Abstract
Increased cardiovascular morbidity and mortality have been extensively documented in patients with chronic kidney disease (CKD). Notably, cardiovascular risk gradually increases with increasing stages of CKD, being the highest among patients with end-stage renal disease. Although the role of hyperlipidemia in the development of cardiovascular disease (CVD) in the general population has been efficiently documented, this relationship has been difficult to establish in patients with CKD. Indeed, non-traditional cardiovascular risk factors (inflammation, increased oxidative stress, vascular calcification, endothelial dysfunction, and anemia) confound the association of dyslipidemia and CVD in the CKD setting. Moreover, lipoprotein abnormalities in CKD substantially differ from those in the general population, with hypertriglyceridemia being the primary characteristic, whereas total and low-density lipoprotein cholesterol are normal or low. To further complicate things, pathologic findings of arterial lesions in CKD consist of calcium-rich atherosclerotic plaques, whereas in classic atherosclerotic disease lipid-laden atheromatous or fibroatheromatous plaques are detected, implying a different pathogenetic mechanism of CKD atherosclerotic disease.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Chronic Kidney Disease
- Coronary Artery Calcification
- Chronic Kidney Disease Patient
- Vascular Calcification
- Peritoneal Dialysis Patient
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
Recent data indicate that the burden of chronic kidney disease (CKD) is steadily increasing in the United States [1]. Cardiovascular (CV) disease (CVD) is the leading cause of morbidity and mortality in patients with CKD [2]. Both decreased glomerular filtration rate (GFR) and increased proteinuria are independent CV risk factors in community-based populations as well as in patients at high CV risk [3–5]. Notably, CVD-associated mortality rates increase progressively with increasing CKD stages and are extremely high in end-stage renal disease (ESRD) patients receiving dialysis (10–30 times higher than age-adjusted CV mortality in the general population) [5–7]. It has been reported that 39 % of incident dialysis patients have ischemic heart disease [8], whereas the annual rate of myocardial infarction and/or angina is approximately 10 % [9]. It is well established that dyslipidemias play a pivotal role in the pathogenesis of CVD in the general population [10]. However, the association of dyslipidemia and CVD in CKD patients is confounded by the presence of the so-called non-traditional CV risk factors (inflammation, vascular calcification, anemia, increased oxidative stress, and endothelial dysfunction), rendering the answer to the question of whether and which CKD patients might benefit from lipid-lowering treatments of major importance [11].
Definition and Classification of CKD
CKD is defined based on the presence of either kidney damage (structural or functional abnormalities other than decreased GFR) or decreased kidney function (GFR <60 mL/min/1.73 m2) for 3 or more months, irrespective of cause. Kidney damage refers to pathologic abnormalities detected by a renal biopsy or imaging studies, urinary albumin excretion >30 mg/day, urinary sediment abnormalities, or renal transplant status. Decreased kidney function refers to a decreased GFR, which is usually estimated (eGFR) using serum creatinine and one of several available equations such as the Modification of Diet in Renal Disease (MDRD) and, more recently, the CKD-Epi formula [12]. The definition and classification of CKD were introduced by the National Kidney Foundation (NKF) Kidney Disease Outcomes Initiative (KDOQI) in 2002 [13] and adopted with minor changes by the international guideline group Kidney Disease Improving Global Outcomes (KDIGO) in 2004 [14]. Until recently, staging of CKD was based on eGFR and a five-stage classification was used (Table 5.1). This classification has been recently modified to an 18-stage classification by adding albuminuria stage, subdivision of stage 3, and the cause of CKD (Table 5.2) [5, 15]. By using this modified 18-stage classification, an improved stratification of CKD progression and its major complications has been achieved, thus providing a better guidance for the monitoring and management of CKD patients. However, as most studies have been based on the original classification, we will refer to this one in this chapter.
Association Between CKD and CVD-Epidemiological Data
It is well established that CV mortality rates increase dramatically with advanced CKD stages. In fact, individuals with CKD are more likely to die of CVD than to develop ESRD [6, 16]. Intriguingly, younger ESRD patients (25–34 years old) exhibit 500-fold greater CV mortality rates than age-matched controls with normal renal function [6]. Accumulating evidence suggests that the link between CKD and increased CV morbidity and mortality holds across populations with various degrees of baseline renal function or CV status.
In the General Population
There are plenty of observations that demonstrate an independent association between diminished GFR or proteinuria and major adverse CV events (MACE) in the general population [17–21].
In a high-quality meta-analysis of general population cohorts including 105,872 participants with urine albumin-to-creatinine ratio (ACR) measurements and 1,128,310 participants with urine protein dipstick measurements, followed for 7.9 years, Matsushita et al. concluded that eGFR less than 60 mL/min/1.73 m2 and ACR 1.1 mg/mmol (10 mg/g) or more independently predict all cause and CV mortality risk in the general population. In studies with dipstick measurement of proteinuria, a trace urine-positive dipstick was also associated with increased all-cause and CV mortality independently of the level of kidney function [19].
Go et al. evaluated 1,120,295 subjects for the risk of death, CV events, and hospitalization relative to various levels of GFR over 2.84 years [22]. The adjusted hazard ratio for death was 1.2 with an eGFR of 45–59 mL/min/1.73 m2 (95 % CI, 1.1–1.2), 1.8 with an eGFR of 30–44 mL/min/1.73 m2 (95 % CI, 1.7–1.9), 3.2 with an eGFR of 15–29 mL/min/1.73 m2 (95 % CI, 3.1–3.4), and 5.9 with an estimated GFR of less than 15 mL/min/1.73 m2 (95 % CI, 5.4–6.5). The adjusted hazard ratio for CV events also increased inversely with the estimated GFR: 1.4 (95 % CI, 1.4–1.5), 2.0 (95 % CI, 1.9–2.1), 2.8 (95 % CI, 2.6–2.9), and 3.4 (95 % CI, 3.1–3.8), respectively. This large study demonstrated effectively the inverse association between GFR and rates of CV morbidity and mortality in patients without a prior history of CVD.
Weiner at al. pooled data from community-based trials including the Atherosclerosis Risk in Communities Study, CV Health Study, Framingham Heart Study, and Framingham Offspring Study; 22,634 subjects were followed for 10 years. CKD was defined by a GFR between 15 and 60 mL/min/1.73 m2. A composite of myocardial infarction, fatal coronary heart disease (CHD), stroke, and death was the primary study outcome. In adjusted analyses, CKD was an independent risk factor for the composite study outcome (hazard ratio, 1.19; 95 % CI, 1.07–1.32) [21].
In Patients with CV Risk Factors or Preexistent CVD
A growing number of studies have shown an association between the decrease in GFR and CV events among patients with known risk factors for CVD or preexistent CVD [2, 4, 23–28]. Van der Velde et al. performed a collaborative meta-analysis of ten cohorts with 266,975 patients with a history of hypertension, diabetes, or CV disease. Hazard ratios for CV mortality at eGFRs of 60, 45, and 15 mL/min/1.73 m2 were 1.11, 1.73, and 3.08, respectively, compared to an eGFR of 95 mL/min/1.73 m2, whereas similar findings were noted for all-cause mortality. There was also an association between albuminuria and risk for overall and CV mortality. The authors concluded that decreased eGFR and higher albuminuria are risk factors for all-cause and CV mortality in high-risk populations, independent of each other and of CV risk factors.
Mann et al. performed a post hoc analysis of the Heart Outcomes and Prevention Evaluation (HOPE) study [29]. The HOPE study included individuals with an objective evidence of vascular disease or diabetes combined with another CV risk factor and was designed to test the benefit of add-on ramipril vs. placebo. Nine hundred and eighty subjects with mild renal insufficiency (serum creatinine ≥ 1.4 mg/dL) and 8,307 subjects with normal renal function (serum creatinine < 1.4 mg/dL) were followed for ≈5 years. The cumulative incidence of the primary outcome (composite of CV death, myocardial infarction, or stroke) was significantly higher in individuals with mild renal insufficiency compared to those with normal renal function (P < 0.001).
Perkovic et al. in the Perindopril Protection against Recurrent Stroke Study (PROGRESS) randomly allocated 6,105 participants with cerebrovascular disease to perindopril-based blood pressure-lowering therapy or placebo. Individuals with CKD were at approximately 1.5-fold greater risk of major vascular events, stroke, and CHD, and were more than twice as likely to die (all P ≤ 0.002).
With respect to the relationship between albuminuria and CKD in patients with CV risk factors or preexistent CKD, Anavekar et al. [30] in a post hoc analysis of the Irbesartan Diabetic Nephropathy Trial (IDNT) showed that the proportion of patients who exhibited the CV composite endpoint (CV death, nonfatal MI, hospitalization for heart failure, stroke, amputation, and coronary and peripheral revascularization) increased progressively with increasing quartiles of baseline urine ACR. This result was confirmed by a multivariate analysis in which albuminuria was an independent risk factor for CV events with a 1.3-fold increased relative risk for each natural log increase of 1 U in urine ACR. In the IDNT study 1,715 subjects with type-2 diabetes, hypertension, and macroalbuminuria were randomized to irbesartan, amlodipine, or placebo for a mean period of 2.6 years. The patients had mean urine ACR of 1,416.2 mg/g. Moreover, the HOPE study investigators evaluated the risk of CV events associated with baseline microalbuminuria (defined as ACR > 2.0 mg/mmol (equivalent to 17.7 mg/g)). In the overall population, microalbuminuria at baseline approximately doubled the relative risk of the primary composite outcome (myocardial infarction, stroke, or CV death), and this effect was significant in both diabetics (relative risk, 1.97) and nondiabetics (relative risk, 1.61) [31]. Moreover, albuminuria was a continuous risk factor for CV events even below the level of microalbuminuria.
Future CV risk in the general population can be modeled in various ways, one typical approach being the Framingham score (though QRISK and other algorithms may be much superior especially for diverse populations). It has been shown that the Framingham score demonstrates poor overall accuracy in predicting cardiac events in individuals with CKD [32], and this might be due to the increased CV and overall mortality rates in these patients. Modification of the Framingham equations might improve their predictive accuracy, yet new models evaluating CV risk in this population should be developed.
