Abstract
Anaplastic thyroid cancer (ATC) has a dismal prognosis, with a median overall survival of only 4.9 months and 1-year survival of 20 %. Stage IVA (intrathyroidal) tumors have the best prognosis, while IVC patients (distant metastases) have the worst. The most favorable prognosis is seen in patients with an incidental focus of ATC, but even these uncommon patients do not all survive, indicating that microscopic metastases are present early and aggressive therapy is required. Unfortunately, the median frequency of stage IVA lesions is only 10 %, compared to 40.1 % and 45.8 % for stages IVB and IVC, respectively. Predictors of survival in many but not all studies include completeness of surgical resection, delivery of higher doses of external radiation, smaller tumor size, and locoregionally confined disease. Younger age is more favorable in some studies, while gender does not affect survival. Historically, most studies using combination therapy have not prolonged survival, although several recent reports using multimodal therapy have shown some promise. In a few studies, elevated white blood cell counts, dyspnea, dysphagia, and higher intensity of uptake on FDG-PET imaging have been associated with a worse prognosis. What remains unexplained is why some patients present with stage IVA disease, while most are more advanced. A better understanding of the critical genetic abnormalities present in each stage is needed in order to design better therapies to improve survival.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
The prognosis for anaplastic thyroid cancer (ATC) is dismal, with survival usually but a few months, unlike that of differentiated thyroid cancers in which patients often live years even with distant metastases. The median overall survival for ATC is only 4.9 months, with a 1-year survival of 20 % [1–44]. All ATC patients are stage IV by American Joint Committee on Cancer (AJCC) criteria. Stage IVA encompasses patients with intrathyroidal ATC and carries the best prognosis but represents a minority of patients at presentation. Stage IVB patients have disease confined to the neck, while IVC requires distant metastases.
Table 100.1 illustrates the frequencies of stage IVA–C disease according to TNM classification. Stage IVA disease is, unfortunately, the least common presentation, with a median of only 10%. The median frequencies of stage IVB and IVC disease are similar, at 40.1 % and 45.8 %, respectively. As expected, the survival rates are highest for stage IVA patients (Table 100.2). Nevertheless, the 2-year survival for intrathyroidal disease is only 62 %. And no one with stage IVC disease lived 2 years [39]. Chen et al. [45] reported similar results when analyzing the Surveillance, Epidemiology, and End Results (SEER) database. They examined outcomes in 241 patients who had surgery performed or recommended and who survived a month or longer. If disease was confined to the thyroid, median survival was 50 % longer (9 vs. 6 months) than if adjacent tissues were involved and three times longer than if the tumor was more advanced (Table 100.3). However, even patients with stage IVA disease had only a 23 % 5-year survival. These findings emphasize that patients with disease seemingly confined to the thyroid also have a high mortality rate. Thus, microscopic metastases are present frequently, and aggressive systemic adjuvant therapy will be required to improve outcomes. The most favorable prognosis is seen when an incidental focus of ATC is identified in a thyroid specimen resected for goiter or differentiated cancer. Akaishi et al. [39] had six such cases; all were alive at 1 year, but one died 9 years later with pulmonary metastases. Pierie et al. [21] reported a 3-year survival of 90 % if ATC was discovered incidentally, while Sugino et al. [22] found 1- and 2-year survivals of 73 % and 46 %, respectively, with incidental ATC. Choi et al. [46] identified microscopic anaplastic foci (MAF) in 0.4 % of 3,587 PTC cases; 5-year cause-specific survival was 64 % in the MAF group and 98 % in the pure PTC cases.
