Abstract
Pectus excavatum is the most common congenital deformity of the chest. The Nuss procedure is minimally invasive surgical correction of this defect, using retrosternal metal bars. The purpose of the present study was to describe a 15-year experience with the Nuss surgery, and to evaluate the long-term clinical results of the procedure. We retrospectively evaluated 239 patients, aged 14–34, who underwent the Nuss surgery in the years 2002–2016. Postoperative complications were observed in 40/236 (16.9%) patients. The most common complication was pneumothorax in 14/239 patients. Less common were the following: wound infection in 4, pleural effusion in 3, allergy to nickel in 1, lung atelectasis in 1, and ventricular failure in 1 patient. Three patients were treated because of severe postoperative pain, and in one case the implant had to be removed. Postoperative complications associated with the number of bars inserted, but not with the patient age or gender. A satisfactory and long lasting corrective effect of surgery was observed in 231/239 (96.7%) of patients. There was no perioperative mortality. We conclude that the Nuss surgery is a safe surgery that demonstrates excellent and long-lasting esthetic results, with a low risk of severe complications.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Pectus excavatum is the most common anterior chest wall deformity, which is characterized by conical depression of the sternum and costal cartilages. The incidence is estimated at 1–8:1000 births and it is 4–6 times more frequent in males (Cobben et al. 2014; Obermeyer and Goretsky 2012). Surgical treatment of pectus excavatum is due mainly to psychosocial issues stemming from the abnormal physical appearance. However, in some cases this defect may result in decreased exercise tolerance, chest pain, or a severe cardiopulmonary dysfunction (Lawson et al. 2011; Malek et al. 2003).
The history of surgical correction of pectus excavatum started at the beginning of twentieth century, but the first techniques were unsatisfactory. In 1949, Mark Rawitch introduced a new open method that required a subperichondrial resection of deformed cartilages, osteotomy of the sternum, and suturing through the sternal bone to maintain a new shape (Rawitch 1949). That procedure quickly gained popularity and was used by physicians for several decades. However, the Rawitch method was characterized by serious invasiveness and a high risk of recurrence. A milestone in the development of surgical treatment of pectus excavatum was the introduction of a new minimally invasive procedure by Donald Nuss in 1998, which consisted of remodeling of the anterior chest wall by employing a retrosternal metal bars, with avoidance of cartilage resection (Nuss et al. 1998). The elasticity of the chest wall is a key factor in this procedure. The process of cartilage depletion ends completely by age 18–20 years, so that treatment after this age becomes more difficult than that in children and adolescents. Currently, the Nuss procedure has become the method of choice for patients with pectus excavatum (Zhang et al. 2015; Kelly et al. 2010).
The aim of the present study was to describe our 15-year experience with the Nuss procedure. We set out to evaluate complications and failures associated with this surgery, paying particular attention to the influence of patient age and the shape and degree of deformity. We further addressed the adverse effects having to do with the poor perception of treatment outcomes by the patient and attempted to demonstrate the ways to prevent and treat surgery failures.
2 Methods
2.1 Patients
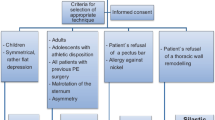
This study was performed in accord with the Declaration of Helsinki for Human Research of the World Medical Association and it was approved by the Bioethics Committee of Wroclaw Medical University in Wroclaw, Poland. The study is retrospective in nature as it is based on the information collected from the patient medical files. It included 236 patients diagnosed with pectus excavatum, including eight patients with a recurrence of pectus excavatum deformity, who were treated surgically by the Nuss procedure in the Department of Thoracic Surgery of the Wrocław Medical University in Wroclaw, Poland in the years 2002–2016. There were 197 (83.5%) men and 39 (16.5%) women. The male-to-female ratio was 5 to 1. Patients’ age ranged from 14 to 34 years (the mean of 17.4 ± 3.8 years). All patients were preoperatively examined. A routine examination consisted of basic laboratory tests, chest X-ray in posterior-anterior and lateral projections, spirometry, and an electrocardiogram. In selected cases, computed tomography was carried out. The majority of patients who were qualified for the Nuss procedure reported the cosmetic reasons as the main cause of a decision to undergo a surgical correction. The patients were subdivided into two groups: Group A – children and adolescents, and Group B adults. The cut-off age for this separation was 18 years. Groups A and B did not differ in terms of gender, quality of deformation, and the number of implants used. Details of patient characteristics are displayed in Table 1.
