Abstract
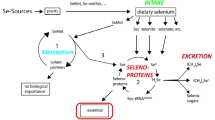
Selenium (Se) is an essential nutrient for humans and animals. Its nutritional functions are discharged by a group of proteins that contain Se in the form of selenocystein (SeCys), which is synthesized by the cotranslational addition of inorganic selenide to tRNA-bound serine [1]. The human selenoproteome consists of 25 selenoproteins [2], including four glutathione peroxidases, which catalyze the reduction of hydroperoxides [3]; three thioredoxin reductases, which are NADPH-dependent flavoenzymes that function in intracellular redox regulation [4]; three iodothyronine 5′-deiodinases, which catalyze the removal of iodine from the thyroid hormones (T4, T3); and selenoproteins P (the major transport form) [5], W (in muscle) [6], and R (methionine sulfoxide reductase) [7]. Because many selenoproteins have antioxidant functions, Se is regarded as important in metabolic protection from cellular-oxidative stress.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
FormalPara Key Points-
Lentil (Lens culinaris L.) is an excellent medium-energy source of protein and several micronutrients including selenium.
-
Lentils produced on high-Se soils contain appreciable amounts of Se, mostly as selenomethionine; consumption of 50–100 g of lentils can satisfy daily Se requirements.
-
Lentils can be enriched in Se through conventional plant breeding and Se-fertilization. Se-biofortification of staple foods may be a sustainable solution to address Se deficiency.
Introduction
Selenium (Se) is an essential nutrient for humans and animals. Its nutritional functions are discharged by a group of proteins that contain Se in the form of selenocystein (SeCys), which is synthesized by the cotranslational addition of inorganic selenide to tRNA-bound serine [1]. The human selenoproteome consists of 25 selenoproteins [2], including four glutathione peroxidases, which catalyze the reduction of hydroperoxides [3]; three thioredoxin reductases, which are NADPH-dependent flavoenzymes that function in intracellular redox regulation [4]; three iodothyronine 5′-deiodinases, which catalyze the removal of iodine from the thyroid hormones (T4, T3); and selenoproteins P (the major transport form) [5], W (in muscle) [6], and R (methionine sulfoxide reductase) [7]. Because many selenoproteins have antioxidant functions, Se is regarded as important in metabolic protection from cellular-oxidative stress.
Two human diseases have been associated with severe endemic Se deficiency: Keshan disease (a cardiomyopathy) and Kashin-Beck disease (an osteoarthropathy). Each occurs in areas of central and northeastern China and eastern Russia in mountainous areas where soil Se levels are very low (<125 μg/kg) and locally produced grains generally contain <40 μg Se/kg. In these areas, the blood Se levels of residents are typically <25 ng/mL. Because Keshan disease is also associated with cardiophilic RNA-viruses, Se deficiency may also increase risks for other diseases with viral etiologies. As many as 100 million people worldwide may be Se-deficient, mainly due to low concentrations of Se in commonly eaten foods [8–10].
Epidemiological studies indicate Se status is inversely associated with cancer risk, and Se-intervention reduces cancer risk through several cellular and metabolic mechanisms in studies with a wide variety of animal tumor models [11, 12]. Supranutritional intakes of Se (ca. 300 μg/day) have been shown to reduce the risk of several types of cancer [13]. Thus, Se-enriched foods may be an effective and sustainable means of increasing Se intakes to support good general health as well as reduce cancer risk. Novel, food-based approaches for increasing Se intakes are needed to support healthful Se nutrition, especially in resource-poor countries with limited access to nutritional supplements. Biofortification of lentils with Se constitutes such an approach.
