Abstract
Dendritic cells (DCs) play a crucial role in initiating immune responses against both foreign pathogens as well as tumors. DCs also control the type, potency, and extent of T-cell responses, contribute to natural killer (NK) and natural killer T-cell (NKT cell) antitumoral activity, as well as to B-cell-mediated immunity. However, antitumor immune responses are often deficient or suboptimal since tumor cells are able to exploit the functional roles of DCs for tumor progression. Suppression, dysfunction, and repolarization of DC function in cancer patients all contribute to the failure of antitumor immune responses and consequent disease progression. Subversion of tumor immunity by altering the tumor immunoenvironment and DC subset distribution and function is mediated by various malignant cell-derived and tumor stroma-derived factors, many of which remain to be identified. Molecular mechanisms of tumor-mediated dysfunction and repolarization of the DC system are under investigation, and several signaling pathways responsible for DC malfunction in cancer have been already described. Here, we summarize findings in the field of DC biology in cancer and discuss the importance of these data for designing novel DC-based vaccination strategies, as well as their applicability for combinatorial therapeutic approaches.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Dendritic cells
- Dendropoiesis
- Immunosuppression
- Regulatory dendritic cells
- Immunomodulation
- Chemoimmunomodulation
1 Introduction
Tumor development and progression are associated with suppression and malfunction of the immune system, of which dendritic cells (DCs) possess many key regulatory functions, especially those related to cytokine production, antigen presentation to naive T cells, and polarization and balancing T-helper subsets. DCs are professional antigen-presenting cells, strategically positioned for bridging innate and adaptive immunity. DCs can initiate T-cell responses against tumors due to their capacity to process and present tumor antigens and stimulate naive T cells. However, less is known about DC differentiation, behavior, and polarization in vivo in tumor-bearing hosts. Although neglected for many years, the importance of the tumor microenvironment in regulating immunology of DC is becoming more defined, as the dual role of DC in cancer was shown to play an important role in cancer progression [1].
Although alterations in DCs in the setting of cancer were described almost two decades ago, characterization of tumor-derived factors responsible for DC dysfunction and the molecular mechanisms of abnormal DC differentiation and activation are still not well understood. An understanding of how the tumor environment regulates the DC system and how it impacts the efficacy of DC vaccines and other immunotherapeutic approaches is far from complete and clinical trials focusing on the protection of DC from the detrimental effects of the tumor microenvironment are constantly being tested. In addition to the tumor/stromal cells and their interactions, the other factors impacting vaccine efficacy in cancer include (a) the psychological stress of both a potentially fatal disease as well as the stress associated with the treatment of the disease (Fig. 5.1) and (b) aging immune system, since more than 60 % of cancer arises in people older than 65 years of age. Thus, in patients with cancer, the DC system functions under the multidirectorial influences of various local and systemic tumor-derived and tumor stroma-derived factors, acute and chronic stress hormones, therapeutic agents and factors, as well as multifaceted conditions associated with aging, infections, autoimmune diseases, and other acute and chronic disorders (Fig. 5.1).
Intrinsic and extrinsic mechanisms of dendritic cell regulation in cancer patients. DC generation, differentiation, polarization, and function are under constant, dynamic, and variable influences and regulatory pathways operating in patients with cancer. This includes numerous tumor-derived factors that affect all stages of DC development and may be represented by cytokines, chemokines, growth factors, prostaglandins, gangliosides, neuropeptides, and many other soluble and membrane-bound molecules on different cell types in the tumor microenvironment. Additional modulation of DC function in cancer may result from psychological stressors associated both with the diagnosis, as well as the effects of the treatment of a potentially fatal illness. Finally, DCs produce different factors, which may modulate cells in an autocrine and paracrine manner, and may also change DC responses to other molecules in the local environment. Thus, an understanding of the complex environmental conditions associated with DC function in cancer is necessary for harnessing the antitumor potential of these unique immunostimulatory and immunoregulatory cells
Modulation of DC generation and function by some of the above-mentioned factors or conditions has been partly described. However, a comprehensive and systematic analysis of the DC system in the tumor environment has not been reported. For instance, both tumor-derived factors (reviewed in [2], [3]) as well as nonmalignant cells in the tumor milieu (reviewed in [4]) have been reported to suppress DC maturation, function, and longevity. Psychological and physical stressors may affect the functional activity of DCs through a variety of hormones, neuromediators, and neuropeptides [5]–[7]. Indeed, modulation of DC maturation and function by glucocorticoids, neuropeptides, and biogenic amines has been described. Glucocorticoid-treated DCs show higher endocytic activity, lower antigen-presenting function, and a lower capacity to secrete cytokines [8]. Norepinephrine can impede interleukin-12 (IL-12) and stimulate IL-10 production in DCs, thus inhibiting their antigen-presenting capability and hampering their motility and chemotaxis [9], [10]. DCs also express receptors for and respond to calcitonin gene-related peptide, neuropeptide Y, opioid peptides, prolactin, bombesin-like peptides, substance P, and other neuropeptides, all of which may be involved in stress-related modulation of immunity [11]–[16].
As shown in Fig. 5.1, surgery, radiation, chemotherapeutic agents, and hormonal therapy might alter DC function and survival [17]–[20]. For example, many chemotherapeutic agents are known to suppress DC activity in therapeutic doses, but they may indirectly or directly upregulate DC maturation and function when used in low- or ultralow doses [21]–[25]. Interestingly, certain factors in the common environment, e.g., nanoparticles, may directly affect DC function in the lung or alter homing and function of other immune cells leading to dysfunction of antitumor immunity and tumor progression. For instance, it has been recently reported that in vivo exposure to single-walled carbon nanotubes (SWCNTs) modifies systemic immunity by modulating DC function [26]. Furthermore, nanomaterials internalized by DCs differently affect their abilities to present antigens to T cells: While C(60)-fullerenes stimulated the antigen-specific major histocompatibility complex (MHC) class I-restricted T-cell response, graphene oxide (GO) impaired the stimulatory potential of DCs [27]. In contrast to C(60)-fullerenes, GO decreased the intracellular levels of low molecular mass polypeptide 7 (LMP7) immunoproteasome subunits required for processing of protein antigens. Interestingly, recent studies show that metastatic establishment and growth of lung carcinoma could be promoted by exposure to SWCNTs [28].
Furthermore, age-related alterations of DC maturity, function, longevity, and subpopulation composition also play a significant role in the ability of the DC system to interact with tumor cells and T cells and induce and maintain an antitumor immune response in patients with cancer (reviewed in [29]). For instance, increased levels of IL-6 and IL-10 repeatedly reported in old individuals might have a direct effect on dendropoiesis (i.e., DC generation) and maturation of DCs and, thus, on their motility and ability to process and present tumor antigens. Finally, exposure to different stimuli induces DCs to produce various endogenous mediators, including arachidonic acid-derived eicosanoids, cytokines, regulatory peptides, and small molecules like nitric oxide (NO). Many secreted products of DCs can act in an autocrine manner and modulate cell function; for instance, autocrine IL-10 can prevent maturation of DCs [30]. Interestingly, aging has been associated with immunological changes (immunosenescence) that mimic changes observed in the setting of chronic stress as well as changes seen with cancer [31], [32]. Thus, there may be common mechanisms of immune alterations in the DC system in cancer, aging, and chronic stress and numerous factors and agents can be involved in abnormal function of DCs in patients with cancer. These and many other issues related to differentiation, function, and clinical application of DCs in cancer have been discussed in the book Dendritic Cells in Cancer [33].
2 Alterations of DCs in Cancer
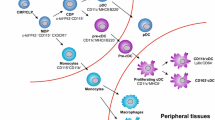
Regardless of the pathways and mechanisms responsible for tumor-associated changes of DCs, functionally three basic subtypes of DC can be seen in tumor-bearing hosts (Fig. 5.2): normal “unaltered” conventional DCs (cDCs) that can initiate and maintain immune responses, including antitumor responses; functionally deficient DCs with suppressed or blocked motility, antigen uptake/processing/presentation, or cytokine production or expression of costimulatory molecules; and regulatory or tolerogenic DCs that inhibit T-cell-mediated immune responses by different means. Because this classification is based strictly on DC function, there are no specific phenotypic markers to distinguish all functional subsets of DC seen in the tumor immunoenvironment [34]. In addition, DC function might be dynamically altered by the local microenvironment and surrounding cells. Functional plasticity of DC is a well-known phenomenon and different functional subsets of DC were repeatedly described in patients with cancer.
Functional subsets of dendritic cells in cancer patients. The presence of numerous cells and factors affecting differentiation of DC precursors and activity of immature and mature DCs in the tumor environment results in formation of three basic functional subsets of DCs: normal “unaltered” immunostimulatory DCs, non-functional or functionally deficient DCs, and protumorigenic regulatory DCs. These DCs might belong to different or similar DC subpopulations (e.g., conventional or plasmacytoid), be on similar or different stages of maturation (e.g., immature, semi-mature, or mature), and express identical or different phenotypic markers (e.g., high or low MHC, CD80, CD86, CD40), but they act as inducers or suppressors of antitumor immune responses depending on the local and systemic environment
From the mechanistic point of view, most of the pathways that are responsible for altered functionality of DC in cancer can be also grouped in four categories: (1) elimination of functional DCs by blocking their production/differentiation/maturation or inducing apoptosis in DC or DC precursors; (2) inhibition of critical function of DCs; (3) polarization of DC subpopulations toward immunosuppressive or tolerogenic DC subsets; and (4) avoidance of the tumor contact with DCs by downregulating the expression of DC-attracting chemokines.
In 1988, Stene et al. revealed that melanoma-associated skin DCs (Langerhans cells) declined in number as melanoma progressed [35]. In 1989, Alcalay et al. described a decreased number and altered morphology of Langerhans cells in squamous cell carcinomas of the skin [36] and showed later (1991) that the antigen-presenting capacity of lymph node cells might be impaired during tumorigenesis [37]. Halliday et al. in 1991 demonstrated that tumor may regulate DC attraction and homing at the tumor site and suggested that yet-unknown factors may inhibit DC function and thus induction of antitumor immunity [38], [39]. In 1992, Becker speculated that outcome of a primary tumor in patients depends on the ability of DCs to enter into tumors and that tumors might be different in their capacity to destroy or prevent DCs from entering the tumor site [40]. He also hypothesized that DCs and tumor cells interacted by releasing cytokines, which abrogate tumor cells or DCs, respectively [41]. In 1993, Tas et al. showed that DCs are functionally abnormal in patients with cancer [42]. Colasante et al. in 1995 studied the role of cytokines in the distribution and differentiation of DC lineage in primary lung carcinomas in humans and concluded on the potential role for granulocyte-macrophage colony-stimulating factor (GM-CSF), tumor necrosis factor-α (TNF-α), IL-1α, and IL-1β in DC modulation [43]. In 1996, Gabrilovich et al. reported that DCs isolated from tumor-bearing mice showed a significantly reduced ability to induce syngeneic tumor-specific cytotoxic T lymphocyte (CTL) and stimulate allogeneic T cells [44] and Chaux et al. revealed that tumor-associated DCs express low levels of costimulatory molecules [45]. Enk et al. in 1997 showed that melanoma-derived factors converted DC antigen-presenting function to tolerance induction against tumor tissue [46] .
Following these initial findings, other teams demonstrated functional suppression in preparation of human CD34-derived and CD14-derived DCs, as well as murine bone marrow-derived DCs by both identified and unidentified tumor-derived factors (Table 5.1). For example, Ninomiya et al. (1999) reported that DCs propagated from patients with hepatocellular carcinoma expressed significantly lower levels of human leukocyte antigen-DR (HLA-DR), had significantly lower capacity to stimulate allogeneic T cells, and produced decreased amounts of IL-12 [47]. In vivo, Lissoni et al. (1999) revealed that the number of circulating DCs in the peripheral blood of cancer patients was also significantly decreased [48], and these results were confirmed by others, e.g., in patients with squamous cell carcinoma of the head and neck (HNSCC) [49], leukemia [50], hepatocellular carcinoma [51], lung cancer [52], and invasive breast cancer [53]. Metastasis development decreased the number of circulating DCs even further [18]. Furthermore, blood monocytes isolated from both patients with glioblastoma and intracranial metastases had significantly reduced expression of granulocyte macrophage colony-stimulating factor receptor (GM-CSFR) and showed a reduced capacity to differentiate into mature DCs [54]. Similar data were reported for other cancers [55]–[58]. Thus, local (at the tumor site) and systemic levels of DC might be markedly lower in cancer patients due to the inhibited or abnormal dendropoiesis [59], i.e., DC generation and differentiation.