Chest deformities were distinguished by the presence of symmetry or lack thereof. The need to use one or two implants was taken as defining the extent of deformation. The majority of corrected deformations were symmetric (Fig. 1). However, chest asymmetry in the sagittal axis was also subject to correction in seven (3%) patients (Fig. 2). Serious distortions in the long sternal axis, with the tumefaction of the sternum manubrium, twisting the sternum over 30–35° or corners of the sterno-costal joint, were corrected by the Ravith procedure. The evaluation of the present study takes into consideration the reasons for early, occurring up to 30 days, postsurgery complications, and all subjective failures in the cosmetic aspects. We further collected detailed medical history consisting of the type of surgical correction, the length of hospitalization, the number of bars used, the clinical course of treatment, and the rate of recurrence.
2.2 Surgical and Postsurgical Interventions
When a defect was visible in the long axis of the sternum, consisting of a depression in the sternum body, hilt of manubrium deformation, sternum twisting over 45°, presence of bumps in the sterno-costal joint, or a large deformation of xiphoid process, the patients were qualified for the Rawitch method. All surgeries were performed under general anesthesia, with tracheal intubation. Figure 3 demonstrates the insertion procedure. Two incisions were made bilaterally at the height of a lower part of the sternum and a guideway was created under the sternum. A measure of a preshaped metal bar was taken for insertion (Fig. 3a). The bar was inserted, semicircular bent directed downward, retrosternally in line with the deepest point of depression (Fig. 3b). Then, the bar was rapidly and forcefully turned over 180° to shore up the sternum depression point (Fig. 3c), and the chest wall layers were sutured, with a drainage tube attached (Fig. 3d). The procedures were carried out under a video-thoracoscopic guidance. In the majority of 236 patients (95.3%) we used one metal bar to obtain a satisfactory correction, but in a few severe and asymmetric cases of chest deformity two metal bars were required (Table 1).
The Nuss procedure for a metal bar implantation: (a) creation a channel guideway under the sternum and taking a measure of a preshaped metal bar for insertion, (b) insertion of the bar retrosternally, (c) forceful rotation of the bar (red arrows) into the correction position to shore up the deformation and (d) suturing and drainage of pleural cavity
Antibiotics were used preoperatively. In the postoperative period, paravertebral anesthesia with 0.25% bupivacaine was routinely used. Most patients were discharged 5–7 days after surgery and were followed-up regularly. The patients were advised to avoid bending, sports, heavy physical activity, or carrying heavy objects to prevent the displacement of bar. Implants were maintained, on average, for 2 years. Then, the bars were removed under general anesthesia through incision along the previous scar.
2.3 Outcome Evaluation
The patients were followed up at 1, 3, 6, 12, and 24 months after surgery. Every complication during treatment or in a postoperative period was reported in the medical history. The results of surgical correction were summarized after removal of bars. A normal position of the sternum after removal of implants was considered a satisfactory result. There was then no need of any additional surgical correction and the patient expressed the acceptance of the final treatment result using a specially designed questionnaire. An unsatisfactory effect of treatment was considered when the deformation recurred after removal of bars, or when there was severe discomfort, pain, or the explicit patient’s dissatisfaction.
Categorical clinical data between the two groups were compared with a Chi squared test. A p-value <0.05 was assumed to define statistically significant differences. Statistical evaluation was performed using Statistica PL v12.0 (StatSoft; Cracow, Poland).
3 Results
The final effect of the Nuss procedure was assessed as satisfactory in 227 (96.2%) of patients. In another nine patients, additional corrective procedures had to be performed to achieve a satisfactory result, due to deformity recurrence in 8 patients and a sternum fracture in one patient. The mean length of hospitalization was 8.3 days. There has been no perioperative mortality. The long-term complications were observed in 40 (16.9%) patients. Complications occurred significantly more often in the patients who required the insertion of two bars due to a severe chest deformity (p < 0.001). The frequency of complications was unrelated to age or gender. Postsurgical complications in patients operated on because of the presence of pectus excavatum are displayed in detail in Table 2.
The most common complication was pneumothorax that occurred in 14 patients (Table 3). These patients were qualified for pleural drainage or were conservatively treated. In three patients, there were seromatous pleural effusions in the postoperative wound. Hemothorax occurred in one patient, which necessitated a redrainage. Wound infection occurred in 4 (10%) cases of complications and responded to antibiotic treatment. Three patients suffered from severe postoperative chest pain that required additional surgical interventions. In two of those patients, intercostal nerve block sufficed to relieve pain, but another patient required surgical removal of a metal bar to relieve neuropathic pain. Bar displacement was noted in three patients. One of those patients required additional stabilizing attachment of the implant to the sternum, and the other two had to undergo a bar removal. We also observed a case allergy to the nickel component of a bar. The implant had to be replaced by metal bar without nickel. In one patient, there was lung atelectasis that was treated by bronchial aspiration and breathing exercise. We also had one case of severe right ventricular failure as a result of pericardial injury cause by the implant during the surgical procedure. A fracture of the sternum occurred in one patient (Fig. 4). The fracture was caused by the inadequate distribution of forces acting on the anterior chest wall, and it required reoperation. We did not observe any statistically significant differences in the frequency of postoperative complication between the adolescent and adult groups.
