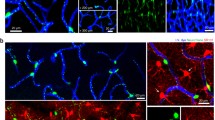
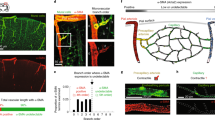
Abstract
The brain’s high energy requirements drive the need for close coupling of local neuronal activity to blood supply. Capillaries have been shown to dilate before arterioles in response to sensory stimulation, pointing to a key role for microvascular pericytes in mediating cerebrovascular dynamics. However, many aspects of these cells’ function remain unknown and even controversial, from their identification, to the mechanism and regulation of their contractility in physiology and disease. Investigating how pericytes regulate vascular diameter is therefore likely to be the subject of many future experiments. Here we provide protocols for three different techniques (ex vivo slice imaging, in vivo imaging, and immunohistochemistry) that are highly valuable for performing such experiments.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bicher HI, Reneau DD, Bruley DF, Knisely MH (1973) Brain oxygen supply and neuronal activity under normal and hypoglycemic conditions. Am J Phys 224(2):275–282. http://ajplegacy.physiology.org/content/224/2/275.abstract. Accessed May 2, 2016
Mergenthaler P, Lindauer U, Dienel GA, Meisel A (2013) Sugar for the brain: the role of glucose in physiological and pathological brain function. Trends Neurosci 36(10):587–597. https://doi.org/10.1016/j.tins.2013.07.001
Sokoloff L (1960) The metabolism of the central nervous system in vivo. In: Handbook of Physiology, Section I, Neurophysiology, vol 3. The Williams & Wilkins co., Philadelphia, pp 1843–1864
Attwell D, Laughlin SB (2001) An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab 21(10):1133–1145. https://doi.org/10.1097/00004647-200110000-00001
Hall C, Howarth C, Kurth-Nelson Z, Mishra A (2016) Interpreting BOLD: towards a dialogue between cognitive and cellular neuroscience. Philos Trans R Soc Lond B Biol Sci 371:20150348
Attwell D, Buchan AM, Charpak S, Lauritzen M, Macvicar BA, Newman EA (2010) Glial and neuronal control of brain blood flow. Nature 468(7321):232–243. https://doi.org/10.1038/nature09613
Lecrux C, Hamel E (2016) Neuronal networks and mediators of cortical neurovascular coupling responses in normal and altered brain states. Philos Trans R Soc Lond B Biol Sci 371(1705):20150350
Lacroix A, Toussay X, Anenberg E et al (2015) COX-2-derived prostaglandin E2 produced by pyramidal neurons contributes to neurovascular coupling in the rodent cerebral cortex. J Neurosci 35(34):11791–11810. https://doi.org/10.1523/JNEUROSCI.0651-15.2015
Gordon GRJ, Choi HB, Rungta RL, Ellis-Davies GCR, MacVicar BA (2008) Brain metabolism dictates the polarity of astrocyte control over arterioles. Nature 456(7223):745–749. https://doi.org/10.1038/nature07525
Uhlirova H, Kılıç K, Tian P et al (2016) Cell type specificity of neurovascular coupling in cerebral cortex. Elife 5:e14315. https://doi.org/10.7554/eLife.14315
Filosa JA, Bonev AD, Straub SV et al (2006) Local potassium signaling couples neuronal activity to vasodilation in the brain. Nat Neurosci 9(11):1397–1403. https://doi.org/10.1038/nn1779
Howarth C (2014) The contribution of astrocytes to the regulation of cerebral blood flow. Front Neurosci 8:103. https://doi.org/10.3389/fnins.2014.00103
Filosa JA, Morrison HW, Iddings JA, Du W, Kim KJ (2016) Beyond neurovascular coupling, role of astrocytes in the regulation of vascular tone. Neuroscience 323:96–109. https://doi.org/10.1016/j.neuroscience.2015.03.064
Lecrux C, Hamel E (2011) The neurovascular unit in brain function and disease. Acta Physiol 203(1):47–59. https://doi.org/10.1111/j.1748-1716.2011.02256.x