Lentil (Lens culinaris L.) is a nutritious grain legume with relatively high protein content (ca. 20–30 %) and moderate energy content (ca. 4.2 kcal/g). Lentil is grown in many countries and is a staple component of diets in Asia and the Middle East as well as vegetarian diets around the world. The Se content of lentil reflects the Se content of the soils in which it is grown. Accordingly, lentils grown on low-Se soils have 22–180 μg Se per g, while lentils grown on the relatively high-Se soils of western Canada, North Dakota, and the USA have 672–1,200 μg Se per g [14] (unpublished data). Such high-Se lentils can supply daily Se needs at the daily consumption rate of 50–100 g. Because lentils can be sourced from high-Se areas, bred for their capacity to retain Se, and produced using Se-fertilization, they can be used to address Se deficiency in lentil consuming countries.
This chapter presents recent results on the Se-enrichment of lentils and implications of the lentil model to other pulses.
Introduction to Lentils
Pulses, including lentil, pea (Pisum sativum L.), and chickpea (Cicer arietinum L.), are high-protein, medium-energy legume crops that are staple foods for millions of people around the world. Lentil is a traditional crop grown mostly in low-rainfall and dryland cropping systems in rotation with cereals, wheat (Triticum sp.), and rice (Oryza sativa L.). Lentils were first grown 8,500 years ago in the Near East; their cultivation has since extended to the Mediterranean, Asia, Europe, and the Western Hemisphere, including Canada and the USA. Current annual world lentil production is approximately four million MT, more than 85 % of which occurs in five regions: south Asia (India, Nepal, and Bangladesh, 32 %), western Canada (29 %), Turkey and northern Syria (18 %), Australia (4 %), and the Midwestern USA (North and South Dakota, Eastern Montana) (3 %) [15]. Canada and the USA are the major lentil producing and exporting countries. North American lentils encompassing several market classes are supplied to more than 100 countries (Table 20.1).
Lentils were introduced to the USA and Canada in the early 1900s; however, agronomic improvement did not begin until the early 1980s. Over the last 40 years, global lentil production has increased by 6.8 % annually [16], and the lentil industry is a now major part of Canadian and the US prairie agriculture with a third of global lentil production now originating in Saskatchewan and the American Midwest [17]. Lentils are typically rotated with wheat or canola (Brassica napus L.) in North America, and with rice or wheat under rain-fed conditions in Asia. Annual lentil production in developing countries is declining, mainly due to low yields obtained from marginal soils with relatively few inputs. In Bangladesh, for example, farmers are shifting from lentils to high-yielding cultivars of rice and wheat. The main drivers of this shift are poor soil conditions, lack of drought tolerance, poor disease resistance, and prohibitively long harvest times in triple rotation cropping systems. In contrast, superior yields and an excellent fit into existing crop rotations have increased Canadian and the US red and green lentil production during the last two decades.
Lentils are important pulse crops in sustainable cropping systems. They provide significant benefits through the fixation of atmospheric nitrogen (N), which improves soil fertility. Generally, grain legumes fix up to 450 kg N per ha. On average, lentils fix N in amounts ranging from 5 to 191 kg N per ha [18], which is in general low compared to other grain legumes. Lentil is well adapted to semiarid, temperate climatic conditions, requiring only 10–12 in. of rain and a soil pH of 7 for high grain yields. Lentils have indeterminate (branching) growth, and plant height can range from 20 to 75 cm. Lentil leaves produce tendrils and flowering begins at the 11th or 12th node stage. Lentil flowers are self-pollinated. Generally, seed pods are <2.5 cm long, containing one or two seeds. Lentils are classified by seed size: large (Chilean) or small seeded (Persian). Seed coats can be red, green, brown, gray, or black, and cotyledons can be red, green, or/and yellow. Lentil grain yields range from 500 to 2,800 kg per ha [19]. Average yields range from 650 kg per ha in India to 1,345 kg per ha in North America [15, 19]; however, yields approaching 2,800 kg per ha are possible for some cultivars with appropriate crop management.