Elimination of functional DCs in cancer may be also associated with the killing of DCs or acceleration of their turnover. Induction of apoptosis in DCs by tumor-derived factors was first reported by Esche et al. in 1999 [60] and confirmed by others [61]–[63]. Furthermore, the results were confirmed by documenting the presence of a significantly higher proportion of apoptotic blood DCs in patients with early-stage breast cancer compared to healthy volunteers [64]. Similarly, tumor-mediated cell death of DC precursors [65] and accelerated early apoptosis of DCs [58], [61], [66] were also reported .
The second type of DC abnormalities in cancer includes the functional deficiency of DCs when compared to cells derived from healthy age-matched controls (Table 5.1). Decreased ability of DCs obtained from cancer patients’ blood or lymph nodes, or DCs cocultured with malignant cells to stimulate allogeneic T cells, uptake, process, and present antigen(s), provide costimulatory signals, migrate toward specific chemokines, and produce IL-12 were repeatedly described for prostate, breast, renal, liver, lung cancer, HNSCC, melanoma, myeloma, leukemia, glioma, neuroblastoma, and other tumor types [65]–[74]. These and other results were also reviewed in [2], [3], [75]–[79] and therefore are not detailed here.
Polarization of DC subtypes represents the third type of the DC aberration in cancer (Table 5.1). For instance, there are substantial numbers of tumor-promoting functional plasmacytoid DCs (pDCs or lymphoid DCs by some classifications, but not cDCs or myeloid DCs) accumulated in tumor ascites in patients with ovarian carcinomas [80]. Similarly, estimating conventional and plasmacytoid subpopulations of DCs in the peritoneal fluid of women with ovarian tumors, Wertel et al. reported that the percentage of pDCs was higher in patients with ovarian cancer than in women with serous cystadenoma [81]. They also reported that the percentage of the peritoneal fluid myeloid DCs was significantly lower in patients with ovarian cancer in comparison to the group of nonmalignant ovarian tumors, while the percentage of the peritoneal fluid lymphoid DCs was higher in patients with ovarian cancer than in the reference group [82]. The presence of pDCs within primary breast tumors correlated with an unfavorable prognosis for patients [83]. Using fresh human breast tumor biopsies, the authors observed increased tumor-associated pDC rates in aggressive breast tumors and showed that these pDCs produced very low amounts of IFN-α. Interestingly, within breast tumors, pDCs colocalized with regulatory T cells (Treg cells); the selective suppression of IFN-α production endowed pDCs with the unique capacity to sustain Foxp3+ Treg expansion [83]. The same team has recently identified transforming growth factor- β (TGF-β) and TNF-α as major soluble factors involved in pDC functional alteration in cancer [84]. These findings indicate that IFN-α-deficient tumor-associated pDCs accumulating in aggressive tumors are involved in the expansion of tumor-associated Treg cells in vivo, contributing to tumor immune tolerance and poor clinical outcome .
The levels of myeloid or cDC subsets in circulation may also be significantly lower, while the number of lymphoid or pDC subsets might vary, as was repetitively reported for patients with different tumor types compared to healthy donors [52], [85], [86]. Interestingly, these alterations were reverted by surgical resection of the tumor or by chemoradiotherapy [53], [85], [87], [88] suggesting that tumor-derived factors are responsible for redirecting DC differentiation (dendropoiesis) in the bone marrow, i.e., systemically. Indeed, microvesicles isolated from plasma of advanced melanoma patients, but not from healthy donors, mediated the effect of tumor on CD14+ monocytes and skewed their differentiation from DCs toward CD14+HLA-DRlow cells with TGF-β-mediated suppressive activity on T-cell functions [89]. A subset of these TGF-β-secreting CD14+HLA-DRlow cells was found to be significantly expanded in the peripheral blood of melanoma patients compared with healthy donors.
Tumor-promoted redirection of dendropoiesis and its repolarization are also associated with increased numbers of immature DCs and the appearance of other related immature cells of myeloid progeny. For example, in addition to having fewer levels of DCs in the peripheral blood, patients with breast and prostate cancer as well as patients with malignant glioma showed significant accumulation of abnormal population of HLA-DR+ immature cells (DR+ICs), which in spite of HLA-DR, CD40, and CD86 expression had reduced capacity to capture antigens and elicited poor proliferation and IFN-γ secretion by T lymphocytes [86]. Immature DCs fail to provide an appropriate costimulatory signal to T cells and might induce tolerance through abortive proliferation or anergy of antigen-specific CD4+ and CD8+ T cells or through the generation of Treg cells that suppress immune responses by producing IL-10 and TGF-β [90]. Immature DCs were found at high levels within tumor-infiltrating leukocytes and increased circulating levels of immature DCs have also been observed in the peripheral blood of patients with lung, breast, head and neck, and esophageal cancer [91]. Immature myeloid precursors of DCs may also suppress T-cell activation as part of a population of myeloid-derived suppressor cells (MDSCs), a heterogeneous population of myeloid cells that comprises immature macrophages, granulocytes, DCs, and myeloid cells at early stages of differentiation, discussed in detail in other chapters of this monograph .
Finally, the last mechanism of decreased number of active DCs associated with the tumor progression is the loss of expression of DC-attracting chemokines at the tumor site (Table 5.1). For instance, it has been demonstrated that HNSCC cells do not express CXCL14 protein and messenger RNA (mRNA), a potent DC-attracting chemokine [92]. This resulted in low chemoattraction of DCs to the tumor bed, low numbers of tumor-associated DCs, and deficient induction of antitumor immunity; however, transduction of CXCL14-negative tumor cells with the CXCL14 gene was associated with increased DC infiltration, an antitumor immune response, and inhibition of tumor growth in vivo. Investigation of the mechanisms of loss of CXCL14 in prostate cancer cells revealed direct evidence for epigenetic regulation of chemokine expression in tumor cells [93]. Interestingly, melanoma cells might utilize an opposite approach and can effectively chemoattract DCs, modulate their phenotype, and, eventually, severely damage DC mobility: Melanoma-conditioned DCs exhibited an increased adhesion capacity to a melanoma cell line in vitro and did not migrate in response to DC chemokines [94]. The explanation for abnormal DC retention inside some human malignant lesions may come from another study where it was found that tumors from patients with hepatocellular carcinoma, colorectal cancer, or pancreatic cancer were producing IL-8 and that this chemokine attracted DCs that uniformly express both IL-8 receptors, CXCR1 and CXCR2 [95].
In summary, abnormal dendropoiesis , DC longevity and function, and DC migration toward or from the tumor site are the key characteristics of the local and systemic DC dysfunction in tumor-bearing hosts that have a crucial role in immune nonresponsiveness to tumors and tumor escape [34] .
3 Mechanisms of Dendritic Cell Dysfunction in Cancer
3.1 Factors
Tumors exploit several strategies to evade immune recognition, including the production of a variety of immunosuppressive/immunomodulating factors , which might specifically block or redirect DC maturation, suppress DC survival, and impair function of DC in the vicinity of tumors [4] (Table 5.2). Historically, the first tumor-derived factor inhibiting DC differentiation in cancer was identified as vascular endothelial growth factor (VEGF) [96]. For instance, in patients affected by colorectal cancer, DC numbers inversely correlated with VEGF serum levels, suggesting a possible effect of this cytokine on the DC compartment. In cultures, the exposure of monocyte-derived DCs to VEGF produced a dramatic alteration of DC differentiation by induction of apoptosis, alteration of DC phenotypic profile, and increased CXCR4 expression [97]. VEGF blocks the functional maturation of DCs from hematopoietic progenitor cells by blocking nuclear factor kappa-B (NF-κB) transcription. The family of VEGF molecules also plays a key role in recruiting immature myeloid cells and immature DCS from the bone marrow to enrich the tumor microenvironment [98].
Tumor-derived TGF-β and IL-10 were shown to be responsible for downregulating CD80 expression on blood DCS in myeloma patients [68]. DC maturation, antigen presentation, and IL-12 production induced by inflammatory cytokines IL-1 and TNF-α or by lipopolysaccharide (LPS) might be inhibited by TGF-β [99]. TGF-β might also induce apoptosis in DCs [100]. Increased levels of IL-10 in serum from patients with hepatocellular carcinoma and tumor progression were shown to correlate with profound numerical deficiencies and immature phenotype of circulating DC subsets [101]. Murine bone marrow-derived DCs that were propagated in IL-10 and TGF-β (so-called alternatively activated DC) expressed low levels of Toll-like receptor 4 (TLR4), MHC class II, CD40, CD80, CD86, IL-12p70, and programmed death-ligand 2 (B7-DC; CD273) and were resistant to maturation [102]. They secreted much higher levels of IL-10 and efficiently expanded functional CD4+CD25+Foxp3+ Treg cells. We have shown earlier that murine colon adenocarcinoma cells produce IL-10 and that IL-10 causes downregulation of CD40 expression on DCs and is responsible for inhibited CD40-dependent IL-12 production by DCs [103]. These and other studies also revealed the tumor-associated in vivo effects of IL-10 on DC function in eliciting a type 1 immune response in both allogeneic and tumor-specific responses [104]. Furthermore, analyzing pancreatic cancer-derived cytokines responsible for inhibition of DC differentiation, Bellone et al. (2006) reported that IL-10, TGF-β, and IL-6, but not VEGF, cooperatively affect DC precursors in a manner consistent with ineffective antitumor immune responses [105]. However, lung squamous cell carcinoma and adenocarcinoma have been shown to use different mediators to induce comparable phenotypic and functional changes in DCs: IL-6 versus IL-10+IL-6+ prostanoids, respectively [106]. Renal cell carcinoma (RCC)-derived IL-6 and VEGF were shown to block the ability of tumor antigen-loaded DCs to induce CTL in the autologous system [107].
To define the pathways limiting DC function in the tumor microenvironment, Sharma et al. assessed the impact of tumor cyclooxygenase (COX)-2 expression on DC activities and reported that inhibition of tumor COX-2 expression or activity could prevent tumor-induced suppression of DC capacity to process and present antigens and secrete IL-12 [108]. COX-1-and COX-2-regulated prostanoids and IL-6 were found to be solely responsible for the hampered differentiation of monocyte-derived and CD34+-precursor-derived DCs by freshly excised solid human tumors (colon, breast, RCC, and melanoma) [109]. An important role for the EP2 receptor in prostaglandin E2 (PGE2)-induced inhibition of DC differentiation and function and the diminished antitumor cellular immune responses in vivo has been also reported [110]. Finally, PGE2 suppressed differentiation of DCs, it is a potent inducer of IL-10 in bone marrow-derived DCs, and PGE2-induced IL-10 is a key regulator of the DC pro-inflammatory phenotype [109].
In addition to these “classic” tumor-derived antidendropoietic factors, other molecules were implicated in tumor-mediated dysfunction of the DC system (Table 5.2). Melanoma, neuroblastoma, RCC, and lung cancer were shown to produce and shed various gangliosides, which may suppress dendropoiesis , inhibit DC function, or induce apoptosis in DCs [63], [69], [111]. Tumor-derived lactic acid is also an important factor modulating the DC phenotype in the tumor environment, which may critically contribute to tumor escape mechanisms [112]. Interestingly, several tumor antigens were recently found to display antidendropoietic properties. Prostate specific antigen (PSA), which is a serine protease, was able to inhibit generation and maturation of DCs from CD34+ hematopoietic precursors, assessed by the levels of expression of CD83, CD80, CD86, and HLA-DR, as well as the ability of DC to induce T-cell proliferation [113]. When cultured with the MUC1 glycoprotein, human monocyte-derived DCs displayed decreased expression of CD86, CD40, CD1d, HLA-DR, and CD83 and were defective in the ability to induce immune responses in both allogeneic and autologous settings. The modified phenotype of MUC1-treated DCs corresponded to an altered balance in IL-12/IL-10 cytokine production with a failure to make IL-12 and induce Th1 responses [114], [115]. Finally, human chorionic gonadotropin (hCG), which serves as an important tumor marker for trophoblastic disease, has been recently shown to upregulate expression of indoleamine-2,3-dioxygenase (IDO) in DCs [116].
