Abstract
Stroke is a major leading cause of death and disability in the human population. The pathology of stroke-induced brain injury involves multifactorial pro-death processes. Among them, inflammation is an important contributor to stroke pathology as indicated by the close association between excessive inflammation and exacerbation of the disease process. Considerable experimental evidence indicates that disease outcome is modulated by several factors including predisposing clinical conditions. Stroke compromises vascular permeability and leads to breakdown of the blood–brain barrier. While the pathology primarily occurs in the CNS, the presence of peripheral immune cells in the infarcted area suggests their potential role in post-ischemic inflammation. Given recent advances highlighting the heterogeneity of peripheral immune cells and diversity of their function, we review neuroimmune interaction in the setting of acute cerebral ischemia, post-ischemic inflammation, and the trafficking of peripheral immune cells to inflamed tissue, with specific focus on the involvement of the class B scavenger receptor, CD36. We discuss CD36 expression and functions, the contribution of the receptor to stroke pathology, its relevance to peripheral inflammatory conditions, and potential strategies to target the CD36-associated neuroinflammatory pathway.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- CD36 Expression
- CD36 Ligand
- Foam Cell Formation
- Glial Fibrillary Acidic Protein Expression
- Experimental Autoimmune Encephalitis
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
CD36, A Multifunctional Scavenger Receptor
Overview
CD36 is an 88 kDa heavily N-linked glycosylated membrane protein [1, 2]. It has to date defied crystallization, so we can only imagine its structure based on protein prediction and modeling. Short intracellular tails extend from the two transmembrane domains (the N-terminal tail results from an uncleaved signal peptide), anchoring the protein, and exposing a large extracellular loop. There is a hydrophobic region that is predicted to dip back towards or into the plasma membrane, and disulfide bonds of the 6 extracellular cysteines constrain the molecule [3, 4]. Two cysteine residues that are palmitoylated characterize both cytoplasmic domains, and both cytoplasmic tails are necessary for efficient plasma membrane CD36 expression [5, 6]. Posttranslational disulfide bond formation, glycosylation, and palmitoylation are all essential in targeting CD36 to the plasma membrane, and the latter is also required for positioning CD36 in caveolae, detergent-resistant membranes, or lipid rafts [4, 7, 8]. In some cell types, expression of caveolin-1 has been shown to be mandatory for plasma membrane targeting of CD36, and disruption of caveolae may affect some CD36-dependent functions [9–11]. The partitioning of CD36 to specific plasma membrane domains may facilitate interaction with signaling partners and interacting proteins that are a requirement for CD36-dependent responses.
The human CD36 gene (including all variants) extends about 77 kb on chromosome 7q11.2 and encodes a predicted protein of 471 amino acids with a predicted molecular weight of 53 kDa (http://www.ncbi.nlm.nih.gov/gene/948). Human CD36 has ten potential N-linked glycosylation sites, and thus the actual molecular weight varies from ~80–100 kDa [12]. The thick complex carbohydrate coat of CD36 may protect it from proteolysis in harsh environments. Variant transcripts and a multitude of single nucleotide polymorphisms (SNP) mostly in noncoding regions have been identified [13, 14]. Mutations which result in absence of CD36 expression in platelets (Type II CD36 deficiency) or in monocytes and platelets (Type I CD36 deficiency) have been found at a frequency of 3–10 % in Asian and African populations and may persist as a result of selective pressure by the malaria parasite [15–18]. There is controversy as to whether absence of CD36 leads to or predisposes to human pathology, or is protective against malaria or other disease states. This may relate to whether CD36 is absent from all cells and tissues or some subset that differs depending upon the particular polymorphism, and the presence or absence of other interacting gene products.
Signaling
CD36 binding sites for oxidized low-density lipoprotein (oxLDL), growth hormone-releasing peptide and the family of thrombospondins (TSPs) are well defined, while the site for fatty acid binding is less precise and has not been tested definitively [19–22]. Other ligands have been assigned to the immunodominant domain (amino acids 155–183) by virtue of antibody blockade [23]. Alternatively, antibody binding to this domain may lead to a disruptive conformational change. There are two potential phosphorylation sites, both on the extracellular face of CD36, threonine 92 and serine 237. To date, phosphorylation at serine 237 has not been observed. However, there is data suggesting important biological consequences with regard to the phosphorylation of threonine 92. On platelets, phosphorylation reduces palmitate uptake and inhibits binding of TSP-1 and perhaps platelet activation [24–26]. Although it was long presumed that the “default” status in resting platelets was the phosphorylated state, recent evidence points towards a low basal level of CD36 phosphorylation in both platelets and microvascular endothelial cells [26]. Thus, why there is little platelet TSP binding and activation remains an open question. Threonine 92 is recognized by protein kinase C and to a lesser extent by protein kinase A [25, 27]. Recent work in transfected cell lines suggests that phosphorylation may also occur intracellularly as a posttranslational modification under certain conditions [26].
CD36 facilitates fatty acid uptake, but not by a classical transporter mechanism. Fatty acids probably bind transiently in the hydrophobic domain, and this facilitates flip-flop across the membrane, followed by rapid esterification or sequestration by cytoplasmic fatty acid binding proteins [28, 29]. Alternatively, CD36 may provide a hydrophobic pore facilitating fatty acid entry into membranes, analogous to the hypothesized mechanism by which scavenger receptor B1 facilitates cholesterol exchange between high-density lipoprotein and cells [30]. In contrast, uptake of oxLDL is via a caveolin-independent endocytic pathway and depends upon CD36-mediated lyn activation of the vav family of guanine exchange factors for Rho family GTPases, for subsequent vesicle maturation [31, 32]. There is recent evidence suggesting that CD36 is also expressed on mitochondrial membranes, but its function remains controversial [33–35]. While all groups consistently show that increased fatty acid oxidation is accompanied by increased CD36-mediated fatty acid uptake at the plasma membrane, there has been no definitive evidence that CD36 plays a direct role in fatty acid delivery into mitochondria.
Expression and Function in the CNS and Periphery
Initially characterized as a platelet receptor for thrombospondin-1 (TSP-1, designated glycoprotein IV), CD36 expression has subsequently been found on many types of cells and tissues [36]. CD36 is expressed on blood cells and cells of the vasculature, including platelets, reticulocytes, monocytes and macrophages, dendritic cells, endothelial cells, and smooth muscle cells [37–44]. It is found in specialized epithelium, including mammary epithelium, retinal pigment epithelium, apical enterocytes of the proximal small intestine, and the proximal tubule epithelium of the kidney [45–50]. CD36 is expressed in insulin-sensitive cells and tissues, including adipocytes, hepatocytes, cardiac and skeletal muscle, and pancreatic beta cell granules [28, 51]. CD36 is also found in taste receptors of the circumvallate papillae and steroidogenic cells of the adrenal, testes, and ovary [48, 52, 53].
The functions of CD36 are dictated by the cell type, circumstances, and ligand (Fig. 18.1). For example, CD36 plays a major role in fatty acid uptake required for production of energy or heat, especially in heart, skeletal muscle, and brown adipose tissue, and also in fat storage in white adipose tissue, and pathologically in liver and muscle [54–56]. CD36 also functions in recognition of malaria parasites and plays a role in fatty acid sensing by taste buds in the mouth and enterocytes in the gut [2, 48]. The uptake of oxLDLs by monocytes/macrophages leads to the formation of “foamy” macrophages and is a key step in atherosclerotic lesion development [57]. In binding the matricellular protein TSPs, CD36 may not only function as an adhesion receptor in platelets and between platelets, monocytes, tumor cells, sickled erythrocytes, and endothelium but can also modulate TGF-β activation, inhibit angiogenesis, and facilitate uptake of apoptotic cells [58]. CD36 is classified as a pattern recognition receptor (PRR) because it recognizes pathogen-associated molecular patterns or PAMPs, and danger or damage-associated molecular patterns also known as DAMPs [59]. These are repetitive motifs found on pathogens, modified phospholipids, or oxidatively denatured cytoplasmic or nuclear constituents that present as nonself [59, 60]. Some examples include oxidatively modified phospholipids in rod outer segments, apoptotic cells and low- and high-density lipoproteins, advanced glycation end products (AGEs), neurotoxic prion protein, amyloid-beta (Αβ), and diacylglycerides in the cell walls of Gram-positive bacteria. The diverse responses by interacting with specific CD36 ligands often converge into endothelial dysfunction and inflammation, common pathological features of cardio- and cerebrovascular diseases.
CD36 as a multifunctional receptor. By recognizing a host of ligands, CD36 elicits myriad responses and the interaction between specific ligands and the receptor results in diverse physiological and pathological responses. CD36 forms a complex with α6β1 integrin and CD47 to elicit downstream function and also acts as a coreceptor for TLRs. Many CD36-associated pathways converge in inflammatory responses. LCFAs long chain fatty acids, TSPs thrombospondins, EC endothelial cells, ox(m)LDLs oxidized (modified) low-density lipoprotein, AGEs advanced glycation end products, Glu-oxLDL Glucose-oxLDL, fAβ fibrillar β amyloid, TLR toll-like receptor
CNS
CD36 is expressed in neurons, microglia, astrocytes, and the endothelium of the blood–brain barrier. CD36 is expressed in neurons found in regions involved in pheromone responses and reproductive behavior. Specifically, it is found in neurons of the pyramidal layer of the ventral hippocampus, CA1 field, amygdalopiriform transition area, the perirhinal cortex, and the ectorhinal cortex [61, 62]. Neurons of the ventromedial hypothalamic nucleus utilize both glucose and fatty acids as signaling molecules and regulate energy homeostasis through central modulation of feeding behavior, hepatic glucose production, and hormone secretion. About 50 % of oleic acid sensing by ventromedial neurons of the hypothalamus was shown to be CD36 dependent, and this sensing was independent of fatty acid metabolism [62]. The mechanism of CD36-dependent fatty acid sensing is presumed to be analogous to that which has been defined in taste buds; CD36 binding by fatty acids is postulated to cause neurotransmitter release by activation of a protein tyrosine kinase and liberation of inositol 1,4,5-triphosphate, leading to calcium-dependent membrane depolarization [62, 63]. Dysfunctional central fatty acid sensing by CD36 may play a role in insulin-resistant states and obesity.
Long chain fatty acid uptake by endothelial cells at the blood–brain barrier is at least partially receptor dependent, and the role of CD36 in this process has been evaluated. The uptake of monounsaturated fatty acids is probably partially CD36 dependent; CD36 knockout mice have a significant decrease in this class of fatty acids, and in cultured blood–brain barrier-derived endothelium, knockdown of CD36 significantly decreased oleic acid uptake [64, 65]. Uptake of polyunsaturated fatty acids is most probably CD36 independent. The role of CD36 in fatty acid uptake within the brain remains to be elucidated.