Lentil production and consumption have been increasing [15]. This is likely due to the convenience and economy of its short cooking time. The nutritional composition of lentils has been reported (Table 20.2); however, composition can vary with genotype and growing location/country. Lentils and peas produced in North America are typically high in Se as well as two other essential micronutrients: iron (Fe) and zinc (Zn). For example, 50 g of lentils or peas can provide a minimum of 25–50 % of the recommended daily allowances (RDA) of these three nutrients. Further, because lentils also contain relatively low concentrations of phytic acid [20], which forms nonabsorbable complexes with divalent cations, it can be expected that the Fe and Zn in lentils should be relatively highly bioavailable. Lentils also have a favorable amino acid profile that complements those of cereal grains to produce a dietary protein mixture of high biologic value. For these reasons, lentils can be an important source of nutrition in grain-based diets. Specifically, we have undertaken to determine the potential of Se-biofortification of lentils to the end of developing lentil as a sustainable, food-based approach to preventing Se deficiency.
Selenium Species in Lentils: Implications for Bioavailability
Plants uptake Se from the soil primarily as selenate or selenite and translocate it to the chloroplast where it follows the sulfur assimilation pathway. Selenate is reduced to selenide, which reacts with serine to form SeCys and is further metabolized to other organic Se forms including selenomethionine (SeMet). Therefore, the major Se-species in lentils have been thought to be SeCys and SeMet, which is predominantly protein-bound [21, 22]. Using synchrotron X-ray absorption spectroscopy (XAS), we found that most (86–95 %) of the Se in Saskatchewan lentils was present in organic forms with the remainder as selenate [22]. Using high performance liquid chromatography-inductively coupled plasma-mass spectrometry (HPLC-ICP-MS), we found that most (69 %) of the organic component was SeMet, with small amounts of SeCys (7 %) and other selenooligopeptides such as γ-glutamylselenocysteine [14]. More than half of the SeCys occurred in the embryo axis [14].
SeMet as the dominant Se-species suggests that lentil-Se is likely to have good biological availability. Free SeMet is well utilized [8], being actively absorbed by the methionine transport system and then trans-selenated to SeCys, which is catabolized to selenide, the obligate form of Se incorporated into selenoproteins [23]. SeMet is also incorporated, as a methionine mimics, in general protein synthesis; this nonspecific incorporation into proteins means that ingestion of SeMet supports significant tissue levels of Se [8, 23]. Because the same phenomenon occurs in plants, SeMet in lentil proteins may determine its bioavailability. We found that Se from Canadian lentils (727 μg Se per kg) was effective in raising blood Se concentrations in healthy Sri Lankan children [24]; children fed with 50 g Se-rich red lentils per day had significantly higher blood Se concentrations 2 h after their meal (82 ppb) compared to children fed with local lentils (64 ppb). Thus, we believe that lentils containing nutritionally significant amounts of bioavailable Se can make important contributions to public health, particularly in South Asia. Such lentils can be provided by sourcing from Se-rich production areas, by selecting lentil cultivars that efficiently utilize soil Se, or by using Se-containing fertilizers.
Sourcing High-Se Lentils
Soils are highly variable with respect to the distribution of Se in forms that can be utilized by plants. Most soils contain between 1.0 and 1.5 μg Se per kg [25]; those with <0.6 μg/kg are considered deficient. The species of Se in soils depend on pH, aeration, organic matter content, texture, microbial activity, and the presence/absence of competitive ions (sulphate, phosphate) and organic compounds such as polysaccharides [26]. Selenate (VI) tends to be the major species in aerobic and neutral to alkaline soils, whereas selenide (-II) and elemental Se (0) dominate in anaerobic soils [27]. In acidic soils, Se is poorly available to plants, occurring mainly as insoluble selenides. In lateritic soils, it binds strongly to Fe to form insoluble and unavailable ferric hydroxide-selenite complexes. Selenate is water-mobile and can be leached from soils, as has occurred in New Zealand and Tasmania [28]. Consequently, some regions of the world have soils with low or unavailable Se, resulting in a food chain that is Se-deficient; other regions with higher amounts of plant-available Se support the production of foods with greater Se contents. Examples of the latter include the major lentil growing regions of Saskatchewan, Canada, which have soil Se concentrations of 37–301 μg/kg [14].
