Abstract
Bacterial cellulose (BC) is a biopolymer with high purity of cellulose and excellent mechanical properties. Increased interest in the use of natural polymer makes BC as an excellent alternative for plant cellulose. Although both celluloses consist of unbranched pellicle with chemically equivalent structure, bacterial cellulose exhibits greater properties and potential in wider applications. The structure of bacterial cellulose that consists only glucose monomer and nanosized cellulose fibres secreted by the bacteria induces it to have high water-holding capacity, high crystallinity, high degree of polymerization and high mechanical strength. Furthermore, the characterization of BC can be certainly altered by incorporation with materials that are not essential for the bacterial growth into the fermentation medium. This unique property of BC opens a new gate for the development of new cellulose nanocomposites with desired properties by the incorporation of selective suitable materials. The BC nanocomposites produced opens new opportunity for various usages of BC in different fields of application in the pharmaceutical, chemical, medical and wastewater treatment plants.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Cellulose from bacteria has more advantages than the cellulose found in plants. These advantages provide plenty room for the use of the bacterial cellulose (BC) in various fields. The most significant advantage of the BC compared to plant cellulose is its high purity. The BC from fermentation is produced in a pure form without lignin and hemicelluloses (Tyagi and Suresh 2015). It is highly hydrophilic with high mechanical strength. As BC is 100% pure and produced in hydrophilic matrix forms, its extensive fibrils and high mechanical strength are maintained throughout the formation. The BC can be produced using different methods and substrates whereby the properties can be modified based on the application. Soluble or insoluble materials added in the fermentation medium, such as enzymes and metallic compound, can directly be incorporated in the cellulose network during the synthesis (Serafica et al. 2002). These properties open the opportunities in many fields mainly in medical field, wastewater treatment, paper and also audio industries.
Addition of other materials that are not needed in the growth of bacteria in the fermentation medium is proven to affect the yield and properties of BC (Ruka et al. 2013). This modification was done either by using in situ or ex situ techniques (Shah et al. 2013). Several researchers have shown the ability to improve the production of BC, while others have successfully altered the properties of the cellulose by adding certain substrates or by manipulating the operating conditions. For example, Park et al. (2013) reported that the addition of magnetite nanoparticles and polyaniline enhanced the thermal stability of BC while da Silva et al. (2016) reported that the addition of polyethylene glycol improved hydrophilicity properties of BC.
This unique ability of BC allowing a series of potential applications as a new novel BC composite can be produced with modified properties based on its function. These applications range from high mechanical strength of hydrogel with addition of genipin (Dayal and Catchmark 2016; Nakayama et al. 2004), incorporation of aloe vera as wound dressing (Saibuatong and Phisalaphong 2010), antimicrobial film with addition of silver nanoparticles (Yang et al. 2012; Wu et al. 2014) to the use of BC as membranes such as cellulose acetate membranes reinforced with BC sheet (Gindl and Keckes 2004). Development of electromagnetic nanocomposite with the incorporation of magnetite nanoparticles and polyaniline was reported by Park et al. (2013). Juncu et al. (2015) suggested the novel uses of BC for drug delivery with the addition of carboxymethyl cellulose. Furthermore, Shanshan et al. (2012) demonstrated that the production of BC membrane in N-methylmorpholine N-oxide had better mechanical and barrier properties.
Undeniably, conservation of the forest, particularly trees, is essential in managing the global warming. However, excessive use of trees for cellulose-based products such as paper, biofuels and construction materials has been continuously depleting the forest resources (deforestation) which lead to global warming problem. At the same time, natural polymers such as cellulose have attracted many attentions due to the effect of environmental pollution of the synthetic polymer (Pei et al. 2013). Previously, plant-derived celluloses were widely used. For example, cellulose fibres have been used as eco-composite plastics in agricultural fields (González-Sánchez et al. 2014), biosorbent for heavy metal removal (O’Connell et al. 2008), nanofiller for biofilm (Salehudin et al. 2014; Slavutsky and Bertuzzi 2014) and other newly developed degradable composites (Piccinno et al. 2015). However, in recent time, BC is started to be used because of its excellent and promising properties (Ashori et al. 2012). Table 1 shows the development of BC and its application starting from dessert or nata in the 1990s followed by various other applications throughout the year.
2 Bacterial Cellulose (BC)
BC is a source of cellulose other than plant cellulose. It is produced by bacteria from many genera such as Acetobacter, Achromobacter, Agrobacterium and Sarcina. However, from that many lists of cellulose producers, the only species that is well known with its capability to produce cellulose in large quantity is Acetobacter genus. While within that species, A. xylinus or Acetobacter xylinum (A. xylinum) is the one that is being used extensively in research and studies (Jonas and Farah 1997).
A. xylinum produces cellulose from glucan chains. The chains extrude into the fermentation medium from A. xylinum pores. These processes are repeated until a bundle of microfibrils is gathered and forms the BC. Medium for BC fermentation can be from any kind of carbon sources or sugars. Acetobacter needs oxygen to produce the BC. Therefore, the production will occur mostly at the surface of the liquid (Schramm and Hestrin 1954, Zahan et al. 2014; Hsieh et al. 2016).