HLA-G molecules, which are normally expressed in cytotrophoblasts and play a key role in maintaining immune tolerance at the maternal–fetal interface, were also reported to be expressed on malignant cells and they can be regulated by hypoxia [117], [118]. As DCs express immunoglobulin-like transcript 4 (ILT4), an inhibitory receptor capable of interacting with HLA-G, they may be tolerized by HLA-G through inhibitory receptor interactions. Indeed, the HLA-G–ILT4 interaction leads to development of tolerogenic DCs with the induction of anergic and immunosuppressive T cells [119].
Finally, human tumors constitutively release endosome-derived microvesicles, transporting a broad array of biologically active molecules with potential modulatory effects on different immune cells. The first evidence that tumor-released microvesicles alter myeloid cell function by impairing monocyte differentiation into DCs and promoting the generation of a myeloid immunosuppressive cell subset was probably published by Valenti et al. [89], [120] and then confirmed by other teams [121], [122].
3.2 Signaling Pathways
Many immunosuppressive factors produced by tumor cells induce signal transducer and activator of transcription 3 (STAT3) activation in DCs, blocking their normal functioning. For instance, treatment of DCs with melanoma-conditioned medium resulted in reduced expression of IL-12, MHC class II, and CD40 due to the increased induction of STAT3 [123]. The immunosuppressive effects of tumor-derived factors on DC differentiation were abrogated in cells from STAT3 knockout mice or by the treatment of DC precursors with a phosphopeptide that binds the STAT3 Src homology 2 (SH2) domain and blocks downstream STAT activation. Furthermore, IL-6-mediated suppression of DC maturation was also abrogated in STAT3-deficient DC precursors, indicating the significance of STAT3 in IL-6-mediated suppression of DC maturation and function [123]. Furthermore, constitutive STAT3 activation in tumor cells was shown to inhibit DC function by the increased induction of STAT3 in immature DCs. Thus, immunosuppression mediated by tumor cells results from a circuit of STAT3 signaling that begins in tumor cells and eventually activates inhibitory STAT3 signaling in DCs in part due to the production of cytokines that increase STAT3 activation in DCs (epidermal growth factor (EGF), VEGF, IL-6, IL-10, granulocyte colony-stimulating factor (G-CSF), macrophage colony-stimulating factor (M-CSF), and GM-CSF) [123]. In addition, STAT3 phosphorylation in DCs was regulated by IL-6 in vivo, and STAT3 was necessary for the IL-6 suppression of DC activation/maturation [124]. Interestingly, CD4+CD25+Foxp3+ Treg cells from tumor-bearing animals may also impede DC function by activating STAT3 and inducing the Smad signaling pathway [125]. The suppression mechanism was also associated with downregulation of activation of the transcription factor NF-κB, required TGF-beta and IL-10, and resulted in strong inhibition of expression of the costimulatory molecules CD80, CD86, and CD40 and the production of TNF-α, IL-12, and chemokine (C-C motif) ligand 5 (CCL5 or RANTES—regulated on activation, normal T cell expressed and secreted) by DCs.
Many STAT family members are developmentally regulated and play a role in DC differentiation and maturation. For instance, the STAT6 signaling pathway is constitutively activated in immature DCs and declines as they differentiate into mature DCs. Downregulation of the STAT6 pathway is accompanied by dramatic induction of suppressors of cytokine signaling 1 (SOCS1), SOCS2, SOCS3, and cytokine-induced SH2-containing protein expression [126]. In contrast, STAT1 signaling is most robust in mature DCs. Thus, it is likely that cytokine-induced maturation of DCs is under feedback regulation by SOCS proteins and that the switch from constitutive activation of the STAT6 pathway in immature DCs to predominant use of STAT1 signals in mature DCs is mediated in part by STAT1-induced SOCS expression [126]. Recent studies also demonstrate that SOCS1 functions as an antigen-presentation attenuator by controlling the tolerogenic state of DCs and the magnitude of antigen presentation [127]. Since SOCS1 restricts DCs’ ability to break self-tolerance and induce antitumor immunity by regulating IL-12 production and signaling, it is quite possible that some products of tumor cells or other cells within the tumor milieu might induce SOCS1 expression in DCs. Although not proven experimentally, this pathway may operate in the tumor microenvironment limiting the ability of DCs to process and present tumor antigens and secrete IL-12.
Another interesting mechanism responsible for tumor-induced downregulation of MHC class II expression in DCs was reported by Choi et al. They found that STAT5 bound to the CIITA pI locus during DC differentiation and that the binding was markedly attenuated by a tumor-conditioned medium or by IL-10 [128]. IL-10 inhibited the expression of type I CIITA during DC differentiation: GM-CSF-mediated histone (H3 and H4) acetylation at the type I promoter (pI) locus of the CIITA gene was markedly increased during DC differentiation and this increase was blocked by IL-10. This suggests that IL-10-mediated MHC class II downregulation results from the inhibition of type I CIITA expression. This inhibition is most likely due to blocking of the STAT5-associated epigenetic modifications of the CIITA pI locus during the entire period of DC differentiation from bone marrow-derived precursors, as opposed to a simple inhibition of MHC class II expression at the immature/mature DC stage.
Wang et al. too speculated that tumor-induced p38 mitogen-activated protein kinase (MAPK) activation and extracellular signal-regulated kinase (ERK) inhibition in DCs might be a new mechanism of tumor evasion [129]. They showed that tumor supernatant-treated DCs were inferior to normal DCs at priming tumor-specific immune responses, but inhibiting p38 MAPK restored the phenotype, cytokine secretion, and function of tumor-treated DCs. Tumor-derived factors activated p38 MAPK and Janus kinase (JNK) but inhibited ERK in DCs. Interestingly, Farrent et al. have recently reported that tumor-mediated myeloid dysregulation may be mediated by Stat3-induced protein kinase C isoform βII (PKCβII) downregulation: they showed that tumors mediate both Stat3 activation and PKCβII downregulation in DC progenitor cells, a process mimicked by the expression of a constitutive active Stat3 mutant [130].
Since many functions of DCs, such as endocytosis, exocytosis, adhesiveness, and motility, depend on actin polymerization and membrane rearrangements, Tourkova et al. analyzed whether small Rho guanosine triphosphatases (GTPases: Cdc42, RhoA, and Rac1/2), which are primarily involved in regulating these functions in DCs [111], might be affected by tumor-derived factors. They found that impaired endocytic activity of DC cocultured with tumor cells was associated with decreased levels of active Cdc42 and Rac1. Transduction of DCs with the dominant negative Cdc42 and Rac1 genes also led to reduced phagocytosis and receptor-mediated endocytosis, while transduction of DCs with the constitutively active Cdc42 and Rac1 genes restored the endocytic activity of DCs that was inhibited by the tumors [131].
Less is known about signaling pathways that control DC longevity and DC sensitivity to tumor-induced cell death. Early studies showed that Bcl-XL, Bcl-2, and mitochondrial cytochrome c release mediate resistance of DCs to tumor-induced apoptosis [132], [133]. Other data demonstrated that downregulation of phosphoinositide 3-kinase (PI3K) is the major facet of tumor-induced DC apoptosis [134]. Interestingly, it is known that some cancer cells have increased production of hydrogen peroxide (H2O2) [135], [136] and, in DCs, hydrogen peroxide activates two key MAPKs, p38 and JNK. Activation of JNK, which is associated with inhibition of tyrosine phosphatases in DCs, is linked to the induction of DC apoptosis [137]. By targeting different antiapoptotic molecules, including FLICE-like inhibitory protein (FLIP), X-linked inhibitor of apoptosis protein or human IAP-like protein (XIAP/hILP), procaspase-9, and heat shock protein 70 (HSP70), Balkir et al. demonstrated that antiapoptotic molecules other than the Bcl-2 family of proteins were involved in tumor-induced apoptosis in DCs [138]. This suggests that tumor-induced apoptosis of DCs is not limited to the mitochondrial pathway of cell death and that both extrinsic and intrinsic apoptotic pathways play a role in DC survival in the tumor microenvironment.
4 Role of DCs in Tumor Escape Mechanisms
A growing body of evidence clearly demonstrates that different subsets of DC are directly and indirectly involved in controlling tumor growth and progression . However, with the realization that the DC lineage represents a varied collection of distinct populations, a question has arisen as to whether certain types of DC are dysregulated in tumor-bearing hosts, or whether the nature of immunological challenge and state of DC maturation define particular facets of innate/acquired/tolerogenic responses in the tumor environment. Numerous studies have revealed that specific DC subsets might be linked to immunological unresponsiveness and/or tolerance to tumor antigens. For instance, the clinical outcome of children with cancer has been shown to correlate with circulating pDC count: Children with high pDC counts at diagnosis showed significantly worse survival than those with low counts and the development of cancer was associated with low number of cDCs [139]. Elevated levels of pDC have been observed as breast cancer disseminates to the bone. The selective depletion of pDCs in mice led to a total abrogation of bone metastasis as well as to an increase in the TH1 antitumor response [140]. Thus, tumor-associated pDCs contribute to the tumor immunosuppressive network. Tumor ascites pDCs induced IL-10+CCR7+CD45RO+CD8+ Treg cells, which significantly suppress myeloid DC-mediated tumor-associated antigen-specific T-cell effector functions through IL-10 [141]. pDCs in tumor-draining lymph nodes might create a local microenvironment that is potently suppressive of host antitumor T-cell responses and this mechanism may be mediated by immunosuppressive IDO.
IDO degrades tryptophan to kynurenine, which is further metabolized to 3-hydroxyanthranilic acid and thus initiates the immunosuppressive pathway of tryptophan catabolism (see Chap. 6 in this monograph). Emerging evidence suggests that Treg cells may be generated de novo against specific tumor-derived antigens, and thus they arise as a direct consequence of antigen presentation in the tumor-draining lymph nodes [142]. IDO can also be expressed within the tumor itself, by tumor cells, or by host stromal cells, where it can inhibit the effector phase of the immune response [143]. Kynurenine pathway enzymes downstream of IDO can initiate tolerogenesis by DCs independently of tryptophan deprivation, as tolerogenic DCs can confer suppressive ability on otherwise immunogenic DCs in an IDO-dependent fashion [144]. IDO, i.e., tryptophan, kynurenine, or 3-hydroxyanthranilic acid, could also induce expression of the tolerogenic molecule HLA-G in DCs [145]. Thus, IDO and HLA-G can cooperate in the immune suppression, since HLA-G-expressing DCs might suppress or alter effector T cells as well. Indeed, activated CD4+ and CD8+ T cells could efficiently acquire immunosuppressive HLA-G from antigen-presenting cells through membrane transfers (a process called trogocytosis) and acquisition of HLA-G immediately reversed T-cell function from effectors to regulatory cells. These Treg cells were able to inhibit proliferative responses through HLA-G that they acquired [146]. Targeting IDO in regulatory DCs (regDCs) may represent a new approach for harnessing DCs in the tumor microenvironment [147] .
In support of the concept that certain DC subpopulations play crucial roles in tumor escape, it was recently reported that tumor expansion could stimulate Treg cells via a specific DC subset. During tumor progression, a subset of DC exhibiting a myeloid immature phenotype may be recruited to draining lymph nodes and selectively promote proliferation of Treg cells in a TGF-β-dependent manner [148]. Importantly, tumor cells are necessary and sufficient to convert DCs into regulatory cells that secrete TGF-β and stimulate Treg cell proliferation. Regulatory DCs in cancer may directly and indirectly maintain antigen-specific and nonspecific T-cell unresponsiveness by controlling T-cell polarization, MDSC and Treg differentiation and activity, and affecting specific microenvironmental conditions in premalignant niches [149].
