Abstract
The aim was to assess the correlation between cerebral autoregulation and outcome. Included were 31 preterm infants, gestational age 26 1/7 to 32 2/7 and <24 h life. Coherence between cerebral total haemoglobin (tHb) or oxygenation index (OI) measured by near-infrared spectrophotometry (NIRS) and systemic heart rate (HR) or arterial blood pressure (MAP) was calculated as a measure of autoregulation. In contrast to previous studies, low coherences in the first 24 h were significantly associated with intraventricular haemorrhage, death or abnormal neurodevelopmental outcome at 18 months or later. We suggest that our results can be explained by the concept of a multi-oscillatory-functions-order.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
- Autoregulation
- Near infrared spectroscopy
- Neurological outcome
- Preterm infants
- Spontaneous oscillations
1 Introduction
In the last two decades mortality for preterm infants has decreased continuously in industrialised countries, but morbidity and long-term cerebral outcome remained stable [1] due to the unchanged incidence of germinal matrix-intraventricular haemorrhage (GMH-IVH) or white brain matter injury. Fluctuations in cerebral blood flow (CBF) are a main factor in the pathogenesis of GMH-IVH [2]. They may be caused by impaired cerebral autoregulation, i.e. changes in blood pressure transmitted to the brain [3, 4]. The coherence between cerebral (tHb or OI measured by NIRS) and systemic (HR or MAP) spontaneous oscillations, is a novel approach to assess autoregulation [5] (Fig. 65.1). A high coherence between cerebral and systemic parameters was interpreted as impaired autoregulation and associated with increased incidence of GMH-IVH [6, 7] or subsequent death [8]. In contrast, Doppler sonography studies showed that CBF is independent of MAP [9, 10]. To clarify these conflicting results, the aim was to determine in very preterm infant the relation between autoregulation during the first 3 days of life and their neurological outcome in the first 10 years.
Simultaneous tracings of total haemoglobin (tHb) and oxygenation index (OI) (above) and mean arterial blood pressure (MAP) and heart rate (HR) (below). Left: Example of coherent tracings of 1-day-old premature neonate (1940 g), who had normal MDIs (89/100; 96/100) and normal ultrasounds. Coherence scores were between 0.9 and 0.99. Right: Incoherent tracings of a 1-day-old premature neonate (1050 g), who had pathologic ultrasound scans and an abnormal MDI at 18 months (62/100). Coherence scores were between 0.14 and 0.39
2 Methods
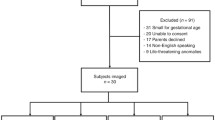
After approval by the ethical committee and parental consent, we enrolled 31 preterm neonates with normal capillary refilling, MAP, HR and without arrhythmias. Included were infants with an intraarterial catheter. Excluded were patients with blood transfusions in the first 6 h, severe sepsis with low blood pressure, severe malformations or severe asphyxia (Sarnat III). Gestational age ranged from 26 1/7 to 32 2/7 weeks (median: 27 2/7), birth weight from 690 to 2440 g (median: 1030) and head circumference from 22.5 to 35.5 (median: 25.60); 14 infants were boys, 17 girls. The median socio-economic index, based on mother’s education and father’s occupation was 6 (from 12), suggesting a normal distribution of the population. 28 infants were delivered by caesarean section and 3 spontaneously. The median APGAR score 5 min postpartum was 7.5 (3–9). The median pH value in the umbilical artery was 7.25 (7.16–7.33). All infants had lung disease diagnosed by clinical examinations and chest radiography, consistent with a respiratory distress syndrome. Five neonates received nasal CPAP, the others were mechanically ventilated for a median time of 7 days. All infants received supplemental oxygen with a mean inspired oxygen fraction of 0.42 ± 0.15 %. Twenty infants received pethidine as analgesic and five diazepam for sedation. In six infants a patent ductus arteriosus was diagnosed and treated with indomethacine after day 2. The following parameters were determined at the time of measurement: Haemoglobin 16.6 ± 2.7 g/100 ml, blood glucose 4.9 ± 2.2 mM, SaO2 91.2 ± 2.3 % and tcpCO2 5.6 ± 1.1 kPa. One infant received dopamine for low MAP.