Predictors of survival have been examined in many studies, using both univariate and multivariate analyses. The most obvious candidates would be size of the tumor, completeness of surgical resection, and lack of distant metastases, and this is borne out in Fig. 100.1 which illustrates the longest survival for patients with stage IVA cancer and shortest with IVC, as well as in Tables 100.2 and 100.3. What remains unexplained is why some patients are more fortunate and their cancer is detected at stage IVA, rather than IVB or IVC. It is recognized that all ATCs contain a plethora of genetic abnormalities responsible for the remarkable aggressiveness of the tumor [47–49]. It will be important to better understand the molecular derangements that contribute to the pathogenesis and progression at each stage of ATC, in order to develop more accurate predictive and therapeutic strategies.
Overall Kaplan-Meier survival in months. Data for historical 50-year Mayo Clinic outcomes for stage IVA, IVB, and IVC anaplastic thyroid cancer are compared to outcomes resulting from our recent practice change combining intensity-modulated radiation therapy with adjuvant and radiosensitizing chemotherapy. IMRT intensity-modulated radiation therapy (Reprinted with permission from Foote et al. [54])
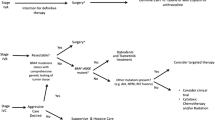
The prognostic factors most commonly evaluated are listed in Table 100.4. Younger patients (<70, 65, or 60 years) have longer survival times in some studies [4, 18, 21, 26, 28, 31, 33, 39, 43, 50], but not in all [13, 15, 24, 30, 32, 34, 35, 42]. Gender, on the other hand, did not influence survival in most studies [15, 24, 28, 31, 42, 43, 50, 51], but did in one [21]. Smaller primary tumors (<5, 6, or 7 cm) portend a more favorable outcome in eight reports [11, 13, 18, 21, 24, 31, 50, 51], but not in four others [28, 32, 39, 43]. The most striking, but not unexpected, predictor is extent of surgical resection, with longer survival observed in 81 % of studies [6, 8, 11, 13–15, 20–24, 27, 28, 30, 36, 39, 41, 50], but not in a minority [29, 31, 32, 43, 51]. Patients with a microscopic focus of ATC have the highest chance of survival, followed by patients with intrathyroidal stage IVA disease and R0 (negative margins) resections. Ito et al. [40] recently reported that stage IVB patients could be subdivided with IVB-a tumors involving the soft tissue, trachea, larynx, recurrent laryngeal nerve, or esophagus and IVB-b tumors which encased the carotid artery or mediastinal vessels or invaded the prevertebral fascia. Median survival was improved from 4.0 to 9.6 months in the IVB-a vs. IVB-b patients. Super-radical resection (trachea/larynx, esophagus/pharynx, major cervical arteries) in stage IVB patients improves survival compared to palliative but not restricted radical surgery [52].
Radiotherapy improved outcomes in 72 % of studies [10, 11, 13, 15, 21, 27–29, 34, 36, 39, 41, 43, 50], particularly in obtaining locoregional control of tumor and reducing death from airway and esophageal complications, but failed to improve survival in others [8, 24, 31, 35, 42]. Distant metastases, as one would predict, carried the worst prognosis in most [4, 6, 15, 26, 28–30, 33, 34, 39, 50, 51] but not all [24, 32] reports. Prior or concurrent differentiated thyroid cancer had an inconsistent effect on survival with a better prognosis in four reports [15, 19, 22, 23], but not in others [4, 14, 31, 51]. Leukocytosis carried a worse prognosis in several [39, 50, 53] but not all [24] studies, and dyspnea [8, 21, 42] and dysphagia [21] at presentation also are worrisome. FDG-PET uptake and intensity are inversely related to survival [44].
The causes of death are generally related to locoregional complications (asphyxia, pneumonia) or distant metastases. As radiotherapy techniques were modified to better accommodate the rapid doubling time of ATC, death rates from airway complications were reduced [3, 10, 13, 15, 27]. In one study, death was attributed to local complications in 37 %, distant metastases in 12 %, and both in 57 % of patients [38].