Another subset of DCs might contribute to neovascularization at the tumor site. Recently, Conejo-Garcia et al. reported that within 3 weeks of culture with tumor cell-conditioned medium, bone marrow-derived DCs could be transdifferentiated into endothelial-like cells in vitro [150]. They also identified a novel leukocyte subset within ovarian carcinoma that coexpressed endothelial and DC markers which may play a role in the formation of blood vessels [151]. Curiel et al. observed high numbers of pDC in malignant ascites of patients with untreated ovarian carcinoma and showed that tumor-associated pDC induced angiogenesis in vivo through production of TNF-α and IL-8 [152]. By contrast, cDCs, which might suppress angiogenesis in vivo through production of IL-12, were absent from malignant ascites. Thus, the tumor may attract pDCs to augment neovascularization while excluding myeloid DC to prevent angiogenesis inhibition .
Thus, one mechanism contributing to immunologic unresponsiveness toward tumors may be presentation of tumor antigens by tolerogenic/regulatory host DCs. Indeed, using bone marrow chimeras in transgenic mice, Mihalyo et al. have recently reported that DCs, but not CD4+CD25+ Treg cells, play a critical role in programming CD4 cell responses to tumor antigens during tumorigenesis [153]. Regulatory DCs could be produced from bone marrow precursors in the presence of GM-CSF, IL-10, TGF-β1, and LPS or TNF-α and they retained their T-cell regulatory property in vitro and in vivo even under inflammatory conditions [154]. Another minor subpopulation of regulatory DCs has been recently described in murine spleen. These splenic CD19+ DCs that did not express the pDC marker acquired potent IDO-dependent T-cell suppressive functions [155].
However, proponents of the “maturation” hypothesis suggest that the maturation state of the DC in the premalignant/inflammatory milieu or in the newly formed tumor setting predicts the development of an antitumor immune response or tumor tolerance. An increased proportion of immature DCs with reduced expression of costimulatory molecules was seen or isolated from tumor mass of patients with RCC, prostate cancer, basal-cell carcinoma, and melanoma or was found in the peripheral blood of patients with breast, head and neck, lung, or esophageal cancer [156]. Similar data have been obtained using several mouse tumor models. The maturation hypothesis was also bolstered by studies showing that in tumor tissues, immature DCs resided within the tumor, whereas mature DCs were located in peritumoral areas [157]. Immature DCs cannot induce antitumor immune responses and, most importantly, immature DCs can induce T-cell tolerance or anergy. Thomachot et al. showed that breast carcinoma cells produce soluble factors (chemokine (C-C motif) ligand 20 (CCL20) and TGF-β), which attract DC precursors in vivo and promote their differentiation into immature DCs with altered functional capacities, and that these altered DCs may contribute to the impaired immune response against the tumor [158]. Similarly, a medium conditioned by human pancreatic carcinoma cells induced monocyte-derived immature DCs with inhibited proliferation, expression of costimulatory molecules (CD80 and CD40) and HLA-DR, and functional activity as assessed by T-cell activation and IL-12p70 production [105]. Immature DCs generated from pancreatic carcinoma patients in advanced stages of the disease similarly showed decreased levels of HLA-DR expression and reduced ability to stimulate T cells. Direct ex vivo flow cytometric analysis of various DC subpopulations in peripheral blood from hepatocellular carcinoma patients revealed an immature phenotype of circulating DCs that was associated with increased IL-10 concentrations in serum and with tumor progression [47], [101] .
To evaluate whether and to what extent the capacity of tumor-infiltrating DCs to drive immunization can be turned off by tumor cells, leading to tumor-specific tolerance rather than immunization, Perrot et al. have characterized the DCs isolated from human non-small cell lung cancer based on the expression of CD11c. All isolated DCs, including CD11chigh myeloid DC, CD11c− pDC, and a third DC subset expressing an intermediate level of CD11c, were immature and displayed poor antigen-presenting function even after TLR stimulation and reduced migratory response toward CCL21 and SDF-1 [159]. Interestingly, CD11cint myeloid DCs, which represented approximately 25 % of total DC in tumor and peritumor tissues, expressed low levels of costimulatory molecules contrasting with high levels of the immunoinhibitory molecule B7-H1. These data suggest that immature tumor-associated DCs have an ability to compromise the tumor-specific immune response in draining lymph nodes in vivo. However, our data demonstrate that immature bone-marrow-derived DCs cannot suppress proliferation of pre-activated T cells without pretreatment with tumor-derived factors. Our recent data also reveal that different tumor cell lines produce soluble factors that induce polarization of cDCs into regulatory DCs, both in vitro and in vivo. These regulatory DCs can suppress proliferation of pre-activated T cells and are phenotypically and functionally different from their precursors as well as the classical immature cDC [160]. Understanding the biology of regDCs and the mechanisms of their formation in the tumor immunoenvironment will provide a new therapeutic target for repolarizing protumorigenic immunoregulatory cells into proimmunogenic effector cells able to induce and support effective antitumor immunity .
In spite of multiple evidence supporting both “subpopulation”-based and “maturation”-based explanations of how the DC system is involved in tumor escape (Table 5.3), additional data suggest that the real situation might be significantly more complex. The first layer of complexity arrives from the results showing that DC subsets may induce both tolerogenic and immunogenic responses depending on the environmental stimuli. For example, although the general thought is that pDCs are usually tolerogenic, it appears that the functional role of pDCs in cancer immunity depends on cytokines that affect the balance between immunity and tolerance in the tumor and lymphoid organ microenvironment. In an analysis of draining lymph nodes in breast cancer, pDCs with a relative increase in IL-12 and interferon-γ (IFN-γ) were associated with a good prognosis, whereas pDCs with a relative increase in IL-10 and IL-4 were associated with a poor prognosis [161]. In confirmation of this conclusion, Kim et al. have reported that although pDCs recruited to the tumor site are implicated in facilitating tumor growth via immune suppression, they can be released from the tumor as a result of cell death caused by primary systemic chemotherapy and can then be activated through TLR9 [162]. Thus, synergistically with cDCs, pDCs may also play a crucial role in mediating cancer immunity. In fact, new results from a recent clinical trial indicate that vaccination with naturally occurring pDCs is feasible, with minimal toxicity, and that in patients with metastatic melanoma, it induces favorable immune responses [163]. Thus, we can conclude that pDCs, as well as myeloid cDCs, have a dual role not only in initiating immune responses but also in inducing tolerance to tumor antigens.
An additional layer of complexity of the DC subset versus the DC maturation problem in cancer comes from the data revealing different maturation patterns of different DC subsets and its differential regulation by other immune cells. For example, analysis of the maturation of human blood-derived cDCs and pDCs activated with TLR ligands in the presence of Treg cells revealed that pre-activated Treg cells strongly suppressed TLR-triggered cDC maturation, as judged by the blocking of costimulatory molecule upregulation and the inhibition of pro-inflammatory cytokine secretion that resulted in poor antigen presentation capacity. Although IL-10 played a prominent role in inhibiting cytokine secretion, suppression of phenotypic maturation required cell–cell contact and was independent of TGF-β and CTLA-4. In contrast, the acquisition of maturation markers and production of cytokines by pDCs triggered by TLR ligands were insensitive to Treg cells[164]. Therefore, human Treg may enlist conventional DCs, but not pDCs for the initiation and the amplification of tolerance in vivo by restraining their maturation after TLR stimulation .
In another study, evidence was provided that maturing cDCs and pDCs express different sets of molecules that drive distinct types of T-cell responses [165]. Although both maturing cDCs and pDCs upregulate the expression of CD80 and CD86, only pDCs upregulate the expression of inducible costimulatory ligand (ICOS-L) and maintain high expression levels upon differentiation into mature DCs. High ICOS-L expression endows maturing pDCs with the ability to induce the differentiation of naive CD4 T cells to produce IL-10 but not the Th2 cytokines IL-4, IL-5, and IL-13. These IL-10-producing T cells are Treg cells, and their generation by ICOS-L is independent of pDC-driven Th1 and Th2 differentiation. Thus, in contrast to cDCs, pDCs are poised to express ICOS-L upon maturation, which leads to the generation of IL-10-producing Treg cells [165].
As such, there is still a confusion in the field as to whether certain DC subpopulations have evolved to fulfill unique immunological roles in cancer (Th1/Th2/Th3/Th17 polarization, Treg induction, tolerance, etc.), or whether distinct DC subsets exist to uniquely respond to tumor-derived stimuli but each participates in maintaining tolerance or immunity in the immature or mature state. It is also somewhat undecided whether some of the diversity in the DC lineage as determined by cell surface-molecule expression represents genuine distinct DC subsets or particular developmental/activation states of the same DC subtype. However, collectively, an emerging view in the field is that DCs control the course of tumor immunity/tolerance on at least three levels: (1) the developmental repertoire of DC lineage populations which can dictate the nature of DC response to a particular stimulus in the tumor microenvironment, (2) the maturation stage of DCs when cells interact with other immune cells or respond to immunological signals (i.e., cytokines, chemokines, and TLR ligands), and (3) the environment within which DCs encounter tumor antigens, as defined by the tissue type, infiltrating leukocytes, and an inflammatory cytokine milieu .
Therefore, DCs in the tumor microenvironment serve as a double-edged sword and, in addition to initiating potent antitumor immune responses, may mediate genomic damage, support neovascularization, block antitumor immunity, and stimulate cancerous cell growth and spreading [149]. The importance of these issues and mechanisms controlling them is significant, as efforts to harness the power of DCs in vaccination strategies against tumors would ultimately aim to identify the correct type of DC for a particular approach and insure that these cells are appropriately activated or protected from tumor influence to elicit the desired response.
5 Concluding Remarks
Numerous experimental and clinical observations discussed above suggest that tumor-induced apoptosis or altered differentiation and function of DCs as well as accumulation of immature DCs or DC precursors with inhibitory and tolerogenic function could impair antitumor immune responses. For patients with cancer, the resulting dysfunction of the DC system would result in marked deficiency in the induction of antitumor immunity, tumor progression, and probably, low response to immunotherapy [34]. This is really important for understanding tumor immunopathology as well as reevaluating tumor immunotherapeutic strategies since DCs prepared from patients with cancer are being evaluated as a cellular vaccine in multiple clinical trials worldwide. However, to date, DC-based immunotherapies have met with limited success for several reasons, including the restricted longevity and efficacy of administered DCs in a suppressive tumor environment. Therefore, alternative approaches, including protection of DC longevity, blockade of tumor-mediated inhibitory pathways, and prevention of DC dysfunction/polarization ex vivo, should be evaluated to potentiate the efficacy of DC-based cancer vaccines. Given that endogenous DCs might be important for fulfilling the potential of various cellular vaccines, gained knowledge in the area of DC immunobiology in cancer may help to find new drugs to selectively block suppressive pathways and restore the original function of DCs.