The BC pellicle is pure and extremely hydrophilic. Therefore, it needs no treatment which makes its original high mechanical strength retained. These unique characteristics of BC open many rooms for new applications as the properties of BC can be changed by manipulating the fermentation process. The BC has more advantages compared to plant cellulose. The most significant advantage is its purity. The BC has no hemicelluloses or lignin as in plant cellulose; thus, less processing steps are required. Besides, its structure is stronger and finer compared to plant cellulose which is due to longer and finer fibre length of BC. Figure 1 shows a microstructure of BC and plant cellulose at 5000 times magnification.
2.1 Synthesis of Bacterial Cellulose
There are several bacteria which are able to produce cellulose such as the strain from genera Acetobacter, Agrobacterium, Pseudomonas, Rhizobium and Sarcina. It has superior properties such as ultrafine network structure, high biodegradability and high mechanical strength as compared to plant cellulose (Tsuchida and Yoshinaga 1997). It is expected to be an alternative for biodegradable biopolymer. According to Lee et al. (2002), cellulose synthesized by the bacterium A. xylinum and plant cellulose is highly crystalline. However, the arrangements of glucosyl units of the crystallites make them differ to each other.
A. xylinum is acetic acid-producing bacterium which is widely used as the model system to study the enzymes and genes involved in cellulose biosynthesis. This species of gram-negative bacteria has high capability to produce cellulose by converting carbon source such as glucose or sucrose into cellulose. It is well known that A. xylinum is an obligate aerobe and forms cellulose at the air/liquid interface in undisturbed cultures. Figure 2 demonstrates the formation of microfibrils by A. xylinum.
Multiple cellulose chain or microfibrils are synthesized in the interior of bacteria cell and spun out of BC export component or nozzles (Iguchi et al. 2011). The cellulose synthesis will continue until a limited condition such as when carbon sources are insufficient or when the BC filled the discs in fermentation using rotary discs reactor (Pa’e 2009; Khairul et al. 2016).
Most cellulose-producing acetic acid bacteria can convert glucose into gluconic acid and ketogluconic acid. The enzyme responsible for the conversion of glucose to gluconic acid is membrane-bound glucose dehydrogenase (GDH) (Hwang et al. 1999). This will simply remove glucose from the fermentation medium and avoid the formation of cellulose (Toru et al. 2005).
2.2 Application of Bacterial Cellulose
BC has similar chemical structure as plant cellulose, but it has higher purity without lignin or hemicelluloses. Therefore, using BC is more economic compared to plant cellulose because it can skip many stages in the production of pure plant cellulose (Jonas and Farah 1997), thus lowering the production cost. Moreover, its distinguishing properties such as ultrafine nanofibre, biodegradability and high mechanical test (Amin et al. 2012) make it as promising materials for many applications.
The interest on applications of BC grows rapidly since the 1990s. Those applications started with the BC usage in its ordinary form and expand to BC composites via the modification of BC using other materials.
3 Modification of Bacterial Cellulose for the Production of Bacterial Cellulose Nanocomposites
The most attractive feature of BC production is the ability to control and modify the properties of the cellulose product while it is being synthesized. Additions of other materials that are not required for bacterial growth to the medium can alter the yield and properties of the cellulose produced (Ruka et al. 2013). With this ability, the chemical composition (Shirai et al. 1994) and properties of BC such as hydrophilicity can be adjusted (Martínez-Sanz et al. 2013). Those improvements are required to enhance the capabilities of BC in order to fulfil the demand in different fields (Shah et al. 2013). The capacity to control and adjust the cellulose characteristic during the synthesis allows the development of new BC composites with better properties than would be conceivable by post-synthetic processing of other sources of cellulose.
Modification of BC to produce BC composites can be done using numerous methods depending on the additives or materials used. The method likewise changes with the required applications. In general, there are two fundamental approaches in modifying BC: ex situ and in situ modification methods.
3.1 Ex Situ Modification of Bacterial Cellulose
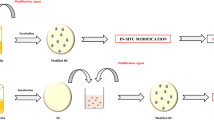
Ex situ modification method can be explained as the modification of pure BC after the cellulose is harvested (Shah et al. 2013). This involves the impregnation of BC with other materials to produce composites. The interaction might be physical or occur through definite hydrogen bonding between the BC and reinforcement materials. Liquid substances and tiny solid particles can easily penetrate and become engrossed inside the porous BC matrix. A schematic diagram showing the synthesis of BC composites using this method is provided in Fig. 3.
The properties of BC are differently affected by the additives used. The major hurdle associated with the ex situ modification method is the size and nature of the additives. Specifically, only submicron to nanosized materials can be impregnated into BC matrix. This is because larger particles cannot enter the BC pores, and hydrophobic materials are not able to combine with BC. Moreover, the structural arrangement of the BC fibril is not always uniform. Therefore, penetrating materials might not be homogeneously distributed inside the BC matrix. Accordingly, there is a need to identify new BC composite synthesis routes to resolve this problem. Table 2 lists previous works on producing BC composites using ex situ modification methods.
