Abstract
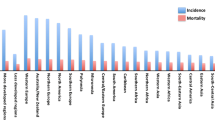
Breast cancer is the most common cause of cancer death among women worldwide, and breast cancer is by far the most frequent cancer among women, with an estimated 1.38 million new cancer cases diagnosed in 2008 (23% of all cancers), and ranks second overall (10.9% of all cancers). It is now the most common cancer both in developed and developing regions with approximately 690,000 new cases estimated for each region (population ratio 1:4). Incidence rates vary from 19.3 per 100,000 women in Eastern Africa to 89.7 per 100,000 women in Western Europe, and are higher (greater than 80 per 100,000) in the developed regions of the world (except Japan) and lower (less than 40 per 100,000) in most of the developing regions [1].
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Breast Cancer
- Breast Cancer Risk
- Hormone Replacement Therapy
- Breast Cancer Incidence
- Reproductive Factor
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Breast cancer is the most common cause of cancer death among women worldwide, and breast cancer is by far the most frequent cancer among women, with an estimated 1.38 million new cancer cases diagnosed in 2008 (23% of all cancers), and ranks second overall (10.9% of all cancers). It is now the most common cancer both in developed and developing regions with approximately 690,000 new cases estimated for each region (population ratio 1:4). Incidence rates vary from 19.3 per 100,000 women in Eastern Africa to 89.7 per 100,000 women in Western Europe, and are higher (greater than 80 per 100,000) in the developed regions of the world (except Japan) and lower (less than 40 per 100,000) in most of the developing countries [1].
The range of mortality rates is much less in (high-incidence) developed regions (approximately 6–19 per 100,000) because of the more favorable survival of breast cancer. As a result, breast cancer ranks as the fifth cause of death from all cancers (458,000 deaths), but it is still the most frequent cause of cancer deaths in women in both developing (269,000 deaths, 12.7% of total) and developed regions, where the estimated 189,000 deaths is almost equal to the estimated number of deaths from lung cancer (188,000 deaths) [1]. Incidence rates are relatively higher in more developed countries, whereas rates are lower but increasing in less developed countries [2]. According to the standardized incidence rates of breast cancer in countries such as France and Belgium, Denmark and the Netherlands, and the United States, as well as in the developed countries of Western Europe, it is the most common cancer in women in developed countries (109.19/100.000; 101.12/100.000; 99.74/100.000; 98.46/100.000; respectively) [1, 3, 4].
Higher breast cancer incidence rates detected in developed countries can be attributed to a variety of reasons, such as higher standards of living, early menarche age, late age of pregnancy and birthing, fewer pregnancies, and also easy accessibility to the use of techniques such as mammography, early diagnosis opportunities, and adequate data recording systems. Breast cancer rates fell over the last 10 years worldwide, and this downward trend, seen especially in developing countries, may be linked to the reduction of the use of hormone replacement therapy (HRT) during menopause [2, 5].
2 Breast Cancer Risk Factors
2.1 Reproductive Factors
Environment, genes, and lifestyle seem to cooperate in increasing or decreasing the probability of development of female breast cancer [6], and this complicated causal relationship can lead to more difficulties in the prevention of the disease. The World Health Organization declared in an August 2005 report that estrogen is carcinogenic in women, and that estrogen exposure is associated with lifelong risk of breast cancer. Studies have also displayed the relationship between endogenous hormone levels and breast cancer [7].
Where the lifetime exposure to endogenous sex hormones is concerned, life stages of women have come to the fore. Younger age at menarche, older age at menopause, late age at first full-term pregnancy, and number of pregnancies have been the subject of various research studies investigating the relationship between the risk of breast cancer and hormones. As a result, it has been proved that women below the age of 12 years at onset of menarche have a higher risk of breast cancer as a result of a longer period of estrogen exposure at the breast epithelium. It seems the same causal mechanism is valid for the older ages of the menopause. For late age onset of menopause, each 1-year delay can result in a 3% increase in the risk of breast cancer [8].
Multiple full-term pregnancies and early age of pregnancy decrease the risk of breast cancers and can be considered as protective situations. Women who experience pregnancy when they are younger than age 20 years carry approximately half of the risk of women whose first pregnancy was older than age 30 years. The mechanism can be linked to early maturation of breast tissue in accordance with pregnancy, with breast-feeding showing a similar protective effect [9–11]. The effects of the reproductive factors are related to the effects of progesterone and estrogen receptors. The relative risk of the reproductive factors is shown in Table 1.1.
2.2 Obesity
Obesity has been associated with a twofold increase in the risk of breast cancer in postmenopausal women, whereas it has been correlated with a reduced incidence of breast cancer among premenopausal women [12].
