Abstract
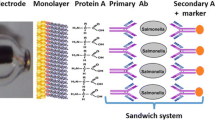
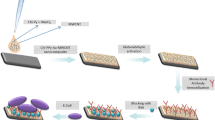
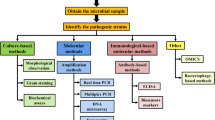
In this chapter methods and protocols for surfaces adapted to electrochemical impedance detection, antibody binding, electrolyte couples used, and instrumentation for EIS Biosensing are presented. Various technical bottlenecks have been overcome in recent years. Other limitations still present in this technique are discussed. We present the most recent applications in food pathogen detection based on EIS methods, as well as using other antibody-based platforms.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Katz E, Willner I (2003) Probing biomolecular interactions at conductive and semiconductive surfaces by impedance spectroscopy: routes to impedimetric immunosensors, DNA sensors and enzyme biosensors. Electroanalysis 15:913–947
Brett CMA, Oliveira Brett AM, Serrano SHP (1999) EIS study of DNA-modified electrodes. Electrochim Acta 44:4233–4239
Davis F, Nabok AV, Seamus PJ (2005) Species differentiation by DNA-modified carbon electrodes using AC impedimetric approach. Biosens Bioelectron 20:1531–1538
Cai W, Peck JR, van der Weide DW et al (2004) Direct electrical detection of hybridization at DNA-modified silicon surface. Biosens Bioelectron 19:1013–1019
Yang WS, Butler JE, Russell JN et al (2007) Direct electrical detection of antibody-antigen binding on diamond and silicon substrates using electrical impedance spectroscopy. Analyst 132:296–306
De Silva MS, Zhang Y, Hesketh PJ et al (1995) Impedance based sensing of the specific binding reaction between Staphylococcus enterotoxin B and its antibody on an ultrathin Pt film. Biosens Bioelectron 10:675–682
Pak SC, Penrose W, Hesketh PJ (2001) An ultrathin platinum film sensor to measure biomolecular binding. Biosens Bioelectron 16:371–379
Mantzila AG, Prodromidis MI (2005) Performance of impedimetric biosensors based on anodically formed Ti/TiO2 electrodes. Electroanalysis 17(20):1878–1885
Mantzila AG, Prodromidis MI (2006) Development and study of anodic Ti/TiO2 electrodes and their potential use as impedimetric immunosensors. Electrochim Acta 51:3537–3542
Ruan CM, Yang L, Li YB (2002) Immunobiosensor chips for detection of Escherichia coli O157:H57 using electrochemical impedance spectroscopy. Anal Chem 74:4814–4820
Corry B, Janelle U, Crawley C (2003) Probing direct binding affinity in electrochemical antibody-based sensors. Anal Chim Acta 496:103–116
Blankespoor R, Limoges B, Shollhorn B et al (2005) Dense monolayers of metal-chelating ligands covalently attached to carbon electrodes electrochemically and their useful application in affinity binding of histidine-tagged proteins. Langmuir 21:3362–3375
Teh HF, Gong H, Dong XD et al (2005) Electrochemical biosensing of DNA with capture probe covalently immobilized onto glassy carbon surface. Anal Chim Acta 551:23–29
Ramesh P, Sampath S (2003) Electrochemical characterization of binderless, recompressed exfoliated graphite electrodes: electron transfer kinetics and diffusion characteristics. Anal Chem 75:6949–6957
Huang Y, Suni II (2008) Degenerate Si as an electrode material for electrochemical biosensors. J Electrochem Soc 155:J350
Radhakrishnan R, Suni II (2016) Antibody regeneration on degenerate Si electrodes for calibration and reuse of impedance biosensors. Sens Biosensing Res 7:20–24
Schoning MJ, Tzarouchas D, Beckers L et al (1996) A highly long term stable silicon pH sensor fabricated by pulsed laser deposition technique. Sensors Actuators B Chem 35:228–233
HuayhuasChipana BC, Gomero JCM, Sotomayor MDPT (2014) Nanostructured screen-printed electrodes modified with self-assembled monolayers for determination of metronidazole in different matrices. J Braz Chem Soc 25:1737–1745
Kumar CSSR (2006) Nanomaterials for biosensors. Wiley-VCH, Weinheim, Germany
Lai RY, Seferos DS, Heeger AJ et al (2006) Comparison of the signaling and stability of electrochemical DNA sensors fabricated from 6- or 11-carbon self-assembled monolayers. Langmuir 22:10796–10800
Patel N, Davies MC, Hartshorne M et al (1997) Immobilization of protein molecules onto homogeneous and mixed carboxylate-terminated self-assembled monolayers. Langmuir 13:6485–6490
Ulman A (1996) Formation and structure of self-assembled monolayers. Chem Rev 96:1533–1554
Rickert J, Gopel W, Beck W et al (1996) A mixed self-assembled monolayer for an impedimetric immunosensors. Biosens Bioelectron 11:757–768
