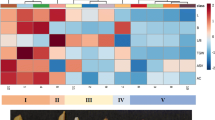
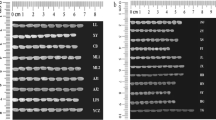
Abstract
Gas chromatograph coupled with mass spectrometer is widely used to profile volatiles and metabolites from the homogenized rice flour obtained from mature grains. Rice grains consist of central endosperm which stores majorly starch and, in addition, accumulate various storage proteins as storage reserves. The outer nutritious aleurone layer stores lipids, sugar alcohols, volatiles, antioxidants, vitamins, and various micronutrients. Once paddy sample is dehulled, milled, and ground cryogenically, the brown rice flour is subjected to extraction of primary metabolites and volatiles using an appropriate extraction method. In metabolite profiling of the liquid extract obtained from the rice sample, mixture is initially subjected to methoxyamination then silylation before being subjected to untargeted metabolite profiling. Peaks obtained are processed for noise reduction and specific signal selection. Volatile compounds are initially extracted using a solid phase adsorbent prior to analysis. All these compounds, metabolites, and volatiles are detected in the mass selective detector by fragmentation at 70 eV ionization energy and the resultant mass spectrum compared with a built-in library of compounds. Data mined from the gas chromatography mass spectrometry analysis are then subjected to post-processing statistical analysis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Sreenivasulu N, Wobus U (2013) Seed-development programs: a systems biology-based comparison between dicots and monocots. Annu Rev Plant Biol 64:189–217
Weckwerth W (2003) Metabolomics in systems biology. Annu Rev Plant Biol 54:669–689
Patti GJ, Yanes O, Siuzdak G (2012) Innovation: metabolomics: the apogee of the omics trilogy. Nat Rev Mol Cell Biol 13(4):263–269
Zhen S, Dong K, Deng X, Zhou J, Xu X, Han C, Zhang W, Xu Y, Wang Z, Yan Y (2016) Dynamic metabolome profiling reveals significant metabolic changes during grain development of bread wheat (triticum aestivum L.). J Sci Food Agric 96(11):3731–3740
Q-q Y, C-q Z, M-l C, D-s Z, J-z C, Wang Q, Li Q-f, H-x Y, Gu M-h, SS-m S, Q-q L (2016) Biofortification of rice with the essential amino acid lysine: molecular characterization, nutritional evaluation, and field performance. J Exp Bot 67:4285–4296
Tian JS, Liu CC, Xiang H, Zheng XF, Peng GJ, Zhang X, Du GH, Qin XM (2015) Investigation on the antidepressant effect of sea buckthorn seed oil through the GC-MS-based metabolomics approach coupled with multivariate analysis. Food Funct 6(11):3585–3592
Lisec J, Schauer N, Kopka J, Willmitzer L, Fernie AR (2006) Gas chromatography mass spectrometry–based metabolite profiling in plants. Nat Prot 1:387–396
Butardo VM Jr, Sreenivasulu N (2016) Tailoring grain storage reserves for a healthier rice diet and its comparative status with other cereals. Int Rev Cell Mol Biol 323:31–70
Calingacion MN, Boualaphanh C, Daygon VD, Anacleto R, Sackville-Hamilton NR, Biais B, Deborde C, Maucourt M, Moing A, Mumm R, de Vos RCH, Erban A, Kopka J, Hansen TH, Laursen KH, Schjoerring JK, Hall RD, Fitzgerald MA (2012) A genomics and multi-platform metabolomics approach to identify new traits of rice quality in traditional and improved varieties. Metabolomics 8:771–783
Kusano M, Fukushima A, Fujita N, Okazaki Y, Kobayashi M, Oitome NF, Ebana K, Saito K (2012) Deciphering starch quality of rice kernels using metabolite profiling and pedigree network analysis. Mol Plant 5:442–451
Bryant RJ, McClung AM (2011) Volatile profiles of aromatic and non-aromatic rice cultivars using SPME/GC-MS. Food Chem 124(2):501–513