A survey of the Se content of lentils grown in six major regions shows considerable variation, with Turkey and Morocco producing very low-Se lentils (<30 μg/kg), Nepal and Australia producing moderate-Se lentils (140–180 μg/kg), and Saskatchewan and North Dakota producing high-Se lentils (<1,600 μg/kg) (Fig. 20.1). These differences reflect differences in soil Se content, with soils in parts of the Northern Great Plains of North America being rich in plant-available forms of Se.
Total Se concentration in lentils from different lentil growing countries (modified with new data from [24]). Number of samples from each region: Syria (n = 64), Nepal (n = 255), Morocco (n = 72), the USA (Pullman, Washington, n = 216), the USA (North Dakota, n = 150), Australia (n = 57), Turkey (n = 74), Canada (n = 912). Lentil seed selenium concentrations of major lentil producing and exporting countries
Utilizing Se-Efficient Cultivars
Lentils grown in the Se-rich soils of Saskatchewan are naturally rich in Se (425–673 μg/kg), with some genotypes having 40–50 % more Se than others [14]. The extra-small genotype, CDC Robin, and two of the large green lentil genotypes, CDC Sedley and CDC Grandora, had the greatest Se concentrations at 612–672 μg/kg. A single 100 g serving of such lentils would provide as much as 77–122 % of an adult’s RDA of Se (Fig. 20.2).
Frequency distribution of Se uptake in lentils grown in Saskatchewan, Canada (data from Thavarajah et al. [14]). %RDA was calculated based on the mean Se concentration across eight location for 100 g of lentils. Variation in lentil genotypes in their ability to enrich seed selenium concentrations and %RDA of selenium from 100 g serving
Enhancing the efficiency of soil-Se uptake by lentils would be possible through selective breeding, using the approach termed “biofortification.” The uptake of soils Se by the lentil plant is likely governed by several genes. The broad sense heritability of Se content in Canadian grown lentils was 40 %, with 4–5-fold differences in Se uptake among genotypes [14]. This suggests it may be worthwhile to screen available lentil germplasm for grain Se levels as a proxy for Se uptake capacity.
Selenium Fertilization
Selenium fertilization can increase crop Se contents. Allaway et al. [29] demonstrated that application of sodium selenate to Se-deficient Oregon, USA, soils increased the Se concentration of alfalfa (Medicago sativa L.) from 0.01–0.04 to 2.6–2.7 mg/kg. Since then, the efficacy of Se-fertilization has been well established as a means to prevent Se deficiencies in North America, Australia, Finland, and New Zealand [8, 30]. Se-containing fertilizers (16 mg/kg as sodium selenate) have been used in Finland since 1984 to elevate Se concentrations in their major food crops, and this practice resulted in increases in the daily Se intake of Finnish consumers from 39 to 110 μg of Se per day [31]. While either selenate or selenite can be used for Se-fertilization, selenate appears to be used by plants with greater efficacy. This has been shown for barley (Hardeum vlugare L.) [32, 33], red clover (Trifolium pretense L.) [34], perennial ryegrass (Lolium perenne L.) [35], and wheat (Triticum aestivum L.) [36]. Accordingly, we have found selenate more effective than selenite for the Se-fertilization of lentils, with application of aqueous solutions containing 2 ppm Se resulting in seed Se concentrations to 2.5–8.7 mg/kg [37] (Table 20.3). The mechanism of selenate uptake by plants was discussed above and plant roots are known to update selenate through the high affinity sulphate transporters [38, 39]. However, mechanism of selenite uptake is not well understood in plants and has yet to be studied in lentil. Arvy [40] suggests that selenite is taken up by plant roots through passive diffusion, yet a recent study indicates that uptake in wheat is an active process mediated through phosphate transporters. We have found that lentil genotypes differ in their responses to soil applications of Se. For example, the small green lentil genotype Eston shows a significantly greater increase in Se uptake than other genotypes in high soil Se environments [37] (Fig. 20.3). Moreover, Se uptake in lentils is dose-dependent, with doses of 5–10 mg/kg increasing lentil Se concentrations 2–4-fold (Fig. 20.3).