CD36 expression was found in a subset of astrocytes in the post-ischemic brain [66]. The expression was temporally and spatially limited, only found 3 days following stroke and in the peri-infarct area, where the glial scar forms. Bao and colleagues subsequently showed a close relationship in the expression of CD36 and glial fibrillary acidic protein (GFAP). Inhibition of CD36 expression coincided with decreased GFAP expression and reduced glial scar, suggesting the involvement of CD36 in injury-induced scar formation [67].
CD36 has been found to play a role in microglial activation induced by amyloid-beta (Αβ) in Alzheimer’s disease (AD) plaque and by the neurotoxic prion protein, leading to secretion of pro-inflammatory cytokines [68–70]. There are apparently multiple mechanisms of microglial activation and downstream signaling that are CD36 dependent. Studies demonstrated that fibrillar amyloid beta (fΑβ) engaged microglia by a complex of receptors that included CD36, scavenger receptor A, α6β1 integrin, CD47, and toll-like receptors (TLRs) 2 and 4. These initiate a signaling cascade that includes p38, src kinase, vav proteins, rac, and reactive oxygen species, leading to cytokine release and phagocytosis [71, 72]. Stewart et al. suggested that TLR 4/6 and CD36 engaged Αβ and activated the inflammasome, leading to release of inflammatory cytokines. This group also found that CD36 was essential to a signaling cascade involving the src kinase fyn phosphorylating p130CAS, a focal adhesion scaffolding protein. This led to recruitment and phosphorylation of pyk2 and paxillin to the leading edge and membrane ruffles, resulting in an increase in microglia migration [73].
Periphery
CD36 is highly expressed in mononuclear phagocytes, including monocytes, macrophages, and dendritic cells [41, 74, 75]. Receptor-mediated uptake of oxidatively modified LDL (mLDL)/oxLDL by monocytes increases transcription of CD36 and several other genes via activation of the nuclear hormone receptor peroxisome proliferator-activated receptor-γ (PPAR-γ) [76]. The activating ligand(s) for PPAR-γ are oxidized phospholipids, such as 9- and 13-HODE, and these and/or their precursor lipids are delivered to the cell within the oxLDL particle. Similarly, in heart and liver, uptake of ligands for other members of the PPAR family is mediated by CD36 and may contribute to cardiac lipotoxicity and hepatic steatosis [77, 78].
On platelets, CD36 expression is variable in the population, and this is attributed to genetic polymorphisms but potentially may also relate to physiological status [79]. For example, both a high-fat diet and insulin resistance upregulate CD36 on monocytes/macrophages and could have similar effect on megakaryocytes. Humans and mice deficient in platelet CD36 do not have a significant phenotype. However, recent studies using pathophysiological ligands of CD36, specifically, oxidized phospholipids and AGEs, support the hypothesis that platelet CD36 can modulate platelet reactivity by transducing prothrombotic signals [80–82]. The pathway in platelets follows a recurring theme: src kinase activation (in platelets, fyn and lyn are associated with CD36 following oxLDL binding), phosphorylation of vav family proteins, and Map kinase (in this case, MKK4), resulting ultimately in jnk activation. AGEs also trigger platelet CD36-mediated jnk2 activation [83]. In addition to activation of signaling pathways, CD36 may foster exchange of phospholipids and fatty acids between platelets and lipoproteins. The type of fatty acid/phospholipid, and whether it is oxidized, may alter platelet membranes rendering them more susceptible to aggregation [84]. Platelet CD36 has been shown to interact with amyloids, and platelet aggregation was mediated by a p38 MAP kinase and thromboxane A2-dependent pathway [85]. Thus, increased platelet expression and sensitization by pathophysiological CD36 ligands may explain platelet hyperreactivity in inflammation and hyperlipidemic and insulin-resistant states, among others, and lead to appreciable thrombosis in response to subthreshold platelet stimulating agents. In contrast to CD36-expressing mice, CD36 KO mice do not show platelet hyperreactivity in response to high-fat diet feeding or insulin-resistant states and have normal thrombosis profiles in experimental in vivo models that enrich for CD36 ligands [80, 84].
Neuroinflammation
Post-ischemic Inflammation
Post-ischemic inflammation is a contributing negative factor in stroke, exacerbating injury, and influencing outcome [86, 87]. Stroke increases inflammatory mediator: free radical, cytokines/chemokines (IL-1β, TNF-α, IL-6, monocyte chemotactic protein (MCP)-1, macrophage inflammatory protein-1, C-C chemokine receptor type 2 (CCR2), and inflammatory proteins (inducible nitric oxide synthase, cyclooxygenase, and matrix metalloproteinases). These mediators increase endothelial expression of adhesion molecules, such as intercellular adhesion molecule (ICAM)-1 and p-selectin, leading to leukocyte arrest and transendothelial migration at the injured site. These mediators either act in concert or converge into an inflammatory response following activation of temporally and spatially separated cascades [88, 89]. A number of studies have shown that targeting specific neuroinflammatory mediators attenuates stroke-induced brain injury [90–92]. Mice deficient in ICAM-1 or p-selectin displayed smaller infarct size compared to wild type [93–95]. Deficiency in either MCP-1 or its cognate receptor, CCR2, also resulted in protection in murine stroke models [96, 97], while increased expression of MCP-1 exacerbated ischemic outcome with enhanced recruitment of inflammatory cells at the injury sites [98]. Despite apparent benefits of attenuating neuroinflammatory pathways in animal models, clinical trials in human stroke, using antibodies against adhesion molecules and neutrophils, were not effective [99, 100].
Recent studies recognized the complexity in targeting neuroinflammatory pathways. Stroke-induced inflammation is a double-edged sword in that it not only is necessary for containment and repair but can also lead to further damage. It has become increasingly clear that peripheral inflammatory status and comorbidities are important factors in neuroinflammation and outcome [101–103]. Understanding the temporal sequence of activation of inflammatory mediators, spatial localization of the cascade in the infarct (core vs. penumbra), and identification of cell types activated remain to be investigated to selectively reduce adverse while preserving beneficial aspects of the inflammatory response.
CD36: A Modulator of the Innate Immune System
In response to an encounter with microbes, the host elicits a rapid, specific, and self-constraining acute inflammatory response to avoid inflammatory-mediated damage to neighboring tissues [104]. This primordial defense response involves initial recognition of the triggers, so-called PAMPs, through pattern recognition receptors (PRRs), which include scavenger receptors. Subsequently, triggered adaptive immune responses lead to resolution to reinstate tissue homeostasis in a timely manner. Sterile inflammation occurs in post-ischemic tissues in the absence of microbes [105, 106]. The triggers of sterile inflammation are elements of damaged tissue, including oxidized lipids and cytoplasmic proteins, DNA, RNA, and proteolyzed or oxidized extracellular matrix components, which, as previously noted, are collectively termed DAMPs. Regardless of microbial or endogenous in nature, PRRs are believed to be involved in recognizing the triggers and eliciting inflammatory responses (Fig. 18.2).
Innate immune receptors resolve pathogen-induced and sterile inflammation. Convergence of innate immune system to resolve inflammation. PAMPs or DAMPs are recognized through pattern recognition receptors such as TLRs and/or CD36. The interactions elicit appropriate downstream responses to resolve infection and repair tissue damage. TLRs toll-like receptors, PAMPs pathogen-associated molecular patterns, DAMPs damage-associated molecular patterns
In addition to playing a role in the endocytic uptake of modified lipoproteins leading to foam cell formation, an important role for monocyte/macrophage CD36 as a PRR in innate immune modulation has emerged [107–109]. This is both in association with and independent of TLRs. CD36 recognizes nonself, and this is one of the oldest and most conserved functions of these receptors, beginning with recognition of apoptotic cells during development as a result of normal homeostasis [110]. The recognition motif, altered fatty acid chains that become hydrophilic and more easily accessed by surface receptors, accounts for the crossover recognition to modified lipoproteins carrying these ligands as a result of oxidative stress [111, 112]. CD36 interaction with apoptotic cells invokes an anti-inflammatory response, consistent with the maintenance of organism status quo. This response involves p38 activation and transcriptional upregulation of the IL-10 promoter by Pbx-1 and Prep-1 [113, 114]. On dendritic cells, CD36 mediates uptake of apoptotic cells and is also involved in cross-presentation of antigens to cytotoxic T cells [41].
In recognition of lipids in cell walls of bacteria, CD36 may play a role not only in endocytosis/phagocytosis but also in TLR signaling responses [73, 115–121] (Fig. 18.1). This is an emerging topic in CD36 biology, and the exact mechanism is still under investigation. One hypothesis is that CD36 acts an accessory protein for TLRs to deliver ligands, while alternative data suggest that CD36 enhances downstream signaling. This may prove to be important in many of the inflammatory responses mediated by CD36, as there is significant crossover between TLR and CD36 ligands, including fatty acids, amyloid-beta, modified LDL, and other PAMPs/DAMPs. Specifically, CD36 acts as a co-receptor for the recognition of bacteria-derived diacylglyceride through a TLR2/6 complex [115, 122]. OxLDL and Αβ trigger inflammatory signaling through TLR4/6 [73]. Abe and colleagues found that CD36 plays a key role in the inflammatory response and tissue damage mediated by TLR2/1, but not TLR2/6, as a result of cerebral ischemia [121]. This differed from the accessory role of CD36 in monocyte/macrophages in the periphery, where it was instead important for TLR 2/6 signaling. The explanation for this difference was not explored and may relate to differences in expression of other TLR accessory proteins. Nonetheless, these studies suggest that CD36 is involved in the pathogenesis of sterile inflammation through convergence with TLR signaling.
CD36: An Inflammatory Receptor
The pro-inflammatory nature of CD36 has been implicated in atherosclerosis, vascular dysfunction, and neurodegenerative diseases [57, 69, 123, 124]. The concept that CD36 is a prototypic inflammatory receptor and contributes to stroke pathology also has been recognized [66, 102, 125]. In addition to elevated CD36 expression, CD36 ligands such as fAβ, mLDL, oxLDL, and TSPs are elevated in the post-ischemic brain [126–130]. Several studies showed CD36 activation is associated with elevated levels of free radicals, IL-1β, TNF-α, IL-6, MCP-1, and CCR2 [66, 102, 131]. CD36 expression was found predominantly on CD11b+ cells within the infarct territory and the presence of the cells occurs throughout the course of infarct development. However, the identity of the CD11b+ cells as to resident microglia versus infiltrated peripheral mononuclear phagocytes has not been explored. Although CD36 expression was not detected in neurons in the injured tissue, the expression was shown in the subsets of GFAP-expressing astrocytes in the glial scar area [66]. As inflammation is essential in glial scar formation [132], the involvement of CD36 in injury-induced scar formation was addressed. The study by Bao and colleagues reported that CD36 expression covaries with GFAP, an intermediated filament in astrocytes [67]. This study identified CD36 as a novel mediator of GFAP expression and glial scar formation and suggested that targeting CD36 may decrease the barring effect of scar tissue to promote regeneration.