In the neonatal brain NIRS [11] measures concentration changes in cerebral oxy-, deoxy-, total haemoglobin (O2Hb, HHb, tHb in μM) and oxygen index (OI = (O2Hb-HHb)/2) [12]. The neonatal sensor of the Critikon Cerebral Oxygenation Monitor 2001 was placed fronto-parietally. It emitted light at 774, 815, 867, 904 nm. The interoptode distance was 35 mm, differential pathlength factor 4.4 [13], and sampling time 0.56 s. NIRS was measured at <6 h for 2 h, at 12–16 h, at 24–28 h and at 68–76 h (if the infant still had additional O2 and invasive MAP). Transcutaneous pO2 (tcpO2, Hellige Oxymonitor), pCO2 (tcpCO2, Hellige Kapnomonitor), SaO2 (Nellcor 200 or Radiometer Oximeter), HR in (1/min) and MAP (in mm Hg from an umbilical artery catheter (Hellige Vicom-SM)) were recorded together with the NIRS data. All were kept within normal ranges.
A cranial ultrasound examination (Acuson 128XP, 7 MHz transducer) was performed at <24 h, 3, 7, 14 days of life and every 2 weeks until discharge. Findings were classified into grade of intraventricular haemorrhages (IVH): 0 = none, 1 = subependymal, 2 = intraventricular, 3 = intraventricular with dilatation, 4 = parenchymal [14]. Grade of parenchymal lesions (PVL): 0 = none, 1 = echodensities persisting more than 10 days, 2 = localized periventricular cysts, 3 = cystic periventricular leukomalacia, and 4 = cystic subcortical leukomalacia [15]. To compare with previous studies two ultrasound groups were formed: (1) Poor ultrasound: IVH grades 3, 4 and/or any PVL and (2) Normal ultrasound: IVH grade 0, 1 or 2 and no PVL.
Developmental follow-up was examined at 9 and 18 months age corrected for prematurity using a standardised neurological test and the Bayley II scales of infant development: psycho-motor-developmental index (PDI) and mental-developmental index (MDI) ≥ 84 were considered normal and < 84 abnormal [16]. Further neurological examinations were performed at 3 years (3 infants) and 10 years (10 infants). Neurological findings were classified abnormal for markedly increased muscle tone (score ≥ 2 on Ashworth scale [17]). Correlation between MDI, PDI or neurological status and coherences were estimated on the basis of the worst outcome in any of the assessments. Neonates who died in the first days of life were classified as abnormal Bayley indices.
All measurements were screened and periods of ≥12 min without artefacts or changes in SaO2 (<5 %) were selected. Groups of 9 samples were aggregated to obtain a sample rate of ~5 s. Coherence spectra were calculated by MATLAB’s cohere function (parameters: nfft = 144, numoverlap = 143, subtraction of ‘mean’) [18]. The coherence was calculated between the tHb or OI and MAP or HR [6, 19]. We calculated the mean coherence for the band between 0 and 0.01 Hz (ultra-low frequency = ULF) and 0–0.1 Hz (low frequency = LF).
We used Spearman’s rho for continuous variables and to compare groups.
3 Results
Since analysis of variance did not show a significant difference between the first three measurement (6, 12 and 24 h), for each infant the coherences of the first 24 h were averaged. The different coherence indices showed a significant linear correlation (p < 0.00001, Fig. 65.2). The grade of correlation was higher for LF compared to ULF. LF and ULF were also highly linearly correlated (p < 0.005). The different coherence indices correlated significantly with the clinical parameters (Table 65.1, Fig. 65.3). Low coherence during the first 24 h of life was consistently associated with unfavourable outcome.