3.2 In Situ Modification of Bacterial Cellulose
The in situ method utilizes the addition of other materials as additives to the BC during its synthesis, which then becomes part of the cellulose structure. Using this method, additives were added to BC fermentation medium at the beginning of the process. A schematic diagram showing the synthesis of BC composites using this method is provided in Fig. 4.
Microfibrils of BC become denser with time and produce a web-shaped structure (Horii et al. 1997; Tang et al. 2010) that can trap various materials added to the medium (Ul-Islam et al. 2012). The encaged materials become part of the BC fibril network, resulting in BC composites. Bacterial composites can be synthesized via static fermentation (Wu et al. 2014; Saibuatong and Phisalaphong 2010), agitated culture (Cheng et al. 2009; Yan et al. 2008) and in vessels with rotating discs called rotary disc reactor (Pa’e et al. 2013; Serafica et al. 2002).
In situ modification of BC is a widely used approach that employs a wide range of modifications in agitation equipment and operating methods. However, this technique has certain limitations that prevent the synthesis of many BC composites. For example, several important antimicrobial agents such as titanium oxide cannot be added directly to the media because of their toxic effects on microorganisms. Moreover, BC composite synthesized through agitation culture cannot be applied as a gel or sheet in biomedical applications. Table 3 lists previous works on producing BC composites using in situ modification methods.
3.3 Characterization of Bacterial Cellulose Nanocomposites
BC composites can be characterized in many different ways. These important properties will vary from application to application. Microstructure study of the BC composites will provide information about the appearance of the cellulose produced under microscopic scale. Field emission scanning electron microscope is usually used to study the microstructure of the BC. The FESEM is a versatile analytical ultrahigh resolution that extends imaging and analytical resolution beyond previously achievable limits. The equipment is well suited for characterization of BC since it provides high magnification up to 30,000 magnification. Since BC consists of multiple nanocellulose networks, equipment with high magnification like FESEM will provide better overview of the samples. It also has a unique in-lens detector for clear topographic imaging in high vacuum mode.
The possible interactions between the components in the BC composites were evaluated by X-ray diffractometer (XRD) and Fourier transform infrared spectroscopy (FTIR). XRD is routinely employed to characterize the phase composition and per cent crystallinity for organic or inorganic materials. It is important to study the crystallinity patterns of BC and BC composites since it influence many of its characteristics including tensile strength, opacity and its swelling ability.
FTIR identifies chemical bonds in a molecule by producing an infrared absorption spectrum. The spectra produce a profile of the sample, a distinctive molecular fingerprint that can be used to screen and scan samples for many different components. Therefore, FTIR is an effective analytical instrument for detecting functional groups in the BC composites and characterizing the difference between native BC.
Tensile properties specify in what way that the materials respond to forces applied in tension. A tensile test is an important test performed to define the modulus of elasticity, elongation and Young’s modulus of native BC and BC composites. In this test, the specimen is carefully prepared and loaded in a very controlled manner while measuring the applied load and the elongation of the specimen over some distance. The results from the testing are used for the selection of materials for various purposes and application considering how it will react under different types of forces.
4 Application of Bacterial Cellulose Nanocomposites in Different Fields
Modification of BC had been done to enhance the properties of native BC and impart some additional properties for certain specific application. A variety of additives materials that had been used lead to the development of many new composite materials design for application on different fields.
4.1 Application in Biomedical Field
The modification of the bacterial cellulose can occur during or after fermentation by introducing selected bioactive material as additive in order to produce bacterial cellulose nanocomposites. This unique nanostructured matrix of composite materials are widely used in biomedical applications (Liyaskina et al. 2017). Modifying bacterial cellulose results in a composite material with better properties such as good mechanical properties and high moisture-keeping properties. These features make modified bacterial cellulose an excellent dressing material for treating different kinds of wounds, burns and ulcers. BC and BC nanocomposites are mostly used in the medical field, including wound healing materials (Legeza et al. 2004; Ul-Islam et al. 2012; Kim et al. 2011), artificial skin, blood vessels (Klemm et al. 2001; Charpentier et al. 2006; Arias et al. 2016), scaffolds for tissue engineering (Watanabe et al. 1998) and drug delivery (Amin et al. 2012; Müller et al. 2013). Table 4 lists different uses of BC nanocomposites in the medical field.
4.2 Application in Electronic Device
The uses of BC nanocomposites have been explored as conducting materials for application in electric and electronic devices. These include sensors, electronic, papers and display devices (Muller et al. 2012; Lee et al. 2012). Previous researchers have revealed the production of BC nanocomposites with conducting properties with addition of CNTs (Yoon et al. 2006), graphene (Feng et al. 2012) and graphite nanoplatelets (Zhou et al. 2013) which are well known as conducting nanomaterial. One of the examples of BC conducting polymer composite was prepared by Muller et al. (2011). In their research, pyrrole was added into BC matrix through in situ oxidative polymerization resulted to 1 S/cm conductance for the composites. Another BC conducting composite was also successfully produced from polyaniline (Pa’e et al. 2013; Muller et al. 2011). The BC–polyaniline nanocomposites prepared were reported to have good conducting properties, thus suitable for the uses as electronic device such as sensor, signal receiver and fuel cell and to guide enviable cell function for tissue engineering applications. Table 5 shows some application of BC nanocomposites in electric and electronic fields.