Steel AB, Levicky RL, Herne TM et al (2000) Immobilization of nucleic acids at solid surfaces: effect of oligonucleotide length on layer assembly. Biophys J 79:975–981
Patolsky F, Katz E, Bardea A et al (1999) Enzyme linked amplified electrochemical sensing of oligonucleotide DNA interactions by means of the precipitation of an insoluble product and using impedance spectroscopy. Langmuir 15:3703–3706
Bain CD, Troughton EB, Tao YT et al (1989) Formation of monolayer films by the spontaneous assembly of organic thiols from solution onto gold. J Am Chem Soc 111:321
Manickam A. 2012 Integrated Impedance Spectroscopy Biosensors. Ph.D. Thesis University of Texas, Austin
Poirier GE, Tarlov MJ, Rushmeier HE (1994) Two-dimensional liquid phase and the p √3 phase of alkanethiol self-assembled monolayers on Au(111). Langmuir 10:3383
Primiceri E, Chiriacò MS, De Feo F et al (2016) A multipurpose biochip for food pathogen detection. Anal Methods 8:3055–3060
Maupas H, Soldatkin AP, Martelet C et al (1997) Direct immunosensing using differential electrochemical measurements of impedimetric variations. J Electroanal Chem 421:165–171
Radhakrishnan R, Pali M, Lee HJ et al (2016) Impedance biosensor incorporating a carboxylate-terminated Bidentate Thiol for antibody immobilization. J Electrochem Soc 163:125–130
Dijksma M, Boukamp BA, Kamp B et al (2002) Effect of hexacyanoferrate(ii/iii) on self-assembled monolayers of thioctic acid and 11-mercaptoundecanoic acid on gold. Langmuir 18:3105
Homola J (2008) Surface Plasmon resonance sensors for detection of chemical and biological species. Chem Rev 108:462–493
Huang J, Hemminger JC (1993) Photooxidation of thiols in self-assembled monolayers on gold. J Am Chem Soc 115:3342–3343
Zamborini FP, Crooks RM (1997) In-situ electrochemical scanning Tunneling microscopy (ECSTM) study of cyanide-induced corrosion of naked and hexadecylmercaptan-passivated Au(111). Langmuir 13:122–126
Srimsombat L, Zhang S, Lee TR (2010) Thermal stability of mono-, Bis-, and Tris-chelating alkanethiol films assembled on gold nanoparticles and evaporated flat gold. Langmuir 26:41–46
Chinwangso P, Jamison AC, Lee TR (2011) Multidentate adsorbates for self-assembled monolayer films. Acc Chem Res 44:511–519
Lee HJ, Jamison AC, Yuan Y et al (2013) Robust carboxylic acid terminated organic thin films and nanoparticle protectants generated from bidentate alkanethiols. Langmuir 29:10432–10439
Huang Y, Bell MC, Suni II (2008) Impedance biosensor for peanut protein Ara h 1. Anal Chem 80:9157–9161
Radhakrishnan R, Poltronieri P (2017) Fluorescence-free biosensor methods in detection of food pathogens with a special focus on Listeria monocytogenes. Biosensors (Basel) 7:63
Cimaglia F, De Lorenzis E, Mezzolla V et al (2016) Detection of L. monocytogenes in enrichment cultures by immunoseparation and immunosensors. IEEE Sensors 16:7045–7052
Morgan H, Green NG (eds) (2003) AC electrokinetics: colloids and nanoparticles. Baldock. Research Studies Press, Philadelphia
Wang D, Sigurdson M, Meinhart CD (2005) Experimental analysis of particle and fluid motion in AC electrokinetics. Exp Fluids 38:1–10
Ahualli S, Jimenez ML, Carrique F et al (2009) AC electrokinetics of concentrated suspensions of soft particles. Langmuir 25:1986–1997
Wu J (2006) Biased AC electro-osmosis for on-chip bioparticle processing. IEEE Trans Nanotechnol 5:84–89
Wu J (2008) Interactions of electrical fields with fluids: laboratory-on-a-chip applications. IET Nanobiotechnol 2:14–27
Castellanos A, Ramos A, Gonzale A et al (2003) Electrohydrodynamics and dielectrophoresis in microsystems: scaling laws. J Phys D Appl Phys 36:2584
Liu X, Yang K, Wadhwa A et al (2011) Development of an AC electrokinetics-based immunoassay system for on-site serodiagnosis of infectious diseases. Sens Actuators, A 171:406–413
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Poltronieri, P., Primiceri, E., Radhakrishnan, R. (2019). EIS-Based Biosensors in Foodborne Pathogen Detection with a Special Focus on Listeria monocytogenes. In: Bridier, A. (eds) Foodborne Bacterial Pathogens. Methods in Molecular Biology, vol 1918. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9000-9_7
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9000-9_7
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-8999-7
Online ISBN: 978-1-4939-9000-9
eBook Packages: Springer Protocols