Cardiovascular Risk Factors in CKD: The Role of Dyslipidemia
Evidence that reduced GFR and increased albuminuria independently and continuously predict higher CV event rates in CKD patients with or without preexistent CVD, prompts for the early detection and abrogation of the responsible factors that predispose these patients to the development of CVD. Apart from the traditional CV risk factors that are defined by epidemiological studies such as the Framingham study and are present in the general population, CKD patients also exhibit a variety of non-traditional risk factors that accelerate and aggravate the development of CVD in this population.
Traditional risk factors include smoking, diabetes, hypertension, left ventricular hypertrophy (LVH), older age, and hyperlipidemia. These factors are highly prevalent in the CKD setting [33], and they tend to increase the risk of CVD in early CKD stages [34]. Moreover, metabolic syndrome, a condition characterized by insulin resistance, elevated serum glucose, hypertension, abdominal obesity, and dyslipidemia, might also play a role in the development of CVD. This syndrome is also frequently detected in patients with CKD [35].
Although hypercholesterolemia is one of the most widely recognized CV risk factors in the general population as well as in patients with preexistent CVD [10, 36–39], this association in CKD has been difficult to establish. In fact, some studies have shown that low cholesterol levels associate with increased mortality in dialysis patients [11, 40, 41], whereas another study failed to detect any association between hyperlipidemia and mortality in nondiabetic stage 3–4 CKD patients [42]. However, this reverse epidemiology of lower cholesterol predicting a higher mortality is likely due to cholesterol-lowering effect of malnutrition and systemic inflammation, both present in CKD patients [11, 43]. Furthermore, the presence of numerous non-traditional risk factors in the CKD setting further confounds the association of CKD and CVD, thus increasing the prevalence of CVD even in patients with mild to moderate CKD [44]. These factors include increased oxidative stress, amplified inflammatory status, anemia, abnormalities in mineral-bone metabolism, endothelial dysfunction, and reduced nitric oxide (NO) activity [45–48].
The role of oxidative stress and inflammation in the development of CVD in CKD has recently been increasingly supported. In patients with CKD the balance between the production of reactive oxygen species (ROS)/free radicals (FR) and antioxidant defenses is shifted towards amplified oxidative stress. The increase in ROS/FR is caused by numerous factors such as uremic toxins, diabetes mellitus, chronic inflammation, and the dialysis treatments per se [49, 50]. Oxidative stress in uremia can be increased through activation of various ROS-producing enzymatic systems such as the reduced nicotinamide adenine dinucleotide (NAD(P)H) oxidase, xanthine oxidase, uncoupled endothelial NO synthase, and myeloperoxidase (MPO) [51, 52]. Among them, NAD(P)H oxidase seems to be the most important source of oxidative stress in vessels [53], whereas MPO, an enzymatic constituent of neutrophils and macrophages, is also highly expressed in atheromatic lesions [54]. MPO might also play a role in accelerated atherosclerosis in dialysis patients as it has been reported to be released from white blood cells during hemodialysis. Oxidative modification of macromolecules such as lipids, proteins, and nucleic acids results in structural and functional changes and accelerates atherosclerosis. In this regard, elevated plasma levels of oxidized low-density lipoprotein (LDL) have been shown to correlate with CHD [55]. Patients with CKD have increased levels of oxidized LDL [56].
Increased oxidative stress also induces the expression of inflammatory biomarkers. In this regard, both CKD and ESRD are characterized by elevated levels of inflammatory markers such as C-reactive protein (CRP), an acute phase protein reactant, TNF-α, IL-6, fibrinogen, factors VIIc and VIIIc, and the adhesion molecules VCAM-1 and ICAM-1 [57–59]. In the MDRD study elevated levels of CRP [60] were associated with an increased risk of all-cause and CV mortality in stage 3 and 4 CKD patients. Moreover, in dialysis patients increased levels of CRP and IL-6 have been associated with a significant increased risk of sudden cardiac death independently of traditional CVD risk factors [47, 61].
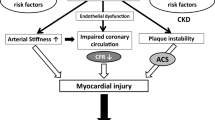
Endothelial dysfunction, which is present in CKD, is an important early event in the pathogenesis of atherosclerosis, contributing to plaque initiation and progression [62]. Microalbuminuria might be a manifestation of impaired endothelial function [63], thus explaining the link between microalbuminuria and increased CV morbidity and mortality reported in many epidemiology studies (see section on “Association Between CKD and CVD-Epidemiological Data”). Reduced NO production seems to be the main culprit for the endothelial dysfunction observed in CKD [64, 65]. In hemodialysis patients, increased levels of asymmetrical dimethylarginine (ADMA), an endogenous inhibitor of NO synthase, have been reported and independently predict overall mortality and CV outcomes [66]. Moreover, in patients with mild to advanced CKD, ADMA level is inversely related to GFR and represents an independent risk factor for progression to ESRD and mortality [67]. ADMA, now considered one of the strongest markers of atherosclerosis [68], is increased under inflammatory conditions and might serve as a link between inflammation and endothelial dysfunction [69]. Finally, the term protein-energy wasting (PEW) has been recently introduced to describe the role of malnutrition, inflammation, and atherosclerosis on the increased mortality observed in patients with ESRD [46, 70].
Yet, it should be noted that the exact pathomechanisms by which these nontraditional risk factors contribute to the development of CVD are still unclear, and various studies report contradictory results. In this regard, although the aforementioned studies demonstrated a positive association between elevated levels of CRP [60] and adverse CV outcomes, no such relationship was detected in the Irbesartan for Diabetic nephropathy trial [71]. Similarly, although coronary artery calcification, which is a frequent finding in CKD patients, has been linked to abnormalities in mineral-bone metabolism [72], studies evaluating the associations between parathyroid hormone, calcium, and phosphorus with coronary artery calcification report conflicting results [73, 74].
Characteristics of Dyslipidemia in CKD
Multiple lipoprotein abnormalities are detected in CKD patients caused by a profound dysregulation in their metabolism (Table 5.3). The primary characteristic of dyslipidemia in CKD is hypertriglyceridemia, with 40–50 % of CKD patients having fasting triglyceride levels greater than 200 mg/dL. Of note, the lipid profile of patients with CKD depends on CKD stage, the presence or not of nephrotic syndrome, and the dialysis modality for ESRD patients [75]. In this regard, apart from the increased triglycerides, in patients with stage 5 CKD total serum cholesterol and LDL are normal or low, whereas high-density lipoprotein (HDL) is decreased [76, 77]. Patients with stage 1–4 CKD might exhibit increased total cholesterol and LDL cholesterol, whereas nephrotic patients are usually characterized by a marked increase in total cholesterol and LDL levels with cholesterol being directly correlated with the degree of albuminuria and indirectly with serum albumin level [78]. Finally, peritoneal dialysis patients are characterized by increased protein losses in the peritoneal fluid effluent, a condition mimicking the nephrotic syndrome [79]. These losses might induce the hepatic production of albumin and lipoproteins, resulting in elevated concentrations of total cholesterol, LDL, and a modified highly atherogenic form of LDL, lipoprotein (a) (Lp(a)) [80, 81]. Moreover, increased insulin levels, which is a consequence of the absorption of glucose from the dialysis fluid, may induce the hepatic synthesis of very low-density lipoprotein (VLDL) and possibly Lp(a) [82].
It should be noted that in CKD, apart from lipoproteins assessed in every day clinical practice, a variety of not routinely measured highly atherogenic lipoprotein fragments accumulate. These include chylomicron remnants, VLDL remnants or intermediate-density lipoproteins (IDL), oxidized LDL, small dense-LDL (sd-LDL), and Lp(a) [75]. The former two are products of chylomicron and VLDL metabolism whose clearance is impaired in CKD. These lipoproteins are prone to oxidization, a process that further increases their atherogenic potential. In fact, Shoji et al. showed that among LDL, HDL, VLDL, IDL, and Lp(a), IDL was the lipoprotein fraction more closely associated with aortic sclerosis in hemodialysis patients [83]. Moreover, chylomicron remnants potentiated endothelium-dependent arterial contraction [84], whereas oxidized VLDL remnants significantly enhanced macrophage cholesterol ester accumulation compared to either VLDL remnants, or oxidized LDL in experimental models of atherosclerosis [85]. It is well known that most clinical trials evaluating the role of lipid-lowering treatments in CKD patients do not assess these lipoprotein fractions, and this might be an explanation for the reported negative results.
In CKD there is an impairment in the distribution of LDL subclasses favoring the predominance of sd-LDL particles [86, 87] (see Table 5.3). These electronegative particles penetrate the endothelial barrier easier than large LDL particles and interact with electropositive intimal proteoglycans [88]. This interaction prolongs their retention in the arterial wall, thus rendering them more susceptible to oxidization by ROS. Indeed, sd-LDL particles appear to be more atherogenic than larger LDL fragments [89, 90]. The effect of the routinely administered lipid-lowering agents, statins on sd-LDL in CKD patients, remains unclear. Of note, a recent study reported that statins decrease sd-LDL in peritoneal dialysis patients but not in hemodialysis patients [91]. Apart from oxidization, under the uremic milieu, LDL also undergoes protein carbamylation. This process has been reported to increase the atherogenic potential of LDL through multiple mechanisms, including the proliferation of smooth muscle cells [92].
Elevated plasma levels of Lp(a) have been detected in patients with CKD [93] (see Table 5.3). Moreover, Lp(a) is now recognized as a risk factor for CV morbidity and mortality in hemodialysis patients [94, 95]. Lp(a) is a modified form of LDL emerging from the covalent binding of apolipoprotein(a) to apolipoprotein B through disulfide linkage [96]. In the general population, elevated serum Lp(a), has been recognized as a risk factor for CVD, whereas the association between Lp(a) and CHD risk seem to be continuous [97, 98]. Lp(a) excess is frequently detected in patients with premature CHD [99], and its levels are also associated with cerebrovascular disease, especially in men [100]. Because of its structural similarity to plasminogen, it has been proposed that Lp(a) may promote thrombogenesis by inhibiting fibrinolysis [101]. Moreover, Lp(a) is capable of binding to macrophage receptors, thus promoting foam cell formation and accelerating atherosclerosis [102]. Lp(a) also enhances LDL susceptibility to oxidization [55] and promotes monocyte attachment to vascular endothelial cells by increasing endothelial ICAM-1 expression [103]. Serum Lp(a) levels are genetically determined and are mostly due to polymorphisms at the apo(a) gene (LPA gene) [104]. These polymorphisms account for a variety of sizes of apo(a) isoforms. An inverse association of the size of apo(a) isoforms and Lp(a) levels has been detected (i.e., subjects with low molecular weight isoforms have higher levels of Lp(a)) [104, 105]. Intriguingly, in dialysis patients, small apo(a) isoform size but not Lp(a) level has been identified as an independent predictor of total and CV mortality [106, 107]. There are no clinical trials evaluating the effects of therapeutic regimes targeting the reduction of Lp(a) on CV morbidity and mortality in the general population or in patients with CKD.