4.3 Application in Wastewater Treatment
In wastewater treatment, BC with its unique properties is suitable to be used as adsorbent for heavy metal removal (Wang et al. 2015; Lu et al. 2010). This includes high tensile strength, porous structure and high water-holding capacity. Besides that, BC has large surface area with many hydroxyl groups in the chain that make it effective for the separation of heavy metals ions (Lu et al. 2013). Most importantly, BC is simple to produce and the modification can be done using various materials and methods. Furthermore, the physiochemical properties of this BC can be controlled by changes in growth conditions or by chemical modification to obtain desired functionality (Pa’e et al. 2011; Sokolnicki et al. 2006; Serafica et al. 2002). These features along with its biocompatibility and low production make BC ideal to be used as eco-friendly biosorbent for heavy metal removal.
However, the BC itself as biosorbent has several disadvantages such as low adsorption capacity, poor selectivity and high hydrophilicity which makes it swell easily in water. Therefore, new functional group is added to BC to modify its properties and improve the activity of BC on adsorption of heavy metal ions in wastewater treatment. Table 6 lists the previous works on the use of BC as biosorbent for heavy metal removal.
5 Conclusion
BC is a unique material; hence, research and development in BC has expanded fast. This expansion of the BC exploration is due to several advantages such as BC’s properties which are suitable for numerous application, easy production process and economical viability. From the foregoing review of the literature, the progress in BC studies continues with its application as nanocomposites. Various synthetic and organic materials were added in situ or ex situ to produce nanocomposites materials with certain properties. At present, the focus is essentially moving towards the reasonable uses of BC and BC nanocomposites as a based material for various medical devices, electronic devices and biosorbent for heavy metal removal. Various uses in many fields are expected to increase the demand on BC and BC nanocomposites production. Thus, efforts that focus on large-scale production need to be highlighted next.
References
Aleshin AN, Berestennikov AS, Krylov PS, Shcherbakov IP, Petrov VN, Trapeznikova IN, Mamalimov RI, Khripunov AK, Tkachenkob AA (2015) Electrical and optical properties of bacterial cellulose films modified with conductive polymer PEDOT/PSS. Synth Met 199:147–151
Amin MCM, Abadi AG, Ahmad N, Katas H, Jamal JA (2012) Bacterial cellulose film coating as drug delivery system: physicochemical, thermal and drug release properties. Sains Malaysiana 41(5):561–568
Arias SL, Shetty AR, Senpan A, Echeverry-Rendón M, Reece LM, JAllain JP (2016) Fabrication of a functionalized magnetic bacterial nanocellulose with iron oxide nanoparticles. J Vis Exp 26:111
Ashori A, Sheykhnazari S, Tabarsa T, Shakeri A, Golalipour M (2012) Bacterial cellulose/silica nanocomposites: preparation and characterization. Carbohydr Polym 90:413–418
Barud HGO, Barud HS, Cavicchioli M, do Amaral TS, de Oliveira Junior OB, Santos DM, de Oliveira Almeida Petersen AL, Celes F, Borges VM, de Oliveira CI, de Oliveira PF, Furtado RA, Tavares DC, Ribeiro SJL (2016) Preparation and characterization of a bacterial cellulose/silk fibroin sponge scaffold for tissue regeneration. Carbohydr Polym 128:41–51
Bertocchi C, Delneri D, Signore S, Weng Z, Bruschi CV (1997) Characterization of microbial cellulose from a high-producing mutagenized Acetobacter pasteurianus strain. Biochem Biophys Acta 1336:211–217
Budhiono A, Rosidi B, Taher H, Iguchi M (1999) Kinetics aspects of bacterial cellulose formation in nata de coco culture system. Carbohydr Polym 40:137–143
Busuioc C, Stroescu M, Stoica-Guzun A, Voicu G, Jinga SI (2016) Fabrication of 3D calcium phosphates based scaffolds using bacterial cellulose as template. Ceram Int 42(14):15449–15458
Charpentier PA, Maguire A, Wan WK (2006) Surface modification of polyester to produce a bacterial cellulose-based vascular prosthetic device. Appl Surf Sci 252:6360–6367
Chen S, Zou Y, Yan Z, Shen W, Shi S, Zhang X, Wang H (2009) Carboxymethylated-bacterial cellulose for copper and lead ion removal. J Hazard Mater 161:1355–1359