Endogenous estrogen in postmenopausal women is primarily found in adipose tissue, secondary to the aromatization of adrenal androgens, with a higher risk of developing breast cancers. A study found that women who gained 55 pounds or more after the age of 18 years had an approximately 55% greater risk of breast cancer compared with those who maintained their weight. A gain of 22 pounds or more after menopause was associated with an 82% greater risk of breast cancer, whereas losing at least 22 pounds after menopause and maintaining weight were associated with a trend toward decreased breast cancer risk [13].
2.3 Diet
The observation that breast cancer rates are much higher in the countries with high-fat diets than underdeveloped countries and Japan, where fat intake is recognized as being much lower, it has been suggested that high-fat diets might increase the risk of breast cancer. However, it was reported in a pooled analysis of seven prospective studies no association was found between fat intake and breast cancer risk among adult women living in more developed countries [14, 15]. These results do not entirely exclude any significant effect of fat on breast cancer, while there would be a considerable error in measurement of fat intake by dietary questionnaires [2].
Excessive fat intake can increase breast cancer risk by increasing the endogenous estrogen levels. Further studies are required to confirm the effect of diet on breast cancer.
2.4 Oral Contraceptive Use
Key et al. reported that the overall effect of oral contraceptive use on the risk of breast cancer among young premenopausal women is low because the small increase in relative risk is acting on a very low background risk. In older premenopausal women, oral contraceptives can be a less favorable choice because of the absolute risk of breast cancer, as well as other health hazards which increase rapidly at ages from 40–50 years [2]. Additionally, in their randomized controlled trial, Rossouw et al. pointed out that after an average follow-up of approximately 5 years, combined oral contraceptive therapy (estrogen plus progesterone) has a 26% higher risk then therapy using only estrogen, and also reported a 15% increase for estrogen plus progestin use for less than 5 years, with a 53% increase for use for more than 5 years [16].
2.5 Hormone Replacement Therapy
The Million Women Study, a cohort study of all quarter of British women between the ages of 50–64 years, was set up especially to investigate the relation between various patterns of HRT use and breast cancer incidence and mortality. It was reported in the study that the use of HRT at that time was associated with an increased risk of incidence and fatal breast cancer; the effect was found to be substantially greater for estrogen-progesterone combinations compared with other types of HRT (relative risks [RR] have been reported; for combined treatment of estrogen-progesterone, RR = 2.00; for estrogen, RR = 1.30; and for Tibolon, RR = 1.45) [17]. Similar findings were reported by Magnusson et al., who found that users of hormonal therapy for menopause are considered at higher risk of breast cancer compared with women who have never used these therapies [18].
The results from the Million Women Study reported that minimal or no overall increase was detected in RR of breast cancer in past users of HRT.
2.6 Smoking
In many cohort studies conducted in recent years, it has been suggested that smoking increases the risk of breast cancer, especially among women who smoked cigarettes for a long period of time or who started smoking at a young age [19, 20]. Additionally, in a larger prospective cohort study by Luo et al., such associations have been further confirmed in postmenopausal women [21].
Additionally, Luo et al. also reported that they observed a significantly increased risk of breast cancer associated with the amount and duration of smoking among nonobese women. The mechanism behind this relation can mainly be attributed to the carcinogenic effects of tobacco smoking on breast tissue [22].
2.7 Alcohol
Smith-Warner et al. reported that alcohol consumption was associated with a linear increase in breast cancer incidence in women [23]. In a similar study, Chen et al. reported that low levels of alcohol consumption were associated with a small increase in breast cancer risk, with the most consistent measure being cumulative alcohol intake throughout the adult life. Alcohol intake both at earlier and later ages of adult life was independently associated with breast cancer risk [24].
2.8 Heredity
Various aspects of genetic transition of cancer can be monitored. Historically, French surgeon Paul Broca had identified a large number of breast cancer host families in 1860 [25]. Since then, researchers have confirmed Broca’s insight. Presently, it has been proved that 5–10% of all breast cancers were a result of mutations in the genes at high risk, such as BRCA1 and BRCA2 (familial risks of breast cancer) [26].