No single therapy has been optimal for ATC. While combination therapy has not improved survival in most studies [3, 8, 10, 21, 22, 24, 31–33, 41, 42] (Table 100.4), this reflects series spanning many decades during which the surgical, radiation, and chemotherapy approaches have evolved. Several studies of aggressive multimodal therapy [4, 17, 34, 40, 43, 50, 54] have shown some promise, at least in stage IVA and IVB ATC. Further experience and refinement of these strategies, and testing of newer agents with benefits demonstrated in preclinical studies [48, 49], will hopefully improve outcome in patients with all stages of ATC.
References
Carcangiu ML, Steeper T, Zampi G, et al. Anaplastic thyroid carcinoma. A study of 70 cases. Am J Clin Pathol. 1985;83:135–58.
Kim JH, Leeper RD. Treatment of locally advanced thyroid carcinoma with combination doxorubicin and radiation therapy. Cancer. 1987;60:2372–5.
Tennvall J, Tallroth E, el Hassan A, et al. Anaplastic thyroid carcinoma. Doxorubicin, hyperfractionated radiotherapy and surgery. Acta Oncol. 1990;29:1025–8.
Venkatesh YS, Ordonez NG, Schultz PN, et al. Anaplastic carcinoma of the thyroid. A clinicopathologic study of 121 cases. Cancer. 1990;66:321–30.
Demeter JG, De Jong SA, Lawrence AM, et al. Anaplastic thyroid carcinoma: risk factors and outcome. Surgery. 1991;110:956–61; discussion 961–3.
Schlumberger M, Parmentier C, Delisle MJ, et al. Combination therapy for anaplastic giant cell thyroid carcinoma. Cancer. 1991;67:564–6.
Wong CS, Van Dyk J, Simpson WJ. Myelopathy following hyperfractionated accelerated radiotherapy for anaplastic thyroid carcinoma. Radiother Oncol. 1991;20:3–9.
Junor EJ, Paul J, Reed NS. Anaplastic thyroid carcinoma: 91 patients treated by surgery and radiotherapy. Eur J Surg Oncol. 1992;18:83–8.
Hadar T, Mor C, Shvero J, et al. Anaplastic carcinoma of the thyroid. Eur J Surg Oncol. 1993;19:511–6.
Levendag PC, De Porre PM, van Putten WL. Anaplastic carcinoma of the thyroid gland treated by radiation therapy. Int J Radiat Oncol Biol Phys. 1993;26:125–8.
Tan RK, Finley 3rd RK, Driscoll D, et al. Anaplastic carcinoma of the thyroid: a 24-year experience. Head Neck. 1995;17:41–7; discussion 47–8.
Chemotherapy Committee, The Japanese Society of Thyroid Surgery. Intensive chemotherapy for anaplastic thyroid carcinoma: combination of cisplatin, doxorubicin, etoposide and peplomycin with granulocyte colony-stimulating factor support. Jpn J Clin Oncol. 1995;25:203–7.
Kobayashi T, Asakawa H, Umeshita K, et al. Treatment of 37 patients with anaplastic carcinoma of the thyroid. Head Neck. 1996;18:36–41.
Passler C, Scheuba C, Prager G, et al. Anaplastic (undifferentiated) thyroid carcinoma (ATC). A retrospective analysis. Langenbecks Arch Surg. 1999;384:284–93.
Voutilainen PE, Multanen M, Haapiainen RK, et al. Anaplastic thyroid carcinoma survival. World J Surg. 1999;23:975–8; discussion 978–9.
Ain KB. Anaplastic thyroid carcinoma: behavior, biology, and therapeutic approaches. Thyroid. 1998;8:715–26.
Busnardo B, Daniele O, Pelizzo MR, et al. A multimodality therapeutic approach in anaplastic thyroid carcinoma: study on 39 patients. J Endocrinol Invest. 2000;23:755–61.
Lam KY, Lo CY, Chan KW, et al. Insular and anaplastic carcinoma of the thyroid: a 45-year comparative study at a single institution and a review of the significance of p53 and p21. Ann Surg. 2000;231:329–38.