Cheng KC, Catchmark JM, Demirci A (2009) Enhanced production of bacterial cellulose by using a biofilm reactor and its material property analysis. J Biol Eng 3(12)
Czaja W, Romanovicz D, Brown RM Jr (2004) Structural investigations of microbial cellulose produced in stationary and agitated culture. Cellulose 11(3):401–411
Czaja W, Krystynowicz A, Bielecki S, Brown RM Jr (2006) Microbial cellulose—the natural power to heal wound. Biomaterials 27:145–151
da Silva R, Sierakowski MR, Bassani HP, Zawadzki SF, Pirich CL, Ono L, de Freitas RA (2016) Hydrophilicity improvement of mercerized bacterial cellulose films by polyethylene glycol. Int J Biol Macromol 86:599–605
Dayal MS, Catchmark JM (2016) Mechanical and structural property analysis of bacterial cellulose composites. Carbohydr Polym 144:447–453
De Wulf P, Joris K, Vandamme EJ (1996) Improved cellulose formation by an Acetobacter xylinum mutant limited in keto gluconate synthesis. J Chem Technol Biotechnol 67:665–672
Evans BR, O’Neill HM, Malyvanh VP, Lee I, Woodward J (2003) Palladium-bacterial cellulose membranes for fuel cells. Biosens Bioelectron 18:917–923
Feng Y, Zhanga X, Shena Y, Yoshino K, Feng W (2012) A mechanically strong, flexible and conductive film based on bacterial cellulose/graphene nanocomposite. Carbohydr Polym 87:644–649
Fijałkowski K, Żywicka A, Drozd R, Niemczyk A, Junka AF, Peitler D, Kordas M, Konopacki M, Szymczyk P, Fray ME, Rakoczy R (2015) A modification of bacterial cellulose through exposure to the rotating magnetic field. Carbohydr Polym 133:52–60
Foresti ML, Vázquez A, Boury B (2017) Applications of bacterial cellulose as precursor of carbon and composites with metal oxide, metal sulfide and metal nanoparticles: a review of recent advances. Carbohydr Polym 157:447–467
Fu L, Zhang Y, Zhang J, Yang G (2011) Bacterial cellulose for skin repair materials. In: Fazel-Rezai R (ed) Biomedical engineering—frontiers and challenges. In-Tech.Rijeka, Croatia
Gao C, Yan T, Du J, He F, Luo H, Wan Y (2014) Introduction of broad spectrum antibacterial properties to bacterial cellulose nanofibers via immobilising ε-polylysine nanocoatings. Food Hydrocolloids 36:204–211
Gelin K, Bodin A, Gatenholm P, Mihranyan A, Edwards K, Strømme M (2007) Characterization of water in bacterial cellulose using dielectric spectroscopy and electron microscopy. Polymer 48(26):7623–7631
George J, Kumar R, Sajeevkumar VA, Ramana KV, Rajamanickam R, Abhishek V, Nadanasabapathy SS (2014) Hybrid HPMC nanocomposites containing bacterial cellulose nanocrystals and silver nanoparticles. Carbohydr Polym 105:285–292
Gindl W, Keckes J (2004) Tensile properties of cellulose acetate butyrate composites reinforced with bacterial cellulose. Compos Sci Technol 64(15):2407–2413
González-Sánchez C, Martínez-Aguirre A, Pérez-García B, Martínez-Urreaga J, de la Orden MU, Fonseca-Valero C (2014) Use of residual agricultural plastics and cellulose fibers for obtaining sustainable eco-composites prevents waste generation. J Clean Prod 83:228–237
Gutierrez J, Tercjak A, Algar I, Retegi A, Mondragon I (2012) Conductive properties of TiO2/bacterial cellulose hybrid fibres. J Colloid Interface Sci 377(1):88–93
Hestrin S, Schramm M (1954) Synthesis of cellulose by Acetobacter xylinum. 2. Preparation of freeze-dried cells capable of polymerizing glucose to cellulose. Biochem J 58(2):345–352
Horii F, Yamamoto H, Hirai A (1997) Microstructural analysis of microfibrils of bacterial cellulose. Macromol Symp 120:197–205
Hsieh JT, Wang MJ, Lai JT, Liu HS (2016) A novel static cultivation of bacterial cellulose production by intermittent feeding strategy. J Taiwan Inst Chem Eng 63:46–51
Hu W, Chen S, Yang Z, Liu L, Wang H (2011) Flexible electrically conductive nanocomposite membrane based on bacterial cellulose and polyaniline. J Phys Chem B 115:8453–8845
Hwang JW, Yang YK, Hwang JK, Pyun YR, Kim YS (1999) Effects of pH and dissolved oxygen on cellulose production by Acetobacter xylinum BRCS in agitated culture. J Biosci Bioeng 88(2):183–188
Iguchi M, Huang HC, Chen LC, Lina SB, Chen HH (2011) Nano-biomaterials application: In situ modification of bacterial cellulose structure by adding HPMC during fermentation. Carbohydr Polym 83:979–987
Jeon S, Yoo YM, Park JW, Kim HJ, Hyun J (2014) Electrical conductivity and optical transparency of bacterial cellulose based composite by static and agitated methods. Curr Appl Phys 14(12):1621–1624