Enager P, Piilgaard H, Offenhauser N et al (2009) Pathway-specific variations in neurovascular and neurometabolic coupling in rat primary somatosensory cortex. J Cereb Blood Flow Metab 29(5):976–986. https://doi.org/10.1038/jcbfm.2009.23
Webb RC (2003) Smooth muscle contraction and relaxation. Adv Physiol Educ 27(1–4):201–206. http://www.ncbi.nlm.nih.gov/pubmed/14627618. Accessed October 3, 2016
Hamilton NB, Attwell D, Hall CN (2010) Pericyte-mediated regulation of capillary diameter: A component of neurovascular coupling in health and disease. Front Neuroenergetics 2:5. https://doi.org/10.3389/fnene.2010.00005
Longden TA, Hill-Eubanks DC, Nelson MT (2015) Ion channel networks in the control of cerebral blood flow. J Cereb Blood Flow Metab 36(3):0271678X15616138. https://doi.org/10.1177/0271678X15616138
Peppiatt CM, Howarth C, Mobbs P, Attwell D (2006) Bidirectional control of CNS capillary diameter by pericytes. Nature 443(7112):700–704. https://doi.org/10.1038/nature05193
Hall CN, Reynell C, Gesslein B et al (2014) Capillary pericytes regulate cerebral blood flow in health and disease. Nature 508:55–60. https://doi.org/10.1038/nature13165
Attwell D, Mishra A, Hall CN, O’Farrell FM, Dalkara T (2016) What is a pericyte? J Cereb Blood Flow Metab 36(2):451–455. https://doi.org/10.1177/0271678X15610340
Hill RA, Tong L, Yuan P, Murikinati S, Gupta S, Grutzendler J (2015) Regional blood flow in the Normal and ischemic brain is controlled by arteriolar smooth muscle cell contractility and not by capillary Pericytes. Neuron 87(1):95–110. https://doi.org/10.1016/j.neuron.2015.06.001
Zimmermann KW (1923) Der feinere Bau der Blutcapillaren. Z Anat Entwicklungsgesch 68(1):29–109. https://doi.org/10.1007/BF02593544
Krueger M, Bechmann I (2010) CNS pericytes: concepts, misconceptions, and a way out. Glia 58(1):1–10. https://doi.org/10.1002/glia.20898
Hartmann DA, Underly RG, Grant RI, Watson AN, Lindner V, Shih AY (2015) Pericyte structure and distribution in the cerebral cortex revealed by high-resolution imaging of transgenic mice. Neurophotonics 2(4):41402. https://doi.org/10.1117/1.NPh.2.4.041402
Mishra A, O’Farrell FM, Reynell C, Hamilton NB, Hall CN, Attwell D (2014) Imaging pericytes and capillary diameter in brain slices and isolated retinae. Nat Protoc 9(2):323–336. https://doi.org/10.1038/nprot.2014.019
Kim KJ, Filosa JA (2012) Advanced in vitro approach to study neurovascular coupling mechanisms in the brain microcirculation. J Physiol 590(7):1757–1770. https://doi.org/10.1113/jphysiol.2011.222778
Kim KJ, Iddings JA, Stern JE et al (2015) Astrocyte contributions to flow/pressure-evoked parenchymal arteriole vasoconstriction. J Neurosci 35(21):8245–8257. https://doi.org/10.1523/JNEUROSCI.4486-14.2015
Nowak L, Bregestovski P, Ascher P, Herbet A, Prochiantz A (1984) Magnesium gates glutamate-activated channels in mouse central neurones. Nature 307(5950):462–465. https://doi.org/10.1038/307462a0
Benveniste M, Mayer ML (1991) Kinetic analysis of antagonist action at N-methyl-D-aspartic acid receptors. Two binding sites each for glutamate and glycine. Biophys J 59(3):560–573. https://doi.org/10.1016/S0006-3495(91)82272-X
Choi DW (1992) Excitotoxic cell death. J Neurobiol 23(9):1261–1276. https://doi.org/10.1002/neu.480230915
Ting JT, Daigle TL, Chen Q, Feng G (2014) Acute brain slice methods for adult and aging animals: application of targeted patch clamp analysis and optogenetics. Methods Mol Biol 1183:221–242. https://doi.org/10.1007/978-1-4939-1096-0_14
Hille B (1971) The permeability of the sodium channel to organic cations in myelinated nerve. J Gen Physiol 58:599–619
MacGregor DG, Chesler M, Rice ME (2001) HEPES Prevents Edema in Rat Brain Slices. Neurosci Lett 303:141–144. https://doi.org/10.1016/S0304-3940(01)01690-1