Patients having these mutations and indicating the inheritance of Mendel (i.e., dominant, recessive, X-linked) defined a hereditary breast cancer. When the family history is suggestive of a hereditary predisposition, but the number or distribution of the cancers is not definitive, then families are described as having familial cancer [27]. Specific genetic alterations have been identified for many of the established hereditary breast cancer syndromes that are responsible for 10% of all breast cancer cases. Hereditary breast-ovarian cancer (HBOC) syndrome is an inherited tendency to develop breast, ovarian, and other cancers. Although most cancers are not inherited, approximately 5% of those with breast cancer and approximately 10% of women with ovarian cancer also possess HBOC. Approximately 80–90% of cases of hereditary breast and ovarian cancers are caused by mutations in the BRCA1 and BRCA2 genes [28]. A study reviewing 22 trials reported that the average cumulative risks among BRCA1-mutation carriers at age 70 years were determined as 65% for breast cancer and 39% for ovarian cancer. The corresponding estimates for BRCA2 were 45% and 11%, respectively [29, 30].
Typically, the BRCA1 mutation carriers increase rapidly, as well as high-grade, estrogen receptor negative breast tumors in premenauposal women. Foulkes et al. also reported that BRCA1 serves as a breast stem cell regulator [31].
Further studies have revealed that women with mutations in either the BRCA1 or BRCA2 gene have a predicted lifetime risk of breast cancer between 37% and 85%, and a lifetime risk of ovarian cancer between 15% and 40% [27, 32, 33].
The p53 tumor suppressor protein can prevent carcinogenesis. In breast cancers, p53 is mutated in almost 30% of cases, with a higher frequency in some tumor subtypes. Tumor p53 mutation is reported to be a factor for good prognosis in some studies, while in others it is a factor for poor prognosis [34]. In breast cancers, the p53 inactivation was 20–30%. The presence of mutations on p53 can demonstrate tumor transition from in situ to invasive carcinoma as a marker. Li-Fraumeni syndrome (LFS) is a classic cancer predisposition disorder that is commonly associated with germ-line mutations of the p53 tumor suppressor gene. Germ-line mutations in p53 predispose to LFS, including childhood sarcomas and brain tumors, as well as early-onset breast cancer [35, 36]. The risk of breast cancer is 50–60% by age 45 years [34] and those in pentaerythritol tetranitrate are responsible for Cowden disease, in which breast cancer is a major feature and the risk of breast cancer is 25–50% [27, 33].
3 Molecular Epidemiology of the Breast Cancer
Breast cancers, which have different clinical properties, pathologic features, morphology, grade, and hormone receptors, are very different biological and clinically heterogeneous groups of diseases. Breast cancer is classified jointly by estrogen receptor (ER) and progesterone receptor (PR) status in human epidermal growth factor receptor-2 (Her-2). Her-positive tumors are observed in 60–70% under the age of 35 years and in 80% over the age of 60 years [1], which also shows the heterogeneous etiology. In all age groups, rates of Her-2 positive and Her-2 negative are quite similar. Risk factors for breast cancer include obesity, lifestyle factors, reproductive factors, as well as some environmental and postmenopausal obesity lifestyle factors. ER and/or PR may indicate a tight relationship [37, 38]; although no relationship between genetics and ER/PR receptor status was seen [39]. Mavaddat et al. reported that they could not find any association between ER-positive and ER-negative receptors, but under the age of 50 years, the ER-negative disease rate is high in relations and family [40].
Gene expression profiling in tumor tissues with polymerase chain reaction suggests that breast cancers may be divided into subtypes consisting of two ER-positive types (luminal A and luminal B) and three ER-negative types (Her-2), expressing basal-like, and unclassified “normal-like”, with distinctive clinical outcomes [41, 42]. Yang et al. [43, 44] and Gaudet et al. [43, 44] reported that they observed significant differences in terms of tumor subtypes for the distribution of age at menarche, age at first full-term birth, and body mass index (BMI) among premenopausal women. Basal-like tumors were related to the youngest age at menarche and highest BMI among premenopausal women, whereas luminal A tumors were associated with the oldest mean age at first full-term birth.
The Carolina Breast Cancer Study also investigated the risk factors that could cause such differences. They reported that a number of risk factors depending on the subtype of tumor were also observed, and the effects were opposed of each other. More importantly, having more than one child, the age of the completed first pregnancy was found to be a protective factor for luminal A, in turn these factors are found as predisposing to basal-like cancers [45]. Millikan et al. reported other well-known risk factors showing a different behavior according to the subgroups. For example, a measure of obesity with high waist-to-hip ratio also increases susceptibility to ER-negative and ER-positive cancers. Younger menarche age, Multiparity, the age at first full-term pregnancy (younger age), increased waist-to-hip ratio are considered as factors increasing the risk of triple-negative breast cancer. However, the number of births and the age at first birth showed no difference between the triple-negative group and the other groups [46].
Cancer stem cell hypothesis shows that breast stem cell populations targeting strategies for prevention and treatment of breast cancer are very efficient, and are shown in Figure 1.1.