Jonas R, Farah LF (1997) Production and application of microbial cellulose. J Polym Degrad Stab 59:101–106
Juncu G, Stoica-Guzun A, Stroescu M, Isopencu G, Jinga SI (2015) Drug release kinetics from carboxymethyl cellulose-bacterial cellulose composite films. Int J Pharm 510(2):485–492
Khairul AZ, Norhayati P, Ida IM (2016) An evaluation of fermentation period and discs rotation speed of rotary discs reactor for bacterial cellulose production. Sains Malaysiana 45(3):393–400
Kim SY, Kim JN, Wee YJ, Park DH, Ryu HW (2006) Production of bacterial cellulose by Gluconacetobacter sp. RKY5 isolated from persimmon vinegar. Appl Biochem Biotechnol 129–132:705–715
Kim J, Cai Z, Lee HS, Choi GS (2011) Preparation and characterization of a bacterial cellulose/chitosan composite for potential biomedical application. J Polym Res 18:739–744
Kirdponpattara S, Khamkeaw A, Sanchavanakit N, Pavasant P, Phisalaphong M (2015) Structural modification and characterization of bacterial cellulose–alginate composite scaffolds for tissue engineering. Carbohydr Polym 132:146–155
Kiziltas EE, Kiziltas A, Rhodes K, Emanetoglu NW, Blumentritt M, Gardner DJ (2016) Electrically conductive nano graphite-filled bacterial cellulose composites. Carbohydr Polym 136:1144–1151
Klemm D, Schumann D, Udhardt U, Marsch S (2001) Bacterial synthesized cellulose—artificial blood vessels for microsurgery. Prog Polym Sci 26:1561–1603
Lee RL, Paul JW, Willem HZ, Isak SP (2002) Microbial cellulose utilization: fundamentals and biotechnology. J Microbiol Mol Biol Rev 66(3):506–577
Lee BH, Kim HJ, Yang HS (2012) Polymerization of aniline on bacterial cellulose and characterization of bacterial cellulose/polyaniline nanocomposite films. Curr Appl Phys 12:75–80
Legeza VI, Galenko-Yaroshevskii VP, Zinov’ev EV, Paramonov BA (2004) Effects of new wound dressings on healing of thermal burns of the skin in acute radiation disease. Bull Exp Biol Med 138:311–315
Li Z, Zhu BJ, Yang JX, Peng K, Zhou BH, Xu RQ, Hu WL, Chen SY, Wang HP (2011) Method for manufacture of bacterial cellulose hydrogel cold pack. CN Patent No 201020239963.4
Lin WC, Lien CC, Yeh HJ, Yu CM, Hsu SH (2013) Bacterial cellulose and bacterial cellulose–chitosan membranes for wound dressing applications. Carbohydr Polym 94(1):603–611
Liyaskina E, Revin V, Paramonova E, Nazarkina M, Pestov N, Revina N, Kolesnikova S (2017) Nanomaterials from bacterial cellulose for antimicrobial wound dressing. J Phys Conf Ser 784:(1)
Lu M, Li YY, Guan XH, Wei DZ (2010) Preparation of bacterial cellulose and its adsorption of Cd2+. J Northeast Univ 31(8):1196–1199
Lu M, Guan XH, Xu X, Wei D (2013) Characteristic and mechanism of Cr(VI) adsorption by ammonium sulfamate-bacterial cellulose in aqueous solutions. Chin Chem Lett 24:253–256
Lu M, Zhang YM, Guan XH, Xu X, Gao T (2014) Thermodynamics and kinetics of adsorption for heavy metal ions from aqueous solutions onto surface amino-bacterial cellulose. Trans Nonferrous Metals Soc China 24:1912–1917
Luo H, Ao H, Li G, Li W, Xiong G, Zhu Y, Wan Y (2017) Advanced nano- and bio-materials: a pharmaceutical approach bacterial cellulose/graphene oxide nanocomposite as a novel drug delivery system. Curr Appl Phys 17(2):249–254
Maneerung T, Tokura S, Rujiravanit R (2008) Impregnation of silver nanoparticles into bacterial cellulose for antimicrobial wound dressing. Carbohydr Polym 72:43–51
Martínez-Sanz M, Lopez-Rubio A, Lagaron JM (2013) High-barrier coated bacterial cellulose nanowhiskers films with reduced moisture sensitivity. Carbohydr Polym 98:1072–1082
Mormino R, Bungay H (2003) Composites of bacterial cellulose and papermade with a rotating disk bioreactor. Appl Microbiol Biotechnol 62:503–506
Muller D, Rambo CR, Recouvreux DOS, Porto LM (2011) Chemical in situ polymerization of polypyrrole on bacterial cellulose nanofibers. Synth Met 161:106–111
Muller D, Mandelli JS, Marins JA, Soares BG (2012) Electrically conducting nanocomposites: preparation and properties of polyaniline (PAni)-coated bacterial cellulose nanofibers (BC). Cellulose 19:1645–1654
Müller A, Ni Z, Hessler N, Wesarg F, Müller FA, Kralisch D, Fischer D (2013) The biopolymer bacterial nanocellulose as drug delivery system: investigation of drug loading and release using the model protein albumin. J Pharm Sci 102:579–592
