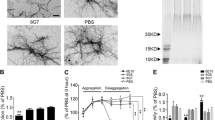
Abstract
Catalytic antibodies (catabodies) hold potential for superior immunotherapy because of their turnover capability and no or minimal induction of inflammatory responses. Catabodies neutralize and remove target antigens more potently than conventional antibodies. Depending on the catalytic rate constant, a single catabody molecule degrades thousands to millions of target molecules over its useful lifespan, whereas conventional antibodies only form reversibly associated, stoichiometric complexes with the target. Thus, removal of the antibody-bound target requires accessory phagocytic cells that ingest the immune complexes, which is usually accompanied by release of inflammatory mediators. In comparison, catabodies bind the target only transiently, and the rapid and direct target destruction reduces the concentration of immune complexes that can activate inflammatory processes. These features are especially pertinent when large target amounts at anatomically vulnerable sites must be removed, e.g., amyloids. We reported specific catabodies to misfolded transthyretin (misTTR) amyloid and amyloid β peptide (Aβ). Accumulation of the oligomeric and fibrillized amyloid TTR forms causes diverse systemic pathologies, including cardiomyopathy, polyneuropathy, and skeletal diseases. Brain Aβ aggregates are thought to cause central nervous system degenerative disease, chiefly Alzheimer’s disease. We describe methods for testing catabody-mediated degradation and dissolution of Aβ and TTR.
The original version of this chapter was revised. An erratum to the chapter can be found at DOI: 10.1007/978-1-4939-7180-0_16
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Williams TL, Serpell LC (2011) Membrane and surface interactions of Alzheimer's Abeta peptide—insights into the mechanism of cytotoxicity. FEBS J 278(20):3905–3917
Sorgjerd K, Klingstedt T, Lindgren M, Kagedal K, Hammarstrom P (2008) Prefibrillar transthyretin oligomers and cold stored native tetrameric transthyretin are cytotoxic in cell culture. Biochem Biophys Res Commun 377(4):1072–1078
Bourgault S, Choi S, Buxbaum JN, Kelly JW, Price JL, Reixach N (2011) Mechanisms of transthyretin cardiomyocyte toxicity inhibition by resveratrol analogs. Biochem Biophys Res Commun 410(4):707–713
Westermark P, Westermark GT, Suhr OB, Berg S (2014) Transthyretin-derived amyloidosis: probably a common cause of lumbar spinal stenosis. Ups J Med Sci 119(3):223–228
Yanagisawa A, Ueda M, Sueyoshi T, Okada T, Fujimoto T, Ogi Y et al (2014) Amyloid deposits derived from transthyretin in the ligamentum flavum as related to lumbar spinal canal stenosis. Mod Pathol 28(2):201–207
Sekijima Y, Uchiyama S, Tojo K, Sano K, Shimizu Y, Imaeda T et al (2011) High prevalence of wild-type transthyretin deposition in patients with idiopathic carpal tunnel syndrome: a common cause of carpal tunnel syndrome in the elderly. Hum Pathol 42(11):1785–1791
Buxbaum JN, Reixach N (2009) Transthyretin: the servant of many masters. Cell Mol Life Sci 66(19):3095–3101
Plante-Bordeneuve V, Said G (2011) Familial amyloid polyneuropathy. Lancet Neurol 10(12):1086–1097
Ueda M, Horibata Y, Shono M, Misumi Y, Oshima T, Su Y et al (2011) Clinicopathological features of senile systemic amyloidosis: an ante- and post-mortem study. Mod Pathol 24(12):1533–1544
Ueda M, Ando Y (2014) Recent advances in transthyretin amyloidosis therapy. Transl Neurodegener 3:19. doi:10.1186/2047-9158-3-19
Oddo S, Caccamo A, Shepherd JD, Murphy MP, Golde TE, Kayed R et al (2003) Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron 39(3):409–421
Spires-Jones TL, Hyman BT (2014) The intersection of amyloid beta and tau at synapses in Alzheimer’s disease. Neuron 82(4):756–771
Jacobson DR, Pastore R, Pool S, Malendowicz S, Kane I, Shivji A et al (1996) Revised transthyretin Ile 122 allele frequency in African-Americans. Hum Genet 98(2):236–238
Plante-Bordeneuve V, Kerschen P (2013) Transthyretin familial amyloid polyneuropathy. Handb Clin Neurol 115:643–658
Cacace R, Sleegers K, Van Broeckhoven C (2016) Molecular genetics of early-onset Alzheimer’s disease revisited. Alzheimers Dement 12(6):733–748
Paul S, Volle DJ, Beach CM, Johnson DR, Powell MJ, Massey RJ (1989) Catalytic hydrolysis of vasoactive intestinal peptide by human autoantibody. Science 244(4909):1158–1162
Gabibov AG, Gololobov GV, Makarevich OI, Schourov DV, Chernova EA, Yadav RP (1994) DNA-hydrolyzing autoantibodies. Appl Biochem Biotechnol 47(2–3):293–302; discussion 303
Ponomarenko NA, Durova OM, Vorobiev, II, Belogurov AA, Jr., Kurkova IN, Petrenko AG, Telegin GB et al (2006) Autoantibodies to myelin basic protein catalyze site-specific degradation of their antigen. Proc Natl Acad Sci U S A 103 (2):281-286
Ponomarenko NA, Durova OM, Vorobiev II, Aleksandrova ES, Telegin GB, Chamborant OG et al (2002) Catalytic antibodies in clinical and experimental pathology: human and mouse models. J Immunol Methods 269(1–2):197–211