Rodriguez JM, Pinero A, Ortiz S, et al. Clinical and histological differences in anaplastic thyroid carcinoma. Eur J Surg. 2000;166:34–8.
Haigh PI, Ituarte PH, Wu HS, et al. Completely resected anaplastic thyroid carcinoma combined with adjuvant chemotherapy and irradiation is associated with prolonged survival. Cancer. 2001;91:2335–42.
Pierie JP, Muzikansky A, Gaz RD, et al. The effect of surgery and radiotherapy on outcome of anaplastic thyroid carcinoma. Ann Surg Oncol. 2002;9:57–64.
Sugino K, Ito K, Mimura T, et al. The important role of operations in the management of anaplastic thyroid carcinoma. Surgery. 2002;131:245–8.
De Crevoisier R, Baudin E, Bachelot A, et al. Combined treatment of anaplastic thyroid carcinoma with surgery, chemotherapy, and hyperfractionated accelerated external radiotherapy. Int J Radiat Oncol Biol Phys. 2004;60:1137–43.
Kihara M, Miyauchi A, Yamauchi A, et al. Prognostic factors of anaplastic thyroid carcinoma. Surg Today. 2004;34:394–8.
Veness MJ, Porter GS, Morgan GJ. Anaplastic thyroid carcinoma: dismal outcome despite current treatment approach. ANZ J Surg. 2004;74:559–62.
Besic N, Hocevar M, Zgajnar J, et al. Prognostic factors in anaplastic carcinoma of the thyroid-a multivariate survival analysis of 188 patients. Langenbecks Arch Surg. 2005;390:203–8.
Goutsouliak V, Hay JH. Anaplastic thyroid cancer in British Columbia 1985–1999: a population-based study. Clin Oncol (R Coll Radiol). 2005;17:75–8.
Kebebew E, Greenspan FS, Clark OH, et al. Anaplastic thyroid carcinoma. Treatment outcome and prognostic factors. Cancer. 2005;103:1330–5.
Wang Y, Tsang R, Asa S, et al. Clinical outcome of anaplastic thyroid carcinoma treated with radiotherapy of once- and twice-daily fractionation regimens. Cancer. 2006;107:1786–92.
Brignardello E, Gallo M, Baldi I, et al. Anaplastic thyroid carcinoma: clinical outcome of 30 consecutive patients referred to a single institution in the past 5 years. Eur J Endocrinol. 2007;156:425–30.
Kim TY, Kim KW, Jung TS, et al. Prognostic factors for Korean patients with anaplastic thyroid carcinoma. Head Neck. 2007;29:765–72.
Miccoli P, Materazzi G, Antonelli A, et al. New trends in the treatment of undifferentiated carcinomas of the thyroid. Langenbecks Arch Surg. 2007;392:397–404.
Yau T, Lo CY, Epstein RJ, et al. Treatment outcomes in anaplastic thyroid carcinoma: survival improvement in young patients with localized disease treated by combination of surgery and radiotherapy. Ann Surg Oncol. 2008;15:2500–5.
Bhatia A, Rao A, Ang KK, et al. Anaplastic thyroid cancer: clinical outcomes with conformal radiotherapy. Head Neck. 2010;32:829–36.
Dandekar P, Harmer C, Barbachano Y, et al. Hyperfractionated accelerated radiotherapy (HART) for anaplastic thyroid carcinoma: toxicity and survival analysis. Int J Radiat Oncol Biol Phys. 2009;74:518–21.
Swaak-Kragten AT, de Wilt JH, Schmitz PI, et al. Multimodality treatment for anaplastic thyroid carcinoma – treatment outcome in 75 patients. Radiother Oncol. 2009;92:100–4.
Vrbic S, Pejcic I, Vrbic M, et al. Therapy of stage IV B anaplastic thyroid carcinoma: single institution experience. J BUON. 2009;14:41–4.