Disorders of VLDL and Chylomicron Metabolism in CKD
Hypertriglyceridemia in CKD is a consequence of impaired VLDL and chylomicron metabolism, which leads to diminished triglyceride clearance [108] (Fig. 5.1a, b). VLDL and chylomicrons are triglyceride-rich lipoproteins that deliver lipids to muscle and adipose tissue for energy production and storage, respectively. Nascent VLDL consists of a apoB100 lipoprotein core, to which cholesterol ester, triglycerides, and phospholipids are bound. Similarly, in nascent chylomicrons, these lipid fractions are packed in apo48. Both nascent VLDL and chylomicrons mature by receiving apoE and apoC from HDL-2. Endothelium-bound lipoprotein lipase (LPL) in the capillaries of skeletal muscle and adipose tissue is responsible for the hydrolysis of triglycerides of VLDL and chylomicron and the disposal of fatty acids to the adjacent myocytes and adipocytes. This process leads to the formation of VLDL remnants (IDL) and chylomicron remnants, which are subsequently cleared by the liver via LDL receptor-related protein (LRP) [109]. However, the bulk of IDL is converted to LDL after being enriched in cholesterol esters by cholesterol ester transfer protein (CETP) and then lysed by hepatic lipase. LDL is then removed by LDL receptor in the hepatic cells. Finally, the fraction of VLDL that has not been lysed by LPL is cleared entirely by VLDL receptor in adipocytes and myocytes [110]. Impaired metabolism of VLDL in CKD is characterized by the downregulation of LPL, hepatic lipase, LPR, and VLDL receptor, leading to the accumulation of triglycerides, VLDL, and IDL and the triglyceride enrichment of LDL (see Fig. 5.1a, b and Table 5.3). In CKD an increase in plasma Apolipoprotein C-III (apoCIII), a potent inhibitor of LPL, has been reported [75]. ApoCIII and apo CII are important components of VLDL and chylomicrons, and apoCIII/apoCII ratio determines the ability of these lipoproteins to activate LPL. Moreover, in CKD-increased plasma levels of pre-β-HDL, an inhibitor of LPL has been reported [111]. Regular heparinization that occurs in dialysis patients might also result in the degradation of tissue-bound LPL [75].
VLDL (a) and chylomicron (b) metabolism. The asterisk denotes downregulation. See text for details. Adapted from [108]
Another mechanism involved in the impaired lipoprotein metabolism in CKD setting is the reduced expression and activity of hepatic lipase [112, 113]. As discussed earlier, hepatic lipase is responsible for the removal of almost all of the remaining triglycerides from IDL, a process crucial for its conversion to LDL. Therefore, hepatic lipase downregulation leads to the accumulation of IDL, triglyceride enrichment of LDL, and hypertriglyceridemia. Hepatic lipase as well as LPL activity might be diminished by calcium accumulation within liver and adipose tissue cells caused by secondary hyperparathyroidism, a common complication of CKD. Of note, parathyroidectomy can restore both hepatic lipase and LPL activity and plasma triglyceride levels in experimental animals and humans with CKD [114, 115]. Moreover, verapamil administration to rats with CKD prevented the development of hypertriglyceridemia and the reduction of hepatic lipase and LPL activity by reducing basal levels of cytosolic calcium [112].
CKD has also been reported to downregulate LRP, thus leading to the atherogenic chylomicron remnants and IDL accumulation [116]. The downregulation of VLDL receptors in the skeletal muscle, heart, and adipose tissue has also been reported in experimental animals with CKD, a condition leading to elevated VLDL and triglycerides [115, 117]. Finally, increased triglyceride synthesis might contribute to the hypertriglyceridemia observed in nephrotic patients and peritoneal dialysis patients but not in the remainder CKD population [118]. This is due to the upregulation of Acyl-CoA:diglycerol acyltransferase, an enzyme that catalyzes the conversion of diglyceride to triglyceride [119].
As discussed earlier, CKD is characterized by increased oxidative stress. In this environment chylomicron remnants, IDL, LDL, sd-LDL, and Lp(a) might undergo oxidization. These oxidized lipoproteins can bind to receptors on macrophages and trigger the release of pro-inflammatory cytokines thus amplifying the inflammatory status of CKD. The uptake of these lipoproteins by the scavenger receptors of arterial wall macrophages results in their transformation to foam cells, the hallmark of the atherosclerotic lesion. In this regard, increased scavenger receptor expression has been reported in CKD patients [120]. The formation of foam cells is also a consequence of impaired cholesterol export mechanisms (see section on “Disorders of HDL Metabolism in CKD”). LDL has also been reported to activate the renin–angiotensin system [121], leading to increased angiotensin II levels and the upregulation of the angiotensin type-I (AT1) receptor. Angiotensin II, in turn, acting through AT1 receptor, stimulates NAD(P)H oxidase and other enzymes, augmenting synthesis of the superoxide anion and proinflammatory mediators that result in endothelial dysfunction and atherosclerosis aggravation.
Disorders of HDL Metabolism in CKD
In CKD, impaired functionality and reduced levels of HDL have been reported [75]. Normally, HDL prevents atherosclerosis by various mechanisms [108, 122–125] (Fig. 5.2): (1) inhibits and reverses the oxidization of lipoproteins by its antioxidant enzyme constituents, paraoxonase-1 (PON-1) and glutathione peroxidase (GPX); (2) removes oxidized phospholipids and endotoxins and disposes them to the liver via apoA1 and lecithin:cholesterol acyltransferase (LCAT); (3) improves endothelial function by inhibiting cellular adhesion molecule expression [126] and increasing eNOS production [127]; (4) reduces inflammation by alleviating oxidative stress and inhibiting cellular adhesion molecule expression; (5) transfers surplus cholesterol and phospholipids from the periphery to the liver (reverse cholesterol transport [RCT]); (6) contributes apoC and apoE to nascent VLDL and chylomicrons, thus facilitating their proper metabolism and removal; (7) facilitates the conversion of highly atherogenic oxidization-prone IDL to LDL via CETP-mediated exchange of cholesterol esters for triglycerides (indirect RCT); (8) exerts antithrombotic effects through its constituent platelet-activating factor (PAF) acetylhydrolase, which inactivates PAF, thus preventing platelet activation and thrombus formation.
Protective role of HDL against atherosclerosis. Reverse cholesterol pathway. The asterisk denotes downregulation, whereas the plus (+) denotes elevation. See text for details. Adapted from [108]
RCT is a multiorgan, multistep process via which excess cholesterol is retrieved from lipid-laden macrophages in the peripheral tissue and then is transported to the liver, where it is processed and excreted in bile and intestine [108, 123, 128, 129] (see Fig. 5.2). Oxidized LDL and other atherogenic lipid fractions are internalized by vascular macrophages through scavenger receptors (SRA1 and LOX1). This leads to their transformation into foam cells and the acceleration of atherosclerosis. In RCT nascent (lipid-poor) HDL binds to ATP-binding cassette transporter type A1 (ABCA1) and ABCG1 on the macrophage cell membrane [123, 130]. Then free cholesterol is actively transferred to the surface of HDL where it is rapidly esterified by LCAT and then sequestered in the core of HDL (mature HDL). Of note, albumin has also been shown to play a role in transferring cellular-free cholesterol from peripheral tissues to the circulating nascent HDL via passive transportation [131]. Thereafter, mature HDL moves to the liver, where it binds to the HDL docking receptor SRB-1. SRB-1 facilitates the unloading of HDL’s lipid content (cholesterol esters, triglycerides, and phospholipids) and subsequently HDL is released to the circulation as lipid-poor HDL to repeat the cycle [132].
CKD is associated with a reduction in serum apoA-I and apoA-II, which are mandatory components of the HDL particle [133, 134]. This mechanism might play a crucial role in the reduction of HDL levels detected in CKD patients. Hypoalbuminemia, which is a result of chronic inflammation, in CKD patients might also contribute to reduced HDL levels [77]. However, the main reason for the impaired HDL cholesterol enrichment and maturation in CKD is LCAT deficiency (see Fig. 5.2 and Table 5.3). LCAT deficiency is a result of decreased production by the liver [135]. LCAT deficiency, apart from preventing the maturation of HDL through the esterification of free cholesterol on its surface, also facilitates HDL degradation by the hepatic endocytic receptor (β-chain of ATP synthase). This receptor has higher affinity for the nascent HDL than the mature one, whereas SRB-1 has higher affinity for mature HDL and, as noted above, facilitates HDL cycle from the liver to peripheral tissues and does not degrade it. Apart from the reduced levels of HDL in CKD, there also seems to exist a decreased affinity to its ABCA-1 macrophage receptors due to its oxidization in the uremic milieu [136, 137]. Accumulating evidence also suggests that HDL under systemic oxidative and inflammatory conditions (as in CKD) might also transform and promote oxidative stress and inflammation [138, 139]. Thus, HDL oxidization might impair the maturation of HDL and RCT in general. As discussed earlier, by inhibiting the formation and increasing the disposal of oxidized lipids, HDL exerts both antioxidant and anti-inflammatory effects. It has been reported that in dialysis patients there is a significant reduction in paraoxonase and GPX [136]. Moreover, the expression of macrophage scavenger receptors SRA1 and LOX1 is upregulated in both experimental models and in patients with CKD, a process that seems to be induced by inflammatory cytokines and oxidized LDL [120, 140]. This, combined with apoA-1 reduction, limits the ability of HDL to prevent or reverse the oxidization of LDL and phospholipids, thus promoting an influx of oxidized LDL in macrophages in the artery wall and facilitating foam cell formation and atherosclerosis. In this context, HDL anti-inflammatory activity has been reported to decrease in the uremic plasma of dialysis patients [141]. Thus, it seems that HDL impaired anti-oxidant and anti-inflammatory properties are both a consequence and a cause of increased oxidative stress and inflammation observed in CKD.