References
Shurin MR, Lotze MT (2009) Dendritic cells in cancer: emergence of the discipline. In: Salter RD, Shurin MR Dendritic cells in cancer. Springer, New York, pp 11–30
Shurin MR, Gabrilovich DI (2001) Regulation of dendritic cell system by tumor. Cancer Res Ther Control 11:65–78
Fricke I, Gabrilovich DI (2006) Dendritic cells and tumor microenvironment: a dangerous liaison. Immunol Investig 35(3–4):459–483
Shurin MR, Shurin GV, Lokshin A, Yurkovetsky ZR, Gutkin DW, Chatta G, Zhong H, Han B, Ferris RL (2006) Intratumoral cytokines/chemokines/growth factors and tumor infiltrating dendritic cells: friends or enemies? Cancer Metastasis Rev 25(3):333–356
Saint-Mezard P, Chavagnac C, Bosset S, Ionescu M, Peyron E, Kaiserlian D, Nicolas JF, Berard F (2003) Psychological stress exerts an adjuvant effect on skin dendritic cell functions in vivo. J Immunol 171(8):4073–4080
Seiffert K, Granstein RD (2006) Neuroendocrine regulation of skin dendritic cells. Ann New York Acad Sci 1088:195–206
Maestroni GJ (2005) Adrenergic modulation of dendritic cells function: relevance for the immune homeostasis. Curr Neurovasc Res 2(2):169–173
Piemonti L, Monti P, Allavena P, Sironi M, Soldini L, Leone BE, Socci C, Di Carlo V (1999) Glucocorticoids affect human dendritic cell differentiation and maturation. J Immunol 162(11):6473–6481
Maestroni GJ, Mazzola P (2003) Langerhans cells beta 2-adrenoceptors: role in migration, cytokine production, Th priming and contact hypersensitivity. J Neuroimmunol 144(1–2):91–99
Maestroni GJ (2000) Dendritic cell migration controlled by alpha 1b-adrenergic receptors. J Immunol 165(12):6743–6747
Lambert RW, Granstein RD (1998) Neuropeptides and Langerhans cells. Exp Dermatol 7(2–3):73–80
Bedoui S, von Horsten S, Gebhardt T (2007) A role for neuropeptide Y (NPY) in phagocytosis: implications for innate and adaptive immunity. Peptides 28(2):373–376
Makarenkova VP, Esche C, Kost NV, Shurin GV, Rabin BS, Zozulya AA, Shurin MR (2001) Identification of delta- and mu-type opioid receptors on human and murine dendritic cells. J Neuroimmunol 117(1–2):68–77
Matera L, Mori M, Galetto A (2001) Effect of prolactin on the antigen presenting function of monocyte-derived dendritic cells. Lupus 10(10):728–734
Makarenkova VP, Shurin GV, Tourkova IL, Balkir L, Pirtskhalaishvili G, Perez L, Gerein V, Siegfried JM, Shurin MR (2003) Lung cancer-derived bombesin-like peptides down-regulate the generation and function of human dendritic cells. J Neuroimmunol 145(1–2):55–67
Marriott I, Bost KL (2001) Expression of authentic substance P receptors in murine and human dendritic cells. J Neuroimmunol 114(1–2):131–141
Cao MD, Chen ZD, Xing Y (2004) Gamma irradiation of human dendritic cells influences proliferation and cytokine profile of T cells in autologous mixed lymphocyte reaction. Cell Biol Int 28(3):223–228
Bellik L, Gerlini G, Parenti A, Ledda F, Pimpinelli N, Neri B, Pantalone D (2006) Role of conventional treatments on circulating and monocyte-derived dendritic cells in colorectal cancer. Clin Immunol 121(1):74–80
Corrales JJ, Almeida M, Burgo R, Mories MT, Miralles JM, Orfao A (2006) Androgen-replacement therapy depresses the ex vivo production of inflammatory cytokines by circulating antigen-presenting cells in aging type-2 diabetic men with partial androgen deficiency. J Endocrinol 189(3):595–604
Schmidt J, Jager D, Hoffmann K, Buchler MW, Marten A (2007) Impact of interferon-alpha in combined chemoradioimmunotherapy for pancreatic adenocarcinoma (CapRI): first data from the immunomonitoring. J Immunother 30(1):108–115
Shurin MR, Naiditch H, Gutkin DW, Umansky V, Shurin GV (2012) ChemoImmunoModulation: immune regulation by the antineoplastic chemotherapeutic agents. Curr Med Chem 19(12):1792–1803
Naiditch H, Shurin MR, Shurin GV (2011) Targeting myeloid regulatory cells in cancer by chemotherapeutic agents. Immunol Res 50(2–3):276–285
Kaneno R, Shurin GV, Kaneno FM, Naiditch H, Luo J, Shurin MR (2011) Chemotherapeutic agents in low noncytotoxic concentrations increase immunogenicity of human colon cancer cells. Cell Oncol (Dordrecht) 34(2):97–106
Nakashima H, Tasaki A, Kubo M, Kuroki H, Matsumoto K, Tanaka M, Nakamura M, Morisaki T, Katano M (2005) Effects of docetaxel on antigen presentation-related functions of human monocyte-derived dendritic cells. Cancer Chemother Pharmacol 55(5):479–487
Shurin GV, Tourkova IL, Kaneno R, Shurin MR (2009) Chemotherapeutic agents in noncytotoxic concentrations increase antigen presentation by dendritic cells via an IL-12-dependent mechanism. J Immunol 183(1):137–144
Tkach AV, Shurin GV, Shurin MR, Kisin ER, Murray AR, Young SH, Star A, Fadeel B, Kagan VE, Shvedova AA (2011) Direct effects of carbon nanotubes on dendritic cells induce immune suppression upon pulmonary exposure. ACS Nano 5(7):5755–5762
Tkach AV, Yanamala N, Stanley S, Shurin MR, Shurin GV, Kisin ER, Murray AR, Pareso S, Khaliullin T, Kotchey GP, Castranova V, Mathur S, Fadeel B, Star A, Kagan VE, Shvedova AA (2012) Graphene oxide, but not fullerenes, targets immunoproteasomes and suppresses antigen presentation by dendritic cells. Small 9:1686–1690
Shvedova AA, Tkach AV, Kisin ER, Khaliullin T, Stanley S, Gutkin DW, Star A, Chen Y, Shurin GV, Kagan VE, Shurin MR (2013) Carbon nanotubes enhance metastatic growth of lung carcinoma via up-regulation of myeloid-derived suppressor cells. Small 9:1691–1695
Shurin MR, Shurin GV, Chatta GS (2007) Aging and the Dendritic Cell System: Implications for Cancer. Crit Rev Oncol Hematol. 64(2):90–105
Corinti S, Albanesi C, la Sala A, Pastore S, Girolomoni G (2001) Regulatory activity of autocrine IL-10 on dendritic cell functions. J Immunol 166(7):4312–4318
Tarazona R, Solana R, Ouyang Q, Pawelec G (2002) Basic biology and clinical impact of immunosenescence. Exp Gerontol 37(2–3):183–189
Bauer ME (2005) Stress, glucocorticoids and ageing of the immune system. Stress 8(1):69–83
Shurin MR, Salter RD (eds) (2009) Dendritic cells in cancer. Springer, New York
Ma Y, Shurin GV, Peiyuan Z, Shurin MR (2013) Dendritic cells in the cancer microenvironment. J Cancer 4(1):36–44
Stene MA, Babajanians M, Bhuta S, Cochran AJ (1988) Quantitative alterations in cutaneous Langerhans cells during the evolution of malignant melanoma of the skin. J Invest Dermatol 91(2):125–128
Alcalay J, Goldberg LH, Wolf JE Jr, Kripke ML (1989) Variations in the number and morphology of Langerhans’ cells in the epidermal component of squamous cell carcinomas. Arch Dermatol 125(7):917–920
Alcalay J, Kripke ML (1991) Antigen-presenting activity of draining lymph node cells from mice painted with a contact allergen during ultraviolet carcinogenesis. J Immunol 146:1717–1721
Halliday GM, Reeve VE, Barnetson RS (1991) Langerhans cell migration into ultraviolet light-induced squamous skin tumors is unrelated to anti-tumor immunity. J Invest Dermatol 97(5):830–834
Halliday GM, Lucas AD, Barnetson RS (1992) Control of Langerhans’ cell density by a skin tumour-derived cytokine. Immunology 77(1):13–18
Becker Y (1992) Anticancer role of dendritic cells (DC) in human and experimental cancers—a review. Anticancer Res 12(2):511–520
Becker Y (1993) Dendritic cell activity against primary tumors: an overview. Vivo 7(3):187–191
Tas MP, Simons PJ, Balm FJ, Drexhage HA (1993) Depressed monocyte polarization and clustering of dendritic cells in patients with head and neck cancer: in vitro restoration of this immunosuppression by thymic hormones. Cancer Immunol Immunother 36(2):108–114
Colasante A, Castrilli G, Aiello FB, Brunetti M, Musiani P (1995) Role of cytokines in distribution and differentiation of dendritic cell/Langerhans’ cell lineage in human primary carcinomas of the lung. Hum Pathol 26(8):866–872
Gabrilovich DI, Nadaf S, Corak J, Berzofsky JA, Carbone DP (1996) Dendritic cells in antitumor immune responses. II. Dendritic cells grown from bone marrow precursors, but not mature DC from tumor-bearing mice, are effective antigen carriers in the therapy of established tumors. Cell Immunol 170(1):111–119
Chaux P, Moutet M, Faivre J, Martin F, Martin M (1996) Inflammatory cells infiltrating human colorectal carcinomas express HLA class II but not B7-1 and B7-2 costimulatory molecules of the T-cell activation. Lab Invest 74(5):975–983
Enk AH, Jonuleit H, Saloga J, Knop J (1997) Dendritic cells as mediators of tumor-induced tolerance in metastatic melanoma. Int J Cancer 73(3):309–316
Ninomiya T, Akbar SM, Masumoto T, Horiike N, Onji M (1999) Dendritic cells with immature phenotype and defective function in the peripheral blood from patients with hepatocellular carcinoma. J Hepatol 31(2):323–331
Lissoni P, Bolis S, Mandala M, Viviani S, Pogliani E, Barni S (1999) Blood concentrations of tumor necrosis factor-alpha in malignant lymphomas and their decrease as a predictor of disease control in response to low-dose subcutaneous immunotherapy with interleukin-2. Int J Biol Markers 14(3):167–171
Sakakura K, Chikamatsu K, Takahashi K, Whiteside TL, Furuya N (2006) Maturation of circulating dendritic cells and imbalance of T-cell subsets in patients with squamous cell carcinoma of the head and neck. Cancer Immunol Immunother 55(2):151–159
Maecker B, Mougiakakos D, Zimmermann M, Behrens M, Hollander S, Schrauder A, Schrappe M, Welte K, Klein C (2006) Dendritic cell deficiencies in pediatric acute lymphoblastic leukemia patients. Leukemia 20(4):645–649
Ormandy LA, Farber A, Cantz T, Petrykowska S, Wedemeyer H, Horning M, Lehner F, Manns MP, Korangy F, Greten TF (2006) Direct ex vivo analysis of dendritic cells in patients with hepatocellular carcinoma. World J Gastroenterol 12(20):3275–3282
Wojas K, Tabarkiewicz J, Jankiewicz M, Rolinski J (2004) Dendritic cells in peripheral blood of patients with breast and lung cancer–a pilot study. Folia Histochem Cytobiol 42(1):45–48
Della Bella S, Gennaro M, Vaccari M, Ferraris C, Nicola S, Riva A, Clerici M, Greco M, Villa ML (2003) Altered maturation of peripheral blood dendritic cells in patients with breast cancer. Br J Cancer 89(8):1463–1472
Ogden AT, Horgan D, Waziri A, Anderson D, Louca J, McKhann GM, Sisti MB, Parsa AT, Bruce JN (2006) Defective receptor expression and dendritic cell differentiation of monocytes in glioblastomas. Neurosurgery 59(4):902–909; discussion 909–910
Hasebe H, Nagayama H, Sato K, Enomoto M, Takeda Y, Takahashi TA, Hasumi K, Eriguchi M (2000) Dysfunctional regulation of the development of monocyte-derived dendritic cells in cancer patients. Biomed Pharmacother 54(6):291–298
