Abstract
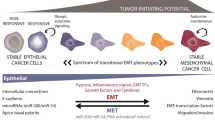
The epithelial-to-mesenchymal transition (EMT) encompasses a complex cascade of events through which a cell transits to reduce its epithelial characteristics and become migratory. Classically, this transition has been considered complete upon loss of molecular markers characteristic of an “epithelial” state and acquisition of those associated with “mesenchymal” cells. Recently, however, evidence from both developmental and cancer EMT contexts suggest that cells undergoing EMT are often heterogeneous, concomitantly expressing both epithelial and mesenchymal markers to varying degrees; rather, cells frequently display a “partial” EMT phenotype and do not necessarily require full “mesenchymalization” to become migratory. Here, we offer a brief perspective on recent important advances in our fundamental understanding of the spectrum of cellular states that occur during partial EMT in the context of development and cancer metastasis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Thiery JP, Acloque H, Huang RY et al (2009) Epithelial-mesenchymal transitions in development and disease. Cell 139:871–890
Aiello NM, Kang Y (2019) Context-dependent EMT programs in cancer metastasis. J Exp Med 216:1016–1026
Nieto MA, Huang RY, Jackson RA et al (2016) Emt: 2016. Cell 166:21–45
Thiery JP, Sleeman JP (2006) Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol 7:131–142
Kalluri R, Weinberg RA (2009) The basics of epithelial-mesenchymal transition. J Clin Invest 119:1420–1428
Campbell K, Casanova J (2016) A common framework for EMT and collective cell migration. Development 143:4291–4300
Campbell K (2018) Contribution of epithelial-mesenchymal transitions to organogenesis and cancer metastasis. Curr Opin Cell Biol 55:30–35
Theveneau E, Mayor R (2012) Neural crest delamination and migration: from epithelium-to-mesenchyme transition to collective cell migration. Dev Biol 366:34–54
Hutchins EJ, Kunttas E, Piacentino ML et al (2018) Migration and diversification of the vagal neural crest. Dev Biol 444(Suppl 1):S98–S109
Pastushenko I, Blanpain C (2019) EMT transition states during tumor progression and metastasis. Trends Cell Biol 29:212–226
Aiello NM, Maddipati R, Norgard RJ et al (2018) EMT subtype influences epithelial plasticity and mode of cell migration. Dev Cell 45(681–695):e684
Lu W, Kang Y (2019) Epithelial-mesenchymal plasticity in cancer progression and metastasis. Dev Cell 49:361–374
Pastushenko I, Brisebarre A, Sifrim A et al (2018) Identification of the tumour transition states occurring during EMT. Nature 556:463–468
Grigore AD, Jolly MK, Jia D et al (2016) Tumor budding: the name is EMT. Partial EMT. J Clin Med 5:E51
Lamouille S, Xu J, Derynck R (2014) Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol 15:178–196
Chang R, Zhang Y, Zhang P et al (2017) Snail acetylation by histone acetyltransferase p300 in lung cancer. Thorac Cancer 8:131–137
Chen A, Wong CS, Liu MC et al (2015) The ubiquitin ligase Siah is a novel regulator of Zeb1 in breast cancer. Oncotarget 6:862–873
Sanchez-Vasquez E, Bronner ME, Strobl-Mazzulla PH (2019) Epigenetic inactivation of miR-203 as a key step in neural crest epithelial-to-mesenchymal transition. Development 146:dev171017
Ma L, Young J, Prabhala H et al (2010) miR-9, a MYC/MYCN-activated microRNA, regulates E-cadherin and cancer metastasis. Nat Cell Biol 12:247–256
Li J, He M, Xu W et al (2019) LINC01354 interacting with hnRNP-D contributes to the proliferation and metastasis in colorectal cancer through activating Wnt/beta-catenin signaling pathway. J Exp Clin Cancer Res 38:161
Zhou Y, Chang R, Ji W et al (2016) Loss of scribble promotes snail translation through translocation of HuR and enhances cancer drug resistance. J Biol Chem 291:291–302
Nagaoka K, Fujii K, Zhang H et al (2016) CPEB1 mediates epithelial-to-mesenchyme transition and breast cancer metastasis. Oncogene 35:2893–2901
Cursons J, Pillman KA, Scheer KG et al (2018) Combinatorial targeting by MicroRNAs co-ordinates post-transcriptional control of EMT. Cell Syst 7(77–91):e77
Bocci F, Jolly MK, Tripathi SC et al (2017) Numb prevents a complete epithelial-mesenchymal transition by modulating notch signalling. J R Soc Interface 14:20170512
He P, Qiu K, Jia Y (2018) Modeling of mesenchymal hybrid epithelial state and phenotypic transitions in EMT and MET processes of cancer cells. Sci Rep 8:14323
Maclean AL, Hong T, Nie Q (2018) Exploring intermediate cell states through the lens of single cells. Curr Opin Syst Biol 9:32–41
Jolly MK, Tripathi SC, Somarelli JA et al (2017) Epithelial/mesenchymal plasticity: how have quantitative mathematical models helped improve our understanding? Mol Oncol 11:739–754
Acknowledgments
We thank members of the Bronner lab for helpful discussions. The authors are supported by the National Institutes of Health K99 DE028592 (E.J. Hutchins), R01DE027538 and P01 HD037105 (M.E. Bronner).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Hutchins, E.J., Bronner, M.E. (2021). A Spectrum of Cell States During the Epithelial-to-Mesenchymal Transition. In: Campbell, K., Theveneau, E. (eds) The Epithelial-to Mesenchymal Transition. Methods in Molecular Biology, vol 2179. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0779-4_1
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0779-4_1
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0778-7
Online ISBN: 978-1-0716-0779-4
eBook Packages: Springer Protocols