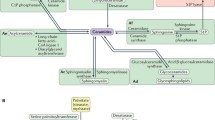
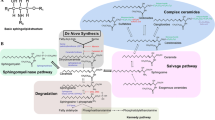
Abstract
The relationship between sphingolipid levels and NAFLD pathology has been recognized for some time. Numerous studies using pharmacological and genetic approaches in vitro and in animal models of NAFLD have demonstrated that modifications to sphingolipid metabolism can attenuate various facets of NAFLD pathology. However, a more precise understanding of the role of sphingolipids and NAFLD pathology is essential to creating therapeutics that target this pathway. This chapter touches on the scale and variety of sphingolipid metabolites at play in NAFLD, which vary widely in their chemical structures and biological functions. With advances in liquid chromatography and tandem mass spectrometry approaches, each of thousands of individual sphingolipid species and sphingolipid metabolites can be identified and precisely quantified. These approaches are beginning to reveal specific sub-classes and species of sphingolipids that change in NAFLD, and as such, enzymes that generate them can be identified and potentially serve as therapeutic targets. Advances in lipidomics technology have been, and will continue to be, critical to these gains in our understanding of NAFLD.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Rotthier A, Auer-Grumbach M, Janssens K et al (2010) Mutations in the SPTLC2 subunit of serine palmitoyltransferase cause hereditary sensory and autonomic neuropathy type I. Am J Hum Genet 87:513–522
Weyler J, Verrijken A, Hornemann T et al (2021) Association of 1-deoxy-sphingolipids with steatosis but not steatohepatitis nor fibrosis in non-alcoholic fatty liver disease. Acta Diabetol 58:319–327
Hornemann T, Richard S, Rutti MF et al (2006) Cloning and initial characterization of a new subunit for mammalian serine-palmitoyltransferase. J Biol Chem 281:37275–37281
Hornemann T, Wei Y, von Eckardstein A (2007) Is the mammalian serine palmitoyltransferase a high-molecular-mass complex? Biochem J 405:157–164
Lone MA, Hülsmeier AJ, Saied EM et al (2020) Subunit composition of the mammalian serine-palmitoyltransferase defines the spectrum of straight and methyl-branched long-chain bases. Proc Natl Acad Sci U S A 117:15591–15598
Hornemann T, Penno A, Rutti MF et al (2009) The SPTLC3 subunit of serine palmitoyltransferase generates short chain sphingoid bases. J Biol Chem 284:26322–26330
Gault CR, Obeid LM, Hannun YA (2010) An overview of sphingolipid metabolism: from synthesis to breakdown. Adv Exp Med Biol 688:1–23
Park WJ, Park JW, Merrill AH et al (2014) Hepatic fatty acid uptake is regulated by the sphingolipid acyl chain length. Biochim Biophys Acta Mol Cell Biol Lipids 1841:1754–1766
Raichur S, Wang ST, Chan PW et al (2014) CerS2 haploinsufficiency inhibits β-oxidation and confers susceptibility to diet-induced steatohepatitis and insulin resistance. Cell Metab 20:687–695
Montefusco DJ, Allegood JC, Spiegel S et al (2018) Non-alcoholic fatty liver disease: insights from sphingolipidomics. Biochem Biophys Res Commun 504:608–616
Summers SA (2020) Ceramides: nutrient signals that drive hepatosteatosis. J Lipid Atheroscler 9:50–65
Zhou H, Summers SA, Birnbaum MJ et al (1998) Inhibition of Akt kinase by cell-permeable ceramide and its implications for ceramide-induced apoptosis. J Biol Chem 273:16568–16575
Summers SA, Garza LA, Zhou H et al (1998) Regulation of insulin-stimulated glucose transporter GLUT4 translocation and Akt kinase activity by ceramide. Mol Cell Biol 18:5457–5464
Chavez JA, Summers SA (2003) Characterizing the effects of saturated fatty acids on insulin signaling and ceramide and diacylglycerol accumulation in 3T3-L1 adipocytes and C2C12 myotubes. Arch Biochem Biophys 419:101–109
Xia JY, Holland WL, Kusminski CM et al (2015) Targeted induction of ceramide degradation leads to improved systemic metabolism and reduced hepatic steatosis. Cell Metab 22:266–278
Turpin SM, Nicholls HT, Willmes DM et al (2014) Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab 20:678–686
Lee AY, Lee JW, Kim JE et al (2017) Dihydroceramide is a key metabolite that regulates autophagy and promotes fibrosis in hepatic steatosis model. Biochem Biophys Res Commun 494:460–469
Guri Y, Colombi M, Dazert E et al (2017) mTORC2 promotes tumorigenesis via lipid synthesis. Cancer Cell 32:807–823.e12
Apostolopoulou M, Gordillo R, Koliaki C et al (2018) Specific hepatic sphingolipids relate to insulin resistance, oxidative stress, and inflammation in nonalcoholic steato hepatitis. Diabetes Care 41:1235–1243