4 Conclusion
Breast cancer should be regarded as one of the most important cancers affecting women in the future, therefore, there is a need for prevention. There would be great benefit of education programs aimed at promoting breastfeeding, at the reduction of abdominal obesity, promoting a healthy lifestyle and healthy nutrition, especially in preventing bad progressive types of breast cancer progression.
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. GLOBOCAN 2008 v1.2, Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 10 [Internet]. Lyon, France: International Agency for Research on Cancer; 2010. Available from: http://globocan.iarc.fr. Accessed 04 Nov 2011.
Key TJ, Verkasalo PK, Banks E. Epidemiology of breast cancer. Lancet Oncol. 2001;2(3):133–40.
Desantis C, Siegel R, Bandi P, Jemal A. Breast cancer statistics, 2011. CA Cancer J Clin. 2011;61(6):409–18. doi:10.3322/caac.20134. Epub 2011 Nov 26.
Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61(4):212–36. Epub 2011 Nov 26.
Héry C, Ferlay J, Boniol M, Autier P. Changes in breast cancer incidence and mortality in middle-aged and elderly women in 28 countries with Caucasian majority populations. Ann Oncol. 2008;19(5):1009–18. Epub 2008 Feb 21.
Hartge P. Genes, hormones, and pathways to breast cancer. N Engl J Med. 2003;348:2352–4.
Dorgan JF, Longcope C, Stephenson Jr HE, Falk RT, Miller R, Franz C, et al. Serum sex hormone levels are related to breast cancer risk in postmenopausal women. Environ Health Perspect. 1997;105 Suppl 3:583–5.
Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53,297 women with breast cancer and 100,239 women without breast cancer from 54 epidemiological studies. Lancet. 1996;347:1713–27.
Kelsey JL, Gammon MD, John EM. Reproductive factors and breast cancer. Epidemiol Rev. 1993;15(1):36–47.
Butler LM, Potischman NA, Newman B, Millikan RC, Brogan D, Gammon MD, et al. Menstrual risk factors and early-onset breast cancer. Cancer Causes Control. 2000;11(5):451–8.
Russo J, Hu YF, Yang X, Russo IH. Developmental, cellular, and molecular basis of human breast cancer (in process citation). J Natl Cancer Inst Monogr. 2000;27:17–37.
McPherson K, Steel CM, Dixon JM. Breast cancer epidemiology, risk factors, and genetics. BMJ. 2000;321:624–8.
Eliassen AH, Colditz GA, Rosner B, et al. Adult weight change and risk of postmenopausal breast cancer. JAMA. 2006;296:193–201.
Hunter DJ, Spiegelman D, Adami H-O, et al. Cohort studies of fat intake and the risk of breast cancer—a pooled analysis. N Engl J Med. 1996;334:356–61.
Davies NJ, Batehup L, Thomas R. The role of diet and physical activity in breast, colorectal, and prostate cancer survivorship: a review of the literature. Br J Cancer. 2011;105 Suppl 1:S52–73. doi:10,1038/bjc.2011.423.
Rossouw JE, Anderson GL, Prentice RL, LaCroix AZ, Kooperberg C, Stefanick ML, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the women’s health initiative randomized controlled trial. JAMA. 2002;288:321–33.
Beral V, Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2003;362(9382):419–27.
Magnusson CM, Baron J, Correia N, et al. Breast-cancer risk following long-term estrogen- and estrogen-progestin replacement therapy. Int J Cancer. 1999;81:339–44.
Cui Y, Miller AB, Rohan TE. Cigarette smoking and breast cancer risk: update of a prospective cohort study. Breast Cancer Res Treat. 2006;100(3):293–9.
Xue F, Willett WC, Rosner BA, et al. Cigarette smoking and the incidence of breast cancer. Arch Intern Med. 2011;171(2):125–33.
Luo J, Margolis KL, Wactawski-Wende J, et al. Association of active and passive smoking with risk of breast cancer among postmenopausal women: a prospective cohort study [electronic article]. BMJ. 2011;342:d1016. doi:10.1136/bmj. d1016.
Luo J, Horn K, Ockene JK, Simon MS, Stefanick ML, Tong E, et al. Interaction between smoking and obesity and the risk of developing breast cancer among postmenopausal women: the Women’s Health Initiative Observational Study. Am J Epidemiol. 2011;174(8):919–28. Epub 2011 Aug 29.
Smith-Warner SA, Spiegelman D, Yaun SS, van den Brandt PA, Folsom AR, Goldbohm RA, et al. Alcohol and breast cancer in women: a pooled analysis of cohort studies. JAMA. 1998;279:535–40.
