Abstract
It is well established that histone posttranslational modifications mediate the control of gene expression played by chromatin. Such modifications are commonly reversible and many alternatives are open to drive transcription of inducible genes.
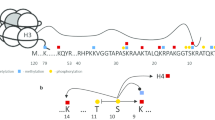
Estrogens govern growth and survival of hormone-sensitive cells by inducing expression of genes important for cell cycle progression and apoptosis. Transcription of estrogen-responsive genes is triggered by the lysine-specific demethylase 1 (LSD1)-dependent demethylation of dimethylated lysine 9 in histone H3 (H3K9me2) that accompanies to local generation of oxygen reactive species (ROS). Production of ROS modifies guanines in neighbor DNA with consequent recruitment of base-excision repair (BER) enzymes and formation of breaks that support creation of bridges between sites that, although distant on linear DNA, establish strategic contacts useful for productive transcription.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Strahl BD, Allis CD (2000) The language of covalent histone modifications. Nature 403:41–45
Mangelsdorf DJ et al (1995) The nuclear receptor superfamily: the second decade. Cell 83:835–839
Cosma MP (2002) Ordered recruitment: gene-specific mechanism of transcription activation. Mol Cell 10:227–236
Jenuwein T, Allis CD (2001) Translating the histone code. Science 293:1074–1080
Perissi V, Rosenfeld MG (2005) Controlling nuclear receptors: the circular logic of cofactor cycles. Nat Rev Mol Cell Bio 6:542–554
Park KJ et al (2005) Formation of an IKKa-dependent transcription complex is required for estrogen receptor-mediated gene activation. Mol Cell 18:71–82
Peters AH et al (2003) Partitioning and plasticity of repressive histone methylation states in mammalian chromatin. Mol Cell 12:1577–1589
Shang Y et al (2000) Cofactor dynamics and sufficiency in estrogen receptor-regulated transcription. Cell 103:843–852
Metivier R et al (2003) Estrogen receptor-a directs ordered, cyclical, and combinatorial recruitment of cofactors on a natural target promoter. Cell 115:751–763
Perillo B et al (2008) DNA oxidation as triggered by H3K9me2 demethylation drives estrogen-induced gene expression. Science 319:202–206
Abbondanza C et al (2011) Highlighting chromosome loops in DNA-picked chromatin (DPC). Epigenetics 6:979–986
Cook PR (1999) Organization of replication and transcription. Science 284:1790–1795
Dejardin J, Kingston RE (2009) Purification of proteins associated with specific genomic loci. Cell 136:175–186
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Di Santi, A., Cernera, G., Migliaccio, A., Perillo, B. (2014). Analysis of Histone Posttranslational Modifications in the Control of Chromatin Plasticity Observed at Estrogen-Responsive Sites in Human Breast Cancer Cells. In: Castoria, G., Auricchio, F. (eds) Steroid Receptors. Methods in Molecular Biology, vol 1204. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-1346-6_6
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1346-6_6
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-1345-9
Online ISBN: 978-1-4939-1346-6
eBook Packages: Springer Protocols