Kumar DP, Caffrey R, Marioneaux J et al (2020) The PPAR α/γ agonist saroglitazar improves insulin resistance and steatohepatitis in a diet induced animal model of nonalcoholic fatty liver disease. Sci Rep 10(1):9330
Wang X, Zhang DM, Gu TT et al (2013) Morin reduces hepatic inflammation-associated lipid accumulation in high fructose-fed rats via inhibiting sphingosine kinase 1/sphingosine 1-phosphate signaling pathway. Biochem Pharmacol 86:1791–1804
Liao CY, Song MJ, Gao Y et al (2018) Hepatocyte-derived lipotoxic extracellular vesicle sphingosine 1-phosphate induces macrophage chemotaxis. Front Immunol 9:2980
Mauer AS, Hirsova P, Maiers JL et al (2017) Inhibition of sphingosine 1-phosphate signaling ameliorates murine nonalcoholic steatohepatitis. Am J Physiol Gastrointest Liver Physiol 312:G300–G313
Geng T, Sutter A, Harland MD et al (2015) SphK1 mediates hepatic inflammation in a mouse model of NASH induced by high saturated fat feeding and initiates proinflammatory signaling in hepatocytes. J Lipid Res 56:2359–2371
Rohrbach TD, Asgharpour A, Maczis MA et al (2019) FTY720/fingolimod decreases hepatic steatosis and expression of fatty acid synthase in diet-induced nonalcoholic fatty liver disease in mice. J Lipid Res 60:1311–1322
Weigert A, Olesch C, Brüne B (2019) Sphingosine-1-phosphate and macrophage biology-how the sphinx tames the big eater. Front Immunol 10:1706
Wang K, Li C, Lin X et al (2020) Targeting alkaline ceramidase 3 alleviates the severity of nonalcoholic steatohepatitis by reducing oxidative stress. Cell Death Dis 11(1):28
Srinivas AN, Suresh D, Santhekadur PK et al (2021) Extracellular vesicles as inflammatory drivers in NAFLD. Front Immunol 11:627424
Dasgupta D, Nakao Y, Mauer AS et al (2020) IRE1A stimulates hepatocyte-derived extracellular vesicles that promote inflammation in mice with steatohepatitis. Gastroenterology 159:1487–1503.e17
Kakazu E, Mauer AS, Yin M et al (2016) Hepatocytes release ceramide-enriched pro-inflammatory extracellular vesicles in an IRE1 α-dependent manner. J Lipid Res 57:233–245
Eguchi A, Feldstein AE (2018) Extracellular vesicles in non-alcoholic and alcoholic fatty liver diseases. Liver Res 2:30–34
Jiang M, Li C, Liu Q et al (2019) Inhibiting ceramide synthesis attenuates hepatic steatosis and fibrosis in rats with non-alcoholic fatty liver disease. Front Endocrinol 10:665
Moles A, Tarrats N, Morales A et al (2010) Acidic sphingomyelinase controls hepatic stellate cell activation and in vivo liver fibrogenesis. Am J Pathol 177:1214–1224
Fucho R, Martínez L, Baulies A et al (2014) ASMase regulates autophagy and lysosomal membrane permeabilization and its inhibition prevents early stage non-alcoholic steatohepatitis. J Hepatol 61:1126–1134
Schwalm S, Pfeilschifter J, Huwiler A (2013) Sphingosine-1-phosphate: a Janus-faced mediator of fibrotic diseases. Biochim Biophys Acta 1831:239–250
Santos WL, Lynch KR (2015) Drugging sphingosine kinases. ACS Chem Biol 10:225–233
Bu S, Asano Y, Bujor A et al (2010) Dihydrosphingosine 1-phosphate has a potent antifibrotic effect in scleroderma fibroblasts via normalization of phosphatase and tensin homolog levels. Arthritis Rheum 62:2117–2126
Bu S, Kapanadze B, Hsu T et al (2008) Opposite effects of dihydrosphingosine 1-phosphate and sphingosine 1-phosphate on transforming growth factor-β/Smad signaling are mediated through the PTEN/PPM1A-dependent pathway. J Biol Chem 283:19593–19602
Engstler AJ, Sellmann C, Jin CJ et al (2017) Treatment with alpha-galactosylceramide protects mice from early onset of nonalcoholic steatohepatitis: role of intestinal barrier function. Mol Nutr Food Res 61
Acknowledgments
The authors wish to acknowledge research support from NIH HL151243 and HL117233, Veterans’ Affairs I01BX000200, all to L.A.C., and P30 CA016059 to Massey Cancer Center at Virginia Commonwealth University. Virginia Commonwealth University also supported this work via the Value and Efficiency Teaching and Research Award (VETAR) to L.A.C., and the McGuire Research Institute/Virginia Commonwealth University Joint Pilot Grant 2019 To D. J. M.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Montefusco, D., Lambert, J., Anderson, A., Allegood, J., Cowart, L.A. (2022). Analysis of the Sphingolipidome in NAFLD. In: Sarkar, D. (eds) Non-Alcoholic Steatohepatitis. Methods in Molecular Biology, vol 2455. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2128-8_22
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2128-8_22
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2127-1
Online ISBN: 978-1-0716-2128-8
eBook Packages: Springer Protocols