Chen WY, Rosner B, Hankinson SE, Colditz GA, Willett WC. Moderate alcohol consumption during adult life, drinking patterns, and breast cancer risk. JAMA. 2011;306(17):1884–90.
Lynch HT, Silva E, Snyder C, Lynch JF. Hereditary breast cancer: part I. Diagnosing hereditary breast cancer syndromes. Breast J. 2008;14(1):3–13. Epub 2007 Dec 11. Review.
Easton DF. http://www.ncbi.nlm.nih.gov/pubmed/12223120. Familial risks of breast cancer. Breast Cancer Res. 2002;4(5):179–81. Epub 2002 Aug 2.
Bradbury AR, Olopade OI. Genetic susceptibility to breast cancer. Rev Endocr Metab Disord. 2007;8(3):255–67. Epub 2007 May 17.
Thull DL, Vogel VG. Recognition and management of hereditary breast cancer syndromes. Oncologist. 2004;9(1):13–24.
Levy-Lahad E, Friedman E. Cancer risks among BRCA1 and BRCA2 mutation carriers. Br J Cancer. 2007;96(1):11–5.
Antoniou A, Pharoah PD, Narod S, Risch HA, Eyfjord JE, Hopper JL, et al. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case Series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet. 2003;72(5):1117–30. Epub 2003 Apr 3.
Foulkes WD. BRCA1 functions as a breast stem cell regulator. J Med Genet. 2004;41(1):1–5.
Struewing JP, Hartge P, Wacholder S, Baker SM, Berlin M, McAdams M, et al. The risk of cancer associated with specific utations of BRCA1 and BRCA2 among Ashkenazi Jews. N Engl J Med. 1997;336:1401–8.
Thompson D, Easton D. The genetic epidemiology of breast cancer genes. J Mammary Gland Biol Neoplasia. 2004;9(3):221–36.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57–70.
Malkin D. Li-fraumeni syndrome. Genes Cancer. 2011;2(4):475–84.
Lalloo F, Varley J, Moran A, Ellis D, O’dair L, Pharoah P, et al. BRCA1, BRCA2 and TP53 mutations in very early-onset breast cancer with associated risks to relatives. Eur J Cancer. 2006;42(8):1143–50. Epub 2006 Apr 27.
Colditz GA, Rosner BA, Chen WY, Holmes MD, Hankinson SE. Risk factors for breast cancer according to estrogen and progesterone receptor status. J Natl Cancer Inst. 2004;96:218–28.
Cotterchio M, Kreiger N, Theis B, Sloan M, Bahl S. Hormonal factors and the risk of breast cancer according to estrogen- and progesterone-receptor subgroup. Cancer Epidemiol Biomarkers Prev. 2003;12:1053–60.
Welsh ML, Buist DS, Aiello Bowles EJ, Anderson ML, Elmore JG, Li CI. Population-based estimates of the relation between breast cancer risk, tumor subtype, and family history. Breast Cancer Res Treat. 2009;114:549–58.
Mavaddat N, Pharoah PD, Blows F, Driver KE, Provenzano E, Thompson D, et al. Familial relative risks for breast cancer by pathological subtype: a population-based cohort study. Breast Cancer Res. 2010;12:R10.1–2.
Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumors. Nature. 2000;406:747–52.
Sorlie T, Tibshirani R, Parker J, et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA. 2003;100:8418–23.
Yang XR, Sherman ME, Rimm DL, Lissowska J, Brinton LA, Peplonska B, et al. Differences in risk factors for breast cancer molecular subtypes in a population-based study. Cancer Epidemiol Biomarkers Prev. 2007;16(3):439–43.
Gaudet MM, Press MF, Haile RW, Lynch CF, Glaser SL, Schildkraut J, et al. Risk factors by molecular subtypes of breast cancer across a population-based study of women 56 years or younger. Breast Cancer Res Treat. 2011;130(2):587–97. Epub 2011 Jun 11.
Carey LA, Perou CM, Livasy CA, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–502.
Millikan RC, Newman B, Tse CK, et al. Epidemiology of basal-like breast cancer. Breast Cancer Res Treat. 2008;109:123–39.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Kitapcioglu, G. (2013). Epidemiology and Etiology of Breast Cancer. In: Haydaroglu, A., Ozyigit, G. (eds) Principles and Practice of Modern Radiotherapy Techniques in Breast Cancer. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-5116-7_1
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5116-7_1
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-5115-0
Online ISBN: 978-1-4614-5116-7
eBook Packages: MedicineMedicine (R0)