Foliar application can be more effective than soil application of Se-fertilizer for increasing crop Se content [41]. Foliar application of Se has been demonstrated effective for the Se-enrichment of wheat [36], barley [41], rice [42], broccoli [43], and chicory [44]. Foliar application of Se has not been investigated for lentils, but there is no reason to expect it will be less effective than application of Se to the soil.
Selenium is not considered an element essential for higher plants. However, benefits have been reported for plants treated with Se [43, 45]. These include increased tuber yield in potato (Solanum tuberosum L.) and increased vegetative growth in rye grass (Lolium perenne L.) and lettuce (Lactuca sativa L.) exposed to UVB radiation [46, 47]. Lyons et al. [45] found a 43 % yield increase in mustard (Brassica rapa L.) in response to a low dose (22 μL of 0.001 M selenite) of Se to the soil, and noted that increased respiratory activity in leaves and flowers may have contributed to the greater seed production.
Conclusion
Lentil is an important food crop in many parts of the world. It also can contribute to the growing global demand for protein foods. Lentil is produced in and exported from regions relatively rich in the essential nutrient Se, which is limiting in several parts of the world. Lentil can also be enriched in Se through the use of Se-fertilizers. Moreover, this pulse crop shows genetic potential for biofortification in Se, which could further enrich Se in seeds produced in marginal soils. Current evidence suggests that lentils so enriched, when consumed in modest amounts (50–100 g/day), can provide daily Se needs. Thus, lentils may be a sustainable food-based approach to malnutrition, including the prevention of Se deficiency. Similar strategies are likely to be efficacious for other pulses.
Abbreviations
- Fe:
-
Iron
- HPLC-ICP-MS:
-
High performance liquid chromatography inductively coupled plasma mass spectrometry
- N:
-
Nitrogen
- RDA:
-
Recommended daily allowance
- Se:
-
Selenium
- SeCys:
-
Selenocystein
- SeMet:
-
Selenomethionine
- XAS:
-
X-ray absorption spectroscopy
- Zn:
-
Zinc
References
Berry MJ, Banu L, Chen Y, Mandel SJ, Kieffer JD, Harney JW, et al. Recognition of a UGA as a selenocysteine codon in Type 1 deiodinase requires sequences in the 3′ untranslated region. Nature (London). 1991;353:273–6.
Kryukov GV, Castellano S, Novoselov SV, Lobanov AV, Zehtab O, Guigo R, et al. Characterization of mammalian selenoproteomes. Science. 2003;300(5624):1439–43.
Brigelius-Flohé R. Glutathione peroxidases and redox-regulated transcription factors. Biol Chem. 2006;387:1329–35.
Tamura T, Stadman TC. A new selenoprotein from human lung adenocarcinoma cells: purification, properties, and thioredoxin reductase activity. Proc Natl Acad Sci U S A. 1996;93:1006–11.
Burk RF, Hill KE. Selenoprotein P-expression, functions and roles in mammals. Biochim Biophys Acta. 2009;1790:1441–7.
Vandeland SC, Beilstein MA, Yeh JY, Ream W, Whanger PD. Rat skeletal muscle selenoprotein W: cDNA clone and mRNA modulation by dietary selenium. Proc Natl Acad Sci U S A. 1995;92:8749–53.
Kim HY, Gladyshev VN. Mineral element composition of Finnish foods. Mol Biol Cell. 2004;15:1055–64.
Combs Jr GF. Selenium in global food systems. Br J Nutr. 2001;85(5):517–47.
Ellis DR, Salt DE. Plants, selenium and human health. Curr Opin Plant Biol. 2003;6(3):273–9.