Comorbidities Influencing CD36 Expression/Function
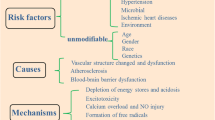
Cardiovascular and cerebrovascular diseases share many prevalent risk factors. These comorbid conditions include hyperlipidemia, insulin resistance associated with metabolic syndrome, obesity and diabetes, impaired vascular function, and hypertension. CD36 expression has been shown to be modulated in comorbid conditions. Since comorbidities increase CD36 expression and a specific set of ligands in a feed-forward manner [76], excessive ligands/receptor interaction associated with risk factors presumably intensify CD36 pathways in disease conditions (Fig. 18.3).
CD36 exacerbates stroke-induced injury as a result of disease modifying risk factors. Following stroke, interaction of CD36 with its ligand occurs in a feed-forward manner. Vascular risk factors and cormodities such as hyperlipidemia, insulin resistance, vascular dysfunction, and hypertension enhance the generation of CD36 ligands and intensify CD36 pathways
Dyslipidemia
Podrez and colleagues reported increased lipid-based CD36 ligands in ApoE KO mice fed a high-fat diet [80]. They demonstrated a profound upregulation of structurally defined, oxidized choline glycerophospholipid species ( ox PC CD36 ), that serve as high-affinity ligands for CD36 in lipoproteins in the plasma of hyperlipidemic mice and also in humans with low HDL levels [133]. The abundance of ox PC CD36 in hyperlipidemia led to vascular foam cell formation, a key event in atherosclerotic lesion development [57]. Compared to controls, hyperlipidemic mice subjected to cerebral ischemia displayed larger infarcts and heightened post-ischemic inflammation [131]. An underlying hypothesis is that “priming” of peripheral mononuclear phagocytes by CD36/ligands prior to stroke might account for the exacerbation. This study also showed higher expression of CD36 in peripheral monocytes/macrophages in hyperlipidemic mice prior to ischemia. Following stroke, the mice displayed elevated CD36 expression, foam cell area, and pro-inflammatory cytokines/chemokines (MCP-1, CCR2, IL-1β, TNFα, and IL-6) in the post-ischemic brain. The absence of CD36 reversed the hyperlipidemia-associated phenotype [131]. Clear indication from the study is CD36’s involvement in hyperlipidemia-induced exacerbation of ischemic inflammation and injury and notably, its peripheral influence on CNS injury development. The possibility that CD36-dependent stroke outcomes could be influenced by the presence of other comorbidities at the time of stroke and also involvement of other factors such as developmental stage (neonatal vs. adult) remain to be investigated.
Insulin Resistance
Insulin resistance associated with diabetes is a predisposing risk factor for stroke as indicated by the fact that 70 % of new stroke victims were previously diagnosed with diabetes, occult diabetes, or were prediabetic with impaired insulin sensitivity [134, 135]. Diabetic conditions promote a chronic pro-inflammatory state that increases the burden of CD36 ligands via modifications of LDL and AGEs, and augments CD36 expression and function. Increased expression of CD36 in monocytes/macrophages not only influences the peripheral inflammatory state but also impacts at the localized site of cerebral ischemic injury.
A potential link between CD36 and impaired insulin sensitivity has been found experimentally in mice. However, in humans, studies are less equivocal, and this may depend upon which cells/tissues are affected by the specific CD36 mutation/SNP, whether the mutation/SNP leads to reduced or increased CD36 expression, and expression of other gene products [136–138]. CD36 KO mice not only have overall increased insulin sensitivity, as a result of increased glucose uptake in muscle, but also show liver-specific insulin resistance as a result of reduced capacity to utilize fatty acids in heart [139, 140]. Further studies showed that CD36 is linked to inflammation in insulin resistance and defective insulin signaling [55, 141]. The increased burden of CD36 ligands in the diabetic state was shown to promote a pro-inflammatory state and a CD36-dependent paracrine loop between adipocytes and macrophages that facilitated chronic inflammation and contributed to insulin resistance common in obesity and dyslipidemia [142].
Moreover, there is abundant evidence that glucose/diabetes modulates CD36 expression and thus impacts CD36 downstream effects. For example, glucose administration upregulates CD36 expression on macrophages [143] and in proximal renal tubular epithelia in humans [144]. Other studies have shown that CD36 expression is increased in monocytes from type 2 diabetic patients [145] and in diabetic mouse hearts [146]. In diabetes associated with atherosclerosis, increased plasma MCP-1 levels were associated with increased monocyte CCR2, CD68, and CD36 and increased vessel wall monocyte number [147]. Liang and colleagues reported increased CD36 protein in macrophages as a response to defective insulin signaling [148]. Glucose was also shown to promote LDL oxidation, and the resulting glucose-oxLDLs stimulated macrophage proliferation in a manner that was dependent on CD36 [149]. Human THP-1 macrophages that were exposed to glucoxidized LDLs increased both CD36 gene expression and accumulation of cholesterol ester (an indicator of foam cell formation) to extents greater than those produced by glycated LDLs or oxLDLs [150].
CD36 has been shown to be localized on insulin-containing granules in human pancreatic beta cells and mediates fatty acid effects on insulin secretion [151]. Handberg and colleagues identified CD36 in plasma (later shown to be contained within microparticles) as a novel marker of insulin resistance [152, 153]. Thus, multiple lines of evidence show that CD36 is modulated by insulin and glucose pathways, and that CD36 has effects on these pathways.
Vascular Dysfunction
The deposition of Aβ in the microvasculature, a hallmark of AD, contributes to oxidative stress and compromises blood–brain barrier (BBB) integrity [154, 155]. Due to the nature of the ligand, many studies on CD36 relevant to fAβ were focused on innate host response and inflammation associated with AD [69, 71, 156]. A key role for CD36 was reported, as CD36 deficiency attenuated fAβ-induced secretion of cytokines, chemokines, and reactive oxygen species in microglia. Macrophage or microglia recruitment into the peritoneum or brain, respectively, in response to injection of fAβ was attenuated in CD36 KO mice [69]. A multi-receptor complex comprised of CD36/α6β1-integrin/CD47 stimulates intracellular tyrosine kinase-based signaling cascades and cellular activation, as detailed previously, which leads to the secretion of pro-inflammatory molecules [72]. In animal models of AD, interaction of Aβ with CD36 causes cerebrovascular oxidative stress and neurovascular dysfunction. The dysfunction was abrogated in the absence of CD36, suggesting that a strategy of CD36 inhibition to normalize cerebrovascular dysfunction might be effective [123, 157]. Lee and colleagues showed an increased level of circulating Aβ in patients with acute ischemic stroke and suggested that the ligand is derived from brain as a consequence of ischemic insult [158].
Hypertension
Hypertension is a major risk factor for stroke [159, 160]. Clinical trials employing antihypertensive agents that aim at reducing blood pressure have been effective in management and prevention [161, 162]. CD36 has been implicated in blood pressure control and modulated by hypertension. Pravenec and colleagues showed that CD36 mutation in the kidney can increase blood pressure and identified renal CD36 as a genetical determinant of blood pressure and risk factor for hypertension [163]. In the stroke prone spontaneous hypertensive rat, BBB impairment was associated with increased CD36 expression in the vessel [164]. Similar to what occurs in diabetics, macrophages from hypertensive subjects show significant increase in CD36 expression, and this was associated with enhanced adhesion to endothelial cells and greater production of ROS [165]. Circulating human endothelial cells also show increased CD36 expression in pulmonary hypertensive states [166]. In hypoxia-induced pulmonary hypertension, CD36 expression increases on intrapulmonary arteries [167]. Human gene association studies have been equivocal with respect to CD36, probably for similar reasons described above in the case of insulin resistance/diabetes.
Neuroimmune Interaction
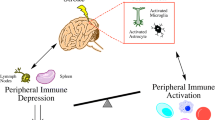
The presence of granulocytes (neutrophils), subsets of T cells, and monocytes/macrophages in the post-stroke brain suggests mobilization of peripheral immune cells to the injured tissue [88, 168]. There has been controversy regarding the order and timing among the types of immune cells for trafficking. An early study reported that neutrophil infiltration occurs prior to macrophages/activated microglia following stroke [169], while others showed that the accumulation of microglia and/or macrophages in the infarct territory precedes neutrophils [170]. Despite disagreement regarding the order of infiltrating cell types, it is believed that the accumulation of peripheral immune cells contributes to injury development during the acute phase of stroke.
MCP-1/CCR2 Axis for Monocyte Trafficking
Experimental autoimmune encephalitis (EAE) in mice is an example of how inflammatory cells impact disease in the CNS and demonstrates the importance of the MCP-1/CCR2 axis in monocyte recruitment. Among the types of infiltrating cells, monocytes was most tightly coupled to neurobehavioral severity in EAE [171]. Specific inhibition of monocyte recruitment reduced EAE lesion progression, while the presence of T cells was independent of disease severity, strongly implicating infiltrating monocytes in EAE pathogenesis, and confirming an earlier finding [172]. Through serial experimental manipulation using parabiosis (suturing a pair of mice to share circulation) and irradiation/bone marrow transplant of stem cells from genetically engineered mice, CCR2, a G-protein-linked membrane receptor, was found to be the essential mediator of monocyte trafficking, as the study showed the absence of monocyte CCR2 profoundly attenuated paralytic progression of the disease [171].
Monocytes exhibit distinct subsets that are reminiscent of macrophage phenotypes [173–175]. The subset that expresses a high level of the hematopoietic cell differentiation antigen Ly-6C (Ly-6Chi) also expresses CCR2. Ly-6Chi (CCR2+) monocytes are specifically recruited to an injury site and become classically activated M1 macrophages. This CCR2+ subset is chemotactic to MCP-1, which is produced in the inflamed tissue. Recruitment of this subset to inflammatory sites is believed to be CCR2 dependent since monocytes from CCR2-deficient mice do not traffic as efficiently into areas of inflammation [176, 177]. The Ly-6Clow monocyte subset expresses CX3CR1, a receptor for CX3CL1 (fractalkine), but is devoid of CCR2 expression. This anti-inflammatory Ly-6Clow (CCR2-/CX3CR1+) subset is recruited to normal tissues and develops into resident M2 macrophages that function in host defense and repair after injury [174].