Examples of correlation of two coherences between cerebral and systemic parameters: tHb_HR versus OI_MAP (left) and tHb_MAP versus OI_HR (right). Each dot represents one infant. All correlations were highly significant (p < 0.00001, Spearman), which indicates that the systemic variables HR and MAP are interchangeable to a high degree. The same is true for the cerebral variables tHb and OI
Nine infants died between 2 and 34 days (median 3 days) due to grade 3/4 IVH (N = 5) and respiratory failure (N = 4). In 12 of 22 surviving infants an abnormal neurology was found at discharge (mostly increased muscle tonus). The number decreased with age: at 9 months 10 of 20 infants, at 18 months 3 of 18 infants and at 10 years 3 of 13 infants. MDI at 18 months was normal in 13 of 19 infants, suspect (83–68) in two and abnormal (<68) in four infants. The respective values for the PDI were: 11, 5 and 3 infants. Neonates with high MDI and/or normal neurological outcome had significantly higher coherences than neonates with low MDI and/or abnormal neurology.
Ultrasound scans showed no lesion in 5 of 31 infants. IVH grade 1/2 was found in 9, and grade 3/4 in 5 infants. PVL grade 1 was observed in 11, grade 3 in 1 infant. Again low coherence was significantly associated with unfavourable IVH grade ≥ 3 (Fig. 65.3). PVL did not correlate with the coherences.
To summarise, in our population of preterm infants a low coherence during the first 24 h of life was consistently associated with unfavourable outcome.
4 Discussion
In this study we demonstrated that low coherences between MAP or HR and OI or tHb in the first 24 h of life in very preterm infants are associated with unfavourable outcome. The coherence-indices were high in 20 of 31 neonates. They showed no significant variability over time, indicating no linear tendency towards lower values during the first 24 h as found by Menke et al. [20]. Nevertheless, in some infants coherence changed during the first 24 h. This may be due to individually different time course of entrainment with environmental rhythms (discussed below). Remarkable is the high correlation between the types of coherences: HR or MAP and OI or tHb yield very similar results.
It was postulated [6] that coherence scores reflect cerebral autoregulation and therefore represent a predictive value for neurological outcome. Previous studies found high ULF coherences (>0.5), i.e. impaired autoregulation, in 17 of 31 neonates, which was associated with a higher incidence of IVH/PVL [6–8, 21, 22]. These results are in contrast to our findings: We found a significant correlation of high coherence values with favourable outcome, not only for IVH but also for later outcome assessments. Our data are consistent, since all outcome parameters showed the same tendency. We were not able to identify differences between the previous and our study that could explain this discrepancy.
In the following the concept of autoregulation has to be discussed in order to possibly explain our results. Traditionally, autoregulation was seen as a myogenic concept, as a cerebral protective mechanism leading to a constriction/dilatation of arteries in response to an excessive increase/decrease of MAP [23]. On the one hand assessing cerebral autoregulation in neonates is difficult, since it is “very easy to obtain completely misleading results” [23]. Autoregulation may vary considerably over short periods of time within the same subject [24]. On the other hand even in adults, the text book concept of autoregulation (i.e. the CBF stable from 50 to 150 mmHg of MAP) is obsolete: today autoregulation merely stabilizes CBF in a range of only 10 mmHg [25]. Recently this led to a more complex view, the metabolic concept, i.e. autoregulation as a response to metabolic demands [24, 26–28]. Also this concept postulates that a high coherence is a sign of impaired autoregulation and thus neither approach is able to explain our results.
Spontaneous oscillations in our frequency range (0–0.1 Hz) may not reflect autoregulation. Since our results indicate that a high coherence is associated with beneficial outcome, let us consider an example where a high coherence was beneficial [29, 30]. The coordination or coherence of different central oscillators, e.g. of neurons in the medulla oblongata, is of great importance for a healthy functioning of the respiratory and sucking functions. A loss of such coherence leads to aspiration pneumonia [31]. Analogously we hypothesize that high coherences indicate a coordination of physiological sub-systems and thus are a sign of health. This concept called multi-oscillatory-functions-order (MOFO) was postulated already in 1980 [32]. It is applicable to a wide spectrum of rhythmically changing physiological processes. The coordination of interacting oscillations may lead to system stabilisation [33, 34]. The following examples illustrate the scope, productivity and relevance of this MOFO approach.