Spallholz JE, Mallory BL, Rhaman MM. Environmental hypothesis: is poor dietary selenium intake an underlying factor for arsenicosis and cancer in Bangladesh and West Bengal, India? Sci Total Environ. 2004;323(1–3):21–32.
Combs Jr GF, Lu J. Selenium as a cancer preventive agent. In: Hatfield DL, Berry MJ, Gladyshev VN, editors. Selenium: its molecular biology and role in human health. 2nd ed. New York: Springer; 2001. p. 205–17.
Jackson MI, Combs Jr GF. Selenium and anticarcinogenesis: underlying mechanisms. Curr Opin Clin Nutr Metab Care. 2008;11:718–26.
Clark LC, Combs Jr GF, Turnbull BW, Slate EH, Chalker DK, Chow J, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA. 1996;276:1957–63.
Thavarajah D, Ruszkowski J, Vandenberg A. High potential for selenium biofortification of lentils (Lens culinaris L.). J Agric Food Chem. 2008;56(22):10747–53.
FAOSTAT Website. Available at http://faostat.fao.org/. Accessed 5 May 2010.
Vandenberg A. Expansion of lentil in Canada. In: Masood A, Kumar S, editors. Milestones in food legume research. Kanpur: IIPR; 2009. p. 58–72.
Northern Pulse Growers Association Website. Available at http://www.northernpulse.com/. Accessed 5 June 2011.
Unkovich MJ, Pate JS. An appraisal of recent field measurements of symbiotic N2 fixation by annual legumes. Field Crop Res. 2000;65:211–28.
Saskatchewan Pulse Growers Association Website. Available at http://www.saskpulse.com/. Accessed 5 June 2011.
Thavarajah P, Thavarajah D, Vandenberg A. Low phytic acid lentils (Lens culinaris L.): a potential solution for increased micronutrient bioavailability. J Agric Food Chem. 2009;57:9044–9.
Olson OE, Novacek EJ, Whitehead EI, Palmer IS. Investigations on selenium in wheat. Phytochemistry. 1970;9:1181–8.
Thavarajah D, Vandenberg A, George GN, Pickering IJ. Chemical form of selenium in naturally selenium-rich lentils (Lens culinaris L.) from Saskatchewan. J Agric Food Chem. 2007;55(18):7337–41.
Rayman MP. The argument for increasing selenium intake. Proc Nutr Soc. 2002;61:203–15.
Thavarajah D, Thavarajah P, Wejesuriya A, Rutzke M, Glahn R, Combs Jr GF, et al. The potential of lentil (Lens culinaris L.) as a whole food for increased selenium, iron, and zinc intake: preliminary results from a three year study. Euphytica. 2011;180:123–8.
Berrow ML, Ure AM. Geological materials and soils. In: Ihnat M, editor. Occurrence and distribution of selenium. Boca Raton, FL: CRC Press; 1989. p. 226–8.
Pezzarossa B, Petruzzelli G, Petacco F, Malorgio F, Ferri T. Chemosphere. Absorption of selenium by Lactuca sativa as affected by carboxymethylcellulose. Chemosphere. 2007;67:322–9.
Terry N, Zayed AM, De Souza MP, Tarun AS. Selenium in higher plants. Annu Rev Plant Physiol Plant Mol Biol. 2000;51:401–32.
Reilly C. Selenium: a new entrant into the functional food arena. Trends Food Sci Technol. 1998;9:114–8.
Allaway WH, Moore DP, Oldfield JE, Muth OH. Movement of physiological levels of selenium from soils through plants to animals. J Nutr. 1966;88:411–8.
Lee GP, Park KW. Quality improvement of ‘Seoul’ celery by selenium in the nutrient solution culture. Acta Hortic. 1999;483:185–92.
Wang WC, Naentoe V, Maekelae AL, Maekelae P. Effect of nationwide selenium supplementation in Finland on selenium status in children with juvenile rheumatoid arthritis. A ten-year follow-up study. Analyst. 1995;120:955–8.
