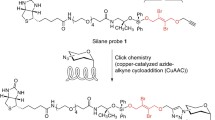
Abstract
Glycosylation is an abundant post-translational modification that alters the fate and function of its substrate proteins. To aid in understanding the significance of protein glycosylation, identification of target proteins is key. As with all proteomics experiments, mass spectrometry has been established as the desired method for substrate identification. However, these approaches require selective enrichment and purification of modified proteins. Chemical reporters in combination with bioorthogonal reactions have emerged as robust tools for identifying post-translational modifications including glycosylation. We provide here a method for the use of bioorthogonal chemical reporters for isolation and identification of glycosylated proteins. More specifically, this protocol is a representative procedure from our own work using an alkyne-bearing O-GlcNAc chemical reporter (GlcNAlk) and a chemically cleavable azido-azo-biotin probe for the identification of O-GlcNAc-modified proteins.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zielinska DF, Gnad F, Wisniewski JR, Mann M (2010) Precision mapping of an in vivo N-glycoproteome reveals rigid topological and sequence constraints. Cell 141(5):897–907
Teo CF et al (2010) Glycopeptide-specific monoclonal antibodies suggest new roles for O-GlcNAc. Nat Chem Biol 6(5):338–343
Saxon E, Bertozzi CR (2000) Cell surface engineering by a modified Staudinger reaction. Science 287(5460):2007–2010
Hang HC, Yu C, Kato DL, Bertozzi CR (2003) A metabolic labeling approach toward proteomic analysis of mucin-type O-linked glycosylation. Proc Natl Acad Sci USA 100(25):14846–14851
Dehnert KW et al (2011) Metabolic labeling of fucosylated glycans in developing zebrafish. ACS Chem Biol 6(6):547–552
Dube DH, Prescher JA, Quang CN, Bertozzi CR (2006) Probing mucin-type O-linked glycosylation in living animals. Proc Natl Acad Sci USA 103(13):4819–4824
Zaro BW, Bateman LA, Pratt MR (2011) Robust in-gel fluorescence detection of mucin-type O-linked glycosylation. Bioorg Med Chem Lett 21(17):5062–5066
Hanson SR et al (2007) Tailored glycoproteomics and glycan site mapping using saccharide-selective bioorthogonal probes. J Am Chem Soc 129(23):7266–7267
Boyce M et al (2011) Metabolic cross-talk allows labeling of O-linked {beta}-N-acetylglucosamine-modified proteins via the N-acetylgalactosamine salvage pathway. Proc Natl Acad Sci USA 108(8):3141–3146
Zaro BW, Yang Y-Y, Hang HC, Pratt MR (2011) Chemical reporters for fluorescent detection and identification of O-GlcNAc-modified proteins reveal glycosylation of the ubiquitin ligase NEDD4-1. Proc Natl Acad Sci USA 108(20):8146–8151
Sprung R et al (2005) Tagging-via-substrate strategy for probing O-GlcNAc modified proteins. J Proteome Res 4(3):950–957
Nandi A et al (2006) Global identification of O-GlcNAc-modified proteins. Anal Chem 78(2):452–458
Clark PM et al (2008) Direct in-gel fluorescence detection and cellular imaging of O-GlcNAc-modified proteins. J Am Chem Soc 130(35):11576–11577
Wang Z et al (2010) Enrichment and site mapping of O-linked N-acetylglucosamine by a combination of chemical/enzymatic tagging, photochemical cleavage, and electron transfer dissociation mass spectrometry. Mol Cell Proteomics 9(1):153–160
Wang Z et al (2010) Extensive crosstalk between O-GlcNAcylation and phosphorylation regulates cytokinesis. Science STKE 3(104):ra2
Yang Y-Y, Ascano JM, Hang HC (2010) Bioorthogonal chemical reporters for monitoring protein acetylation. J Am Chem Soc 132(11):3640–3641
Fonović M, Verhelst SHL, Sorum MT, Bogyo M (2007) Proteomics evaluation of chemically cleavable activity-based probes. Mol Cell Proteomics 6(10):1761–1770
Verhelst SHL, Fonović M, Bogyo M (2007) A mild chemically cleavable linker system for functional proteomic applications. Angew Chem Int Ed 46(8):1284–1286
Denny JB, Blobel G (1984) 125I-labeled crosslinking reagent that is hydrophilic, photoactivatable, and cleavable through an azo linkage. Proc Natl Acad Sci USA 81(17):5286–5290
Gurcel C et al (2008) Identification of new O-GlcNAc modified proteins using a click-chemistry-based tagging. Anal Bioanal Chem 390(8):2089–2097
Saxon E et al (2002) Investigating cellular metabolism of synthetic azidosugars with the Staudinger ligation. J Am Chem Soc 124(50):14893–14902
Rabuka D, Hubbard SC, Laughlin ST, Argade SP, Bertozzi CR (2006) A chemical reporter strategy to probe glycoprotein fucosylation. J Am Chem Soc 128(37):12078–12079
Hsu T-L et al (2007) Alkynyl sugar analogs for the labeling and visualization of glycoconjugates in cells. Proc Natl Acad Sci USA 104(8):2614–2619
Goon S, Schilling B, Tullius MV, Gibson BW, Bertozzi CR (2003) Metabolic incorporation of unnatural sialic acids into Haemophilus ducreyi lipooligosaccharides. Proc Natl Acad Sci USA 100(6):3089–3094
Acknowledgments
We thank Leslie Bateman for careful reading of the manuscript. This work was supported by the University of Southern California and the American Cancer Society Grant IRG-58-007-51 (to M.R.P.) and the Ellison Medical Foundation and the National Institutes of Health and General Medical Sciences Grant 1RO1GM087544 O1A2 (to H.C.H.).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Zaro, B.W., Hang, H.C., Pratt, M.R. (2013). Incorporation of Unnatural Sugars for the Identification of Glycoproteins. In: Kohler, J., Patrie, S. (eds) Mass Spectrometry of Glycoproteins. Methods in Molecular Biology, vol 951. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-146-2_5
Download citation
DOI: https://doi.org/10.1007/978-1-62703-146-2_5
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-145-5
Online ISBN: 978-1-62703-146-2
eBook Packages: Springer Protocols