, Bilgin LK, Cagatay A, et al. A rare complication after transcanalicular dacryocystorhinostomy: tissue necrosis and nasal-cutaneous fistula. Ophthal Plast Reconstr Surg. 2011;27(5):112–3.
Yeniad B, Tuncer S, Kir N, et al. Orbital infarction syndrome after transcanalicular dacryocystorhinostomy with diode laser. Ophthalmic Surg Lasers Imaging. 2012;25:43.
Financial Disclosures
The author has no financial interest in any of the materials or equipment used in the chapter.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kaynak, P. (2019). Transcanalicular Laser-Assisted Dacryocystorhinostomy. In: Cohen, A., Burkat, C. (eds) Oculofacial, Orbital, and Lacrimal Surgery. Springer, Cham. https://doi.org/10.1007/978-3-030-14092-2_46
Download citation
DOI: https://doi.org/10.1007/978-3-030-14092-2_46
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-14090-8
Online ISBN: 978-3-030-14092-2
eBook Packages: MedicineMedicine (R0)