Abstract
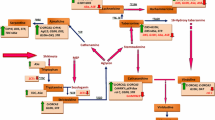
Madagascar periwinkle (Catharanthus roseus, family Apocynaceae) is a reservoir of more than 130 monoterpene indole alkaloids (MIAs) including the famous anti-neoplastic dimeric MIAs vinblastine and vincristine, and anti-hypertensive monomeric MIAs ajmalicine and serpentine. Understanding the biosynthetic steps and regulatory factors leading to the formation of MIAs is crucial for rational engineering to achieve targeted enhancement of different MIAs. Due to its highly recalcitrant nature, C. roseus is considered genetically non-tractable for transformation at the whole-plant level. Though few reports have demonstrated tissue culture-mediated regeneration and transformation of C. roseus at whole-plant level recently, the efficiency and reproducibility of these protocols have been a major challenge. To overcome this, we have developed a tissue-culture-independent Agrobacterium-mediated in planta transformation method in C. roseus. Using this method, we were able to efficiently generate stable transgenic plants without relying on the cumbersome methods of tissue-culture regeneration and transformation. Moreover, the transformed plants obtained through this in planta method exhibited stability in subsequent generations. Our method is useful not only for the elucidation of biosynthetic and regulatory steps involved in MIA formation through transgenic plant approach but also for metabolic engineering at the whole-plant level in C. roseus.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kellner F, Kim J, Clavijo BJ, Hamilton JP, Childs KL, Vaillancourt B, Cepela J, Habermann M, Steuernagel B, Clissold L, McLay K (2015) Genome-guided investigation of plant natural product biosynthesis. Plant J 82(4):680–692
Zhao L, Sander GW, Shanks JV (2013) Perspectives of the metabolic engineering of terpenoid indole alkaloids in Catharanthus roseus hairy roots. Adv Biochem Eng Biotechnol 134:23–54
Zhu X, Zeng X, Sun C, Chen S (2014) Biosynthetic pathway of terpenoid indole alkaloids in Catharanthus roseus. Front Med 8(3):285–293
Sottomayor M, De Pinto MC, Salema R, DiCosmo F, Pedreoo MA, Ros Barcelo A (1996) The vacuolar localization of a basic peroxidase isoenzyme responsible for the synthesis of α-31, 41-anhydrovinblastine in Catharanthus roseus (L.) G. Don Leaves. Plant Cell Environ 19(6):761–767
Costa MM, Hilliou F, Duarte P, Pereira LG, Almeida I, Leech M, Memelink J, Barceló AR, Sottomayor M (2008) Molecular cloning and characterization of a vacuolar class III peroxidase involved in the metabolism of anticancer alkaloids in Catharanthus roseus. Plant Physiol 146(2):403–417
Noble RL (1990) The discovery of the vinca alkaloids — chemotherapeutic agents against cancer. Biochem Cell Biol 68(12):1344–1351
Kuboyama T, Yokoshima S, Tokuyama H, Fukuyama T (2004) Stereocontrolled total synthesis of (+)-vincristine. Proc Natl Acad Sci 101(33):11966–11970
Jacobs DI, Snoeijer W, Hallard D, Verpoorte R (2004) The Catharanthus alkaloids: pharmacognosy and biotechnology. Curr Med Chem 11(5):607–628
Ishikawa H, Elliott GI, Velcicky J, Choi Y, Boger DL (2006) Total synthesis of (−)-and ent-(+)-vindoline and related alkaloids. J Am Chem Soc 128(32):10596–10612
Zárate R, Verpoorte R (2007) Strategies for the genetic modification of the medicinal plant Catharanthus roseus (L.) G. Don. Phytochem Rev 6(2–3):475–491
Guirimand G, Burlat V, Oudin A, Lanoue A, St-Pierre B, Courdavault V (2009) Optimization of the transient transformation of Catharanthus roseus cells by particle bombardment and its application to the subcellular localization of hydroxymethylbutenyl 4-diphosphate synthase and geraniol 10-hydroxylase. Plant Cell Rep 28(8):1215–1234