Pedersen AE, Thorn M, Gad M, Walter MR, Johnsen HE, Gaarsdal E, Nikolajsen K, Buus S, Claesson MH, Svane IM (2005) Phenotypic and functional characterization of clinical grade dendritic cells generated from patients with advanced breast cancer for therapeutic vaccination. Scand J Immunol 61(2):147–156
Neves AR, Ensina LF, Anselmo LB, Leite KR, Buzaid AC, Camara-Lopes LH, Barbuto JA (2005) Dendritic cells derived from metastatic cancer patients vaccinated with allogeneic dendritic cell-autologous tumor cell hybrids express more CD86 and induce higher levels of interferon-gamma in mixed lymphocyte reactions. Cancer Immunol Immunother 54(1):61–66
Onishi H, Morisaki T, Kuroki H, Matsumoto K, Baba E, Kuga H, Tanaka M, Katano M (2005) Evaluation of a dysfunctional and short-lived subset of monocyte-derived dendritic cells from cancer patients. Anticancer Res 25(5):3445–3451
Shurin MR (1999) Regulation of dendropoiesis in cancer. Clin Immunol Newslett 19(10/11):135–139
Esche C, Lokshin A, Shurin GV, Gastman BR, Rabinowich H, Watkins SC, Lotze MT, Shurin MR (1999) Tumor’s other immune targets: dendritic cells. J Leukoc Biol 66(2):336–344
Kiertscher SM, Luo J, Dubinett SM, Roth MD (2000) Tumors promote altered maturation and early apoptosis of monocyte-derived dendritic cells. J Immunol 164(3):1269–1276
Yang T, Witham TF, Villa L, Erff M, Attanucci J, Watkins S, Kondziolka D, Okada H, Pollack IF, Chambers WH (2002) Glioma-associated hyaluronan induces apoptosis in dendritic cells via inducible nitric oxide synthase: implications for the use of dendritic cells for therapy of gliomas. Cancer Res 62(9):2583–2591
Peguet-Navarro J, Sportouch M, Popa I, Berthier O, Schmitt D, Portoukalian J (2003) Gangliosides from human melanoma tumors impair dendritic cell differentiation from monocytes and induce their apoptosis. J Immunol 170(7):3488–3494
Pinzon-Charry A, Maxwell T, McGuckin MA, Schmidt C, Furnival C, Lopez JA (2006) Spontaneous apoptosis of blood dendritic cells in patients with breast cancer. Breast Cancer Res 8(1):R5
Katsenelson NS, Shurin GV, Bykovskaia SN, Shogan J, Shurin MR (2001) Human small cell lung carcinoma and carcinoid tumor regulate dendritic cell maturation and function. Mod Pathol 14(1):40–45
Onishi H, Morisaki T, Baba E, Kuga H, Kuroki H, Matsumoto K, Tanaka M, Katano M (2002) Dysfunctional and short-lived subsets in monocyte-derived dendritic cells from patients with advanced cancer. Clin Immunol 105(3):286–295
Aalamian M, Pirtskhalaishvili G, Nunez A, Esche C, Shurin GV, Huland E, Huland H, Shurin MR (2001) Human prostate cancer regulates generation and maturation of monocyte-derived dendritic cells. Prostate 46(1):68–75
Brown RD, Pope B, Murray A, Esdale W, Sze DM, Gibson J, Ho PJ, Hart D, Joshua D (2001) Dendritic cells from patients with myeloma are numerically normal but functionally defective as they fail to up-regulate CD80 (B7–1) expression after huCD40LT stimulation because of inhibition by transforming growth factor-beta1 and interleukin-10. Blood 98(10):2992–2998
Shurin GV, Shurin MR, Bykovskaia S, Shogan J, Lotze MT, Barksdale EM Jr (2001) Neuroblastoma-derived gangliosides inhibit dendritic cell generation and function. Cancer Res 61(1):363–369
Ratta M, Fagnoni F, Curti A, Vescovini R, Sansoni P, Oliviero B, Fogli M, Ferri E, Della Cuna GR, Tura S, Baccarani M, Lemoli RM (2002) Dendritic cells are functionally defective in multiple myeloma: the role of interleukin-6. Blood 100(1):230–237
Satthaporn S, Robins A, Vassanasiri W, El-Sheemy M, Jibril JA, Clark D, Valerio D, Eremin O (2004) Dendritic cells are dysfunctional in patients with operable breast cancer. Cancer Immunol Immunother 53(6):510–518
Song EY, Shurin MR, Tourkova IL, Chatta G, Shurin GV (2004) Human renal cell carcinoma inhibits dendritic cell maturation and functions. Urologe A 43(Suppl 3):128–130
Kichler-Lakomy C, Budinsky AC, Wolfram R, Hellan M, Wiltschke C, Brodowicz T, Viernstein H, Zielinski CC (2006) Deficiences in phenotype expression and function of dentritic cells from patients with early breast cancer. Eur J Med Res 11(1):7–12
Ataera H, Hyde E, Price KM, Stoitzner P, Ronchese F (2011) Murine melanoma-infiltrating dendritic cells are defective in antigen presenting function regardless of the presence of CD4CD25 regulatory T cells. PloS One 6(3):e17515
Vicari AP, Caux C, Trinchieri G (2002) Tumour escape from immune surveillance through dendritic cell inactivation. Semin Cancer Biol 12(1):33–42
Shurin GV, Yurkovetsky ZR, Shurin MR (2003) Tumor-induced dendritic cell dysfunction. In: A. Ochoa (ed) Mechanisms of Tumor Escape from the Immune Response. Taylor and Francis, London, pp 112–138
Yang L, Carbone DP (2004) Tumor-host immune interactions and dendritic cell dysfunction. Adv Cancer Res 92:13–27
Eisendle K, Wolf D, Gastl G, Kircher-Eibl B (2005) Dendritic cells from patients with chronic myeloid leukemia: functional and phenotypic features. Leuk Lymphoma 46(5):663–670
Pinzon-Charry A, Maxwell T, Lopez JA (2005) Dendritic cell dysfunction in cancer: a mechanism for immunosuppression. Immunol Cell Biol 83(5):451–461
Zou W, Machelon V, Coulomb-L’Hermin A, Borvak J, Nome F, Isaeva T, Wei S, Krzysiek R, Durand-Gasselin I, Gordon A, Pustilnik T, Curiel DT, Galanaud P, Capron F, Emilie D, Curiel TJ (2001) Stromal-derived factor-1 in human tumors recruits and alters the function of plasmacytoid precursor dendritic cells. Nat Med 7(12):1339–1346
Wertel F, Polak G, Rolinski J, Barczynski B, Kotarski J (2006) Myeloid and lymphoid dendritic cells in the peritoneal fluid of women with ovarian cancer. Adv Med Sci 51:174–177
Wertel I, Polak G, Bednarek W, Barczynski B, Rolinski J, Kotarski J (2008) Dendritic cell subsets in the peritoneal fluid and peripheral blood of women suffering from ovarian cancer. Cytometry 74(4):251–258
Sisirak V, Faget J, Gobert M, Goutagny N, Vey N, Treilleux I, Renaudineau S, Poyet G, Labidi-Galy SI, Goddard-Leon S, Durand I, Le Mercier I, Bajard A, Bachelot T, Puisieux A, Puisieux I, Blay JY, Menetrier-Caux C, Caux C, Bendriss-Vermare N (2012) Impaired IFN-alpha production by plasmacytoid dendritic cells favors regulatory T-cell expansion that may contribute to breast cancer progression. Cancer Res 72(20):5188–5197
Sisirak V, Vey N, Goutagny N, Renaudineau S, Malfroy M, Thys S, Treilleux I, Labidi-Galy SI, Bachelot T, Dezutter-Dambuyant C, Menetrier-Caux C, Blay JY, Caux C, Bendriss-Vermare N (2013) Breast cancer-derived transforming growth factor-beta and tumor necrosis factor-alpha compromise interferon-alpha production by tumor-associated plasmacytoid dendritic cells. Int J Cancer 133(3):771–778
Hoffmann TK, Muller-Berghaus J, Ferris RL, Johnson JT, Storkus WJ, Whiteside TL (2002) Alterations in the frequency of dendritic cell subsets in the peripheral circulation of patients with squamous cell carcinomas of the head and neck. Clin Cancer Res 8(6):1787–1793
Pinzon-Charry A, Ho CS, Laherty R, Maxwell T, Walker D, Gardiner RA, O’Connor L, Pyke C, Schmidt C, Furnival C, Lopez JA (2005) A population of HLA-DR+ immature cells accumulates in the blood dendritic cell compartment of patients with different types of cancer. Neoplasia 7(12):1112–1122
Takahashi K, Toyokawa H, Takai S, Satoi S, Yanagimoto H, Terakawa N, Araki H, Kwon AH, Kamiyama Y (2006) Surgical influence of pancreatectomy on the function and count of circulating dendritic cells in patients with pancreatic cancer. Cancer Immunol Immunother 55(7):775–784
Yanagimoto H, Takai S, Satoi S, Toyokawa H, Takahashi K, Terakawa N, Kwon AH, Kamiyama Y (2005) Impaired function of circulating dendritic cells in patients with pancreatic cancer. Clin Immunol 114(1):52–60
Valenti R, Huber V, Filipazzi P, Pilla L, Sovena G, Villa A, Corbelli A, Fais S, Parmiani G, Rivoltini L (2006) Human tumor-released microvesicles promote the differentiation of myeloid cells with transforming growth factor-beta-mediated suppressive activity on T lymphocytes. Cancer Res 66(18):9290–9298
Kim R, Emi M, Tanabe K (2006) Functional roles of immature dendritic cells in impaired immunity of solid tumour and their targeted strategies for provoking tumour immunity. Clin Exp Immunol 146(2):189–196
Lizee G, Radvanyi LG, Overwijk WW, Hwu P (2006) Improving antitumor immune responses by circumventing immunoregulatory cells and mechanisms. Clin Cancer Res 12(16):4794–4803
Shurin GV, Ferris RL, Tourkova IL, Perez L, Lokshin A, Balkir L, Collins B, Chatta GS, Shurin MR (2005) Loss of new chemokine CXCL14 in tumor tissue is associated with low infiltration by dendritic cells (DC), while restoration of human CXCL14 expression in tumor cells causes attraction of DC both in vitro and in vivo. J Immunol 174(9):5490–5498
Song EY, Shurin MR, Tourkova IL, Gutkin DW, Shurin GV (2010) Epigenetic mechanisms of promigratory chemokine CXCL14 regulation in human prostate cancer cells. Cancer Res 70(11):4394–4401
Remmel E, Terracciano L, Noppen C, Zajac P, Heberer M, Spagnoli GC, Padovan E (2001) Modulation of dendritic cell phenotype and mobility by tumor cells in vitro. Hum Immunol 62(1):39–49
Feijoo E, Alfaro C, Mazzolini G, Serra P, Penuelas I, Arina A, Huarte E, Tirapu I, Palencia B, Murillo O, Ruiz J, Sangro B, Richter JA, Prieto J, Melero I (2005) Dendritic cells delivered inside human carcinomas are sequestered by interleukin-8. Int J Cancer 116(2):275–281
Gabrilovich DI, Chen HL, Girgis KR, Cunningham HT, Meny GM, Nadaf S, Kavanaugh D, Carbone DP (1996) Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med 2(10):1096–1103
Della Porta M, Danova M, Rigolin GM, Brugnatelli S, Rovati B, Tronconi C, Fraulini C, Russo Rossi A, Riccardi A, Castoldi G (2005) Dendritic cells and vascular endothelial growth factor in colorectal cancer: correlations with clinicobiological findings. Oncology 68(2–3):276–284
Kim R, Emi M, Tanabe K, Arihiro K (2006) Tumor-driven evolution of immunosuppressive networks during malignant progression. Cancer Res 66(11):5527–5536
Geissmann F, Revy P, Regnault A, Lepelletier Y, Dy M, Brousse N, Amigorena S, Hermine O, Durandy A (1999) TGF-beta 1 prevents the noncognate maturation of human dendritic Langerhans cells. J Immunol 162(8):4567–4575
Ito M, Minamiya Y, Kawai H, Saito S, Saito H, Nakagawa T, Imai K, Hirokawa M, Ogawa J (2006) Tumor-derived TGFbeta-1 induces dendritic cell apoptosis in the sentinel lymph node. J Immunol 176(9):5637–5643
Beckebaum S, Zhang X, Chen X, Yu Z, Frilling A, Dworacki G, Grosse-Wilde H, Broelsch CE, Gerken G, Cicinnati VR (2004) Increased levels of interleukin-10 in serum from patients with hepatocellular carcinoma correlate with profound numerical deficiencies and immature phenotype of circulating dendritic cell subsets. Clin Cancer Res 10(21):7260–7269