Based on the evidence that HDL level is reduced in CKD patients, the design of treatment strategies targeted to increase HDL levels seems plausible. However, efforts to raise HDL in the clinical setting have unexpectedly resulted in unfavorable CV outcomes. CETP inhibitors are a novel class of compounds that are very effective in increasing plasma HDL [142]. However, despite a meaningful increase of plasma HDL, the administration of the CETP inhibitor torcetrapib was early terminated in patients at high risk of coronary events due to increased CV events and overall mortality [143]. This negative outcome was probably related to an off-target effect indicated by increased arterial blood pressure in the treatment group, although a possible torcetrapib adverse effect could not be ruled out. Moreover, in another RCT, torcetrapib failed to halt the progression of coronary atherosclerosis or improve carotid intimal thickness [144]. These discouragingly negative outcomes might be a result of the accumulation of the highly atherogenic IDL [145]. As discussed earlier, CETP plays a crucial role in the conversion of IDL to LDL by promoting the transfer of cholesterol esters from IDL to LDL in exchange of triglycerides [108]. Therefore, CETP inhibition might result in the accumulation of IDL and the acceleration of atherosclerosis, especially in CKD patients in whom IDL clearance is impaired due to LRP-1 and hepatic lipase deficiency. Moreover, neither low HDL nor high CETP activity was associated with CV events in hemodialysis patients over a 48-month observation period [146], implying that functional changes in HDL might play a more important role in atherosclerosis progression. Thus, in CKD, the absolute increase in HDL levels might not be enough to prevent CVD, as there also exist qualitative changes in the HDL molecule impairing its composition, maturation process, as well as its antioxidant and anti-inflammatory properties [136, 139, 147].
Pathophysiology of CVD in the General Population and in the CKD Setting
Atherosclerosis is a chronic, complex, and progressive inflammatory process of the vascular wall of large and medium-sized arteries. Although the exact pathomechanism of this process remains unclear, dyslipidemia and abnormal immune response to endothelial damage with inflammatory recruitment of monocytes and the formation of foam cells seem to play a central role in the development of the atherosclerotic lesions [148, 149]. The chronic inflammation of the vascular wall results in multifocal plaque development. Furthermore, intraplaque hemorrhage, lipid deposition, proliferation of neovessels, and plaque remodeling all contribute to atherosclerotic plaque formation [149, 150]. Although most plaques remain asymptomatic, some progress to luminal obstruction, whereas a few are vulnerable to thrombosis, leading to acute atherothrombotic events such as acute myocardial infarction and stroke [149, 151].
Intriguingly, the pathogenesis and, consequently, the pathologic findings of arterial lesions in CKD patients differ substantially from patients with preserved renal function with classic atherosclerotic disease. In the latter, arterial lesions consist of lipid-laden atheromatous or fibroatheromatous plaques, whereas in the former, atherosclerotic plaques are rich in calcium deposits and fibrous tissue and exhibit a prominent thickening of the intima and media of the vessel wall, resulting in lumen narrowing [152, 153]. Calcium deposition in CKD may occur in the intima (as in classic atherosclerotic plaques) or in the medial layer, where it increases vascular stiffness as well as in cardiac valves [154]. In fact, coronary artery calcification has been reported to be significantly more frequent in predialysis patients compared with matched control subjects with no renal impairment (40 % vs. 13 %) [155]. The calcium content of atherosclerotic lesions in advanced CKD stages is in some occasions so high that many of these patients’ vessels can be readily delineated on simple plain radiograms. The most devastating demonstration of vascular calcification presented in dialysis patients is calciphylaxis or calcific uremic arteriopathy. This life-threatening condition is characterized by extensive microvascular calcification accompanied by intimal proliferation and thrombosis leading to non-healing skin ulcers, necrosis, secondary infection, and sepsis [156]. Although the exact pathomechanisms contributing to vascular calcification remain unclear, elevated calcium (Ca) × phosphate (P) product facilitating the precipitation of Ca and P along with the induction of calcification promoters and the reduction of calcification inhibitors seem to play a major role in this process [157, 158]. Under these conditions, vascular smooth muscle cells acquire an osteoblast phenotype, thus promoting hydroxyapatite formation in the media resulting in vascular calcification. Vascular calcification promotes vascular stiffening. Aortic stiffening combined with anemia and hypertension, which are common in CKD patients, result in the development of LVH. The combination of LVH and tissue calcification may result in myocardial fibrosis and conduction abnormalities that predispose to potentially lethal arrhythmias [152, 153, 159]. Indeed, arrhythmias or cardiac arrest seem to be more common death causes in CKD patients (they account for approximately 60 % of all cardiac deaths in dialysis patients) than myocardial infarction or stroke, which represent typical atherosclerotic diseases [160].
Conclusion
It is well established that reduced GFR and albuminuria predict CV event rates in a continuous and independent manner. This relation between CVD and CKD, apart from the presence of non-traditional CV risk factors such as oxidative stress and inflammation, which are prominent in CKD, has also been attributed to the substantial variations of lipid abnormalities and dyslipidemia characteristics in CKD. Indeed, hypertriglyceridemia is the hallmark of CKD dyslipidemia, whereas total and LDL cholesterol are normal or low. Moreover, a variety of highly atherogenic lipoprotein fragments, which are minimally affected by classic lipid-lowering treatments, such as chylomicron remnants, IDL, sd-LDL, oxidized LDL, and Lp(α), are present in CKD, further aggravating atherosclerotic lesions. Finally, the pathogenesis of arterial lesions in CKD differs substantially from the pathogenesis of classical atherosclerotic disease. In this regard, calcium-rich atherosclerotic plaques with prominent thickening of the intima and media of the vessel wall are the pathologic hallmarks of CKD atherosclerotic lesions, whereas lipid-laden atheromatous or fibroatheromatous plaques are detected in classic atherosclerotic disease. Therefore, it is not surprising that lipid-lowering strategies alone seem to have no meaningful effect in ameliorating CVD in CKD patients, especially in those with advanced renal failure [161, 162]. Thus, an adequately designed therapeutic regime apart from modifying lipoprotein levels (i.e., by the use of a statin) should also probably include agents that reduce oxidative stress, inflammation, and vascular calcification. Therefore, combined treatments targeted at inhibiting or reversing multilevel pathogenetic mechanisms responsible for CVD in CKD will pave the way for the effective management of dyslipidemia and CVD in this fragile population.
References
Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, et al. Prevalence of chronic kidney disease in the United States. JAMA. 2007;298(17):2038–47. PubMed PMID: 17986697.
Foley RN, Collins AJ. End-stage renal disease in the United States: an update from the United States Renal Data System. J Am Soc Nephrol. 2007;18(10):2644–8. PubMed PMID: 17656472.
Leoncini G, Viazzi F, Pontremoli R. Overall health assessment: a renal perspective. Lancet. 2010;375(9731):2053–4. PubMed PMID: 20483450. Epub 2010/05/21.eng.
van der Velde M, Matsushita K, Coresh J, Astor BC, Woodward M, Levey A, et al. Lower estimated glomerular filtration rate and higher albuminuria are associated with all-cause and cardiovascular mortality. A collaborative meta-analysis of high-risk population cohorts. Kidney Int. 2011;79(12):1341–52. PubMed PMID: 21307840. Epub 2011/02/11.eng.
Levey AS, de Jong PE, Coresh J, El Nahas M, Astor BC, Matsushita K, et al. The definition, classification, and prognosis of chronic kidney disease: a KDIGO Controversies Conference report. Kidney Int. 2011;80(1):17–28. PubMed PMID: 21150873. Epub 2010/12/15.eng.
Sarnak MJ, Levey AS, Schoolwerth AC, Coresh J, Culleton B, Hamm LL, et al. Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Hypertension. 2003;42(5):1050–65. PubMed PMID: 14604997.
Manjunath G, Tighiouart H, Coresh J, Macleod B, Salem DN, Griffith JL, et al. Level of kidney function as a risk factor for cardiovascular outcomes in the elderly. Kidney Int. 2003;63(3):1121–9. PubMed PMID: 12631096.
Cheung AK, Sarnak MJ, Yan G, Berkoben M, Heyka R, Kaufman A, et al. Cardiac diseases in maintenance hemodialysis patients: results of the HEMO Study. Kidney Int. 2004;65(6):2380–9. PubMed PMID: 15149351. Epub 2004/05/20.eng.
United States Renal Data System. Excerpts from USRDS 2009 Annual Data Report. U.S. Department of Health and Human Services. The National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases. Am J Kidney Dis. 2010;55(Suppl 1):S1.
Law MR, Wald NJ, Thompson SG. By how much and how quickly does reduction in serum cholesterol concentration lower risk of ischaemic heart disease? BMJ. 1994;308(6925):367–72. PubMed PMID: 8043072.
Liu Y, Coresh J, Eustace JA, Longenecker JC, Jaar B, Fink NE, et al. Association between cholesterol level and mortality in dialysis patients: role of inflammation and malnutrition. JAMA. 2004;291(4):451–9. PubMed PMID: 14747502.
Stevens LA, Padala S, Levey AS. Advances in glomerular filtration rate-estimating equations. Curr Opin Nephrol Hypertens. 2010;19(3):298–307. PubMed PMID: 20393287. Epub 2010/04/16.eng.
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–266. PubMed PMID: 11904577. Epub 2002/03/21.eng.
Levey AS, Eckardt KU, Tsukamoto Y, Levin A, Coresh J, Rossert J, et al. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2005;67(6):2089–100. PubMed PMID: 15882252. Epub 2005/05/11.eng.
Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1–150.
Tonelli M, Wiebe N, Culleton B, House A, Rabbat C, Fok M, et al. Chronic kidney disease and mortality risk: a systematic review. J Am Soc Nephrol. 2006;17(7):2034–47. PubMed PMID: 16738019.
Muntner P, He J, Hamm L, Loria C, Whelton PK. Renal insufficiency and subsequent death resulting from cardiovascular disease in the United States. J Am Soc Nephrol. 2002;13(3):745–53. PubMed PMID: 11856780. Epub 2002/02/22.eng.