Fractured relations (i.e. 3.55:1) of oscillations in reaction time were a sign of immaturity [32] and were followed by integer relations (i.e. 3:1) considered as mature. This constitutes evidence of von Holst’s “principle of absolute and relative coordination”, i.e. the maturation from fractured to integer relations [35]. This experiment was expanded to other physiologic parameters, such as HR, respiratory rate, short-term memory, auditory-motor reaction time or those recorded by electro-oculogram. Sinz defined a coupling ratio i.e., the ratio between realised and possible frequency synchronisations. This ratio was shown to have diagnostic value for infants with congenital heart disease, patients with neurosis, office workers and students listening to music [32]. Similar efforts were continued by others [36–41]. All these findings indicate that synchronization is actually a sign of health and maturity. In addition, high short-term coherence values between HR, respiration and MAP were reduced by severe brain disorders [41, 42]. This can be explained by an impaired central autonomic coordination. Thus a reduced coherence, (or ‘uncoupling’ [37] or ‘decomplexification’ [38]) indicates a pathological process. It is expected that the more the coupling decreases, the poorer the outcome, because uncoupling between lungs, heart and the vascular system decreases their functional performance.
Summarizing our findings, all outcome parameters showed the same tendency: Favourable outcome was associated with high coherence. According to the MOFO approach, the high coherences in our study indicate a strong coordination of physiologic sub-systems, which is a sign of stability and explains the favourable short and long-term outcome. This MOFO evolves in the first months of life [29, 30, 43]. The autoregulation concept is not necessarily erroneous. Probably the concept of autoregulation does not apply to oscillations in our frequency range of up to 0.1 Hz. They are understandable by a MOFO concept. Future studies should investigate the areas of validity of autoregulation and MOFO approaches.
Almost all of our neonates were mechanically ventilated. Possibly this affected our coherence values by entrainment [39, 40]. A methodological problem of calculating the coherence is the intermittently low amplitude of spontaneous MAP variations. In a previous study MAP variations of 25 % were observed, but the cerebral spectral power was <2.5 % in a third of the subjects [27]. It was suggested that variations in MAP of >10 % are necessary to reduce noise to an acceptable level [23]. Therefore, in our recordings, we selected periods fulfilling this criterion.
Many studies demonstrate both the productivity of a MOFO-like approach and the lack of knowledge concerning coupling phenomena. Our results are internally consistent and support the MOFO hypothesis. They demonstrate that it is necessary to overcome the concept of autoregulation for the coherence analysis of spontaneous oscillations in the frequency range of 0–0.1 Hz.
The first ultrasound examination in our study was performed <24 h and IVH usually appeared later. Thus low coherence precedes brain damage. This is of potentially high clinical value, because there may be time to prevent IVH.
5 Conclusion
In contrast to previous studies, low coherences between systemic and cerebral parameters in the first 24 h of life were associated with IVH, death, abnormal MDI and abnormal neurological outcome. Similar predictive results were obtained with coherences using OI and tHb as cerebral variables and MAP and HR as systemic variables. The time of measurement in the first 24 h of life did not influence coherences significantly. According to a MOFO approach, high coherences reflect a high coordination of all physiological cycles. We propose that high coherences are an indicator of maturation and integrity.