Verma P, Mathur AK (2011) Direct shoot bud organogenesis and plant regeneration from pre-plasmolysed leaf explants in Catharanthus roseus. Plant Cell Tiss Org 106(3):401–408
Pan Q, Wang Q, Yuan F, Xing S, Zhao J, Choi YH, Verpoorte R, Tian Y, Wang G, Tang K (2012) Overexpression of ORCA3 and G10H in Catharanthus roseus plants regulated alkaloid biosynthesis and metabolism revealed by NMR-metabolomics. PLoS One 7(8):e43038
Wang Y, van Kronenburg B, Menzel T, Maliepaard C, Shen X, Krens F (2012) Regeneration and agrobacterium-mediated transformation of multiple lily cultivars. Plant Cell Tiss Org 111(1):113–122
Kumar SR, Shilpashree HB, Nagegowda DA (2018) Terpene moiety enhancement by overexpression of geranyl (geranyl) diphosphate synthase and geraniol synthase elevates monomeric and dimeric monoterpene indole alkaloids in transgenic Catharanthus roseus. Front Plant Sci 9:942
Supartana P, Shimizu T, Nogawa M, Shioiri H, Nakajima T, Haramoto N, Nozue M, Kojima M (2006) Development of simple and efficient in planta transformation method for wheat (Triticum aestivum L.) using Agrobacterium tumefaciens. J Biosci Bioeng 102(3):162–170
Shah SH, Ali S, Jan SA, Ali GM (2015) Piercing and incubation method of in planta transformation producing stable transgenic plants by overexpressing DREB1A gene in tomato (Solanum lycopersicum Mill.). Plant Cell Tiss Org 120(3):1139–1157
Ben-Amar A, Cobanov P, Buchholz G, Mliki A, Reustle G (2013) In planta agro-infiltration system for transient gene expression in grapevine (Vitis spp.). Acta Physiol Plant 35(11):3147–3156
Yellisetty V, Reddy LA, Mandapaka M (2015) In planta transformation of sorghum (Sorghum bicolor (L.) Moench) using TPS1 gene for enhancing tolerance to abiotic stresses. J Genet 94(3):425–434
Keshamma E, Rohini S, Sankara Rao K, Madhusudhan B, Udaya Kumar M (2008) Tissue culture-independent in planta transformation strategy: an Agrobacterium tumefaciens-mediated gene transfer method to overcome recalcitrance in cotton (Gossypium hirsutum L.). J Cotton Sci 12(3):264–272
Jaganath B, Subramanyam K, Mayavan S, Karthik S, Elayaraja D, Udayakumar R, Manickavasagam M, Ganapathi A (2014) An efficient in planta transformation of Jatropha curcas (L.) and multiplication of transformed plants through in vivo grafting. Protoplasma 251(3):591–601
Kumar SR, Rai A, Bomzan DP, Kumar K, Hemmerlin A, Dwivedi V, Godbole RC, Barvkar V, Shanker K, Shilpashree HB, Bhattacharya A (2020) A plastid-localized bona fide geranylgeranyl diphosphate synthase plays a necessary role in monoterpene indole alkaloid biosynthesis in Catharanthus roseus. Plant J 103(1):248–265
Acknowledgments
This work was supported by the Department of Biotechnology (Govt. of India) supported project (GAP-272: BT/PR6109/AGII/106/857/2012) to D.A.N. D.P.B is the recipient of Research Fellowship from University Grant Commission (UGC) and enrolled under Academy of Scientific and Innovative Research (AcSIR). Shilpashree H.B is the recipient of ICMR JRF fellowship and enrolled under CIMAP-JNU PhD program. The institutional communication number for this article is CIMAP/PUB/2021/APR/21.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Bomzan, D.P., Shilpashree, H.B., Nagegowda, D.A. (2022). Agrobacterium-Mediated in Planta Transformation in Periwinkle. In: Courdavault, V., Besseau, S. (eds) Catharanthus roseus. Methods in Molecular Biology, vol 2505. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2349-7_22
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2349-7_22
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2348-0
Online ISBN: 978-1-0716-2349-7
eBook Packages: Springer Protocols