Peça J, Feliciano C, Ting JT et al (2011) Shank3 mutant mice display autistic-like behaviours and striatal dysfunction. Nature 472(7344):437–442. https://doi.org/10.1038/nature09965
Brain Slice Methods. (2016). http://www.brainslicemethods.com/#!protective-recovery-method/c1uf. Accessed May 3, 2016
Zhu X, Bergles DE, Nishiyama A (2008) NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development 135(1):145–157. https://doi.org/10.1242/dev.004895
Stallcup WB (2002) The NG2 proteoglycan: past insights and future prospects. J Neurocytol 31(6–7):423–435. https://doi.org/10.1023/A:1025731428581
Lund-Andersen H, Hertz L (1970) Effects of potassium and of glutamate on swelling and on sodium and potassium content in brain-cortex slices from adult rats. Exp Brain Res 11(2):199–212. https://doi.org/10.1007/BF00234323
Li K (2008) The image stabilizer plugin for ImageJ. http://www.cs.cmu.edu/~kangli/code/Image_Stabilizer.html. Accessed August 19, 2016
Thevenaz P, Ruttimann UE, Unser M (1998) A pyramid approach to subpixel registration based on intensity. IEEE Trans Image Process 7(1):27–41. https://doi.org/10.1109/83.650848
Tseng Q, Wang I, Duchemin-Pelletier E et al (2011) A new micropatterning method of soft substrates reveals that different tumorigenic signals can promote or reduce cell contraction levels. Lab Chip 11(13):2231. https://doi.org/10.1039/c0lc00641f
Schechner JS, Braverman IM (1992) Synchronous vasomotion in the human cutaneous microvasculature provides evidence for central modulation. Microvasc Res 44(1):27–32. https://doi.org/10.1016/0026-2862(92)90099-B
Kleinfeld D, Mitra PP, Helmchen F, Denk W (1998) Fluctuations and stimulus-induced changes in blood flow observed in individual capillaries in layers 2 through 4 of rat neocortex. Proc Natl Acad Sci U S A 95(26):15741–15746. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=28114&tool=pmcentrez&rendertype=abstract. Accessed May 5, 2016
Berg-Johnsen J, Langmoen IA (1992) The effect of isoflurane on excitatory synaptic transmission in the rat hippocampus. Acta Anaesthesiol Scand 36(4):350–355. https://doi.org/10.1111/j.1399-6576.1992.tb03480.x
Helmchen F, Kleinfeld D (2008) In Vivo measurements of blood flow and glial cell function with two-photon laser-scanning microscopy. Methods Enzymol 444:231–254. https://doi.org/10.1016/S0076-6879(08)02810-3
Summers PM, Taylor ZJ, Shih AY (2014) Two-photon imaging of cerebral Vasodynamics in awake mice during health and disease. In: Weigert R (ed) Advances in intravital microscopy. Springer Netherlands, Vol Dordrecht, pp 25–43. https://doi.org/10.1007/978-94-017-9361-2
Drew PJ, Shih AY, Driscoll JD et al (2010) Chronic optical access through a polished and reinforced thinned skull. Nat Methods 7(12):981–984. https://doi.org/10.1038/nmeth.1530
Xu H-T, Pan F, Yang G, Gan W-B (2007) Choice of cranial window type for in vivo imaging affects dendritic spine turnover in the cortex. Nat Neurosci 10(5):549–551. https://doi.org/10.1038/nn1883
Klohs J, Rudin M, Shimshek DR, Beckmann N (2014) Imaging of cerebrovascular pathology in animal models of Alzheimer’s disease. Front Aging Neurosci 6:32. https://doi.org/10.3389/fnagi.2014.00032
Holtmaat A, Bonhoeffer T, Chow DK et al (2009) Long-term, high-resolution imaging in the mouse neocortex through a chronic cranial window. Nat Protoc 4(8):1128–1144. https://doi.org/10.1038/nprot.2009.89
Goldey GJ, Roumis DK, Glickfeld LL et al (2014) Removable cranial windows for long-term imaging in awake mice. Nat Protoc 9(11):2515–2538. https://doi.org/10.1038/nprot.2014.165
LASA (2010) Guiding Principles for Preparing for and Undertaking Aseptic Surgery. A report by the LASA Education, Training and Ethics section. (M. Jennings and M. Berdoy eds.). http://www.lasa.co.uk/publications.html