Lan YY, Wang Z, Raimondi G, Wu W, Colvin BL, de Creus A, Thomson AW (2006) “Alternatively activated” dendritic cells preferentially secrete IL-10, expand Foxp3+CD4+ T cells, and induce long-term organ allograft survival in combination with CTLA4-Ig. J Immunol 177(9):5868–5877
Shurin MR, Yurkovetsky ZR, Tourkova IL, Balkir L, Shurin GV (2002) Inhibition of CD40 expression and CD40-mediated dendritic cell function by tumor-derived IL-10. Int J Cancer 101(1):61–68
Yang AS, Lattime EC (2003) Tumor-induced interleukin 10 suppresses the ability of splenic dendritic cells to stimulate CD4 and CD8 T-cell responses. Cancer Res 63(9):2150–2157
Bellone G, Carbone A, Smirne C, Scirelli T, Buffolino A, Novarino A, Stacchini A, Bertetto O, Palestro G, Sorio C, Scarpa A, Emanuelli G, Rodeck U (2006) Cooperative induction of a tolerogenic dendritic cell phenotype by cytokines secreted by pancreatic carcinoma cells. J Immunol 177(5):3448–3460
Avila-Moreno F, Lopez-Gonzalez JS, Galindo-Rodriguez G, Prado-Garcia H, Bajana S, Sanchez-Torres C (2006) Lung squamous cell carcinoma and adenocarcinoma cell lines use different mediators to induce comparable phenotypic and functional changes in human monocyte-derived dendritic cells. Cancer Immunol Immunother 55(5):598–611
Cabillic F, Bouet-Toussaint F, Toutirais O, Rioux-Leclercq N, Fergelot P, de la Pintiere CT, Genetet N, Patard JJ, Catros-Quemener V (2006) Interleukin-6 and vascular endothelial growth factor release by renal cell carcinoma cells impedes lymphocyte-dendritic cell cross-talk. Clin Exp Immunol 146(3):518–523
Sharma S, Stolina M, Yang SC, Baratelli F, Lin JF, Atianzar K, Luo J, Zhu L, Lin Y, Huang M, Dohadwala M, Batra RK, Dubinett SM (2003) Tumor cyclooxygenase 2-dependent suppression of dendritic cell function. Clin Cancer Res 9(3):961–968
Sombroek CC, Stam AG, Masterson AJ, Lougheed SM, Schakel MJ, Meijer CJ, Pinedo HM, van den Eertwegh AJ, Scheper RJ, de Gruijl TD (2002) Prostanoids play a major role in the primary tumor-induced inhibition of dendritic cell differentiation. J Immunol 168(9):4333–4343
Yang L, Yamagata N, Yadav R, Brandon S, Courtney RL, Morrow JD, Shyr Y, Boothby M, Joyce S, Carbone DP, Breyer RM (2003) Cancer-associated immunodeficiency and dendritic cell abnormalities mediated by the prostaglandin EP2 receptor. J Clin Invest 111(5):727–735
Tourkova IL, Shurin GV, Chatta GS, Perez L, Finke J, Whiteside TL, Ferrone S, Shurin MR (2005) Restoration by IL-15 of MHC class I antigen-processing machinery in human dendritic cells inhibited by tumor-derived gangliosides. J Immunol 175(5):3045–3052
Gottfried E, Kunz-Schughart LA, Ebner S, Mueller-Klieser W, Hoves S, Andreesen R, Mackensen A, Kreutz M (2006) Tumor-derived lactic acid modulates dendritic cell activation and antigen expression. Blood 107(5):2013–2021
Aalamian M, Tourkova IL, Chatta GS, Lilja H, Huland E, Huland H, Shurin GV, Shurin MR (2003) Inhibition of dendropoiesis by tumor derived and purified prostate specific antigen. J Urol 170(5):2026–2030
Rughetti A, Pellicciotta I, Biffoni M, Backstrom M, Link T, Bennet EP, Clausen H, Noll T, Hansson GC, Burchell JM, Frati L, Taylor-Papadimitriou J, Nuti M (2005) Recombinant tumor-associated MUC1 glycoprotein impairs the differentiation and function of dendritic cells. J Immunol 174(12):7764–7772
Carlos CA, Dong HF, Howard OM, Oppenheim JJ, Hanisch FG, Finn OJ (2005) Human tumor antigen MUC1 is chemotactic for immature dendritic cells and elicits maturation but does not promote Th1 type immunity. J Immunol 175(3):1628–1635
Ueno A, Cho S, Cheng L, Wang J, Hou S, Nakano H, Santamaria P, Yang Y (2007) Transient upregulation of IDO in dendritic cells by human chorionic gonadotropin downregulates autoimmune diabetes. Diabetes 56(6):1686–1693
Wilczynski JR (2006) Cancer and pregnancy share similar mechanisms of immunological escape. Chemotherapy 52(3):107–110
Mouillot G, Marcou C, Zidi I, Guillard C, Sangrouber D, Carosella ED, Moreau P (2007) Hypoxia Modulates HLA-G Gene Expression in Tumor Cells. Hum Immunol 68(4):277–285
Ristich V, Liang S, Zhang W, Wu J, Horuzsko A (2005) Tolerization of dendritic cells by HLA-G. Eur J Immunol 35(4):1133–1142
Valenti R, Huber V, Iero M, Filipazzi P, Parmiani G, Rivoltini L (2007) Tumor-released microvesicles as vehicles of immunosuppression. Cancer Res 67(7):2912–2915
Yu S, Liu C, Su K, Wang J, Liu Y, Zhang L, Li C, Cong Y, Kimberly R, Grizzle WE, Falkson C, Zhang HG (2007) Tumor exosomes inhibit differentiation of bone marrow dendritic cells. J Immunol 178(11):6867–6875
Marton A, Vizler C, Kusz E, Temesfoi V, Szathmary Z, Nagy K, Szegletes Z, Varo G, Siklos L, Katona RL, Tubak V, Howard OM, Duda E, Minarovits J, Nagy K, Buzas K (2012) Melanoma cell-derived exosomes alter macrophage and dendritic cell functions in vitro. Immunol Lett 148(1):34–38
Wang T, Niu G, Kortylewski M, Burdelya L, Shain K, Zhang S, Bhattacharya R, Gabrilovich D, Heller R, Coppola D, Dalton W, Jove R, Pardoll D, Yu H (2004) Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat Med 10(1):48–54
Park SJ, Nakagawa T, Kitamura H, Atsumi T, Kamon H, Sawa S, Kamimura D, Ueda N, Iwakura Y, Ishihara K, Murakami M, Hirano T (2004) IL-6 regulates in vivo dendritic cell differentiation through STAT3 activation. J Immunol 173(6):3844–3854
Larmonier N, Marron M, Zeng Y, Cantrell J, Romanoski A, Sepassi M, Thompson S, Chen X, Andreansky S, Katsanis E (2007) Tumor-derived CD4(+)CD25(+) regulatory T cell suppression of dendritic cell function involves TGF-beta and IL-10. Cancer Immunol Immunother 56(1):48–59
Jackson SH, Yu CR, Mahdi RM, Ebong S, Egwuagu CE (2004) Dendritic cell maturation requires STAT1 and is under feedback regulation by suppressors of cytokine signaling. J Immunol 172(4):2307–2315
Evel-Kabler K, Song XT, Aldrich M, Huang XF, Chen SY (2006) SOCS1 restricts dendritic cells’ ability to break self tolerance and induce antitumor immunity by regulating IL-12 production and signaling. J Clin Invest 116(1):90–100
Choi YE, Yu HN, Yoon CH, Bae YS (2009) Tumor-mediated down-regulation of MHC class II in DC development is attributable to the epigenetic control of the CIITA type I promoter. Eur J Immunol 39(3):858–868
Wang S, Yang J, Qian J, Wezeman M, Kwak LW, Yi Q (2006) Tumor evasion of the immune system: inhibiting p38 MAPK signaling restores the function of dendritic cells in multiple myeloma. Blood 107(6):2432–2439
Farren MR, Carlson LM, Lee KP (2010) Tumor-mediated inhibition of dendritic cell differentiation is mediated by down regulation of protein kinase C beta II expression. Immunol Res 46(1–3):165–176
Tourkova IL, Shurin GV, Wei S, Shurin MR (2007) Small Rho GTPases mediate tumor-induced inhibition of endocytic activity of dendritic cells. J. Immunol. 178(12):7787–7793
Pirtskhalaishvili G, Shurin GV, Esche C, Cai Q, Salup RR, Bykovskaia SN, Lotze MT, Shurin MR (2000) Cytokine-mediated protection of human dendritic cells from prostate cancer-induced apoptosis is regulated by the Bcl-2 family of proteins. Br J Cancer 83(4):506–513
Esche C, Shurin GV, Kirkwood JM, Wang GQ, Rabinowich H, Pirtskhalaishvili G, Shurin MR (2001) Tumor necrosis factor-alpha-promoted expression of Bcl-2 and inhibition of mitochondrial cytochrome c release mediate resistance of mature dendritic cells to melanoma-induced apoptosis. Clin Cancer Res 7(Suppl 3):974s–979s
Kanto T, Kalinski P, Hunter OC, Lotze MT, Amoscato AA (2001) Ceramide mediates tumor-induced dendritic cell apoptosis. J Immunol 167(7):3773–3784
Szatrowski TP, Nathan CF (1991) Production of large amounts of hydrogen peroxide by human tumor cells. Cancer Res 51(3):794–798
Lopez-Lazaro M (2007) Excessive superoxide anion generation plays a key role in carcinogenesis. Int J Cancer 120(6):1378–1380
Handley ME, Thakker M, Pollara G, Chain BM, Katz DR (2005) JNK activation limits dendritic cell maturation in response to reactive oxygen species by the induction of apoptosis. Free Radic Biol Med 38(12):1637–1652
Balkir L, Tourkova IL, Makarenkova VP, Shurin GV, Robbins PD, Yin XM, Chatta G, Shurin MR (2004) Comparative analysis of dendritic cells transduced with different anti-apoptotic molecules: sensitivity to tumor-induced apoptosis. J Gene Med 6(5):537–544
Vakkila J, Thomson AW, Vettenranta K, Sariola H, Saarinen-Pihkala UM (2004) Dendritic cell subsets in childhood and in children with cancer: relation to age and disease prognosis. Clin Exp Immunol 135(3):455–461
Sawant A, Ponnazhagan S (2013) Role of plasmacytoid dendritic cells in breast cancer bone dissemination. Oncoimmunology 2(2):e22983
Wei S, Kryczek I, Zou L, Daniel B, Cheng P, Mottram P, Curiel T, Lange A, Zou W (2005) Plasmacytoid dendritic cells induce CD8+ regulatory T cells in human ovarian carcinoma. Cancer Res 65(12):5020–5026
Munn DH, Mellor AL (2006) The tumor-draining lymph node as an immune-privileged site. Immunol Rev 213:146–158
Munn DH (2006) Indoleamine 2,3-dioxygenase, tumor-induced tolerance and counter-regulation. Curr Opin Immunol 18(2):220–225
Belladonna ML, Grohmann U, Guidetti P, Volpi C, Bianchi R, Fioretti MC, Schwarcz R, Fallarino F, Puccetti P (2006) Kynurenine pathway enzymes in dendritic cells initiate tolerogenesis in the absence of functional IDO. J Immunol 177(1):130–137
Lopez AS, Alegre E, LeMaoult J, Carosella E, Gonzalez A (2006) Regulatory role of tryptophan degradation pathway in HLA-G expression by human monocyte-derived dendritic cells. Mol Immunol 43(14):2151–2160
Lemaoult J, Caumartin J, Daouya M, Favier B, Rond SL, Gonzalez A, Carosella ED (2007) Immune regulation by pretenders: cell-to-cell transfers of HLA-G make effector T cells act as regulatory cells. Blood 109(5):2040–2048
Shurin MR, Naiditch H, Zhong H, Shurin GV (2011) Regulatory dendritic cells: new targets for cancer immunotherapy. Cancer Biol Ther 11(11):988–992
Ghiringhelli F, Puig PE, Roux S, Parcellier A, Schmitt E, Solary E, Kroemer G, Martin F, Chauffert B, Zitvogel L (2005) Tumor cells convert immature myeloid dendritic cells into TGF-beta-secreting cells inducing CD4+CD25+ regulatory T cell proliferation. J Exp Med 202(7):919–929
Ma Y, Shurin GV, Gutkin DW, Shurin MR (2012) Tumor associated regulatory dendritic cells. Semin Cancer Biol 22(4):298–306