Zeng Y-X, Hu C-Y, Lu Y-G, Li J-Q, Liu X-D (2009) Abnormalities occurring during female gametophyte development result in the diversity of abnormal embryo sacs and leads to abnormal fertilization in indica/japonica hybrids in rice. J Integr Plant Biol 51(1):3–12
Kim JK, Park S-Y, Lim S-H, Yeo Y, Cho HS, Ha S-H (2013) Comparative metabolic profiling of pigmented rice (Oryza sativa L.) cultivars reveals primary metabolites are correlated with secondary metabolites. J Cereal Sci 57:14–20
Matsuda F, Okazaki Y, Oikawa A, Kusano M, Nakabayashi R, Kikuchi J, J-i Y, Ebana K, Yano M, Saito K (2012) Dissection of genotype–phenotype associations in rice grains using metabolome quantitative trait loci analysis. The Plant J 70:624–636
Zhou XH, Dong Y, Xiao X, Wang Y, Xu Y, Xu B, Shi WD, Zhang Y, Zhu LJ, Liu QQ (2011) A 90-day toxicology study of high-amylose transgenic rice grain in Sprague–Dawley rats. Food Chem Toxic 49(12):3112–3118
Vu NT, Shimada H, Kakuta Y, Nakashima T, Ida H, Omori T, Nishi A, Satoh H, Kimura M (2008) Biochemical and crystallographic characterization of the starch branching enzyme I (BEI) from Oryza sativa L. Biosci Biotech Biochem 72(11):2858–2866
C-y L, Lai VMF, Shen M-C (2004) Changes in retrogradation properties of rice starches with amylose content and molecular properties. Cereal Chem 81(3):392–398
Calingacion M, Fang L, Quiatchon-Baeza L, Mumm R, Riedel A, Hall RD, Fitzgerald M (2015) Delving deeper into technological innovations to understand differences in rice quality. Rice 8:43
Annison G, Illman RJ, Topping DL (2003) Acetylated, propionylated or butyrylated starches raise large bowel short-chain fatty acids preferentially when fed to rats. J Nutr 133(11):3523–3528
Champagne ET, Bett-Garber KL, Grimm CC, McClung AM, Moldenhauer KA, Linscombe S, McKenzie KS, Barton FE (2001) Near-infrared reflectance analysis for prediction of cooked rice texture. Cereal Chem 78(3):358–362
Laguerre M, Mestres C, Davrieux F, Ringuet J, Boulanger R (2007) Rapid discrimination of scented rice by solid-phase microextraction, mass spectrometry, and mutivariate analysis used as a mass sensor. J Agric Food Chem 55:1077–1083
Redestig H, Kusano M, Ebana K, Kobayashi M, Oikawa A, Okazaki Y, Matsuda F, Arita M, Fujita N, Saito K (2011) Exploring molecular backgrounds of quality traits in rice by predictive models based on high-coverage metabolomics. BMC Syst Biol 5:176
Zhou J, Ma C, Xu H, Yuan K, Lu X, Zhu Z, Wu Y, Xu G (2009) Metabolic profiling of transgenic rice with cryIAc and sck genes: an evaluation of unintended effects at metabolic level by using GC-FID and GC-MS. J Chromatogr B: Analyt Technol Biomed Life Sci 877:725–732
Frank T, Meuleye BS, Miller A, Shu Q-Y, Engel K-H (2007) Metabolite profiling of two low phytic acid (lpa) rice mutants. J Agric Food Chem 55:11011–11019
Heuberger AL, Lewis MR, Chen M-H, Brick MA, Leach JE, Ryan EP (2010) Metabolomic and functional genomic analyses reveal varietal differences in bioactive compounds of cooked rice. PLoS One 5(9):e12915
Yang DS, Shewfelt RL, Lee K-S, Kays SJ (2008) Comparison of odor-active compounds from six distinctly different rice flavor types. J Agric Food Chem 56(8):2780–2787
Verhoeven HA, Jonker H, RCH DV, Hall RD (2012) Solid phase micro-extraction GC–MS analysis of natural volatile components in melon and Rice. In: Hardy NW, Hall RD (eds) Plant metabolomics: methods and protocols. Humana Press, Totowa, NJ, pp 85–99
Agilent Technologies, Inc (2013) Agilent fiehn GC/MS metabolomics RTL library a complete GC/MS solution for metabolite identification. Agilent Technologies
Soria AC, Sanz ML, Villamiel M (2009) Determination of minor carbohydrates in carrot (Daucus carota L.) by GC-MS. Food Chem 114:758–762
