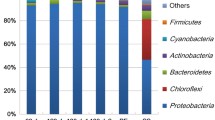
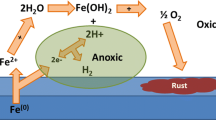
Abstract
Advances in high-throughput next-generation sequencing (NGS) technology for direct sequencing of environmental DNA (i.e., shotgun metagenomics) are transforming the field of microbiology. NGS technologies are now regularly being applied in comparative metagenomic studies, which provide the data for functional annotations, taxonomic comparisons, community profile, and metabolic reconstructions. For example, comparative metagenomic analysis of corroded pipes unveiled novel insights on the bacterial populations associated with the sulfur and nitrogen cycle, which may be directly or indirectly implicated in concrete wastewater pipe corrosion. The objective of this chapter is to describe the steps involved in the taxonomic and functional analysis of metagenome datasets from biofilm involved in microbial-induced concrete corrosion (MICC).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
USEPA (United States Environmental Protection Agency) (2009) State of technology review report on rehabilitation of wastewater collection and water distribution systems. Office of Research and Development, Cincinnati, OH, EPA/600/R-09/048
USEPA (United States Environmental Protection Agency) (2002) State of technology review report on rehabilitation of wastewater collection and water distribution systems. USEPA Urban Watershed Management Branch, Edison, NJ, EPA/600/JA-02/226
Mori T, Nonaka T, Tazaki K et al (1992) Interactions of nutrients, moisture, and pH on microbial corrosion of concrete sewer pipes. Water Res 26:29–37
Vollertsen J, Nielsen AH, Jensen HS et al (2008) Corrosion of concrete sewers—the kinetics of hydrogen sulfide oxidation. Sci Total Environ 394:162–170
Zhang L, De Schryver P, De Gusseme B et al (2008) Chemical and biological technologies for hydrogen sulfide emission control in sewer systems: a review. Water Res 42:1–12
Vincke E, Boon N, Verstraete W (2001) Analysis of the microbial communities on corroded concrete sewer pipes—a case study. Appl Microbiol Biotechnol 57:776–785
Okabe S, Ito T, Satoh H (2003) Sulfate-reducing bacterial community structure and their contribution to carbon mineralization in a wastewater biofilm growing under microaerophilic conditions. Appl Microbiol Biotechnol 63:322–334
Okabe S, Odagiri M, Ito T, Satoh H (2007) Succession of sulfur-oxidizing bacteria in the microbial community on corroding concrete in sewer systems. Appl Environ Microbiol 73:971–980
Satoh H, Odagiri M, Ito T, Okabe S (2009) Microbial community structures and in situ sulfate-reducing and sulfur-oxidizing activities in biofilms developed on mortar specimens in a corroded sewer system. Water Res 43:4729–4739
Santo Domingo JW, Revetta RP, Iker B et al (2011) Molecular survey of concrete sewer biofilm microbial communities. Biofouling 27:993–1001
Jones WJ (2010) High-throughput sequencing and metagenomics. Estuaries Coasts 33:944–952
Simon C, Daniel R (2009) Achievements and new knowledge unraveled by metagenomic approaches. Appl Microbiol Biotechnol 85:265–276
Gilbert JA, Meyer F, Bailey MJ (2011) The future of microbial metagenomics (or is ignorance bliss?). ISME J 5:777–779
Thomas T, Gilbert J, Meyer F (2012) Metagenomics—a guide from sampling to data analysis. Microb Inform Exp 2:3
Shokralla S, Spall JL, Gibson JF, Hajibabaei M (2012) Next-generation sequencing technologies for environmental DNA research. Mol Ecol 21:1794–1805
Meyer F, Paarmann D, D'Souza M et al (2008) The metagenomics RAST server—a public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinformatics 9:386
Sun S, Chen J, Li W et al (2011) Community cyberinfrastructure for advanced microbial ecology research and analysis: the CAMERA resource. Nucleic Acids Res 39:D546–D551
Markowitz VM, Chen I-MA, Chu K et al (2012) IMG/M: the integrated metagenome data management and comparative analysis system. Nucleic Acids Res 40:D123–D129
Gerlach W, Jünemann S, Tille F et al (2009) WebCARMA: a web application for the functional and taxonomic classification of unassembled metagenomic reads. BMC Bioinformatics 10:430
Huson DH, Mitra S, Ruscheweyh H-J et al (2011) Integrative analysis of environmental sequences using MEGAN4. Genome Res 21:1552–1560
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Parks DH, Beiko RG (2010) Identifying biologically relevant differences between metagenomic communities. Bioinformatics 26:715–721
Prakash T, Taylor TD (2012) Functional assignment of metagenomic data: challenges and applications. Brief Bioinform 13:711–727
Gomez-Alvarez V, Revetta RP, Santo Domingo JW (2012) Metagenome analyses of corroded concrete wastewater pipe biofilms reveal a complex microbial system. BMC Microbiol 12:122