Secreted by microvascular endothelial cells, monocytes/macrophages, and astrocytes upon injury [98, 178–180], MCP-1 is a member of the CC chemokine family and functions in the trafficking of CCR2-expressing monocytes into an injury site. Previous work has established the importance of the MCP-1/CCR2 axis in monocyte/macrophage trafficking in cerebral ischemia. Stroke increases MCP-1 expression in the affected hemisphere. The overexpression of MCP-1 increases infarct volume and enhances the recruitment of monocytes to the injury site [98]. The absence of CCR2 or MCP-1 reduces infarct size [96, 97]. In the absence of CD36, CD36 ligands and injury-induced CC and CXC chemokine production were attenuated [102, 131]. In other disease models that involve recruitment of classically activated M1 macrophages, such as obesity and atherosclerosis, the absence of CD36 is associated with decreased monocyte/macrophage migration/infiltration and reduced overall numbers of macrophages [141, 181–183], suggesting MCP-1/CCR2 as a major chemokine/ receptor axis for immune cell trafficking.
CD36 in Monocyte/Macrophage Trafficking
Studies indicate the involvement of CD36 in cell mortality and mobility, an important function in cell trafficking. CD36 has been shown to signal through the P130Cas complex to the actin cytoskeleton and regulate microglial migration [184]. Harb and colleagues addressed the role of CD36 in regulating mononuclear phagocyte trafficking to pro-inflammatory atherosclerotic lesions. This study showed that inhibition of CD36 attenuated macrophage accumulation in atherosclerotic lesions, and this was associated with reduced expression of MCP-1 [182]. Cell polarization is essential for migration and mobility of leukocytes. Thus, studies by Park et al. showing that oxLDL/CD36 interaction induced loss of cell polarity and reduced macrophage migration through a vav-Rac-myosin II pathway provide a mechanistic framework to consider CD36 actions [185]. This work explains why macrophages become trapped in areas rich in CD36 ligands and promote further inflammation.
CD36 ligands are elevated in hyperlipidemic conditions and in injured tissues where oxidative or damaged products from cells are released [80, 107]. Through the uptake of lipid-based ligands and foam cell formation in hyperlipidemic conditions, monocyte/macrophage CD36 has been shown to play a role in atherosclerosis and stroke pathology [102, 131, 182, 186]. In a recent study, Kim and colleagues showed that infiltrating immune cells from the periphery are the major source of CD36 in the post-ischemic brain and contribute to stroke-induced brain injury in a hyperlipidemic condition. Mice receiving CD36-deficient bone marrow showed attenuated infarct volume and MCP-1 and CCR2 expression in the brain. The reverse transplantation study (transplantation of CD36-expressing bone marrow-derived cells to CD36 KO mice) showed no increase in infarct volume. The study suggested that CD36 in both host and periphery is required for peripheral CD36 to exert its effect on the hyperlipidemia-induced exacerbation in stroke injury. The underlying mechanism of the exacerbation presumably is that CD36 regulates immune cell trafficking via modulation of the expression of MCP-1 and CCR2 [102].
Targeting CD36 to Attenuate Inflammation
In light of the receptor’s pro-inflammatory properties, downregulation of CD36 has been suggested as a strategy to reduce inflammation-associated cerebro- and cardiovascular diseases including altherosclerosis and stroke. Several pharmacological agents were identified to attenuate CD36 expression and function. The antioxidant, α-tocopherol, reduces expression of CD36 and the uptake of oxLDL into macrophages [187–190]. Statins downregulate CD36 expression and suppress oxLDL uptake [187, 191, 192] and subsequently prevent oxLDL-induced macrophage foam cell formation [193]. Hexarelin is a member of the hexapeptide growth hormone-releasing peptide family and binds to CD36 and inhibits its expression [20]. Treatment of mice with hexarelin or a structurally related analogue, EP80317, resulted in a marked decrease in atherosclerotic lesions [194]. Using a high-throughput screening approach for CD36 antagonists based on competition in an oxLDL-binding assay, salvianolic acid B (SAB) was identified as a CD36 inhibitor [195]. SAB is a water-soluble polyphenolic antioxidant isolated from Danshen, a Chinese herb that has been used for the prevention and treatment of atherosclerosis and stroke in Asian countries. The specificity and efficacy of SAB in the inhibition of CD36-mediated lipid uptake were confirmed by binding studies for the physical interaction of SAB with CD36. SAB reduces oxLDL-induced CD36 gene expression in cultured cell lines and primary macrophages. Moreover, SAB reduces CD36 gene expression and lipid uptake into macrophages in hyperlipidemic ApoE KO mice [196].
Due to the issues regarding developmental compensatory changes with germ line deletions, investigation on the efficacy of CD36 inhibitors has been complemented by genetic approaches. Besides finding from CD36 KO mice that displayed attenuated stroke-induced inflammation and brain injury [66, 125], effects of a new class of antioxidants peptide, SS31, has been tested against cerebral ischemia [197]. Mice treated with SS31 peptides had attenuated ischemia-induced glutathione (GSH) depletion in the cortex and showed smaller infarct size. The absence of stroke-induced glutathione depletion and no effect on infarct volume in CD36 KO mice treated with the peptide suggested that the protection occurred through the downregulation of CD36 pathways. Because CD36 is a multi-ligand and multifunctional receptor and its expression occurs in a positive feed-forward manner that promotes its functions, targeting at the level of the receptor by interrupting the feed-forward loop to downregulate the CD36 pathway (a multimodal approach) has been suggested [125].
Conclusion
CD36 is an inflammatory receptor that is at the junction of cardio and cerebral vascular disease. Defining the role of CD36 in the CNS and periphery through neuroimmune interactions has been an important emerging area in understanding the pathophysiology of brain injury as a result of cerebral ischemia. CD36 expression is altered in peripheral inflammatory conditions, including obesity, insulin resistance, hyperlipidemia, and hypertension, which also increase stroke incidence either singly or through clustering of these risk factors. Accumulating evidence suggests that CD36 regulates injury-induced mobilization of peripheral immune cells and influences the outcome of stroke. Thus, stroke-induced injury is viewed as the summation of intrinsic ischemic insult and peripheral influences through the neuroimmune interaction. As less favorable outcomes are predicted in patients with various risk factors, targeting CD36-associated pathways may modulate the neuroinflammatory responses in comorbid conditions and serve as a potential approach to limit secondary expansion of primary injury in the setting of acute ischemia.
Abbreviations
- AGE:
-
Advanced glycation end product
- BBB:
-
Blood–brain barrier
- CCR2:
-
c–c chemokine receptor type 2
- DAMP:
-
Damage-associated molecular pattern
- EAE:
-
Experimental autoimmune encephalitis
- fΑβ:
-
Fibrillar beta-amyloid
- ICAM:
-
Intercellular adhesion molecule
- MCP-1:
-
Monocyte chemotactic protein-1
- mLDL:
-
Modified low-density lipoprotein
- oxLDL:
-
Oxidized low-density lipoprotein
- ox PC CD36 :
-
Oxidized choline glycerophospholipid species
- PAMP:
-
Pathogen-associated molecular pattern
- PPAR-γ:
-
Peroxisome proliferator-activated receptor-γ
- PRR:
-
Pattern recognition receptor
- TLR:
-
Toll-like receptors
- TSPs:
-
Thrombospondins
- SAB:
-
Salvianolic acid B
References
Gruarin P, De Monte L, Alessio M (2000) CD36 folding revealed by conformational epitope expression is essential for cytoadherence of Plasmodium falciparum-infected red blood cells. Parasite Immunol 22(7):349–60
Oquendo P, Hundt E, Lawler J, Seed B (1989) CD36 directly mediates cytoadherence of Plasmodium falciparum parasitized erythrocytes. Cell 58(1):95–101
Rasmussen JT, Berglund L, Rasmussen MS, Petersen TE (1998) Assignment of disulfide bridges in bovine CD36. Eur J Biochem 257(2):488–94
Gruarin P, Sitia R, Alessio M (1997) Formation of one or more intrachain disulphide bonds is required for the intracellular processing and transport of CD36. Biochem J 328(Pt 2):635–42
Jochen A, Hays J (1993) Purification of the major substrate for palmitoylation in rat adipocytes: N-terminal homology with CD36 and evidence for cell surface acylation. J Lipid Res 34(10):1783–92
Tao N, Wagner SJ, Lublin DM (1996) CD36 is palmitoylated on both N- and C-terminal cytoplasmic tails. J Biol Chem 271(37):22315–20
Eyre NS, Cleland LG, Tandon NN, Mayrhofer G (2007) Involvement of the C-terminal cytoplasmic domain in the plasma membrane localization of FAT/CD36 and its ability to mediate long-chain fatty acid uptake. J Lipid Res 48:528–42
Eyre NS, Cleland LG, Tandon NN, Mayrhofer G (2007) Importance of the carboxyl terminus of FAT/CD36 for plasma membrane localization and function in long-chain fatty acid uptake. J Lipid Res 48(3):528–42
Ring A, Le Lay S, Pohl J, Verkade P, Stremmel W (2006) Caveolin-1 is required for fatty acid translocase (FAT/CD36) localization and function at the plasma membrane of mouse embryonic fibroblasts. Biochim Biophys Acta 1761(4):416–23