Sarnak MJ, Levey AS, Schoolwerth AC, Coresh J, Culleton B, Hamm LL, et al. Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Circulation. 2003;108(17):2154–69. PubMed PMID: 14581387. Epub 2003/10/29.eng.
Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, de Jong PE, et al. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet. 2010;375(9731):2073–81. PubMed PMID: 20483451. Epub 2010/05/21.eng.
Tonelli M, Muntner P, Lloyd A, Manns BJ, James MT, Klarenbach S, et al. Using proteinuria and estimated glomerular filtration rate to classify risk in patients with chronic kidney disease: a cohort study. Ann Intern Med. 2011;154(1):12–21. PubMed PMID: 21200034. Epub 2011/01/05.eng.
Weiner DE, Tighiouart H, Amin MG, Stark PC, MacLeod B, Griffith JL, et al. Chronic kidney disease as a risk factor for cardiovascular disease and all-cause mortality: a pooled analysis of community-based studies. J Am Soc Nephrol. 2004;15(5):1307–15. PubMed PMID: 15100371. Epub 2004/04/22.eng.
Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296–305. PubMed PMID: 15385656. Epub 2004/09/24.eng.
Rahman M, Pressel S, Davis BR, Nwachuku C, Wright Jr JT, Whelton PK, et al. Cardiovascular outcomes in high-risk hypertensive patients stratified by baseline glomerular filtration rate. Ann Intern Med. 2006;144(3):172–80. PubMed PMID: 16461961. Epub 2006/02/08.eng.
Perkovic V, Ninomiya T, Arima H, Gallagher M, Jardine M, Cass A, et al. Chronic kidney disease, cardiovascular events, and the effects of perindopril-based blood pressure lowering: data from the PROGRESS study. J Am Soc Nephrol. 2007;18(10):2766–72. PubMed PMID: 17804673. Epub 2007/09/07.eng.
Schneider CA, Ferrannini E, Defronzo R, Schernthaner G, Yates J, Erdmann E. Effect of pioglitazone on cardiovascular outcome in diabetes and chronic kidney disease. J Am Soc Nephrol. 2008;19(1):182–7. PubMed PMID: 18057215. Epub 2007/12/07.eng.
Segura J, Campo C, Gil P, Roldan C, Vigil L, Rodicio JL, et al. Development of chronic kidney disease and cardiovascular prognosis in essential hypertensive patients. J Am Soc Nephrol. 2004;15(6):1616–22. PubMed PMID: 15153573. Epub 2004/05/22.eng.
Knobler H, Zornitzki T, Vered S, Oettinger M, Levy R, Caspi A, et al. Reduced glomerular filtration rate in asymptomatic diabetic patients: predictor of increased risk for cardiac events independent of albuminuria. J Am Coll Cardiol. 2004;44(11):2142–8. PubMed PMID: 15582311. Epub 2004/12/08.eng.
Kong AP, So WY, Szeto CC, Chan NN, Luk A, Ma RC, et al. Assessment of glomerular filtration rate in addition to albuminuria is important in managing type II diabetes. Kidney Int. 2006;69(2):383–7. PubMed PMID: 16408130. Epub 2006/01/13.eng.
Mann JF, Gerstein HC, Pogue J, Bosch J, Yusuf S. Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: the HOPE randomized trial. Ann Intern Med. 2001;134(8):629–36. PubMed PMID: 11304102. Epub 2001/04/17.eng.
Anavekar NS, Gans DJ, Berl T, Rohde RD, Cooper W, Bhaumik A, et al. Predictors of cardiovascular events in patients with type 2 diabetic nephropathy and hypertension: a case for albuminuria. Kidney Int Suppl. 2004;92:S50–5. PubMed PMID: 15485418.Epub 2004/10/16.eng.
Mann JF, Yi QL, Gerstein HC. Albuminuria as a predictor of cardiovascular and renal outcomes in people with known atherosclerotic cardiovascular disease. Kidney Int Suppl. 2004;92:S59–62. PubMed PMID: 15485420. Epub 2004/10/16.eng.
Weiner DE, Tighiouart H, Elsayed EF, Griffith JL, Salem DN, Levey AS, et al. The Framingham predictive instrument in chronic kidney disease. J Am Coll Cardiol. 2007;50(3):217–24. PubMed PMID: 17631213. Epub 2007/07/17.eng.
Muntner P, He J, Astor BC, Folsom AR, Coresh J. Traditional and nontraditional risk factors predict coronary heart disease in chronic kidney disease: results from the atherosclerosis risk in communities study. J Am Soc Nephrol. 2005;16(2):529–38. PubMed PMID: 15625072. Epub 2004/12/31.eng.
Shlipak MG, Fried LF, Cushman M, Manolio TA, Peterson D, Stehman-Breen C, et al. Cardiovascular mortality risk in chronic kidney disease: comparison of traditional and novel risk factors. JAMA. 2005;293(14):1737–45. PubMed PMID: 15827312. Epub 2005/04/14.eng.
Kobayashi S, Maesato K, Moriya H, Ohtake T, Ikeda T. Insulin resistance in patients with chronic kidney disease. Am J Kidney Dis. 2005;45(2):275–80. PubMed PMID: 15685504. Epub 2005/02/03.eng.
Sacks FM, Pfeffer MA, Moye LA, Rouleau JL, Rutherford JD, Cole TG, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N Engl J Med. 1996;335(14):1001–9. PubMed PMID: 8801446.
Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet. 2002;360(9326):7–22. PubMed PMID: 12114036.
Sever PS, Dahlof B, Poulter NR, Wedel H, Beevers G, Caulfield M, et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial-Lipid Lowering Arm (ASCOT-LLA): a multicentre randomised controlled trial. Drugs. 2004;64 Suppl 2:43–60. PubMed PMID: 15765890.
Rossouw JE, Lewis B, Rifkind BM. The value of lowering cholesterol after myocardial infarction. N Engl J Med. 1990;323(16):1112–9. PubMed PMID: 2215579.
Kilpatrick RD, McAllister CJ, Kovesdy CP, Derose SF, Kopple JD, Kalantar-Zadeh K. Association between serum lipids and survival in hemodialysis patients and impact of race. J Am Soc Nephrol. 2007;18(1):293–303. PubMed PMID: 17167113. Epub 2006/12/15.eng.
Iseki K, Yamazato M, Tozawa M, Takishita S. Hypocholesterolemia is a significant predictor of death in a cohort of chronic hemodialysis patients. Kidney Int. 2002;61(5):1887–93. PubMed PMID: 11967041. Epub 2002/04/23.eng.
Chawla V, Greene T, Beck GJ, Kusek JW, Collins AJ, Sarnak MJ, et al. Hyperlipidemia and long-term outcomes in nondiabetic chronic kidney disease. Clin J Am Soc Nephrol. 2010;5(9):1582–7. PubMed PMID: 20558558. Epub 2010/06/19.eng.
Kalantar-Zadeh K, Block G, Humphreys MH, Kopple JD. Reverse epidemiology of cardiovascular risk factors in maintenance dialysis patients. Kidney Int. 2003;63(3):793–808. PubMed PMID: 12631061. Epub 2003/03/13.eng.
Schiffrin EL, Lipman ML, Mann JF. Chronic kidney disease: effects on the cardiovascular system. Circulation. 2007;116(1):85–97. PubMed PMID: 17606856. Epub 2007/07/04.eng.
Kaysen GA, Eiserich JP. The role of oxidative stress-altered lipoprotein structure and function and microinflammation on cardiovascular risk in patients with minor renal dysfunction. J Am Soc Nephrol. 2004;15(3):538–48. PubMed PMID: 14978155. Epub 2004/02/24.eng.
Weiner DE, Tighiouart H, Elsayed EF, Griffith JL, Salem DN, Levey AS, et al. The relationship between nontraditional risk factors and outcomes in individuals with stage 3 to 4 CKD. Am J Kidney Dis. 2008;51(2):212–23. PubMed PMID: 18215699. Epub 2008/01/25.eng.
Parekh RS, Plantinga LC, Kao WH, Meoni LA, Jaar BG, Fink NE, et al. The association of sudden cardiac death with inflammation and other traditional risk factors. Kidney Int. 2008;74(10):1335–42. PubMed PMID: 18769368. Epub 2008/09/05.eng.
Kendrick J, Chonchol MB. Nontraditional risk factors for cardiovascular disease in patients with chronic kidney disease. Nat Clin Pract Nephrol. 2008;4(12):672–81. PubMed PMID: 18825155. Epub 2008/10/01.eng.
Canaud B, Cristol J, Morena M, Leray-Moragues H, Bosc J, Vaussenat F. Imbalance of oxidants and antioxidants in haemodialysis patients. Blood Purif. 1999;17(2–3):99–106. PubMed PMID: 10449867. Epub 1999/08/18.eng.
Small DM, Coombes JS, Bennett N, Johnson DW, Gobe GC. Oxidative stress, anti-oxidant therapies and chronic kidney disease. Nephrology (Carlton). 2012;17(4):311–21. PubMed PMID: 22288610. Epub 2012/02/01.eng.
Himmelfarb J, Stenvinkel P, Ikizler TA, Hakim RM. The elephant in uremia: oxidant stress as a unifying concept of cardiovascular disease in uremia. Kidney Int. 2002;62(5):1524–38. PubMed PMID: 12371953. Epub 2002/10/10.eng.
Vaziri ND, Dicus M, Ho ND, Boroujerdi-Rad L, Sindhu RK. Oxidative stress and dysregulation of superoxide dismutase and NADPH oxidase in renal insufficiency. Kidney Int. 2003;63(1):179–85. PubMed PMID: 12472781. Epub 2002/12/11.eng.
Touyz RM, Yao G, Quinn MT, Pagano PJ, Schiffrin EL. p47phox associates with the cytoskeleton through cortactin in human vascular smooth muscle cells: role in NAD(P)H oxidase regulation by angiotensin II. Arterioscler Thromb Vasc Biol. 2005;25(3):512–8. PubMed PMID: 15618548. Epub 2004/12/25.eng.
Daugherty A, Dunn JL, Rateri DL, Heinecke JW. Myeloperoxidase, a catalyst for lipoprotein oxidation, is expressed in human atherosclerotic lesions. J Clin Invest. 1994;94(1):437–44. PubMed PMID: 8040285. Epub 1994/07/01.eng.