References
Ruegger C, Hegglin M, Adams M et al (2012) Population based trends in mortality, morbidity and treatment for very preterm- and very low birth weight infants over 12 years. BMC Pediatr 12:17
Wigglesworth JS (1979) Prevention of cerebral hemorrhage in preterm infants. Lancet 2:256
Lou HC, Lassen NA, Friis-Hansen B (1979) Impaired autoregulation of cerebral blood flow in the distressed newborn infant. J Pediatr 94:118–121
Pryds O (1991) Control of cerebral circulation in the high-risk neonate. Ann Neurol 30:321–329
De Smet D, Vanderhaegen J, Naulaers G et al (2009) New measurements for assessment of impaired cerebral autoregulation using near-infrared spectroscopy. Adv Exp Med Biol 645:273–278
Tsuji M, Saul JP, du Plessis A et al (2000) Cerebral intravascular oxygenation correlates with mean arterial pressure in critically ill premature infants. Pediatrics 106:625–632
Alderliesten T, Lemmers PM, Smarius JJ et al (2013) Cerebral oxygenation, extraction, and autoregulation in very preterm infants who develop peri-intraventricular hemorrhage. J Pediatr 162:698–704 e2
Wong FY, Leung TS, Austin T et al (2008) Impaired autoregulation in preterm infants identified by using spatially resolved spectroscopy. Pediatrics 121:e604–e611
Pellicer A, Valverde E, Gaya F et al (2001) Postnatal adaptation of brain circulation in preterm infants. Pediatr Neurol 24:103–109
Tyszczuk L, Meek J, Elwell C et al (1998) Cerebral blood flow is independent of mean arterial blood pressure in preterm infants undergoing intensive care. Pediatrics 102:337–341
Wray S, Cope M, Delpy DT et al (1988) Characterization of the near infrared absorption spectra of cytochrome aa3 and haemoglobin for the non-invasive monitoring of cerebral oxygenation. Biochim Biophys Acta 933:184–192
Fukui Y, Ajichi Y, Okada E (2003) Monte Carlo prediction of near-infrared light propagation in realistic adult and neonatal head models. Appl Optics 42:2881–2887
Wyatt JS, Cope M, Delpy DT et al (1990) Measurement of optical path length for cerebral near-infrared spectroscopy in newborn infants. Dev Neurosci 12:140–144
Papile LA, Burstein J, Burstein R et al (1978) Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr 92:529–534
de Vries LS, Eken P, Dubowitz LM (1992) The spectrum of leukomalacia using cranial ultrasound. Behav Brain Res 49:1–6
Maas YG, Mirmiran M, Hart AA et al (2000) Predictive value of neonatal neurological tests for developmental outcome of preterm infants. J Pediatr 137:100–106
Damiano DL, Quinlivan JM, Owen BF et al (2002) What does the Ashworth scale really measure and are instrumented measures more valid and precise? Dev Med Child Neurol 44:112–118
Morren G, Lemmerling P, van Huffel S et al (2001) Detection of the autoregulation in the brain using a novel subspace-based technique. In: Proceedings of the 23rd annual international conference of the IEEE Engineering in Medicine and Biology Society (EMBC 2001), Istanbul
von Siebenthal K, Beran J, Wolf M et al (1999) Cyclical fluctuations in blood pressure, heart rate and cerebral blood volume in preterm infants. Brain Dev 21:529–534
Menke J, Michel E, Hillebrand S et al (1997) Cross-spectral analysis of cerebral autoregulation dynamics in high risk preterm infants during the perinatal period. Pediatr Res 42:690–699
Volpe JJ (2001) Neurobiology of periventricular leukomalacia in the premature infant. Pediatr Res 50:553–562
O’Leary H, Gregas MC, Limperopoulos C et al (2009) Elevated cerebral pressure passivity is associated with prematurity-related intracranial hemorrhage. Pediatrics 124:302–309
Panerai RB (1998) Assessment of cerebral pressure autoregulation in humans--a review of measurement methods. Physiol Meas 19:305–338
Panerai RB, Eames PJ, Potter JF (2003) Variability of time-domain indices of dynamic cerebral autoregulation. Physiol Meas 24:367–381