Ylaranta T. Effect of applied selenite and selenate on the selenium content of barley (Hordeum vlugare). Ann Agric Fenn. 1983;22:164–74.
Gupta UC, Winter KA, Sunderson JB. Selenium content of barley as influenced by selenite and selenate enriched fertilizers. Commun Soil Sci Plant Anal. 1993;24:1165–70.
Flemming GA. Some factor affecting the uptake of selenium by plants. Iris J Agric Res. 1962;1:131–8.
Poggi V, Arcioni A, Filippini P, Pifferi PG. Foliar application of selenite and selenate to potato (Solanum tuberosum): effect of a ligand agent on selenium content of tubers. J Agric Food Chem. 2000;48:4749–51.
Lyons GH, Judson GJ, Ortiz-Monasterio I, Genc Y, Stangoulis JCR, Graham RD. Selenium in Australia: selenium status and biofortification of wheat for better health. J Trace Elem Med Biol. 2005;19:75–82.
Thavarajah D, Vandenberg A. Genetic improvement of bioavailable selenium content in lentil seeds. Final report, Agriculture Development Fund (project # 20070186), Saskatchewan Pulse Growers Association (project # BRE0802), 28 Feb 2011.
White PJ, Bowen HC, Parmaguru P, Fritz M, Spracklen WP, Spiby RE, et al. Interactions between selenium and sulphur nutrition in Arabidopsis thaliana. J Exp Bot. 2004;55:1927–37.
Li HF, McGrath SP, Zhao FJ. Selenium uptake, translocation and speciation in wheat supplied with selenate or selenite. New Phytol. 2008;178:92–102.
Arvy MP. Selenate and selenite uptake and translocation in bean plant (Phaseolus vulgaris). J Exp Bot. 1993;44:1083–7.
Gupta UC, MacLeod JA. Effect of various sources of selenium fertilization on the selenium concentration of feed crops. Can J Soil Sci. 1994;74:285–90.
Fang Y, Wang L, Xin Z, Zhao L, An X, Hu Q. Effect of foliar application of zinc, selenium and iron fertilizers on nutrients concentration and yield of rice grain in China. J Agric Food Chem. 2008;56:2079–84.
Finley JW. Selenium accumulation in plant foods. Nutr Rev. 2005;1:196–202.
Germ M, Stibilj V, Osvald J, Kreft I. Effect of selenium foliar application on chicory (Cichorium intybus L.). J Agric Food Chem. 2007;55:795–8.
Lyons GH, Gene Y, Soole K, Stangoulis JCR, Liu F, Graham RD. Selenium increases seed production in Brassica. Plant Soil. 2009;318:73–80.
Turakainen M, Hartikainen H, Seppanen MM. Effects of selenium treatments on potato (Solanum tuberosum L.) growth and concentration of soluble sugars and starch. J Agric Food Chem. 2004;52:5378–82.
Hartikainen H, Xue T. The promotive effect of selenium on plant growth as triggered by ultraviolet irradiation. J Environ Qual. 1999;28:1372–5.
Bhatty RS. Composition and quality of lentil (Lens culinaris Medik): a review. Can Inst Food Sci Technol. 1988;21(2):144–60.
Acknowledgements
The authors thank members, collaborators, and funding agencies of the North Dakota State University Pulse Quality and Nutrition Program (2010–present), Fargo, ND, USA and the University of Saskatchewan Pulse Biofortification Team (2007–2010), Saskatoon, SK, Canada.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Thavarajah, D., Thavarajah, P., Combs, G.F. (2013). Lentils (Lens culinaris L.) as a Source of Dietary Selenium. In: Preedy, V., Srirajaskanthan, R., Patel, V. (eds) Handbook of Food Fortification and Health. Nutrition and Health. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4614-7076-2_20
Download citation
DOI: https://doi.org/10.1007/978-1-4614-7076-2_20
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4614-7075-5
Online ISBN: 978-1-4614-7076-2
eBook Packages: MedicineMedicine (R0)