Tsimikas S, Brilakis ES, Miller ER, McConnell JP, Lennon RJ, Kornman KS, et al. Oxidized phospholipids, Lp(a) lipoprotein, and coronary artery disease. N Engl J Med. 2005;353(1):46–57. PubMed PMID: 16000355. Epub 2005/07/08.eng.
Maggi E, Bellazzi R, Falaschi F, Frattoni A, Perani G, Finardi G, et al. Enhanced LDL oxidation in uremic patients: an additional mechanism for accelerated atherosclerosis? Kidney Int. 1994;45(3):876–83. PubMed PMID: 8196291. Epub 1994/03/01.eng.
Rao M, Wong C, Kanetsky P, Girndt M, Stenvinkel P, Reilly M, et al. Cytokine gene polymorphism and progression of renal and cardiovascular diseases. Kidney Int. 2007;72(5):549–56. PubMed PMID: 17579660. Epub 2007/06/21.eng.
Jofre R, Rodriguez-Benitez P, Lopez-Gomez JM, Perez-Garcia R. Inflammatory syndrome in patients on hemodialysis. J Am Soc Nephrol. 2006;17(12 Suppl 3):S274–80. PubMed PMID: 17130274. Epub 2006/11/30.eng.
Vaziri ND, Oveisi F, Ding Y. Role of increased oxygen free radical activity in the pathogenesis of uremic hypertension. Kidney Int. 1998;53(6):1748–54. PubMed PMID: 9607208. Epub 1998/06/02.eng.
Menon V, Greene T, Wang X, Pereira AA, Marcovina SM, Beck GJ, et al. C-reactive protein and albumin as predictors of all-cause and cardiovascular mortality in chronic kidney disease. Kidney Int. 2005;68(2):766–72. PubMed PMID: 16014054. Epub 2005/07/15.eng.
Honda H, Qureshi AR, Heimburger O, Barany P, Wang K, Pecoits-Filho R, et al. Serum albumin, C-reactive protein, interleukin 6, and fetuin a as predictors of malnutrition, cardiovascular disease, and mortality in patients with ESRD. Am J Kidney Dis. 2006;47(1):139–48. PubMed PMID: 16377395. Epub 2005/12/27.eng.
Endemann DH, Schiffrin EL. Endothelial dysfunction. J Am Soc Nephrol. 2004;15(8):1983–92. PubMed PMID: 15284284. Epub 2004/07/31.eng.
Stehouwer CD, Smulders YM. Microalbuminuria and risk for cardiovascular disease: analysis of potential mechanisms. J Am Soc Nephrol. 2006;17(8):2106–11. PubMed PMID: 16825333. Epub 2006/07/11.eng.
Wever R, Boer P, Hijmering M, Stroes E, Verhaar M, Kastelein J, et al. Nitric oxide production is reduced in patients with chronic renal failure. Arterioscler Thromb Vasc Biol. 1999;19(5):1168–72. PubMed PMID: 10323766. Epub 1999/05/14.eng.
Passauer J, Pistrosch F, Bussemaker E, Lassig G, Herbrig K, Gross P. Reduced agonist-induced endothelium-dependent vasodilation in uremia is attributable to an impairment of vascular nitric oxide. J Am Soc Nephrol. 2005;16(4):959–65. PubMed PMID: 15728785. Epub 2005/02/25.eng.
Zoccali C, Bode-Boger S, Mallamaci F, Benedetto F, Tripepi G, Malatino L, et al. Plasma concentration of asymmetrical dimethylarginine and mortality in patients with end-stage renal disease: a prospective study. Lancet. 2001;358(9299):2113–7. PubMed PMID: 11784625. Epub 2002/01/11.eng.
Ravani P, Tripepi G, Malberti F, Testa S, Mallamaci F, Zoccali C. Asymmetrical dimethylarginine predicts progression to dialysis and death in patients with chronic kidney disease: a competing risks modeling approach. J Am Soc Nephrol. 2005;16(8):2449–55. PubMed PMID: 15944335. Epub 2005/06/10.eng.
Cooke JP. Asymmetrical dimethylarginine: the Uber marker? Circulation. 2004;109(15):1813–8. PubMed PMID: 15096461. Epub 2004/04/21.eng.
Antoniades C, Demosthenous M, Tousoulis D, Antonopoulos AS, Vlachopoulos C, Toutouza M, et al. Role of asymmetrical dimethylarginine in inflammation-induced endothelial dysfunction in human atherosclerosis. Hypertension. 2011;58(1):93–8. PubMed PMID: 21518967. Epub 2011/04/27.eng.
de Mutsert R, Grootendorst DC, Axelsson J, Boeschoten EW, Krediet RT, Dekker FW. Excess mortality due to interaction between protein-energy wasting, inflammation and cardiovascular disease in chronic dialysis patients. Nephrol Dial Transplant. 2008;23(9):2957–64. PubMed PMID: 18400817. Epub 2008/04/11.eng.
Friedman AN, Hunsicker LG, Selhub J, Bostom AG. C-reactive protein as a predictor of total arteriosclerotic outcomes in type 2 diabetic nephropathy. Kidney Int. 2005;68(2):773–8. PubMed PMID: 16014055. Epub 2005/07/15.eng.
Covic A, Kothawala P, Bernal M, Robbins S, Chalian A, Goldsmith D. Systematic review of the evidence underlying the association between mineral metabolism disturbances and risk of all-cause mortality, cardiovascular mortality and cardiovascular events in chronic kidney disease. Nephrol Dial Transplant. 2009;24(5):1506–23. PubMed PMID: 19001560. Epub 2008/11/13.eng.
Tomiyama C, Higa A, Dalboni MA, Cendoroglo M, Draibe SA, Cuppari L, et al. The impact of traditional and non-traditional risk factors on coronary calcification in pre-dialysis patients. Nephrol Dial Transplant. 2006;21(9):2464–71. PubMed PMID: 16735378. Epub 2006/06/01.eng.
Dellegrottaglie S, Saran R, Gillespie B, Zhang X, Chung S, Finkelstein F, et al. Prevalence and predictors of cardiovascular calcium in chronic kidney disease (from the Prospective Longitudinal RRI-CKD Study). Am J Cardiol. 2006;98(5):571–6. PubMed PMID: 16923438. Epub 2006/08/23.eng.
Vaziri ND. Dyslipidemia of chronic renal failure: the nature, mechanisms, and potential consequences. Am J Physiol Renal Physiol. 2006;290(2):F262–72. PubMed PMID: 16403839. Epub 2006/01/13.eng.
Attman PO, Alaupovic P. Lipid abnormalities in chronic renal insufficiency. Kidney Int Suppl. 1991;31:S16–23. PubMed PMID: 2046265.
Vaziri ND, Moradi H. Mechanisms of dyslipidemia of chronic renal failure. Hemodial Int. 2006;10(1):1–7. PubMed PMID: 16441821.
Kaysen GA, Gambertoglio J, Felts J, Hutchison FN. Albumin synthesis, albuminuria and hyperlipemia in nephrotic patients. Kidney Int. 1987;31(6):1368–76. PubMed PMID: 3613408.
Attman PO, Samuelsson O, Johansson AC, Moberly JB, Alaupovic P. Dialysis modalities and dyslipidemia. Kidney Int Suppl. 2003;84:S110–2. PubMed PMID: 12694322. Epub 2003/04/16.eng.
Heimburger O, Stenvinkel P, Berglund L, Tranoeus A, Lindholm B. Increased plasma lipoprotein(a) in continuous ambulatory peritoneal dialysis is related to peritoneal transport of proteins and glucose. Nephron. 1996;72(2):135–44. PubMed PMID: 8684516. Epub 1996/01/01.eng.
Wheeler DC. Abnormalities of lipoprotein metabolism in CAPD patients. Kidney Int Suppl. 1996;56:S41–6. PubMed PMID: 8914053. Epub 1996/11/01.eng.
Johansson AC, Samuelsson O, Attman PO, Haraldsson B, Moberly J, Knight-Gibson C, et al. Dyslipidemia in peritoneal dialysis—relation to dialytic variables. Perit Dial Int. 2000;20(3):306–14. PubMed PMID: 10898048. Epub 2000/07/18.eng.
Shoji T, Nishizawa Y, Kawagishi T, Kawasaki K, Taniwaki H, Tabata T, et al. Intermediate-density lipoprotein as an independent risk factor for aortic atherosclerosis in hemodialysis patients. J Am Soc Nephrol. 1998;9(7):1277–84. PubMed PMID: 9644639. Epub 1998/06/30.eng.
Grieve DJ, Avella MA, Botham KM, Elliott J. Chylomicron remnants potentiate phenylephrine-induced contractions of rat aorta by an endothelium-dependent mechanism. Atherosclerosis. 2000;151(2):471–80. PubMed PMID: 10924724. Epub 2000/08/05.eng.
Whitman SC, Sawyez CG, Miller DB, Wolfe BM, Huff MW. Oxidized type IV hypertriglyceridemic VLDL-remnants cause greater macrophage cholesteryl ester accumulation than oxidized LDL. J Lipid Res. 1998;39(5):1008–20. PubMed PMID: 9610767. Epub 1998/06/04.eng.
Rajman I, Harper L, McPake D, Kendall MJ, Wheeler DC. Low-density lipoprotein subfraction profiles in chronic renal failure. Nephrol Dial Transplant. 1998;13(9):2281–7. PubMed PMID: 9761510. Epub 1998/10/07.eng.
Quaschning T, Krane V, Metzger T, Wanner C. Abnormalities in uremic lipoprotein metabolism and its impact on cardiovascular disease. Am J Kidney Dis. 2001;38(4 Suppl 1):S14–9. PubMed PMID: 11576915. Epub 2001/09/29.eng.
de Graaf J, Hak-Lemmers HL, Hectors MP, Demacker PN, Hendriks JC, Stalenhoef AF. Enhanced susceptibility to in vitro oxidation of the dense low density lipoprotein subfraction in healthy subjects. Arterioscler Thromb. 1991;11(2):298–306. PubMed PMID: 1998647. Epub 1991/03/01.eng.
St-Pierre AC, Cantin B, Dagenais GR, Mauriege P, Bernard PM, Despres JP, et al. Low-density lipoprotein subfractions and the long-term risk of ischemic heart disease in men: 13-year follow-up data from the Quebec Cardiovascular Study. Arterioscler Thromb Vasc Biol. 2005;25(3):553–9. PubMed PMID: 15618542. Epub 2004/12/25.eng.