Pohl J, Ring A, Ehehalt R, Schulze-Bergkamen H, Schad A, Verkade P et al (2004) Long-chain fatty acid uptake into adipocytes depends on lipid raft function. Biochemistry 43(14):4179–87
Pohl J, Ring A, Korkmaz U, Ehehalt R, Stremmel W (2005) FAT/CD36-mediated long-chain fatty acid uptake in adipocytes requires plasma membrane rafts. Mol Biol Cell 16(1):24–31
Hoosdally SJ, Andress EJ, Wooding C, Martin CA, Linton KJ (2009) The Human Scavenger Receptor CD36: glycosylation status and its role in trafficking and function. J Biol Chem 284(24): 16277–88
Wyler B, Daviet L, Bortkiewicz H, Bordet JC, McGregor JL (1993) Cloning of the cDNA encoding human platelet CD36: comparison to PCR amplified fragments of monocyte, endothelial and HEL cells. Thromb Haemost 70(3):500–5
Taylor KT, Tang Y, Sobieski DA, Lipsky RH (1993) Characterization of two alternatively spliced 5′-untranslated exons of the human CD36 gene in different cell types. Gene 133(2):205–12
Yamamoto N, Akamatsu N, Sakuraba H, Yamazaki H, Tanoue K (1994) Platelet glycoprotein IV (CD36) deficiency is associated with the absence (type I) or the presence (type II) of glycoprotein IV on monocytes. Blood 83(2):392–7
Lee K, Godeau B, Fromont P, Plonquet A, Debili N, Bachir D et al (1999) CD36 deficiency is frequent and can cause platelet immunization in Africans. Transfusion 39(8):873–9
Aitman TJ, Cooper LD, Norsworthy PJ, Wahid FN, Gray JK, Curtis BR et al (2000) Malaria susceptibility and CD36 mutation. Nature 405(6790):1015–6
Serghides L, Smith TG, Patel SN, Kain KC (2003) CD36 and malaria: friends or foes? Trends Parasitol 19(10):461–9
Pearce SF, Roy P, Nicholson AC, Hajjar DP, Febbraio M, Silverstein RL (1998) Recombinant glutathione S-transferase/CD36 fusion proteins define an oxidized low density lipoprotein-binding domain. J Biol Chem 273(52):34875–81
Demers A, McNicoll N, Febbraio M, Servant M, Marleau S, Silverstein R et al (2004) Identification of the growth hormone-releasing peptide binding site in CD36: a photoaffinity cross-linking study. Biochem J 382(Pt 2):417–24
Pearce SF, Wu J, Silverstein RL (1995) Recombinant GST/CD36 fusion proteins define a thrombospondin binding domain. Evidence for a single calcium-dependent binding site on CD36. J Biol Chem 270(7):2981–6
Baillie AGS, Coburn CT, Abumrad NA (1996) Reversible binding of long-chain fatty acids to purified FAT, the adipose CD36 homolog. J Membr Biol 153(1):75–81
Navazo MD, Daviet L, Savill J, Ren Y, Leung LL, McGregor JL (1996) Identification of a domain (155–183) on CD36 implicated in the phagocytosis of apoptotic neutrophils. J Biol Chem 271(26):15381–5
Guthmann F, Maehl P, Preiss J, Kolleck I, Rustow B (2002) Ectoprotein kinase-mediated phosphorylation of FAT/CD36 regulates palmitate uptake by human platelets. Cell Mol Life Sci 59(11):1999–2003
Asch AS, Liu I, Briccetti FM, Barnwell JW, Kwakye-Berko F, Dokun A et al (1993) Analysis of CD36 binding domains: ligand specificity controlled by dephosphorylation of an ectodomain. Science (New York, NY 262(5138):1436–40
Chu LY, Silverstein RL (2012) CD36 ectodomain phosphorylation blocks thrombospondin-1 binding: structure-function relationships and regulation by protein kinase C. Arterioscler Thromb Vasc Biol 32(3):760–7
Ho M, Hoang HL, Lee KM, Liu N, MacRae T, Montes L et al (2005) Ectophosphorylation of CD36 regulates cytoadherence of Plasmodium falciparum to microvascular endothelium under flow conditions. Infect Immun 73(12):8179–87
Abumrad NA, el-Maghrabi MR, Amri EZ, Lopez E, Grimaldi PA (1993) Cloning of a rat adipocyte membrane protein implicated in binding or transport of long-chain fatty acids that is induced during preadipocyte differentiation. Homology with human CD36. J Biol Chem 268(24):17665–8
Schwenk RW, Holloway GP, Luiken JJ, Bonen A, Glatz JF (2010) Fatty acid transport across the cell membrane: regulation by fatty acid transporters. Prostaglandins Leukot Essent Fatty Acids 82(4–6):149–54
Papale GA, Nicholson K, Hanson PJ, Pavlovic M, Drover VA, Sahoo D (2010) Extracellular hydrophobic regions in scavenger receptor BI play a key role in mediating HDL-cholesterol transport. Arch Biochem Biophys 496(2):132–9
Sun B, Boyanovsky BB, Connelly MA, Shridas P, van der Westhuyzen DR, Webb NR (2007) Distinct mechanisms for OxLDL uptake and cellular trafficking by class B scavenger receptors CD36 and SR-BI. J Lipid Res 48(12):2560–70
Zeng Y, Tao N, Chung KN, Heuser JE, Lublin DM (2003) Endocytosis of oxidized low density lipoprotein through scavenger receptor CD36 utilizes a lipid raft pathway that does not require caveolin-1. J Biol Chem 278(46):45931–6
King KL, Stanley WC, Rosca M, Kerner J, Hoppel CL, Febbraio M (2007) Fatty acid oxidation in cardiac and skeletal muscle mitochondria is unaffected by deletion of CD36. Arch Biochem Biophys 467(2):234–8
Campbell SE, Tandon NN, Woldegiorgis G, Luiken JJ, Glatz JF, Bonen A (2004) A novel function for fatty acid translocase (FAT)/CD36: involvement in long chain fatty acid transfer into the mitochondria. J Biol Chem 279(35):36235–41
Bezaire V, Bruce CR, Heigenhauser GJ, Tandon NN, Glatz JF, Luiken JJ et al (2006) Identification of fatty acid translocase on human skeletal muscle mitochondrial membranes: essential role in fatty acid oxidation. Am J Physiol Endocrinol Metab 290(3):E509–15
Tandon NN, Lipsky RH, Burgess WH, Jamieson GA (1989) Isolation and characterization of platelet glycoprotein IV (CD36). J Biol Chem 264(13):7570–5
Joneckis CC, Ackley RL, Orringer EP, Wayner EA, Parise LV (1993) Integrin alpha 4 beta 1 and glycoprotein IV (CD36) are expressed on circulating reticulocytes in sickle cell anemia. Blood 82(12):3548–55
Endemann G, Stanton LW, Madden KS, Bryant CM, White RT, Protter AA (1993) CD36 is a receptor for oxidized low density lipoprotein. J Biol Chem 268(16):11811–6
Huh HY, Lo SK, Yesner LM, Silverstein RL (1995) CD36 induction on human monocytes upon adhesion to tumor necrosis factor-activated endothelial cells. J Biol Chem 270(11):6267–71
Rouabhia M, Jobin N, Doucet R Jr, Bergeron J, Auger FA (1994) The skin immune system: CD36(+)-dendritic epidermal cell–a putative actor in posttransplant immunological events. Transplant Proc 26(6):3482
Albert ML, Pearce SF, Francisco LM, Sauter B, Roy P, Silverstein RL et al (1998) Immature dendritic cells phagocytose apoptotic cells via alphavbeta5 and CD36, and cross-present antigens to cytotoxic T lymphocytes. J Exp Med 188(7):1359–68
Swerlick RA, Lee KH, Wick TM, Lawley TJ (1992) Human dermal microvascular endothelial but not human umbilical vein endothelial cells express CD36 in vivo and in vitro. J Immunol 148(1):78–83
Lim HJ, Lee S, Lee KS, Park JH, Jang Y, Lee EJ et al (2006) PPARgamma activation induces CD36 expression and stimulates foam cell like changes in rVSMCs. Prostaglandins Other Lipid Mediat 80(3–4):165–74
Li W, Febbraio M, Reddy SP, Yu DY, Yamamoto M, Silverstein RL (2010) CD36 participates in a signaling pathway that regulates ROS formation in murine VSMCs. J Clin Investig 120(11):3996–4006
Clezardin P, Frappart L, Clerget M, Pechoux C, Delmas PD (1993) Expression of thrombospondin (TSP1) and its receptors (CD36 and CD51) in normal, hyperplastic, and neoplastic human breast. Cancer Res 53(6):1421–30
Ryeom SW, Sparrow JR, Silverstein RL (1996) CD36 participates in the phagocytosis of rod outer segments by retinal pigment epithelium. J Cell Sci 109(Pt 2):387–95
Lobo MV, Huerta L, Ruiz-Velasco N, Teixeiro E, de la Cueva P, Celdran A et al (2001) Localization of the lipid receptors CD36 and CLA-1/SR-BI in the human gastrointestinal tract: towards the identification of receptors mediating the intestinal absorption of dietary lipids. J Histochem Cytochem 49(10):1253–60
Laugerette F, Passilly-Degrace P, Patris B, Niot I, Febbraio M, Montmayeur JP et al (2005) CD36 involvement in orosensory detection of dietary lipids, spontaneous fat preference, and digestive secretions. J Clin Investig 115(11):3177–84
Zhang X, Fitzsimmons RL, Cleland LG, Ey PL, Zannettino AC, Farmer EA et al (2003) CD36/fatty acid translocase in rats: distribution, isolation from hepatocytes, and comparison with the scavenger receptor SR-B1. Lab Invest 83(3):317–32
Baines RJ, Chana RS, Hall M, Febbraio M, Kennedy D, Brunskill NJ (2012) CD36 mediates proximal tubular binding and uptake of albumin and is upregulated in proteinuric nephropathies. Am J Physiol Renal Physiol 303(7):F1006–14
Van Nieuwenhoven FA, Verstijnen CP, Abumrad NA, Willemsen PH, Van Eys GJ, Van der Vusse GJ et al (1995) Putative membrane fatty acid translocase and cytoplasmic fatty acid-binding protein are co-expressed in rat heart and skeletal muscles. Biochem Biophys Res Commun [Comparative Study] 207(2):747–52
Gillot I, Jehl-Pietri C, Gounon P, Luquet S, Rassoulzadegan M, Grimaldi P et al (2005) Germ cells and fatty acids induce translocation of CD36 scavenger receptor to the plasma membrane of Sertoli cells. J Cell Sci 118(Pt 14):3027–35
Zibara K, Malaud E, McGregor JL (2002) CD36 mRNA and protein expression levels are significantly increased in the heart and testis of apoE deficient mice in comparison to wild type (C57BL/6). J Biomed Biotechnol 2(1):14–21
Coburn CT, Knapp FF Jr, Febbraio M, Beets AL, Silverstein RL, Abumrad NA (2000) Defective uptake and utilization of long chain fatty acids in muscle and adipose tissues of CD36 knockout mice. J Biol Chem 275(42):32523–9