Nakayama A, Kakugo A, Gong JP, Osada Y, Takai M, Erata T, Kawano S (2004) High mechanical strength double-network hydrogel with bacterial cellulose. Adv Funct Mater 14:1124–1128
Naritomi T, Kouda T, Yano H, Yoshinaga F (1998) Effect of ethanol on bacterial cellulose production from fructose in continuous culture. J Ferment Bioeng 85(6):598–603
O’Connell DW, Birkinshaw C, O’Dwyer TF (2008) Heavy metal adsorbents prepared from the modification of cellulose: a review. Biores Technol 99:6709–6724
Pa’e N (2009) Rotary discs reactor for enhanced production microbial cellulose. Master thesis, Universiti Teknologi Malaysia, Skudai
Pa’e N, Zahan KA, Muhamad II (2011) Production of biopolymer from acetobacter xylinum using different fermentation methods. Int J Eng Technol (IJET-IJEN) 11(5):90–98
Pa’e N, Zahan KA, Muhamada II, Kok FS (2013) Modified fermentation for production of bacterial cellulose/polyaniline as conductive biopolymer material. Jurnal Teknologi 62(2):21–23
Park M, Cheng J, Choi J, Kim J, Hyun J (2013) Electromagnetic nanocomposite of bacterial cellulose using magnetite nanoclusters and polyaniline. Colloids Surf B 102:238–242
Pavaloiu RD, Stoica-Guzun A, Stroescu M, Jinga SI, Dobre T (2014) Composite films of poly(vinyl alcohol)–chitosan–bacterial cellulose for drug controlled release. Int J Biol Macromol 68:117–124
Paximada P, Dimitrakopoulou EA, Tsouko E, Koutinas AA, Fasseas C, Mandala IG (2016) Structural modification of bacterial cellulose fibrils under ultrasonic irradiation. Carbohydr Polym 150:5–12
Pei Y, Yang J, Liu P, Xu M, Zhang X, Zhang L (2013) Fabrication, properties and bioapplications of cellulose/collagen hydrolysate composite films. Carbohydr Polym 92:1752–1760
Piccinno F, Hischier R, Saba A, Mitrano D, Seeger S, Som C (2015) Multi-perspective application selection: a method to identify sustainable applications for new materials using the example of cellulose nanofiber reinforced composites. J Clean Prod 112:1199–1210
Pircher N, Veigel S, Aignea N, Nedelecc JM, Rosenau T, Liebner F (2014) Reinforcement of bacterial cellulose aerogels with biocompatible polymers. Carbohydr Polym 111:505–513
Ruka DR, Simon GP, Deana KM (2013) In situ modifications to bacterial cellulose with the water insoluble polymer poly-3-hydroxybutyrate. Carbohydr Polym 92:1717–1723
Sai H, Fu R, Xing L, Xiang J, Li Z, Li F, Zhang T (2015) Surface modification of bacterial cellulose aerogels web-like skeleton for oil/water separation. ACS Appl Mater Interfaces 7(13):7373–7381
Saibuatong O, Phisalaphong M (2010) Novo aloe vera—bacterial cellulose composite film from biosynthesis. Carbohydr Polym 79:455–460
Salehudin MH, Salleh E, Muhamad II, Mamat SNH (2014) Starch-based biofilm reinforced with empty fruit bunch cellulose nanofibre. Mater Res Innovations 18:322–325
Schramm M, Hestrin S (1954) Factors affecting production of cellulose at the air/liquid interface of a culture of Acetobacter xylinum. Microbiology 11:123–129
Serafica G, Mormino R, Bungay H (2002) Inclusion of solid particles in bacterial cellulose. Appl Microbiol Biotechnol 58:756–760
Shah J, Brown RM Jr (2005) Towards electronic paper displays made from microbial cellulose. Appl Microbiol Biotechnol 66(4):352–355
Shah N, Ul-Islam M, Khattak WA, Park JK (2013) Overview of bacterial cellulose composites: a multipurpose advanced material. Carbohydr Polym 98:1585–1598
Shanshan G, Jianqing W, Zhengwei J (2012) Preparation of cellulose films from solution of bacterial cellulose in NMMO. Carbohydr Polym 87(2):1020–1025
Shirai A, Takahashi M, Kaneko H, Nishimura S, Ogawa M, Nishi N, Tokura S (1994) Biosynthesis of a novel polysaccharide by Acetobacter xylinum. Int J Biol Macromol 16(6):297–300
Slavutsky MA, Bertuzzi MA (2014) Water barrier properties of starch films reinforced with cellulosenanocrystals obtained from sugarcane bagasse. Carbohydr Polym 110:53–61
Sokolnicki AM, Fisher RJ, Harrah TP, Kaplan DL (2006) Permeability of bacterial cellulose membranes. J Membr Sci 272(1–2):15–27
Son HJ, Heo MS, Kim YG, Lee SJ (2001) Optimization of fermentation conditions for the production of bacterial cellulose by a newly isolated Acetobacter sp. A9 in shaking cultures. Biotechnol Appl Biochem 33(1):1–3
Son HJ, Kim HG, Kim KK, Kim HS, Kim YG, Lee SJ (2003) Increased production of bacterial cellulose by Acetobacter sp. V6 in synthetic media under shaking culture conditions. Biores Technol 86(3):215–219