LeMoine DM, Bergdall VK, Freed C (2015) Performance analysis of exam gloves used for aseptic rodent surgery. J Am Assoc Lab Anim Sci 54(3):311–316. http://www.ncbi.nlm.nih.gov/pubmed/26045458. Accessed August 22, 2016
Dombeck DA, Khabbaz AN, Collman F, Adelman TL, Tank DW (2007) Imaging large-scale neural activity with cellular resolution in awake, Mobile Mice. Neuron 56(1):43–57. https://doi.org/10.1016/j.neuron.2007.08.003
Mostany R, Portera-Cailliau C (2008) A method for 2-photon imaging of blood flow in the neocortex through a cranial window. J Vis Exp 12:678. https://doi.org/10.3791/678
Shih AY, Driscoll JD, Pesavento MJ, Kleinfeld D (2014) Two-photon microscopy to measure blood flow and concurrent brain cell activity. In: Weber B, Helmchen F (eds) Optical Imaging of Neocrotical Dynamics, Neuromethods, vol 85. Humana Press, Totowa, NJ, pp 273–290. https://doi.org/10.1007/978-1-62703-785-3
Shih AY, Driscoll JD, Drew PJ, Nishimura N, Schaffer CB, Kleinfeld D (2012) Two-photon microscopy as a tool to study blood flow and neurovascular coupling in the rodent brain. J Cereb Blood Flow Metab 32(7):1277–1309. https://doi.org/10.1038/jcbfm.2011.196
Santisakultarm TP, Cornelius NR, Nishimura N et al (2012) In vivo two-photon excited fluorescence microscopy reveals cardiac- and respiration-dependent pulsatile blood flow in cortical blood vessels in mice. Am J Physiol Heart Circ Physiol 302(7):H1367–H1377. https://doi.org/10.1152/ajpheart.00417.2011
Nizar K, Uhlirova H, Tian P et al (2013) In vivo stimulus-induced vasodilation occurs without IP3 receptor activation and may precede astrocytic calcium increase. J Neurosci 33(19):8411–8422. https://doi.org/10.1523/JNEUROSCI.3285-12.2013
Gao Y-R, Drew PJ (2014) Determination of vessel cross-sectional area by thresholding in radon space. J Cereb Blood Flow Metab 34(7):1180–1187. https://doi.org/10.1038/jcbfm.2014.67
Armulik A, Genové G, Betsholtz C (2011) Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell 21(2):193–215. https://doi.org/10.1016/j.devcel.2011.07.001
Káradóttir R, Attwell D (2006) Combining patch-clamping of cells in brain slices with immunocytochemical labeling to define cell type and developmental stage. Nat Protoc 1(4):1977–1986. https://doi.org/10.1038/nprot.2006.261
Sicard K, Shen Q, Brevard ME et al (2003) Regional cerebral blood flow and BOLD responses in conscious and anesthetized rats under basal and hypercapnic conditions: implications for functional MRI studies. J Cereb Blood Flow Metab 23(4):472–481. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2989608&tool=pmcentrez&rendertype=abstract. Accessed May 5, 2016
Kapinya KJ, Prass K, Dirnagl U (2002) Isoflurane induced prolonged protection against cerebral ischemia in mice: a redox sensitive mechanism? Neuroreport 13(11):1431–1435. http://www.ncbi.nlm.nih.gov/pubmed/12167768. Accessed May 5, 2016
Acknowledgments
This work was supported by funding from the Physiological Society, the Royal Society, and the University of Sussex Junior Research Associate scheme. We would like to thank Devin Clarke and Orla Bonnar for comments on the manuscript, David Attwell for providing the NG2/DsRed mice to start our colony, and Leon Lagnado for practical support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Boyd, K., Hammond-Haley, M., Vroman, R., Hall, C.N. (2021). Imaging Pericytes and the Regulation of Cerebral Blood Flow. In: Péault, B.M. (eds) Pericytes. Methods in Molecular Biology, vol 2235. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1056-5_7
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1056-5_7
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1055-8
Online ISBN: 978-1-0716-1056-5
eBook Packages: Springer Protocols