Siironen P, Hagstrom J, Maenpaa HO, et al. Anaplastic and poorly differentiated thyroid carcinoma: therapeutic strategies and treatment outcome of 52 consecutive patients. Oncology. 2010;79:400–8.
Akaishi J, Sugino K, Kitagawa W, et al. Prognostic factors and treatment outcomes of 100 cases of anaplastic thyroid carcinoma. Thyroid. 2011;21:1183–9.
Ito KI, Hanamura T, Murayama K, et al. Multimodality therapeutic outcomes in anaplastic thyroid carcinoma: improved survival in subgroups of patients with localized primary tumors. Head Neck. 2012;34:230–7.
McIver B, Hay ID, Giuffrida DF, et al. Anaplastic thyroid carcinoma: a 50-year experience at a single institution. Surgery. 2001;130:1028–34.
Tashima L, Mitzner R, Durvesh S, et al. Dyspnea as a prognostic factor in anaplastic thyroid carcinoma. Eur Arch Otorhinolaryngol. 2012;269:1251–5.
Sherman EJ, Lim SH, Ho AL, et al. Concurrent doxorubicin and radiotherapy for anaplastic thyroid cancer: a critical re-evaluation including uniform pathologic review. Radiother Oncol. 2011;101:425–30.
Poisson T, Deandreis D, Leboulleux S, et al. 18F-fluorodeoxyglucose positron emission tomography and computed tomography in anaplastic thyroid cancer. Eur J Nucl Med Mol Imaging. 2010;37:2277–85.
Chen J, Tward JD, Shrieve DC, et al. Surgery and radiotherapy improves survival in patients with anaplastic thyroid carcinoma: analysis of the surveillance, epidemiology, and end results 1983–2002. Am J Clin Oncol. 2008;31:460–4.
Choi JY, Hwang BH, Jung KC, et al. Clinical significance of microscopic anaplastic focus in papillary thyroid carcinoma. Surgery. 2013;154:106–10.
Smallridge RC, Copland JA. Anaplastic thyroid carcinoma: pathogenesis and emerging therapies. Clin Oncol. 2010;22:486–97.
Smallridge RC, Marlow LA, Copland JA. Anaplastic thyroid cancer: molecular pathogenesis and emerging therapies. Endocr Relat Cancer. 2009;16:17–44.
Abate E, Smallridge R. Managing anaplastic thyroid cancer. Expert Rev Endocrinol Metab. 2011;6:793–809.
Sugitani I, Miyauchi A, Sugino K, et al. Prognostic factors and treatment outcomes for anaplastic thyroid carcinoma: ATC research consortium of Japan cohort study of 677 patients. World J Surg. 2012;36:1247–54.
Segerhammar I, Larsson C, Nilsson IL, et al. Anaplastic carcinoma of the thyroid gland: treatment and outcome over 13 years at one institution. J Surg Oncol. 2012;106:981–6.
Sugitani I, Hasegawa Y, Sugasawa M, et al. Super-radical surgery for anaplastic thyroid carcinoma: a large cohort study using the anaplastic thyroid carcinoma research consortium of Japan database. Head Neck. 2014;36:328–33.
Sugitani I, Kasai N, Fujimoto Y, et al. Prognostic factors and therapeutic strategy for anaplastic carcinoma of the thyroid. World J Surg. 2001;25:617–22.
Foote RL, Molina JR, Kasperbauer JL, et al. Enhanced survival in locoregionally confined anaplastic thyroid carcinoma: a single-institution experience using aggressive multimodal therapy. Thyroid. 2011;21:25–30.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this chapter
Cite this chapter
Smallridge, R.C., Abate, E.G. (2016). Anaplastic Thyroid Carcinoma: Prognosis. In: Wartofsky, L., Van Nostrand, D. (eds) Thyroid Cancer. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-3314-3_100
Download citation
DOI: https://doi.org/10.1007/978-1-4939-3314-3_100
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-3312-9
Online ISBN: 978-1-4939-3314-3
eBook Packages: MedicineMedicine (R0)