Abstract
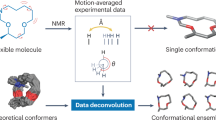
Heterogeneous and dynamic biomolecular complexes play a central role in many cellular processes but are poorly understood due to experimental challenges in characterizing their structural ensembles. To address these difficulties, we developed a hybrid methodology that combines X-ray crystallography with ensemble selections typically used in NMR studies to determine structural ensembles of heterogeneous biomolecular complexes. The method, termed READ, for residual electron and anomalous density, enables the visualization of heterogeneous conformational ensembles of complexes within crystals. Here we present a detailed protocol for performing the ensemble selections to construct READ ensembles. From a diverse pool of binding poses, a selection scheme is used to determine a subset of conformations that maximizes agreement with the X-ray data. Overall, READ is a general approach for obtaining a high-resolution view of dynamic protein-protein complexes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Keskin O, Gursoy A, Ma B et al (2008) Principles of protein-protein interactions: what are the preferred ways for proteins to interact? Chem Rev 108(4):1225–1244. https://doi.org/10.1021/cr040409x
Wright PE, Dyson HJ (2015) Intrinsically disordered proteins in cellular signalling and regulation. Nat Rev Mol Cell Biol 16(1):18–29. https://doi.org/10.1038/nrm3920
Fraser JS, van den Bedem H, Samelson AJ et al (2011) Accessing protein conformational ensembles using room-temperature X-ray crystallography. Proc Natl Acad Sci U S A 108(39):16247–16252. https://doi.org/10.1073/pnas.1111325108
Salmon L, Blackledge M (2015) Investigating protein conformational energy landscapes and atomic resolution dynamics from nmr dipolar couplings: a review. Rep Prog Phys 78(12):126601. https://doi.org/10.1088/0034-4885/78/12/126601
Salmon L, Yang S, Al-Hashimi HM (2014) Advances in the determination of nucleic acid conformational ensembles. Annu Rev Phys Chem 65:293–316. https://doi.org/10.1146/annurev-physchem-040412-110059
Venditti V, Egner TK, Clore GM (2016) Hybrid approaches to structural characterization of conformational ensembles of complex macromolecular systems combining nmr residual dipolar couplings and solution x-ray scattering. Chem Rev 116(11):6305–6322. https://doi.org/10.1021/acs.chemrev.5b00592
Jensen MR, Zweckstetter M, Huang JR et al (2014) Exploring free-energy landscapes of intrinsically disordered proteins at atomic resolution using nmr spectroscopy. Chem Rev 114(13):6632–6660. https://doi.org/10.1021/cr400688u
Nodet G, Salmon L, Ozenne V et al (2009) Quantitative description of backbone conformational sampling of unfolded proteins at amino acid resolution from nmr residual dipolar couplings. J Am Chem Soc 131(49):17908–17918. https://doi.org/10.1021/ja9069024
Salmon L, Bascom G, Andricioaei I et al (2013) A general method for constructing atomic-resolution rna ensembles using nmr residual dipolar couplings: the basis for interhelical motions revealed. J Am Chem Soc 135(14):5457–5466. https://doi.org/10.1021/ja400920w
Horowitz S, Salmon L, Koldewey P et al (2016) Visualizing chaperone-assisted protein folding. Nat Struct Mol Biol 23(7):691–697. https://doi.org/10.1038/nsmb.3237
Wals K, Ovaa H (2014) Unnatural amino acid incorporation in E. coli: current and future applications in the design of therapeutic proteins. Front Chem 2:15. https://doi.org/10.3389/fchem.2014.00015
Santrucek J, Strohalm M, Kadlcik V et al (2004) Tyrosine residues modification studied by maldi-tof mass spectrometry. Biochem Biophys Res Commun 323(4):1151–1156. https://doi.org/10.1016/j.bbrc.2004.08.214
Kmiecik S, Gront D, Kolinski M et al (2016) Coarse-grained protein models and their applications. Chem Rev 116(14):7898–7936. https://doi.org/10.1021/acs.chemrev.6b00163
Salmon L, Ahlstrom LS, Horowitz S et al (2016) Capturing a dynamic chaperone-substrate interaction using nmr-informed molecular modeling. J Am Chem Soc 138(31):9826–9839. https://doi.org/10.1021/jacs.6b02382
Ozenne V, Bauer F, Salmon L et al (2012) Flexible-meccano: a tool for the generation of explicit ensemble descriptions of intrinsically disordered proteins and their associated experimental observables. Bioinformatics 28(11):1463–1470. https://doi.org/10.1093/bioinformatics/bts172
Adcock SA, McCammon JA (2006) Molecular dynamics: survey of methods for simulating the activity of proteins. Chem Rev 106(5):1589–1615. https://doi.org/10.1021/cr040426m
Miller BL, Goldberg DE (1995) Genetic algorithms, tournament selection, and the effects of noise. Complex Syst 9(3):193–212
Afonine PV, Grosse-Kunstleve RW, Echols N et al (2012) Towards automated crystallographic structure refinement with phenix.Refine. Acta Crystallogr D D68:352–367. https://doi.org/10.1107/S0907444912001308
Winn MD, Ballard CC, Cowtan KD et al (2011) Overview of the ccp4 suite and current developments. Acta Crystallogr D Biol Crystallogr 67(Pt 4):235–242. https://doi.org/10.1107/S0907444910045749
Murshudov GN, Vagin AA, Dodson EJ (1997) Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr D Biol Crystallogr 53(Pt 3):240–255. https://doi.org/10.1107/S0907444996012255
Grosse-Kunstleve RW, Sauter NK, Moriarty NW et al (2002) The computational crystallography toolbox: crystallographic algorithms in a reusable software framework. J Appl Crystallogr 35:126–136. https://doi.org/10.1107/S0021889801017824
Press WH (2007) Numerical recipes: the art of scientific computing, 3rd edn. Cambridge University Press, Cambridge
Acknowledgment
This work was supported by the National Institutes of Health (R01-GM102829 to J.C.A.B. and K99/R00-GM120388 to S.H.). J.C.A.B. is a Howard Hughes Medical Investigator. The authors would like to thank S. Rocchio for comments on the manuscript and M. Mourao for useful discussions. The authors would also like to thank C. Stockbridge and the LSA-IT development team for the assistance in coding.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Salmon, L., Ahlstrom, L.S., Bardwell, J.C.A., Horowitz, S. (2018). Selecting Conformational Ensembles Using Residual Electron and Anomalous Density (READ). In: Marsh, J. (eds) Protein Complex Assembly. Methods in Molecular Biology, vol 1764. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7759-8_31
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7759-8_31
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7758-1
Online ISBN: 978-1-4939-7759-8
eBook Packages: Springer Protocols