Tang W, Jia S, Jia Y, Yang H (2010) The influence of fermentation conditions and post-treatment methods on porosity of bacterial cellulose membrane. World J Microbiol Biotechnol 26:125–131
Toru S, Kazunori T, Masaya K, Tetsuya M, Takaaki N, Shingeru M, Kenji K (2005) Cellulose production from glucose using a glucose dehydrogenase gene (gdh)-deficient mutant of Gluconacetobacter xylinus and its use for bioconversion of sweet potato pulp. J Biosci Bioeng 99(4):415–422
Tsuchida T, Yoshinaga F (1997) Production of bacterial cellulose by agitation culture system. J Pure Appl Chem 69(11):2453–2458
Tyagi N, Suresh S (2015) Production of cellulose from sugarcane molasses using Gluconacetobacter intermedius SNT-1: optimization & characterization. J Clean Prod 112:71–80
Ul-Islam M, Khan T, Park JK (2012) Nanoreinforced bacterial cellulose–montmorillonite composites for biomedical applications. Carbohydr Polym 89(4):1189–1197
Ummartyotin S, Juntaro J, Sain M, Manuspiya H (2012) Development of transparent bacterial cellulose nanocomposite film as substrate for flexible organic light emitting diode (OLED) display. Ind Crops Prod 35:92–97
Vandamme EJ, De Baets S, Vanbaelen A, Joris K, De Wulf P (1998) Improved production of bacterial cellulose and its application potential. Polym Degrad Stab 59:93–99
Wang J, Lu X, Ng PF, Lee K, Fei B, Xin JH, Wu J (2015) Polyethylenimine coated bacterial cellulose nanofiber membrane and application as adsorbent and catalyst. J Colloid Interface Sci 440:32–38
Watanabe K, Tabuchi M, Morinaga Y, Yoshinaga F (1998) Structural features and properties of bacterial cellulose produced in agitated culture. Cellulose 5:187–200
Wu YB, Yu SH, Mi FL, Wu CW, Shyu SS, Peng CK, Chao AC (2004) Preparation and characterization on mechanical and antibacterial properties of chitosan/cellulose blends. Carbohydr Polym 57(4):435–440
Wu J, Zheng Y, Song W, Luan J, Wen X, Wu Z, Chen X, Wang Q, Guo S (2014) In situ synthesis of silver-nanoparticles/bacterial cellulose composites for slow-released antimicrobial wound dressing. Carbohydr Polym 102:762–771
Yamanaka S, Watanabe K, Kitamura N, Iguchi M, Mitsuhashi S, Nishi Y, Uryu M (1989) The structure and mechanical properties of sheets prepared from bacterial cellulose. J Mater Sci 24:3141–3145
Yan Z, Chen S, Wang H, Wang B, Jiang J (2008) Biosynthesis of bacterial cellulose/multi-walled carbon nanotubes in agitated culture. Carbohydr Polym 74:659–665
Yang G, Xie J, Hong F, Cao Z, Yang X (2012) Antimicrobial activity of silver nanoparticle impregnated bacterial cellulose membrane: effect of fermentation carbon sources of bacterial cellulose. Carbohydr Polym 87:839–845
Yoon SH, Jin HJ, Kook MC, Pyun YR (2006) Electrically conductive bacterial cellulose by incorporation of carbon nanotubes. Biomacromolecules 7:1280–1284
Zahan KA, Pa’e N, Muhamad II (2014) Process parameter for fermentation in rotary discs reactor for optimum microbial cellulose production using response surface methodology. Bioresources 9(2):1858–1872
Zhang Z, Zhang J, Zhao X, Yang F (2015) Core-sheath structured porous carbon nanofiber composite anode material derived from bacterial cellulose/polypyrrole as an anode for sodium-ion batteries. Carbon 95:552–559
Zhang F, Tang Y, Yang Y, Zhang X, Lee CS (2016) In-situ assembly of three-dimensional MoS2 nanoleaves/carbon nanofiber composites derived from bacterial cellulose as flexible and binder-free anodes for enhanced lithium-ion batteries. Electrochim Acta 211:404–410
Zhou T, Chen D, Jiu J, Nge TT (2013) Electrically conductive bacterial cellulose composite membranes produced by the incorporation of graphite nanoplatelets in pristine bacterial cellulose membranes. Express Polym Lett 7:756–766
Zhu H, Jia S, Wan T, Jia Y, Yang H, Li J, Yan L, Zhong C (2011) Biosynthesis of spherical Fe3O4/Bacterial cellulose nanocomposites as adsorbents for heavy metal ions. Carbohydr Polym 86:1558–1564
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pa’e, N., Muhamad, I.I., Hashim, Z., Yusof, A.H.M. (2019). Bacterial Cellulose Nanocomposites. In: Sanyang, M., Jawaid, M. (eds) Bio-based Polymers and Nanocomposites . Springer, Cham. https://doi.org/10.1007/978-3-030-05825-8_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-05825-8_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-05824-1
Online ISBN: 978-3-030-05825-8
eBook Packages: EngineeringEngineering (R0)