Ruiz-Matute AI, Hernández-Hernández O, Rodríguez-Sánchez S, Sanz ML, Martínez-Castro I (2011) Derivatization of carbohydrates for GC and GC–MS analyses. J Chromatogr B Analyt Technol Biomed Life Sci 879:1226–1240
Lou Q, Ma C, Wen W, Zhou J, Chen L, Feng F, Xu X, Lu X, Luo L, Mei H, Xu G (2011) Profiling and association mapping of grain metabolites in a subset of the core collection of Chinese rice Germplasm (Oryza sativa L.). J Agric Food Chem 59:9257–9264
Chang WT, Lin DL, Liu RH (2001) Isotopic analogs as internal standards for quantitative analyses by GC/MS—evaluation of cross-contribution to ions designated for the analyte and the isotopic internal standard. Forensic Sci Int 121(3):174–182
Chen M, Rao RSP, Zhang Y, Zhong CX, Thelen JJ (2014) A modified data normalization method for GC-MS-based metabolomics to minimize batch variation. Springerplus 3:439
Winnike JH, Wei X, Knagge KJ, Colman SD, Gregory SG, Zhang X (2015) Comparison of GC-MS and GC×GC-MS in the analysis of human serum samples for biomarker discovery. J Proteome Res 14(4):1810–1817
Zhao X, Zhang G, Wang Y, Zhang F, Wang W, Zhang W, Fu B, Xu J, Li Z (2016) Metabolic profiling and physiological analysis of a novel Rice introgression line with broad leaf size. PLoS One 10(12):e0145646
Li J, Dong F, Xu J, Liu X, Li Y, Shan W, Zheng Y (2011) Enantioselective determination of triazole fungicide simeconazole in vegetables, fruits, and cereals using modified QuEChERS (quick, easy, cheap, effective, rugged and safe) coupled to gas chromatography/tandem mass spectrometry. Anal Chim Acta 702(1):127–135
Tranchida PQ, Zoccali M, Schipilliti L, Sciarrone D, Dugo P, Mondello L (2013) Solid-phase microextraction with fast GC combined with a high-speed triple quadrupole mass spectrometer for targeted and untargeted food analysis. J Sep Sci 36(13):2145–2150
Matthews J, Gemme S, Huebschmann H-J, Llorente C, Jimenez R, Sreenivasulu N (2015) Metabolomics of rice genotypes using GC-MS/MS. ThermoScientific Application Note 1–9
Schauer N, Steinhauser D, Strelkov S, Schomburg D, Allison G, Moritz T, Lundgren K, Roessner-Tunali U, Forbes MG, Willmitzer L, Fernie AR, Kopka J (2005) GC–MS libraries for the rapid identification of metabolites in complex biological samples. FEBS Lett 579(6):1332–1337
Agilent Technologies, Inc (2005) Automated mass spectral deconvolution and identification system AMDIS 2.64, in Released December, NIST
Cuadros-Inostroza Á, Caldana C, Redestig H, Kusano M, Lisec J, Peña-Cortés H, Willmitzer L, Hannah MA (2009) TargetSearch-a bioconductor package for the efficient preprocessing of GC-MS metabolite profiling data. BMC Bioinformatics 10:428
van den Berg RA, Hoefsloot HCJ, Westerhuis JA, Smilde AK, van der Werf MJ (2006) Centering, scaling, and transformations: improving the biological information content of metabolomics data. BMC Genomics 7:142
Acknowledgments
This work has been supported under the CGIAR thematic area Global Rice Agri-Food System CRP, RICE, Stress-Tolerant Rice for Africa and South Asia (STRASA) Phase III funding.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Llorente, C., Jimenez, R., Jackie, Brotman, Y., Fernie, A.R., Sreenivasulu, N. (2019). Rice Grain Quality Benchmarking Through Profiling of Volatiles and Metabolites in Grains Using Gas Chromatography Mass Spectrometry. In: Sreenivasulu, N. (eds) Rice Grain Quality. Methods in Molecular Biology, vol 1892. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8914-0_11
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8914-0_11
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8912-6
Online ISBN: 978-1-4939-8914-0
eBook Packages: Springer Protocols