Paul S, Planque SA, Nishiyama Y, Hanson CV, Massey RJ (2012) Nature and nurture of catalytic antibodies. Adv Exp Med Biol 750:56–75
Planque SA, Nishiyama Y, Sonoda S, Lin Y, Taguchi H, Hara M et al (2015) Specific amyloid beta clearance by a catalytic antibody construct. J Biol Chem 290(16):10229–10241
Planque S, Mitsuda Y, Taguchi H, Salas M, Morris MK, Nishiyama Y et al (2007) Characterization of gp120 hydrolysis by IgA antibodies from humans without HIV infection. AIDS Res Hum Retrovir 23(12):1541–1554
Hifumi E, Morihara F, Hatiuchi K, Okuda T, Nishizono A, Uda T (2008) Catalytic features and eradication ability of antibody light-chain UA15-L against Helicobacter pylori. J Biol Chem 283(2):899–907
Cho S, Youn HN, Hoang PM, Cho S, Kim KE, Kil EJ et al (2015) Preventive Activity against Influenza (H1N1) Virus by intranasally delivered RNA-hydrolyzing antibody in respiratory epithelial cells of mice. Virus 7(9):5133–5144
Tomin A, Dumych T, Tolstyak Y, Kril I, Mahorivska I, Bila E et al (2015) Desialylation of dying cells with catalytically active antibodies possessing sialidase activity facilitate their clearance by human macrophages. Clin Exp Immunol 179(1):17–23
Kou J, Yang J, Lim JE, Pattanayak A, Song M, Planque S et al (2015) Catalytic immunoglobulin gene delivery in a mouse model of Alzheimer’s disease: prophylactic and therapeutic applications. Mol Neurobiol 51(1):43–56
Planque SA, Nishiyama Y, Hara M, Sonoda S, Murphy SK, Watanabe K et al (2014) Physiological IgM class catalytic antibodies selective for transthyretin amyloid. J Biol Chem 289(19):13243–13258
Paul S, Tramontano A, Gololobov G, Zhou YX, Taguchi H, Karle S et al (2001) Phosphonate ester probes for proteolytic antibodies. J Biol Chem 276(30):28314–28320
Taguchi H, Planque S, Sapparapu G, Boivin S, Hara M, Nishiyama Y et al (2008) Exceptional amyloid beta peptide hydrolyzing activity of nonphysiological immunoglobulin variable domain scaffolds. J Biol Chem 283(52):36724–36733
Nishiyama Y, Taguchi H, Hara M, Planque SA, Mitsuda Y, Paul S (2014) Metal-dependent amyloid beta-degrading catalytic antibody construct. J Biotechnol 180:17–22
Hifumi E, Honjo E, Fujimoto N, Arakawa M, Nishizono A, Uda T (2012) Highly efficient method of preparing human catalytic antibody light chains and their biological characteristics. FASEB J 26(4):1607–1615
Matsuura K, Ohara K, Munakata H, Hifumi E, Uda T (2006) Pathogenicity of catalytic antibodies: catalytic activity of Bence Jones proteins from myeloma patients with renal impairment can elicit cytotoxic effects. Biol Chem 387(5):543–548
Timofeeva AM, Buneva VN, Nevinsky GA (2015) Systemic lupus erythematosus: molecular cloning and analysis of 22 individual recombinant monoclonal kappa light chains specifically hydrolyzing human myelin basic protein. J Mol Recognit 28(10):614–627
Bezuglova AM, Konenkova LP, Doronin BM, Buneva VN, Nevinsky GA (2011) Affinity and catalytic heterogeneity and metal-dependence of polyclonal myelin basic protein-hydrolyzing IgGs from sera of patients with systemic lupus erythematosus. J Mol Recognit 24(6):960–974
Nevinsky GA, Buneva VN (2002) Human catalytic RNA- and DNA-hydrolyzing antibodies. J Immunol Methods 269(1–2):235–249
Wootla B, Christophe OD, Mahendra A, Dimitrov JD, Repesse Y, Ollivier V et al (2011) Proteolytic antibodies activate factor IX in patients with acquired hemophilia. Blood 117(7):2257–2264
Taguchi H, Planque S, Nishiyama Y, Symersky J, Boivin S, Szabo P et al (2008) Autoantibody-catalyzed hydrolysis of amyloid beta peptide. J Biol Chem 283(8):4714–4722
Sapparapu G, Planque S, Mitsuda Y, McLean G, Nishiyama Y, Paul S (2012) Constant domain-regulated antibody catalysis. J Biol Chem 287(43):36096–36104
Gao QS, Paul S (1995) Molecular cloning of antiground-state proteolytic antibody fragments. Methods Mol Biol 51:281–296
Echan LA, Speicher DW (2002) Protein detection in gels using fixation. Curr Protoc Protein Sci:10–15
Paul S (1994) Catalytic activity of anti-ground state antibodies, antibody subunits, and human autoantibodies. Appl Biochem Biotechnol 47(2–3):241–253. discussion 253-245
Jan A, Hartley DM, Lashuel HA (2010) Preparation and characterization of toxic Abeta aggregates for structural and functional studies in Alzheimer's disease research. Nat Protoc 5(6):1186–1209
Acknowledgments
This work was supported in part by the SENS Research Foundation and NIH grants R01AG025304 and 5U01AG033183. Stephanie Planque and Sudhir Paul have a financial interest in Covalent Bioscience, Inc. and patents concerning catalytic antibodies.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Meretoja, V.V., Paul, S., Planque, S.A. (2017). Hydrolysis and Dissolution of Amyloids by Catabodies. In: Kaveri, S., Bayry, J. (eds) Natural Antibodies. Methods in Molecular Biology, vol 1643. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7180-0_9
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7180-0_9
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7179-4
Online ISBN: 978-1-4939-7180-0
eBook Packages: Springer Protocols