Willie CK, Tzeng YC, Fisher JA et al (2014) Integrative regulation of human brain blood flow. J Physiol 592(5):841–859
Panerai RB, Carey BJ, Potter JF (2003) Short-term variability of cerebral blood flow velocity responses to arterial blood pressure transients. Ultrasound Med Biol 29:31–38
Giller CA, Mueller M (2003) Linearity and non-linearity in cerebral hemodynamics. Med Eng Phys 25:633–646
Caicedo A, Naulaers G, Wolf M et al (2012) Assessment of the myogenic and metabolic mechanism influence in cerebral autoregulation using near-infrared spectroscopy. Adv Exp Med Biol 737:37–44
Lohr B, Siegmund R (1999) Ultradian and circadian rhythms of sleep-wake and food-intake behavior during early infancy. Chronobiol Int 16:129–148
Korte J, Wulff K, Oppe C et al (2001) Ultradian and circadian activity-rest rhythms of preterm neonates compared to full-term neonates using actigraphic monitoring. Chronobiol Int 18:697–708
Hellbrugge T (1967) Chronophysiology of the child. Verh Dtsch Ges Inn Med 73:895–921
Sinz R (1980) Chronopsychophysiologie. Akademie, Berlin
Gonze D, Halloy J, Goldbeter A (2002) Robustness of circadian rhythms with respect to molecular noise. Proc Natl Acad Sci U S A 99:673–678
Goldbeter A (2002) Computational approaches to cellular rhythms. Nature 420:238–245
von Holst E (1939) Die relative Koordination als Phänomen und als Methode zentralnervöser Funktionsanalyse. Ergeb Physiol 42:228–306
Biswal B, Hudetz AG, Yetkin FZ et al (1997) Hypercapnia reversibly suppresses low-frequency fluctuations in the human motor cortex during rest using echo-planar MRI. J Cereb Blood Flow Metab 17:301–308
Godin PJ, Buchman TG (1996) Uncoupling of biological oscillators: a complementary hypothesis concerning the pathogenesis of multiple organ dysfunction syndrome. Crit Care Med 24:1107–1116
Goldstein B, Fiser DH, Kelly MM et al (1998) Decomplexification in critical illness and injury: relationship between heart rate variability, severity of illness, and outcome. Crit Care Med 26:352–357
Larsen PD, Galletly DC (2001) Cardioventilatory coupling in heart rate variability: the value of standard analytical techniques. Br J Anaesth 87:819–826
Censi F, Calcagnini G, Cerutti S (2002) Coupling patterns between spontaneous rhythms and respiration in cardiovascular variability signals. Comput Methods Programs Biomed 68:37–47
Zwiener U, Schelenz C, Bramer S et al (2003) Short-term dynamics of coherence between respiratory movements, heart rate, and arterial pressure fluctuations in severe acute brain disorders. Physiol Res 52:517–524
Arnold M, Miltner WH, Witte H et al (1998) Adaptive AR modeling of nonstationary time series by means of Kalman filtering. IEEE Trans Biomed Eng 45:553–562
Mirmiran M, Baldwin RB, Ariagno RL (2003) Circadian and sleep development in preterm infants occurs independently from the influences of environmental lighting. Pediatr Res 53:933–938
Acknowledgements
We thank Dr. Vera Dietz and Matthias Keel for their help during the measurements and Dr. Daniel Haensse for help during data analysis.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media, New York
About this paper
Cite this paper
Stammwitz, A., von Siebenthal, K., Bucher, H.U., Wolf, M. (2016). Can the Assessment of Spontaneous Oscillations by Near Infrared Spectrophotometry Predict Neurological Outcome of Preterm Infants?. In: Elwell, C.E., Leung, T.S., Harrison, D.K. (eds) Oxygen Transport to Tissue XXXVII. Advances in Experimental Medicine and Biology, vol 876. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-3023-4_65
Download citation
DOI: https://doi.org/10.1007/978-1-4939-3023-4_65
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-3022-7
Online ISBN: 978-1-4939-3023-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)