Prosser JI (2010) Replicate or lie. Environ Microbiol 12:1806–1810
Lennon JT (2011) Replication, lies and lesser-known truths regarding experimental design in environmental microbiology. Environ Microbiol 13:1383–1386
Gomez-Alvarez V, Teal TK, Schmidt TM (2009) Systematic artifacts in metagenomes from complex microbial communities. ISME J 3:1314–1317
Zhou J, Bruns MA, Tiedje JM (1996) DNA recovery from soils of diverse composition. Appl Environ Microbiol 62:316–322
De Filippo C, Ramazzotti M, Fontana P, Cavalieri D (2012) Bioinformatic approaches for functional annotation and pathway inference in metagenomics data. Brief Bioinform 13:696–710
Li W (2009) Analysis and comparison of very large metagenomes with fast clustering and functional annotation. BMC Bioinformatics 10:359–367
Langmead B, Trapnell C, Pop M, Salzberg SL (2009) Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10:R25
Cox MP, Peterson DA, Biggs PJ (2010) SolexaQA: at-a-glance quality assessment of Illumina second-generation sequencing data. BMC Bioinformatics 11:485
Huse SM, Huber JA, Morrison HG et al (2007) Accuracy and quality of massively parallel DNA pyrosequencing. Genome Biol 8:R143
Keegan KP, Trimble WL, Wilkening J et al (2012) A platform-independent method for detecting errors in metagenomic sequencing data: DRISEE. PLoS Comput Biol 8:e1002541
Gomez-Alvarez V, Revetta RP, Santo Domingo JW (2012) Metagenomic analyses of drinking water receiving different disinfection treatments. Appl Environ Microbiol 78:6095–6102
Bengtsson J, Hartmann M, Unterseher M et al (2012) Megraft: a software package to graft ribosomal small subunit (16S/18S) fragments onto full-length sequences for accurate species richness and sequencing depth analysis in pyrosequencing-length metagenomes and similar environmental datasets. Res Microbiol 163:407–412
Bengtsson J, Eriksson KM, Hartmann M et al (2011) Metaxa: a software tool for automated detection and discrimination among ribosomal small subunit (12S/16S/18S) sequences of archaea, bacteria, eukaryotes, mitochondria, and chloroplasts in metagenomes and environmental sequencing datasets. Antonie Van Leeuwenhoek 100:471–475
Kanehisa M, Goto S (2000) KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30
Beszteri B, Temperton B, Frickenhaus S, Giovannoni SJ (2010) Average genome size: a potential source of bias in comparative metagenomics. ISME J 4:1075–1077
Raes J, Korbel JO, Lercher MJ et al (2007) Prediction of effective genome size in metagenomic samples. Genome Biol 8:R10
Frank JA, Sørensen SJ (2011) Quantitative metagenomic analyses based on average genome size normalization. Appl Environ Microbiol 77:2513–2521
Mitra S, Rupek P, Richter DC et al (2011) Functional analysis of metagenomes and metatranscriptomes using SEED and KEGG. BMC Bioinformatics 12:S21
Gill SR, Pop M, Deboy RT et al (2006) Metagenomic analysis of the human distal gut microbiome. Science 312:1355–1359
Michie MG (1982) Use of the Bray-Curtis similarity measure in cluster analysis of foraminiferal data. Math Geol 14:661–667
Wickelmaier F (2003) An introduction to MDS: reports from the Sound Quality Research Unit (SQRU) No. 7, 1–26. http://homepages.uni-tuebingen.de/florian.wickelmaier/pubs/Wickelmaier2003SQRU.pdf
McKenna JE (2003) An enhanced cluster analysis program with bootstrap significance testing for ecological community analysis. Environ Model Software 18:205–220
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14
Smoot ME, Ono K, Ruscheinski J et al (2011) Cytoscape 2.8: new features for data integration and network visualization. Bioinformatics 27:431–432
Acknowledgements
The United States Environmental Protection Agency (USEPA) through the Office of Research and Development funded this research. R.P. Revetta and J.W. Santo Domingo of the USEPA participated in design and coordination of the study. It has been subjected to the Agency’s peer and administrative review and has been approved for external publication. Any opinions expressed in this manuscript are of the authors and do not necessarily reflect the official positions and policies of USEPA. Any mention of trade names or commercial products does not constitute endorsement or recommendation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Gomez-Alvarez, V. (2014). Biofilm-Growing Bacteria Involved in the Corrosion of Concrete Wastewater Pipes: Protocols for Comparative Metagenomic Analyses. In: Donelli, G. (eds) Microbial Biofilms. Methods in Molecular Biology, vol 1147. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-0467-9_23
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0467-9_23
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-0466-2
Online ISBN: 978-1-4939-0467-9
eBook Packages: Springer Protocols