Kennedy DJ, Kuchibhotla S, Westfall KM, Silverstein RL, Morton RE, Febbraio M (2010) A CD36-dependent pathway enhances macrophage and adipose tissue inflammation and impairs insulin signalling. Cardiovasc Res 89(3):604–13
Zhou J, Febbraio M, Wada T, Zhai Y, Kuruba R, He J et al (2008) Hepatic fatty acid transporter Cd36 is a common target of LXR, PXR, and PPARgamma in promoting steatosis. Gastroenterology 134(2):556–67
Febbraio M, Podrez EA, Smith JD, Hajjar DP, Hazen SL, Hoff HF et al (2000) Targeted disruption of the class B scavenger receptor CD36 protects against atherosclerotic lesion development in mice. J Clin Investig 105(8):1049–56
Febbraio M, Hajjar DP, Silverstein RL (2001) CD36: a class B scavenger receptor involved in angiogenesis, atherosclerosis, inflammation, and lipid metabolism. J Clin Investig 108(6):785–91
Gordon S (2002) Pattern recognition receptors: doubling up for the innate immune response. Cell 111(7):927–30
Miller YI, Chang MK, Binder CJ, Shaw PX, Witztum JL (2003) Oxidized low density lipoprotein and innate immune receptors. Curr Opin Lipidol 14(5):437–45
Glezer I, Bittencourt JC, Rivest S (2009) Neuronal expression of Cd36, Cd44, and Cd83 antigen transcripts maps to distinct and specific murine brain circuits. J Comp Neurol 517(6):906–24
Le Foll C, Irani BG, Magnan C, Dunn-Meynell AA, Levin BE (2009) Characteristics and mechanisms of hypothalamic neuronal fatty acid sensing. Am J Physiol Regul Integr Comp Physiol 297(3):R655–64
El-Yassimi A, Hichami A, Besnard P, Khan NA (2008) Linoleic acid induces calcium signaling, Src kinase phosphorylation, and neurotransmitter release in mouse CD36-positive gustatory cells. J Biol Chem 283(19):12949–59
Mitchell RW, Edmundson CL, Miller DW, Hatch GM (2009) On the mechanism of oleate transport across human brain microvessel endothelial cells. J Neurochem 110(3):1049–57
Song BJ, Elbert A, Rahman T, Orr SK, Chen CT, Febbraio M et al (2010) Genetic ablation of CD36 does not alter mouse brain polyunsaturated fatty acid concentrations. Lipids 45(4):291–9
Cho S, Park EM, Febbraio M, Anrather J, Park L, Racchumi G et al (2005) The class B scavenger receptor CD36 mediates free radical production and tissue injury in cerebral ischemia. J Neurosci 25(10):2504–12
Bao Y, Qin L, Kim E, Bhosle S, Guo H, Febbraio M et al (2012) CD36 is involved in astrocyte activation and astroglial scar formation. J Cereb Blood Flow Metab 32(8):1567–77
El Khoury J, Hickman SE, Thomas CA, Cao L, Silverstein SC, Loike JD (1996) Scavenger receptor-mediated adhesion of microglia to beta-amyloid fibrils. Nature 382(6593):716–9
El Khoury JB, Moore KJ, Means TK, Leung J, Terada K, Toft M et al (2003) CD36 mediates the innate host response to beta-amyloid. J Exp Med 197(12):1657–66
Kouadir M, Yang L, Tan R, Shi F, Lu Y, Zhang S et al (2012) CD36 participates in PrP(106–126)-induced activation of microglia. PLoS ONE 7(1):e30756
Bamberger ME, Harris ME, McDonald DR, Husemann J, Landreth GE (2003) A cell surface receptor complex for fibrillar beta-amyloid mediates microglial activation. J Neurosci 23(7):2665–74
Wilkinson B, Koenigsknecht-Talboo J, Grommes C, Lee CY, Landreth G (2006) Fibrillar beta-amyloid-stimulated intracellular signaling cascades require Vav for induction of respiratory burst and phagocytosis in monocytes and microglia. J Biol Chem 281(30):20842–50
Stewart CR, Stuart LM, Wilkinson K, van Gils JM, Deng J, Halle A et al (2010) CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat Immunol 11(2):155–61
Endemann G, Stanton LW, Madden KS, Bryant CM, White RT, Protter AA (1993) CD36 is a receptor for oxidized low density lipoprotein. J Biol Chem 268(16):11811–6
Huh HY, Lo SK, Yesner LM, Silverstein RL (1995) CD36 induction on human monocytes upon adhesion to tumor necrosis factor-activated endothelial cells. J Biol Chem 270(11):6267–71
Tontonoz P, Nagy L, Alvarez JG, Thomazy VA, Evans RM (1998) PPARgamma promotes monocyte/macrophage differentiation and uptake of oxidized LDL. Cell 93(2):241–52
Yang J, Sambandam N, Han X, Gross RW, Courtois M, Kovacs A et al (2007) CD36 deficiency rescues lipotoxic cardiomyopathy. Circulation Res 100(8):1208–17
Ito M, Suzuki J, Tsujioka S, Sasaki M, Gomori A, Shirakura T et al (2007) Longitudinal analysis of murine steatohepatitis model induced by chronic exposure to high-fat diet. Hepatol Res 37(1):50–7
Ghosh A, Murugesan G, Chen K, Zhang L, Wang Q, Febbraio M et al (2011) Platelet CD36 surface expression levels affect functional responses to oxidized LDL and are associated with inheritance of specific genetic polymorphisms. Blood 117(23):6355–66
Podrez EA, Byzova TV, Febbraio M, Salomon RG, Ma Y, Valiyaveettil M et al (2007) Platelet CD36 links hyperlipidemia, oxidant stress and a prothrombotic phenotype. Nat Med 13(9):1086–95
Chen K, Febbraio M, Li W, Silverstein RL (2008) A specific CD36-dependent signaling pathway is required for platelet activation by oxidized low-density lipoprotein. Circulation Res 102(12):1512–9
Chen K, Li W, Major J, Rahaman SO, Febbraio M, Silverstein RL (2011) Vav guanine nucleotide exchange factors link hyperlipidemia and a prothrombotic state. Blood 117(21):5744–50
Zhu W, Li W, Silverstein RL (2012) Advanced glycation end products induce a prothrombotic phenotype in mice via interaction with platelet CD36. Blood 119(25):6136–44
Korporaal SJ, Van Eck M, Adelmeijer J, Ijsseldijk M, Out R, Lisman T et al (2007) Platelet activation by oxidized low density lipoprotein is mediated by CD36 and scavenger receptor-A. Arterioscler Thromb Vasc Biol 27(11):2476–83
Herczenik E, Bouma B, Korporaal SJ, Strangi R, Zeng Q, Gros P et al (2007) Activation of human platelets by misfolded proteins. Arterioscler Thromb Vasc Biol 27(7):1657–65
Iadecola C, Cho S, Feuerstein GZ, Hallenbeck J (2004) Cerebral Ischemia and Inflammation. In Stroke: Pathophysiology, Diagnosis, and Management. 883–94
Dirnagl U, Iadecola C, Moskowitz MA (1999) Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 22(9):391–7
Amantea D, Nappi G, Bernardi G, Bagetta G, Corasaniti MT (2009) Post-ischemic brain damage: pathophysiology and role of inflammatory mediators. FEBS J 276(1):13–26
Tan KT, Lip GY, Blann AD (2003) Post-stroke inflammatory response: effects of stroke evolution and outcome. Curr Atheroscler Rep 5(4):245–51
del Zoppo G, Ginis I, Hallenbeck JM, Iadecola C, Wang X, Feuerstein GZ (2000) Inflammation and stroke: putative role for cytokines, adhesion molecules and iNOS in brain response to ischemia. Brain Pathol (Zurich, Switzerland) 10(1):95–112
Barone FC, Feuerstein GZ (1999) Inflammatory mediators and stroke: new opportunities for novel therapeutics. J Cereb Blood Flow Metab 19(8):819–34
Becker KJ (1998) Inflammation and acute stroke. Curr Opin Neurol 11(1):45–9
Connolly ES Jr, Winfree CJ, Prestigiacomo CJ, Kim SC, Choudhri TF, Hoh BL et al (1997) Exacerbation of cerebral injury in mice that express the P-selectin gene: identification of P-selectin blockade as a new target for the treatment of stroke. Circulation Res 81(3):304–10
Connolly ES Jr, Winfree CJ, Springer TA, Naka Y, Liao H, Yan SD et al (1996) Cerebral protection in homozygous null ICAM-1 mice after middle cerebral artery occlusion. Role of neutrophil adhesion in the pathogenesis of stroke. J Clin Invest 97(1):209–16
Soriano SG, Lipton SA, Wang YF, Xiao M, Springer TA, Gutierrez-Ramos JC et al (1996) Intercellular adhesion molecule-1-deficient mice are less susceptible to cerebral ischemia-reperfusion injury. Ann Neurol 39(5):618–24
Hughes PM, Allegrini PR, Rudin M, Perry VH, Mir AK, Wiessner C (2002) Monocyte chemoattractant protein-1 deficiency is protective in a murine stroke model. J Cereb Blood Flow Metab 22(3):308–17
Dimitrijevic OB, Stamatovic SM, Keep RF, Andjelkovic AV (2007) Absence of the chemokine receptor CCR2 protects against cerebral ischemia/reperfusion injury in mice. Stroke 38(4):1345–53
Chen Y, Hallenbeck JM, Ruetzler C, Bol D, Thomas K, Berman NE et al (2003) Overexpression of monocyte chemoattractant protein 1 in the brain exacerbates ischemic brain injury and is associated with recruitment of inflammatory cells. J Cereb Blood Flow Metab 23(6):748–55
Enlimomab Acute Stroke Trial investigators (2001) Use of anti-ICAM-1 therapy in ischemic stroke: results of the Enlimomab Acute Stroke Trial. Neurology 57(8):1428–34
Becker KJ (2002) Anti-leukocyte antibodies: LeukArrest (Hu23F2G) and Enlimomab (R6.5) in acute stroke. Curr Med Res Opin 18(Suppl 2):s18–22
Doyle KP, Buckwalter MS (2012) The double-edged sword of inflammation after stroke: what sharpens each edge? Ann Neurol 71(6):729–31
Kim E, Febbraio M, Bao Y, Tolhurst AT, Epstein JM, Cho S (2012) CD36 in the periphery and brain synergizes in stroke injury in hyperlipidemia. Ann Neurol 71(6):753–64
Gliem M, Mausberg AK, Lee JI, Simiantonakis I, van Rooijen N, Hartung HP et al (2012) Macrophages prevent hemorrhagic infarct transformation in murine stroke models. Ann Neurol 71(6):743–52
Akira S, Uematsu S, Takeuchi O (2006) Pathogen recognition and innate immunity. Cell 124(4):783–801
Bamboat ZM, Balachandran VP, Ocuin LM, Obaid H, Plitas G, DeMatteo RP (2010) Toll-like receptor 9 inhibition confers protection from liver ischemia-reperfusion injury. Hepatology 51(2):621–32
Bamboat ZM, Ocuin LM, Balachandran VP, Obaid H, Plitas G, DeMatteo RP (2010) Conventional DCs reduce liver ischemia/reperfusion injury in mice via IL-10 secretion. J Clin Invest 120(2):559–69