Austin MA, King MC, Vranizan KM, Krauss RM. Atherogenic lipoprotein phenotype. A proposed genetic marker for coronary heart disease risk. Circulation. 1990;82(2):495–506. PubMed PMID: 2372896. Epub 1990/08/01.eng.
Clementi A, Kim JC, Floris M, Cruz DN, Garzotto F, Zanella M, et al. Statin therapy is associated with decreased small, dense low-density lipoprotein levels in patients undergoing peritoneal dialysis. Contrib Nephrol. 2012;178:111–5. PubMed PMID: 22652726. Epub 2012/06/02.eng.
Ok E, Basnakian AG, Apostolov EO, Barri YM, Shah SV. Carbamylated low-density lipoprotein induces death of endothelial cells: a link to atherosclerosis in patients with kidney disease. Kidney Int. 2005;68(1):173–8. PubMed PMID: 15954906. Epub 2005/06/16.eng.
Kuboyama M, Ageta M, Ishihara T, Fujiura Y, Kashio N, Ikushima I. Serum lipoprotein(a) concentration and Apo(a) isoform under the condition of renal dysfunction. J Atheroscler Thromb. 2003;10(5):283–9. PubMed PMID: 14718745. Epub 2004/01/14.eng.
Cressman MD, Heyka RJ, Paganini EP, O’Neil J, Skibinski CI, Hoff HF. Lipoprotein(a) is an independent risk factor for cardiovascular disease in hemodialysis patients. Circulation. 1992;86(2):475–82. PubMed PMID: 1386292. Epub 1992/08/01.eng.
Koda Y, Nishi S, Suzuki M, Hirasawa Y. Lipoprotein(a) is a predictor for cardiovascular mortality of hemodialysis patients. Kidney Int Suppl. 1999;71:S251–3. PubMed PMID: 10412791. Epub 1999/07/21.eng.
Steyrer E, Durovic S, Frank S, Giessauf W, Burger A, Dieplinger H, et al. The role of lecithin: cholesterol acyltransferase for lipoprotein (a) assembly. Structural integrity of low density lipoproteins is a prerequisite for Lp(a) formation in human plasma. J Clin Invest. 1994;94(6):2330–40.
Erqou S, Kaptoge S, Perry PL, Di Angelantonio E, Thompson A, White IR, et al. Lipoprotein(a) concentration and the risk of coronary heart disease, stroke, and nonvascular mortality. JAMA. 2009;302(4):412–23. PubMed PMID: 19622820. Epub 2009/07/23.eng.
Bennet A, Di Angelantonio E, Erqou S, Eiriksdottir G, Sigurdsson G, Woodward M, et al. Lipoprotein(a) levels and risk of future coronary heart disease: large-scale prospective data. Arch Intern Med. 2008;168(6):598–608. PubMed PMID: 18362252. Epub 2008/03/26.eng.
Genest Jr JJ, Martin-Munley SS, McNamara JR, Ordovas JM, Jenner J, Myers RH, et al. Familial lipoprotein disorders in patients with premature coronary artery disease. Circulation. 1992;85(6):2025–33. PubMed PMID: 1534286. Epub 1992/06/11.eng.
Ohira T, Schreiner PJ, Morrisett JD, Chambless LE, Rosamond WD, Folsom AR. Lipoprotein(a) and incident ischemic stroke: the Atherosclerosis Risk in Communities (ARIC) study. Stroke. 2006;37(6):1407–12. PubMed PMID: 16675734. Epub 2006/05/06.eng.
Loscalzo J, Weinfeld M, Fless GM, Scanu AM. Lipoprotein(a), fibrin binding, and plasminogen activation. Arteriosclerosis. 1990;10(2):240–5. PubMed PMID: 2138452. Epub 1990/03/01.eng.
Zioncheck TF, Powell LM, Rice GC, Eaton DL, Lawn RM. Interaction of recombinant apolipoprotein(a) and lipoprotein(a) with macrophages. J Clin Invest. 1991;87(3):767–71. PubMed PMID: 1825665. Epub 1991/03/01.eng.
Takami S, Yamashita S, Kihara S, Ishigami M, Takemura K, Kume N, et al. Lipoprotein(a) enhances the expression of intercellular adhesion molecule-1 in cultured human umbilical vein endothelial cells. Circulation. 1998;97(8):721–8. PubMed PMID: 9498534. Epub 1998/03/14.eng.
Boerwinkle E, Leffert CC, Lin J, Lackner C, Chiesa G, Hobbs HH. Apolipoprotein(a) gene accounts for greater than 90% of the variation in plasma lipoprotein(a) concentrations. J Clin Invest. 1992;90(1):52–60. PubMed PMID: 1386087. Epub 1992/07/01.eng.
Bowden JF, Pritchard PH, Hill JS, Frohlich JJ. Lp(a) concentration and apo(a) isoform size. Relation to the presence of coronary artery disease in familial hypercholesterolemia. Arterioscler Thromb. 1994;14(10):1561–8. PubMed PMID: 7918305. Epub 1994/10/01.eng.
Kronenberg F, Neyer U, Lhotta K, Trenkwalder E, Auinger M, Pribasnig A, et al. The low molecular weight apo(a) phenotype is an independent predictor for coronary artery disease in hemodialysis patients: a prospective follow-up. J Am Soc Nephrol. 1999;10(5):1027–36. PubMed PMID: 10232689. Epub 1999/05/08.eng.
Longenecker JC, Klag MJ, Marcovina SM, Powe NR, Fink NE, Giaculli F, et al. Small apolipoprotein(a) size predicts mortality in end-stage renal disease: the CHOICE study. Circulation. 2002;106(22):2812–8. PubMed PMID: 12451008. Epub 2002/11/27.eng.
Vaziri ND, Norris K. Lipid disorders and their relevance to outcomes in chronic kidney disease. Blood Purif. 2011;31(1–3):189–96. PubMed PMID: 21228589. Epub 2011/01/14.eng.
Herz J, Strickland DK. LRP: a multifunctional scavenger and signaling receptor. J Clin Invest. 2001;108(6):779–84. PubMed PMID: 11560943. Epub 2001/09/19.eng.
Takahashi S, Kawarabayasi Y, Nakai T, Sakai J, Yamamoto T. Rabbit very low density lipoprotein receptor: a low density lipoprotein receptor-like protein with distinct ligand specificity. Proc Natl Acad Sci U S A. 1992;89(19):9252–6. PubMed PMID: 1384047. Epub 1992/10/01.eng.
Cheung AK, Parker CJ, Ren K, Iverius PH. Increased lipase inhibition in uremia: identification of pre-beta-HDL as a major inhibitor in normal and uremic plasma. Kidney Int. 1996;49(5):1360–71. PubMed PMID: 8731101. Epub 1996/05/01.eng.
Akmal M, Perkins S, Kasim SE, Oh HY, Smogorzewski M, Massry SG. Verapamil prevents chronic renal failure-induced abnormalities in lipid metabolism. Am J Kidney Dis. 1993;22(1):158–63. PubMed PMID: 8322779. Epub 1993/07/01.eng.
Sato T, Liang K, Vaziri ND. Protein restriction and AST-120 improve lipoprotein lipase and VLDL receptor in focal glomerulosclerosis. Kidney Int. 2003;64(5):1780–6. PubMed PMID: 14531811. Epub 2003/10/09.eng.
Lacour B, Roullet JB, Liagre AM, Jorgetti V, Beyne P, Dubost C, et al. Serum lipoprotein disturbances in primary and secondary hyperparathyroidism and effects of parathyroidectomy. Am J Kidney Dis. 1986;8(6):422–9. PubMed PMID: 3812471. Epub 1986/12/01.eng.
Liang K, Oveisi F, Vaziri ND. Role of secondary hyperparathyroidism in the genesis of hypertriglyceridemia and VLDL receptor deficiency in chronic renal failure. Kidney Int. 1998;53(3):626–30. PubMed PMID: 9507207. Epub 1998/03/21.eng.
Kim C, Vaziri ND. Down-regulation of hepatic LDL receptor-related protein (LRP) in chronic renal failure. Kidney Int. 2005;67(3):1028–32. PubMed PMID: 15698441. Epub 2005/02/09.eng.
Vaziri ND, Liang K. Down-regulation of VLDL receptor expression in chronic experimental renal failure. Kidney Int. 1997;51(3):913–9. PubMed PMID: 9067930. Epub 1997/03/01.eng.
Vaziri ND, Kim CH, Dang B, Zhan CD, Liang K. Downregulation of hepatic acyl-CoA:diglycerol acyltransferase in chronic renal failure. Am J Physiol Renal Physiol. 2004;287(1):F90–4. PubMed PMID: 15010358. Epub 2004/03/11.eng.
Vaziri ND, Kim CH, Phan D, Kim S, Liang K. Up-regulation of hepatic Acyl CoA: diacylglycerol acyltransferase-1 (DGAT-1) expression in nephrotic syndrome. Kidney Int. 2004;66(1):262–7. PubMed PMID: 15200432. Epub 2004/06/18.eng.
Chmielewski M, Bryl E, Marzec L, Aleksandrowicz E, Witkowski JM, Rutkowski B. Expression of scavenger receptor CD36 in chronic renal failure patients. Artif Organs. 2005;29(8):608–14. PubMed PMID: 16048476. Epub 2005/07/29.eng.
Park SY, Song CY, Kim BC, Hong HK, Lee HS. Angiotensin II mediates LDL-induced superoxide generation in mesangial cells. Am J Physiol Renal Physiol. 2003;285(5):F909–15. PubMed PMID: 12837686. Epub 2003/07/03.eng.
Rader DJ. Molecular regulation of HDL metabolism and function: implications for novel therapies. J Clin Invest. 2006;116(12):3090–100. PubMed PMID: 17143322. Epub 2006/12/05.eng.
Fielding CJ, Fielding PE. Cellular cholesterol efflux. Biochim Biophys Acta. 2001;1533(3):175–89. PubMed PMID: 11731329. Epub 2001/12/04.eng.
Kaysen GA. Hyperlipidemia in chronic kidney disease. Int J Artif Organs. 2007;30(11):987–92. PubMed PMID: 18067100. Epub 2007/12/11.eng.