Conejo-Garcia JR, Benencia F, Courreges MC, Kang E, Mohamed-Hadley A, Buckanovich RJ, Holtz DO, Jenkins A, Na H, Zhang L, Wagner DS, Katsaros D, Caroll R, Coukos G (2004) Tumor-infiltrating dendritic cell precursors recruited by a beta-defensin contribute to vasculogenesis under the influence of Vegf-A. Nat Med 10(9):950–958
Conejo-Garcia JR, Buckanovich RJ, Benencia F, Courreges MC, Rubin SC, Carroll RG, Coukos G (2005) Vascular leukocytes contribute to tumor vascularization. Blood 105(2):679–681
Curiel TJ, Cheng P, Mottram P, Alvarez X, Moons L, Evdemon-Hogan M, Wei S, Zou L, Kryczek I, Hoyle G, Lackner A, Carmeliet P, Zou W (2004) Dendritic cell subsets differentially regulate angiogenesis in human ovarian cancer. Cancer Res 64(16):5535–5538
Mihalyo MA, Hagymasi AT, Slaiby AM, Nevius EE, Adler AJ (2007) Dendritic cells program non-immunogenic prostate-specific T cell responses beginning at early stages of prostate tumorigenesis. Prostate 67(5):536–546
Sato K, Yamashita N, Baba M, Matsuyama T (2003) Regulatory dendritic cells protect mice from murine acute graft-versus-host disease and leukemia relapse. Immunity 18(3):367–379
Mellor AL, Baban B, Chandler PR, Manlapat A, Kahler DJ, Munn DH (2005) Cutting edge: CpG oligonucleotides induce splenic CD19+ dendritic cells to acquire potent indoleamine 2,3-dioxygenase-dependent T cell regulatory functions via IFN Type 1 signaling. J Immunol 175(9):5601–5605
Gabrilovich D (2004) Mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat Rev Immunol 4(12):941–952
Bell D, Chomarat P, Broyles D, Netto G, Harb GM, Lebecque S, Valladeau J, Davoust J, Palucka KA, Banchereau J (1999) In breast carcinoma tissue, immature dendritic cells reside within the tumor, whereas mature dendritic cells are located in peritumoral areas. J Exp Med 190(10):1417–1426
Thomachot MC, Bendriss-Vermare N, Massacrier C, Biota C, Treilleux I, Goddard S, Caux C, Bachelot T, Blay JY, Menetrier-Caux C (2004) Breast carcinoma cells promote the differentiation of CD34+ progenitors towards 2 different subpopulations of dendritic cells with CD1a(high)CD86(-)Langerin- and CD1a(+)CD86(+)Langerin+ phenotypes. Int J Cancer 110(5):710–720
Perrot I, Blanchard D, Freymond N, Isaac S, Guibert B, Pacheco Y, Lebecque S (2007) Dendritic cells infiltrating human non-small cell lung cancer are blocked at immature stage. J Immunol 178(5):2763–2769
Shurin GV, Ouellette CE, Shurin MR (2012) Regulatory dendritic cells in the tumor immunoenvironment. Cancer Immunol Immunother 61(2):223–230
Cox K, North M, Burke M, Singhal H, Renton S, Aqel N, Islam S, Knight SC (2005) Plasmacytoid dendritic cells (PDC) are the major DC subset innately producing cytokines in human lymph nodes. J Leukoc Biol 78(5):1142–1152
Kim R, Emi M, Tanabe K, Arihiro K (2007) Potential functional role of plasmacytoid dendritic cells in cancer immunity. Immunology 121(2):149–157
Tel J, Aarntzen EH, Baba T, Schreibelt G, Schulte BM, Benitez-Ribas D, Boerman OC, Croockewit S, Oyen WJ, van Rossum M, Winkels G, Coulie PG, Punt CJ, Figdor CG, de Vries IJ (2013) Natural human plasmacytoid dendritic cells induce antigen-specific T-cell responses in melanoma patients. Cancer Res 73(3):1063–1075
Houot R, Perrot I, Garcia E, Durand I, Lebecque S (2006) Human CD4+CD25high regulatory T cells modulate myeloid but not plasmacytoid dendritic cells activation. J Immunol 176(9):5293–5298
Ito T, Yang M, Wang YH, Lande R, Gregorio J, Perng OA, Qin XF, Liu YJ, Gilliet M (2007) Plasmacytoid dendritic cells prime IL-10-producing T regulatory cells by inducible costimulator ligand. J Exp Med 204(1):105–115
Ishida T, Oyama T, Carbone DP, Gabrilovich DI (1998) Defective function of Langerhans cells in tumor-bearing animals is the result of defective maturation from hemopoietic progenitors. J Immunol 161(9):4842–4851
Shurin GV, Aalamian M, Pirtskhalaishvili G, Bykovskaia S, Huland E, Huland H, Shurin MR (2001) Human prostate cancer blocks the generation of dendritic cells from CD34+ hematopoietic progenitors. Eur Urol 39 Suppl 4:37–40
Tourkova IL, Yamabe K, Foster B, Chatta G, Perez L, Shurin GV, Shurin MR (2004) Murine prostate cancer inhibits both in vivo and in vitro generation of dendritic cells from bone marrow precursors. Prostate 59(2):203–213
Melichar B, Savary C, Kudelka AP, Verschraegen C, Kavanagh JJ, Edwards CL, Platsoucas CD, Freedman RS (1998) Lineage-negative human leukocyte antigen-DR+ cells with the phenotype of undifferentiated dendritic cells in patients with carcinoma of the abdomen and pelvis. Clin Cancer Res 4(3):799–809
Schwaab T, Schned AR, Heaney JA, Cole BF, Atzpodien J, Wittke F, Ernstoff MS (1999) In vivo description of dendritic cells in human renal cell carcinoma. J Urol 162(2):567–573
Savary CA, Grazziutti ML, Melichar B, Przepiorka D, Freedman RS, Cowart RE, Cohen DM, Anaissie EJ, Woodside DG, McIntyre BW, Pierson DL, Pellis NR, Rex JH (1998) Multidimensional flow-cytometric analysis of dendritic cells in peripheral blood of normal donors and cancer patients. Cancer Immunol Immunother 45(5):234–240
Walker SR, Aboka A, Ogagan PD, Barksdale EM Jr (2005) Murine neuroblastoma attenuates dendritic cell cysteine cysteine receptor 7 (CCR7) expression. J Pediatr Surg 40(6):983–987
Pirtskhalaishvili G, Shurin GV, Gambotto A, Esche C, Wahl M, Yurkovetsky ZR, Robbins PD, Shurin MR (2000) Transduction of dendritic cells with Bcl-xL increases their resistance to prostate cancer-induced apoptosis and antitumor effect in mice. J Immunol 165(4):1956–1964
Weber F, Byrne SN, Le S, Brown DA, Breit SN, Scolyer RA, Halliday GM (2005) Transforming growth factor-beta1 immobilises dendritic cells within skin tumours and facilitates tumour escape from the immune system. Cancer Immunol Immunother 54(9):898–906
Walker SR, Ogagan PD, DeAlmeida D, Aboka AM, Barksdale EM, Jr. (2006) Neuroblastoma impairs chemokine-mediated dendritic cell migration in vitro. J Pediatr Surg 41(1):260–265
Whiteside TL, Stanson J, Shurin MR, Ferrone S (2004) Antigen-processing machinery in human dendritic cells: up-regulation by maturation and down-regulation by tumor cells. J Immunol 173(3):1526–1534
Nestle FO, Burg G, Fah J, Wrone-Smith T, Nickoloff BJ (1997) Human sunlight-induced basal-cell-carcinoma-associated dendritic cells are deficient in T cell co-stimulatory molecules and are impaired as antigen-presenting cells. Am J Pathol 150(2):641–651
Ferrari S, Malugani F, Rovati B, Porta C, Riccardi A, Danova M (2005) Flow cytometric analysis of circulating dendritic cell subsets and intracellular cytokine production in advanced breast cancer patients. Oncol Rep 14(1):113–120
Ochoa AC, Zea AH, Hernandez C, Rodriguez PC (2007) Arginase, prostaglandins, and myeloid-derived suppressor cells in renal cell carcinoma. Clin Cancer Res 13(2 Pt 2):721s–726s
Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J, Divino CM, Chen SH (2006) Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res 66(2):1123–1131
Frey AB (2006) Myeloid suppressor cells regulate the adaptive immune response to cancer. J Clin Invest 116(10):2587–2590
Starnes T, Rasila KK, Robertson MJ, Brahmi Z, Dahl R, Christopherson K, Hromas R (2006) The chemokine CXCL14 (BRAK) stimulates activated NK cell migration: implications for the downregulation of CXCL14 in malignancy. Exp Hematol 34(8):1101–1105
Menetrier-Caux C, Montmain G, Dieu MC, Bain C, Favrot MC, Caux C, Blay JY (1998) Inhibition of the differentiation of dendritic cells from CD34(+) progenitors by tumor cells: role of interleukin-6 and macrophage colony-stimulating factor. Blood 92(12):4778–4791
Huang B, Lei Z, Zhao J, Gong W, Liu J, Chen Z, Liu Y, Li D, Yuan Y, Zhang GM, Feng ZH (2007) CCL2/CCR2 pathway mediates recruitment of myeloid suppressor cells to cancers. Cancer Lett 252(1):86–92
Stanford A, Chen Y, Zhang XR, Hoffman R, Zamora R, Ford HR (2001) Nitric oxide mediates dendritic cell apoptosis by downregulating inhibitors of apoptosis proteins and upregulating effector caspase activity. Surgery 130(2):326–332
Nefedova Y, Huang M, Kusmartsev S, Bhattacharya R, Cheng P, Salup R, Jove R, Gabrilovich D (2004) Hyperactivation of STAT3 is involved in abnormal differentiation of dendritic cells in cancer. J Immunol 172(1):464–474
Nefedova Y, Gabrilovich DI (2007) Targeting of Jak/STAT pathway in antigen presenting cells in cancer. Curr Cancer Drug Targets 7(1):71–77
Gabrilovich DI, Cheng P, Fan Y, Yu B, Nikitina E, Sirotkin A, Shurin M, Oyama T, Adachi Y, Nadaf S, Carbone DP, Skoultchi AI (2002) H1(0) histone and differentiation of dendritic cells. A molecular target for tumor-derived factors. J Leukoc Biol 72(2):285–296
Capobianco A, Rovere-Querini P, Rugarli C, Manfredi AA (2006) Melanoma cells interfere with the interaction of dendritic cells with NK/LAK cells. Int J Cancer 119(12):2861–2869
Hou DY, Muller AJ, Sharma MD, DuHadaway J, Banerjee T, Johnson M, Mellor AL, Prendergast GC, Munn DH (2007) Inhibition of indoleamine 2,3-dioxygenase in dendritic cells by stereoisomers of 1-methyl-tryptophan correlates with antitumor responses. Cancer Res 67(2):792–801
Chang CC, Ciubotariu R, Manavalan JS, Yuan J, Colovai AI, Piazza F, Lederman S, Colonna M, Cortesini R, Dalla-Favera R, Suciu-Foca N (2002) Tolerization of dendritic cells by T(S) cells: the crucial role of inhibitory receptors ILT3 and ILT4. Nat Immunol 3(3):237–243
Curiel TJ, Wei S, Dong H, Alvarez X, Cheng P, Mottram P, Krzysiek R, Knutson KL, Daniel B, Zimmermann MC, David O, Burow M, Gordon A, Dhurandhar N, Myers L, Berggren R, Hemminki A, Alvarez RD, Emilie D, Curiel DT, Chen L, Zou W (2003) Blockade of B7-H1 improves myeloid dendritic cell-mediated antitumor immunity. Nat Med 9(5):562–567
Taieb J, Chaput N, Menard C, Apetoh L, Ullrich E, Bonmort M, Pequignot M, Casares N, Terme M, Flament C, Opolon P, Lecluse Y, Metivier D, Tomasello E, Vivier E, Ghiringhelli F, Martin F, Klatzmann D, Poynard T, Tursz T, Raposo G, Yagita H, Ryffel B, Kroemer G, Zitvogel L (2006) A novel dendritic cell subset involved in tumor immunosurveillance. Nat Med 12(2):214–219
Ghiringhelli F, Apetoh L, Housseau F, Kroemer G, Zitvogel L (2007) Links between innate and cognate tumor immunity. Curr Opin Immunol 19(2):224–231
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Shurin, M., Keskinov, A., Chatta, G. (2014). Immunobiology of Dendritic Cells in Cancer. In: Gabrilovich, D., Hurwitz, A. (eds) Tumor-Induced Immune Suppression. Springer, New York, NY. https://doi.org/10.1007/978-1-4899-8056-4_5
Download citation
DOI: https://doi.org/10.1007/978-1-4899-8056-4_5
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4899-8055-7
Online ISBN: 978-1-4899-8056-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)