Greenberg ME, Sun M, Zhang R, Febbraio M, Silverstein R, Hazen SL (2006) Oxidized phosphatidylserine-CD36 interactions play an essential role in macrophage-dependent phagocytosis of apoptotic cells. J Exp Med 203(12):2613–25
Ren Y, Silverstein RL, Allen J, Savill J (1995) CD36 gene transfer confers capacity for phagocytosis of cells undergoing apoptosis. J Exp Med 181(5):1857–62
Savill J (1997) Recognition and phagocytosis of cells undergoing apoptosis. Br Med Bull 53(3):491–508
Franc NC, Dimarcq JL, Lagueux M, Hoffmann J, Ezekowitz RA (1996) Croquemort, a novel Drosophila hemocyte/macrophage receptor that recognizes apoptotic cells. Immunity 4(5):431–43
Greenberg ME, Li XM, Gugiu BG, Gu X, Qin J, Salomon RG et al (2008) The lipid whisker model of the structure of oxidized cell membranes. J Biol Chem 283(4):2385–96
Hazen SL (2008) Oxidized phospholipids as endogenous pattern recognition ligands in innate immunity. J Biol Chem 283:15527–31
Voll RE, Herrmann M, Roth EA, Stach C, Kalden JR, Girkontaite I (1997) Immunosuppressive effects of apoptotic cells. Nature 390(6658):350–1
Chung EY, Liu J, Homma Y, Zhang Y, Brendolan A, Saggese M et al (2007) Interleukin-10 expression in macrophages during phagocytosis of apoptotic cells is mediated by homeodomain proteins Pbx1 and Prep-1. Immunity 27(6):952–64
Hoebe K, Georgel P, Rutschmann S, Du X, Mudd S, Crozat K et al (2005) CD36 is a sensor of diacylglycerides. Nature 433(7025):523–7
Stuart LM, Deng J, Silver JM, Takahashi K, Tseng AA, Hennessy EJ et al (2005) Response to Staphylococcus aureus requires CD36-mediated phagocytosis triggered by the COOH-terminal cytoplasmic domain. J Cell Biol 170(3):477–85
Drage MG, Pecora ND, Hise AG, Febbraio M, Silverstein RL, Golenbock DT et al (2009) TLR2 and its co-receptors determine responses of macrophages and dendritic cells to lipoproteins of Mycobacterium tuberculosis. Cellular Immunol 258(1):29–37
Triantafilou M, Gamper FG, Lepper PM, Mouratis MA, Schumann C, Harokopakis E et al (2007) Lipopolysaccharides from atherosclerosis-associated bacteria antagonize TLR4, induce formation of TLR2/1/CD36 complexes in lipid rafts and trigger TLR2-induced inflammatory responses in human vascular endothelial cells. Cell Microbiol 9(8):2030–9
Triantafilou M, Gamper FG, Haston RM, Mouratis MA, Morath S, Hartung T et al (2006) Membrane sorting of toll-like receptor (TLR)-2/6 and TLR2/1 heterodimers at the cell surface determines heterotypic associations with CD36 and intracellular targeting. J Biol Chem 281(41):31002–11
Seimon TA, Nadolski MJ, Liao X, Magallon J, Nguyen M, Feric NT et al (2010) Atherogenic lipids and lipoproteins trigger CD36-TLR2-dependent apoptosis in macrophages undergoing endoplasmic reticulum stress. Cell Metab 12(5):467–82
Abe T, Shimamura M, Jackman K, Kurinami H, Anrather J, Zhou P et al (2010) Key role of CD36 in Toll-like receptor 2 signaling in cerebral ischemia. Stroke 41(5):898–904
Jimenez-Dalmaroni MJ, Xiao N, Corper AL, Verdino P, Ainge GD, Larsen DS et al (2009) Soluble CD36 ectodomain binds negatively charged diacylglycerol ligands and acts as a co-receptor for TLR2. PLoS ONE 4(10):e7411
Park L, Wang G, Zhou P, Zhou J, Pitstick R, Previti ML et al (2011) Scavenger receptor CD36 is essential for the cerebrovascular oxidative stress and neurovascular dysfunction induced by amyloid-beta. Proc Natl Acad Sci U S A 108(12):5063–8
Su X, Maguire-Zeiss KA, Giuliano R, Prifti L, Venkatesh K, Federoff HJ (2007) Synuclein activates microglia in a model of Parkinson’s disease. Neurobiol Aging 29:1690–701
Cho S, Kim E (2009) CD36: a multi-modal target for acute stroke therapy. J Neurochem 109(Suppl 1):126–32
Kim E, Tolhurst AT, Qin LY, Chen XY, Febbraio M, Cho S (2008) CD36/fatty acid translocase, an inflammatory mediator, is involved in hyperlipidemia-induced exacerbation in ischemic brain injury. J Neurosci 28(18):4661–70
Fitch MT, Silver J (1997) Activated macrophages and the blood–brain barrier: inflammation after CNS injury leads to increases in putative inhibitory molecules. Exp Neurol 148(2):587–603
Nihashi T, Inao S, Kajita Y, Kawai T, Sugimoto T, Niwa M et al (2001) Expression and distribution of beta amyloid precursor protein and beta amyloid peptide in reactive astrocytes after transient middle cerebral artery occlusion. Acta Neurochir (Wien) 143(3):287–95
Hayashi T, Noshita N, Sugawara T, Chan PH (2003) Temporal profile of angiogenesis and expression of related genes in the brain after ischemia. J Cereb Blood Flow Metab 23(2):166–80
Pilitsis JG, Coplin WM, O’Regan MH, Wellwood JM, Diaz FG, Fairfax MR et al (2003) Measurement of free fatty acids in cerebrospinal fluid from patients with hemorrhagic and ischemic stroke. Brain Res 985(2):198–201
Uno M, Kitazato KT, Nishi K, Itabe H, Nagahiro S (2003) Raised plasma oxidised LDL in acute cerebral infarction. J Neurol Neurosurg Psychiatry 74(3):312–6
Shie FS, Neely MD, Maezawa I, Wu H, Olson SJ, Jurgens G et al (2004) Oxidized low-density lipoprotein is present in astrocytes surrounding cerebral infarcts and stimulates astrocyte interleukin-6 secretion. Am J Pathol 164(4):1173–81
Podrez EA, Poliakov E, Shen Z, Zhang R, Deng Y, Sun M et al (2002) Identification of a novel family of oxidized phospholipids that serve as ligands for the macrophage scavenger receptor CD36. J Biol Chem 277(41):38503–16
Kernan WN, Inzucchi SE (2004) Type 2 diabetes mellitus and insulin resistance: stroke prevention and management. Curr Treat Options Neurol 6(6):443–50
Kernan WN, Inzucchi SE, Viscoli CM, Brass LM, Bravata DM, Shulman GI et al (2003) Impaired insulin sensitivity among nondiabetic patients with a recent TIA or ischemic stroke. Neurology 60(9):1447–51
Gautam S, Banerjee M (2011) The macrophage Ox-LDL receptor, CD36 and its association with type II diabetes mellitus. Mol Genet Metab [Review] 102(4):389–98
Yanai H, Chiba H, Morimoto M, Jamieson GA, Matsuno K (2000) Type I CD36 deficiency in humans is not associated with insulin resistance syndrome. Thromb Haemost 83(5):786
Wang Y, Zhou XO, Zhang Y, Gao PJ, Zhu DL (2012) Association of the CD36 gene with impaired glucose tolerance, impaired fasting glucose, type-2 diabetes, and lipid metabolism in essential hypertensive patients. Genet Mol Res 11(3):2163–70
Goudriaan JR, Dahlmans VE, Teusink B, Ouwens DM, Febbraio M, Maassen JA et al (2003) CD36 deficiency increases insulin sensitivity in muscle, but induces insulin resistance in the liver in mice. J lipid Res 44(12):2270–7
Kuang M, Febbraio M, Wagg C, Lopaschuk GD, Dyck JR (2004) Fatty acid translocase/CD36 deficiency does not energetically or functionally compromise hearts before or after ischemia. Circulation 109(12):1550–7
Nicholls HT, Kowalski G, Kennedy DJ, Risis S, Zaffino LA, Watson N et al (2011) Hematopoietic cell-restricted deletion of CD36 reduces high-fat diet-induced macrophage infiltration and improves insulin signaling in adipose tissue. Diabetes 60(4):1100–10
Kennedy DJ, Kuchibhotla S, Westfall KM, Silverstein RL, Morton RE, Febbraio M (2011) A CD36-dependent pathway enhances macrophage and adipose tissue inflammation and impairs insulin signalling. Cardiovasc Res 89(3):604–13
Griffin E, Re A, Hamel N, Fu C, Bush H, McCaffrey T et al (2001) A link between diabetes and atherosclerosis: glucose regulates expression of CD36 at the level of translation. Nat Med 7(7):840–6
Susztak K, Ciccone E, McCue P, Sharma K, Bottinger EP (2005) Multiple metabolic hits converge on CD36 as novel mediator of tubular epithelial apoptosis in diabetic nephropathy. PLoS Med 2(2):e45
Sampson MJ, Davies IR, Braschi S, Ivory K, Hughes DA (2003) Increased expression of a scavenger receptor (CD36) in monocytes from subjects with Type 2 diabetes. Atherosclerosis 167(1):129–34
Greenwalt DE, Scheck SH, Rhinehart-Jones T (1995) Heart CD36 expression is increased in murine models of diabetes and in mice fed a high fat diet. J Clin Investig 96(3):1382–8
Mine S, Okada Y, Tanikawa T, Kawahara C, Tabata T, Tanaka Y (2006) Increased expression levels of monocyte CCR2 and monocyte chemoattractant protein-1 in patients with diabetes mellitus. Biochem Biophys Res Commun 344(3):780–5
Liang CP, Han S, Okamoto H, Carnemolla R, Tabas I, Accili D et al (2004) Increased CD36 protein as a response to defective insulin signaling in macrophages. J Clin Investig 113(5):764–73
Lamharzi N, Renard CB, Kramer F, Pennathur S, Heinecke JW, Chait A et al (2004) Hyperlipidemia in concert with hyperglycemia stimulates the proliferation of macrophages in atherosclerotic lesions: potential role of glucose-oxidized LDL. Diabetes 53(12):3217–25
Lam MC, Tan KC, Lam KS (2004) Glycoxidized low-density lipoprotein regulates the expression of scavenger receptors in THP-1 macrophages. Atherosclerosis 177(2):313–20
Noushmehr H, D’Amico E, Farilla L, Hui H, Wawrowsky KA, Mlynarski W et al (2005) Fatty acid translocase (FAT/CD36) is localized on insulin-containing granules in human pancreatic beta-cells and mediates fatty acid effects on insulin secretion. Diabetes 54(2):472–81
Handberg A, Levin K, Hojlund K, Beck-Nielsen H (2006) Identification of the oxidized low-density lipoprotein scavenger receptor CD36 in plasma: a novel marker of insulin resistance. Circulation 114(11):1169–76
Alkhatatbeh MJ, Mhaidat NM, Enjeti AK, Lincz LF, Thorne RF (2011) The putative diabetic plasma marker, soluble CD36, is non-cleaved, non-soluble and entirely associated with microparticles. J Thromb Haemost 9(4):844–51