Tsimihodimos V, Dounousi E, Siamopoulos KC. Dyslipidemia in chronic kidney disease: an approach to pathogenesis and treatment. Am J Nephrol. 2008;28(6):958–73. PubMed PMID: 18612199. Epub 2008/07/10.eng.
McGrath KC, Li XH, Puranik R, Liong EC, Tan JT, Dy VM, et al. Role of 3beta-hydroxysteroid-delta 24 reductase in mediating antiinflammatory effects of high-density lipoproteins in endothelial cells. Arterioscler Thromb Vasc Biol. 2009;29(6):877–82. PubMed PMID: 19325144. Epub 2009/03/28.eng.
Bisoendial RJ, Hovingh GK, Levels JH, Lerch PG, Andresen I, Hayden MR, et al. Restoration of endothelial function by increasing high-density lipoprotein in subjects with isolated low high-density lipoprotein. Circulation. 2003;107(23):2944–8. PubMed PMID: 12771001. Epub 2003/05/29.eng.
Wang X, Rader DJ. Molecular regulation of macrophage reverse cholesterol transport. Curr Opin Cardiol. 2007;22(4):368–72. PubMed PMID: 17556891. Epub 2007/06/09.eng.
Wang X, Collins HL, Ranalletta M, Fuki IV, Billheimer JT, Rothblat GH, et al. Macrophage ABCA1 and ABCG1, but not SR-BI, promote macrophage reverse cholesterol transport in vivo. J Clin Invest. 2007;117(8):2216–24. PubMed PMID: 17657311. Epub 2007/07/28.eng.
Oram JF, Vaughan AM. ATP-binding cassette cholesterol transporters and cardiovascular disease. Circ Res. 2006;99(10):1031–43. PubMed PMID: 17095732. Epub 2006/11/11.eng.
Zhao Y, Marcel YL. Serum albumin is a significant intermediate in cholesterol transfer between cells and lipoproteins. Biochemistry. 1996;35(22):7174–80. PubMed PMID: 8679545. Epub 1996/06/04.eng.
Acton S, Rigotti A, Landschulz KT, Xu S, Hobbs HH, Krieger M. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science. 1996;271(5248):518–20. PubMed PMID: 8560269. Epub 1996/01/26.eng.
Attman PO, Samuelsson O, Alaupovic P. Lipoprotein metabolism and renal failure. Am J Kidney Dis. 1993;21(6):573–92. PubMed PMID: 8503411. Epub 1993/06/01.eng.
Vaziri ND, Deng G, Liang K. Hepatic HDL receptor, SR-B1 and Apo A-I expression in chronic renal failure. Nephrol Dial Transplant. 1999;14(6):1462–6. PubMed PMID: 10383008. Epub 1999/06/26.eng.
Liang K, Kim CH, Vaziri ND. HMG-CoA reductase inhibition reverses LCAT and LDL receptor deficiencies and improves HDL in rats with chronic renal failure. Am J Physiol Renal Physiol. 2005;288(3):F539–44. PubMed PMID: 15507547. Epub 2004/10/28.eng.
Moradi H, Pahl MV, Elahimehr R, Vaziri ND. Impaired antioxidant activity of high-density lipoprotein in chronic kidney disease. Transl Res. 2009;153(2):77–85. PubMed PMID: 19138652. Epub 2009/01/14.eng.
Shao B, Oda MN, Oram JF, Heinecke JW. Myeloperoxidase: an inflammatory enzyme for generating dysfunctional high density lipoprotein. Curr Opin Cardiol. 2006;21(4):322–8. PubMed PMID: 16755201. Epub 2006/06/07.eng.
Van Lenten BJ, Reddy ST, Navab M, Fogelman AM. Understanding changes in high density lipoproteins during the acute phase response. Arterioscler Thromb Vasc Biol. 2006;26(8):1687–8. PubMed PMID: 16857958. Epub 2006/07/22.eng.
Ansell BJ, Navab M, Hama S, Kamranpour N, Fonarow G, Hough G, et al. Inflammatory/antiinflammatory properties of high-density lipoprotein distinguish patients from control subjects better than high-density lipoprotein cholesterol levels and are favorably affected by simvastatin treatment. Circulation. 2003;108(22):2751–6. PubMed PMID: 14638544. Epub 2003/11/26.eng.
Moradi H, Yuan J, Ni Z, Norris K, Vaziri ND. Reverse cholesterol transport pathway in experimental chronic renal failure. Am J Nephrol. 2009;30(2):147–54. PubMed PMID: 19321994. Epub 2009/03/27.eng.
Vaziri ND, Moradi H, Pahl MV, Fogelman AM, Navab M. In vitro stimulation of HDL anti-inflammatory activity and inhibition of LDL pro-inflammatory activity in the plasma of patients with end-stage renal disease by an apoA-1 mimetic peptide. Kidney Int. 2009;76(4):437–44. PubMed PMID: 19471321. Epub 2009/05/28.eng.
van der Steeg WA, Kuivenhoven JA, Klerkx AH, Boekholdt SM, Hovingh GK, Kastelein JJ. Role of CETP inhibitors in the treatment of dyslipidemia. Curr Opin Lipidol. 2004;15(6):631–6. PubMed PMID: 15529021. Epub 2004/11/06.eng.
Barter PJ, Caulfield M, Eriksson M, Grundy SM, Kastelein JJ, Komajda M, et al. Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med. 2007;357(21):2109–22. PubMed PMID: 17984165. Epub 2007/11/07.eng.
Bots ML, Visseren FL, Evans GW, Riley WA, Revkin JH, Tegeler CH, et al. Torcetrapib and carotid intima-media thickness in mixed dyslipidaemia (RADIANCE 2 study): a randomised, double-blind trial. Lancet. 2007;370(9582):153–60. PubMed PMID: 17630038. Epub 2007/07/17.eng.
Vaziri ND, Navab M, Fogelman AM. HDL metabolism and activity in chronic kidney disease. Nat Rev Nephrol. 2010;6(5):287–96. PubMed PMID: 20308998. Epub 2010/03/24.eng.
Seiler S, Schlitt A, Jiang XC, Ulrich C, Blankenberg S, Lackner KJ, et al. Cholesteryl ester transfer protein activity and cardiovascular events in patients with chronic kidney disease stage V. Nephrol Dial Transplant. 2008;23(11):3599–604. PubMed PMID: 18503096. Epub 2008/05/27.eng.
Navab M, Reddy ST, Van Lenten BJ, Anantharamaiah GM, Fogelman AM. The role of dysfunctional HDL in atherosclerosis. J Lipid Res. 2009;50(Suppl):S145–9. PubMed PMID: 18955731. Epub 2008/10/29.eng.
Libby P. Inflammation in atherosclerosis. Nature. 2002;420(6917):868–74. PubMed PMID: 12490960. Epub 2002/12/20.eng.
Falk E. Pathogenesis of atherosclerosis. J Am Coll Cardiol. 2006;47(8 Suppl):C7–12. PubMed PMID: 16631513. Epub 2006/04/25.eng.
Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352(16):1685–95. PubMed PMID: 15843671. Epub 2005/04/22.eng.
Davies MJ. The pathophysiology of acute coronary syndromes. Heart. 2000;83(3):361–6. PubMed PMID: 10677422. Epub 2000/03/04.eng.
Schwarz U, Buzello M, Ritz E, Stein G, Raabe G, Wiest G, et al. Morphology of coronary atherosclerotic lesions in patients with end-stage renal failure. Nephrol Dial Transplant. 2000;15(2):218–23. PubMed PMID: 10648668. Epub 2000/01/29.eng.
London GM, Parfrey PS. Cardiac disease in chronic uremia: pathogenesis. Adv Ren Replace Ther. 1997;4(3):194–211. PubMed PMID: 9239425. Epub 1997/07/01.eng.
Qunibi WY. Reducing the burden of cardiovascular calcification in patients with chronic kidney disease. J Am Soc Nephrol. 2005;16 Suppl 2:S95–102. PubMed PMID: 16251249. Epub 2005/10/28.eng.
Russo D, Palmiero G, De Blasio AP, Balletta MM, Andreucci VE. Coronary artery calcification in patients with CRF not undergoing dialysis. Am J Kidney Dis. 2004;44(6):1024–30. PubMed PMID: 15558523. Epub 2004/11/24.eng.
Budisavljevic MN, Cheek D, Ploth DW. Calciphylaxis in chronic renal failure. J Am Soc Nephrol. 1996;7(7):978–82. PubMed PMID: 8829111. Epub 1996/07/01.eng.
Ketteler M, Schlieper G, Floege J. Calcification and cardiovascular health: new insights into an old phenomenon. Hypertension. 2006;47(6):1027–34. PubMed PMID: 16618842. Epub 2006/04/19.eng.
Covic A, Kanbay M, Voroneanu L, Turgut F, Serban DN, Serban IL, et al. Vascular calcification in chronic kidney disease. Clin Sci (Lond). 2010;119(3):111–21. PubMed PMID: 20443781. Epub 2010/05/07.eng.
Parfrey PS, Foley RN. The clinical epidemiology of cardiac disease in chronic renal failure. J Am Soc Nephrol. 1999;10(7):1606–15. PubMed PMID: 10405218. Epub 1999/07/15.eng.
United States Rena Data System 2007. Mortality and cause of death. www.usrds.org/2007/ref/H_morte_07.pdf.
Heymann EP, Kassimatis TI, Goldsmith DJ. Dyslipidemia, statins, and CKD patients’ outcomes—review of the evidence in the post-sharp era. J Nephrol. 2012;25(4):460–72. PubMed PMID: 22641572. Epub 2012/05/30.eng.
Kassimatis TI, Konstantinopoulos PA. Rosuvastatin in patients undergoing hemodialysis. N Engl J Med. 2009;361(1):93; author reply 4–5. PubMed PMID: 19579277. Epub 2009/07/07.eng.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Kassimatis, T., Goldsmith, D. (2014). CVD in CKD: Focus on the Dyslipidemia Problem. In: Covic, A., Kanbay, M., Lerma, E. (eds) Dyslipidemias in Kidney Disease. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-0515-7_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0515-7_5
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-0514-0
Online ISBN: 978-1-4939-0515-7
eBook Packages: MedicineMedicine (R0)