Hartz AM, Bauer B, Soldner EL, Wolf A, Boy S, Backhaus R et al (2011) Amyloid-beta contributes to blood–brain barrier leakage in transgenic human amyloid precursor protein mice and in humans with cerebral amyloid angiopathy. Stroke 43:514–23
Carrano A, Hoozemans JJ, van der Vies SM, Rozemuller AJ, van Horssen J, de Vries HE (2011) Amyloid Beta induces oxidative stress-mediated blood–brain barrier changes in capillary amyloid angiopathy. Antioxid Redox Signal 15(5):1167–78
Moore KJ, El Khoury J, Medeiros LA, Terada K, Geula C, Luster AD et al (2002) A CD36-initiated signaling cascade mediates inflammatory effects of beta-amyloid. J Biol Chem 277(49):47373–9
Wilkinson K, Boyd JD, Glicksman M, Moore KJ, El Khoury J (2011) A high content drug screen identifies ursolic acid as an inhibitor of amyloid beta protein interactions with its receptor CD36. J Biol Chem 286(40):34914–22
Lee PH, Bang OY, Hwang EM, Lee JS, Joo US, Mook-Jung I et al (2005) Circulating beta amyloid protein is elevated in patients with acute ischemic stroke. J Neural Transm 112(10):1371–9
Amenta F, Di Tullio MA, Tomassoni D (2003) Arterial hypertension and brain damage–evidence from animal models (review). Clin Exp Hypertens 25(6):359–80
Droste DW, Ritter MA, Dittrich R, Heidenreich S, Wichter T, Freund M et al (2003) Arterial hypertension and ischaemic stroke. Acta Neurol Scand 107(4):241–51
Bornstein N, Silvestrelli G, Caso V, Parnetti L (2006) Arterial hypertension and stroke prevention: an update. Clin Exp Hypertens 28(3–4):317–26
Amenta F, Mignini F, Rabbia F, Tomassoni D, Veglio F (2002) Protective effect of anti-hypertensive treatment on cognitive function in essential hypertension: analysis of published clinical data. J Neurol Sci 203–204:147–51
Pravenec M, Churchill P, Chuchill MC, Viklicky O, Kazdova L, Aitman TJ (2008) Identification of renal CD36 as a determinant of blood pressure and risk for hypertension. Nature Genetics 40:952–4
Ueno M, Nakagawa T, Nagai Y, Nishi N, Kusaka T, Kanenishi K et al (2011) The expression of CD36 in vessels with blood–brain barrier impairment in a stroke-prone hypertensive model. Neuropathol Appl Neurobiol 37(7):727–37
Zapolska-Downar D, Siennicka A, Chelstowski K, Widecka K, Goracy I, Halasa M et al (2006) Is there an association between angiotensin-converting enzyme gene polymorphism and functional activation of monocytes and macrophage in young patients with essential hypertension? J Hypertens 24(8):1565–73
Bull HA, Brickell PM, Dowd PM (1994) Src-related protein tyrosine kinases are physically associated with the surface antigen CD36 in human dermal microvascular endothelial cells. FEBS Lett 351(1):41–4
Kwapiszewska G, Wilhelm J, Wolff S, Laumanns I, Koenig IR, Ziegler A et al (2005) Expression profiling of laser-microdissected intrapulmonary arteries in hypoxia-induced pulmonary hypertension. Respir Res 6:109
Schilling M, Besselmann M, Leonhard C, Mueller M, Ringelstein EB, Kiefer R (2003) Microglial activation precedes and predominates over macrophage infiltration in transient focal cerebral ischemia: a study in green fluorescent protein transgenic bone marrow chimeric mice. Exp Neurol 183(1):25–33
Stevens SL, Bao J, Hollis J, Lessov NS, Clark WM, Stenzel-Poore MP (2002) The use of flow cytometry to evaluate temporal changes in inflammatory cells following focal cerebral ischemia in mice. Brain Res 932(1–2):110–9
Gelderblom M, Leypoldt F, Steinbach K, Behrens D, Choe CU, Siler DA et al (2009) Temporal and spatial dynamics of cerebral immune cell accumulation in stroke. Stroke 40(5):1849–57
Ajami B, Bennett JL, Krieger C, McNagny KM, Rossi FM (2011) Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat Neurosci 14(9):1142–9
Bauer J, Huitinga I, Zhao W, Lassmann H, Hickey WF, Dijkstra CD (1995) The role of macrophages, perivascular cells, and microglial cells in the pathogenesis of experimental autoimmune encephalomyelitis. Glia 15(4):437–46
Gordon S (2007) Macrophage heterogeneity and tissue lipids. J Clin Investig 117(1):89–93
Auffray C, Sieweke MH, Geissmann F (2009) Blood monocytes: development, heterogeneity, and relationship with dendritic cells. Ann Rev Immunol 27:669–92
Geissmann F, Manz MG, Jung S, Sieweke MH, Merad M, Ley K (2010) Development of monocytes, macrophages, and dendritic cells. Science (New York, NY); 327(5966):656–61
Nahrendorf M, Swirski FK, Aikawa E, Stangenberg L, Wurdinger T, Figueiredo JL et al (2007) The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J Exp Med 204(12):3037–47
Swirski FK, Nahrendorf M, Etzrodt M, Wildgruber M, Cortez-Retamozo V, Panizzi P et al (2009) Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science (New York, NY 325(5940):612–6
Kim JS, Gautam SC, Chopp M, Zaloga C, Jones ML, Ward PA et al (1995) Expression of monocyte chemoattractant protein-1 and macrophage inflammatory protein-1 after focal cerebral ischemia in the rat. J Neuroimmunol 56(2):127–34
Minami M, Satoh M (2003) Chemokines and their receptors in the brain: pathophysiological roles in ischemic brain injury. Life Sci 74(2–3):321–7
Che X, Ye W, Panga L, Wu DC, Yang GY (2001) Monocyte chemoattractant protein-1 expressed in neurons and astrocytes during focal ischemia in mice. Brain Res 902(2):171–7
Kennedy DJ, Kuchibhotla SD, Guy E, Park YM, Nimako G, Vanegas D et al (2009) Dietary cholesterol plays a role in CD36-mediated atherogenesis in LDLR-knockout mice. Arterioscl Thromb Vasc biol 29(10):1481–7
Harb D, Bujold K, Febbraio M, Sirois MG, Ong H, Marleau S (2009) The role of the scavenger receptor CD36 in regulating mononuclear phagocyte trafficking to atherosclerotic lesions and vascular inflammation. Cardiovasc Res 83:42–51
Kuchibhotla S, Vanegas D, Kennedy DJ, Guy E, Nimako G, Morton RE et al (2008) Absence of CD36 protects against atherosclerosis in ApoE knock-out mice with no additional protection provided by absence of scavenger receptor A I/II. Cardiovasc Res 78:185–96
Stuart LM, Bell SA, Stewart CR, Silver JM, Richard J, Goss JL et al (2007) CD36 signals to the actin cytoskeleton and regulates microglial migration via a p130Cas complex. J Biol Chem 282(37):27392–401
Park YM, Drazba JA, Vasanji A, Egelhoff T, Febbraio M, Silverstein RL (2012) Oxidized LDL/CD36 interaction induces loss of cell polarity and inhibits macrophage locomotion. Mol Biol Cell 23(16):3057–68
Park YM, Febbraio M, Silverstein RL (2009) CD36 modulates migration of mouse and human macrophages in response to oxidized LDL and may contribute to macrophage trapping in the arterial intima. J Clin Investig 119(1):136–45
Fuhrman B, Volkova N, Aviram M (2002) Oxidative stress increases the expression of the CD36 scavenger receptor and the cellular uptake of oxidized low-density lipoprotein in macrophages from atherosclerotic mice: protective role of antioxidants and of paraoxonase. Atherosclerosis 161(2):307–16
Venugopal SK, Devaraj S, Jialal I (2004) RRR-alpha-tocopherol decreases the expression of the major scavenger receptor, CD36, in human macrophages via inhibition of tyrosine kinase (Tyk2). Atherosclerosis 175(2):213–20
Ricciarelli R, Zingg JM, Azzi A (2000) Vitamin E reduces the uptake of oxidized LDL by inhibiting CD36 scavenger receptor expression in cultured aortic smooth muscle cells. Circulation 102(1):82–7
Munteanu A, Zingg JM, Ogru E, Libinaki R, Gianello R, West S et al (2004) Modulation of cell proliferation and gene expression by alpha-tocopheryl phosphates: relevance to atherosclerosis and inflammation. Biochem Biophys Res Commun 318(1):311–6
Han J, Zhou X, Yokoyama T, Hajjar DP, Gotto AM Jr, Nicholson AC (2004) Pitavastatin downregulates expression of the macrophage type B scavenger receptor, CD36. Circulation 109(6):790–6
Puccetti L, Sawamura T, Pasqui AL, Pastorelli M, Auteri A, Bruni F (2005) Atorvastatin reduces platelet-oxidized-LDL receptor expression in hypercholesterolaemic patients. Eur J Clin Investig 35(1):47–51
Akisato Y, Ishii I, Kitahara M, Tamaki T, Saito Y, Kitada M (2008) Effect of pitavastatin on macrophage cholesterol metabolism. Yakugaku Zasshi 128(3):357–63
Marleau S, Harb D, Bujold K, Avallone R, Iken K, Wang Y et al (2005) EP 80317, a ligand of the CD36 scavenger receptor, protects apolipoprotein E-deficient mice from developing atherosclerotic lesions. Faseb J 19(13):1869–71
Wang L, Bao Y, Yang Y, Wu Y, Chen X, Si S et al (2010) Discovery of antagonists for human scavenger receptor CD36 via an ELISA-like high-throughput screening assay. J Biomol Screen 15(3):239–50
Bao Y, Wang L, Xu Y, Yang Y, Si S, Cho S et al (2012) Salvianolic acid B inhibits macrophage uptake of modified low density lipoprotein (mLDL) in a scavenger receptor CD36-dependent manner. Atherosclerosis 223(1):152–9
Cho S, Szeto HH, Kim E, Kim H, Tolhurst AT, Pinto JT (2007) A novel cell-permeable antioxidant peptide, SS31, attenuates ischemic brain injury by down-regulating CD36. J Biol Chem 282(7):4634–42
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Cho, S., Febbraio, M. (2014). CD36: An Inflammatory Mediator in Acute Brain Injury. In: Chen, J., Hu, X., Stenzel-Poore, M., Zhang, J. (eds) Immunological Mechanisms and Therapies in Brain Injuries and Stroke. Springer Series in Translational Stroke Research, vol 6. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-8915-3_18
Download citation
DOI: https://doi.org/10.1007/978-1-4614-8915-3_18
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-8914-6
Online ISBN: 978-1-4614-8915-3
eBook Packages: MedicineMedicine (R0)