Overall, additional surgical interventions were performed in 9 (3.8%) patients. The interventions consisted of 13 reoperations and one intraoperative correction. The main cause for additional surgery was a recurrence of deformity. The most common intervention was the removal of a metal bar which was done in five cases. In four patients, deformity was reoperated by the Rawitch method. A recurrence of deformation was not observed in patients below 16 years of age.
4 Discussion
Cosmetic effects after corrective thoracic cage surgery using the Nuss procedure are quite satisfactory. According to various authors, the surgery is rated as success in over 90% of cases (Park et al. 2004; Dzielicki et al. 2006). The complications that arise after surgery are transient, reversible, and non-life threatening, even though some of them may be cumbersome to the patient. The percantage of complications and failures noted in the present study was 16.3%, which is comparable to those listed in other studies performed in the groups of patients numbering more than 200 subjects of varied age, such as 16.1% (Park et al. 2004), 18.7% (Umuroglu et al. 2013), or 35.0% (Pawlak et al. 2016). The frequency of complications decreases with the acquisition of surgery experience.
Qualification of a given occurrence as a surgery complication or failure depends on the subjective evaluation of an author. That might be exemplified by a study of Pawlak et al. (2016) describing a large group of 680 regularly monitored patients, in which, apart from the 35% complication rate, carefully collected information on all cosmetic effects is listed. A comparable 37% complication and failure rate has been reported in an earlier study of Antonoff et al. (2009). Different qualification criteria could account for a relatively low incidence (6.4%) of reported complications in a multicenter study, covering a group of 777 patients in Saxena et al.’s (1999) metaanalysis.
The most frequently described and relatively easy to control early complications are the pleural disorders of pneumothorax and effusion. In the present study, pneumothorax accounted for 34% of postsurgery complications, and it required drainage only in one half of the instances. Small air sacks, located at the top of the pleura, underwent a spontaneous resolution. There is a wide spread of data in the literature on the occurrence of pneumothorax, ranging from 4.2% to 16.1% of cases (Umuroglu et al. 2013; Wu et al. 2016; Park et al. 2004; Jacobs et al. 2002). Sometimes, pneumothorax has not considered as a complication at all (Kelly et al. 2013). Pleural effusion has been reported in 2.5% to 10% of cases (Jo et al. 2003; Schaarschmidt et al. 2002). In the present study, small pleural effusions in the diaphragm angle appeared only in three cases, corresponding to 7.5% of all complications. Intrapleural bleeding or pleural hematoma requires surgical intervention. There was one such case that required pleural drainage. The literature describes single cases treated with thoraco-centesis (Park et al. 2004) or requiring thoracotomy (Bilgi et al. 2017). Pleural complications can be avoided, to an extent, by leaving a thin catheter at the top of the pleural cavity for 2–3 days after surgery.
The displacement of a correction plate can be a serious complication of the Nuss procedure, with the frequency ranging from 1% to 33% of cases as described in various studies (Wu et al. 2016; Kelly et al. 2013; Park et al. 2004; Fonkalsrud 2003; Hebra et al. 2001; Engum et al. 2000). To ensure a metal bar stability, recommend to avoid the use of absorbable stitches for the implant fixation. It also is recommended to place one suture to a rib in the parietal region. Schaarschmidt et al’s (2002) approach with 14 absorbable figure of 8 sutures around the bar placed directly on the ribs in a submuscular position gives some protection against the bar displacement. In the present study, bar displacement took place in three cases, constituting 7.5% of complications. In one of those cases, it was caused by the erroneous repeat rotations of the bar during its fitting to the chest. Another serious complication of the surgery was a wider than necessary tunnel for two implants positioned too close to each other. Yet another case of implant displacement was a failure of the patient to follow the postoperative recommendations.
Although operative access in the Nuss procedure is achieved through small incisions, local complications occur in the surgical wound. These complications are often described as a serous effusion, which occurs in 1% to 6.5% of cases (Jo et al. 2003). In the present study, the effusion occurred in 3 cases, which constitutes 7.5% of all complications observed. All were treated with locally, without having to remove the implant. Some of the wound exudates could result from a priori unrecognized allergic reaction to nickel. In the present study, in most cases, no allergy tests on metals were performed. We observed postsurgery allergic reactions in one patients, i.e., 2.5% of all complications. The Nuss surgical procedure afflicts pain, reported in up to 5.0% of cases, which is usually greater than that after the Rawitch or Leonard procedure (Antonoff et al. 2009). It becomes a complication requiring intervention if it is not noticeably reduced after the perioperative period (Hebra et al. 2001; Engum et al. 2000). In the present study, pain persisted over 30 days after surgery in three patients; 7.5% of all complications. In two of those cases, pain was controlled by intercostal nerve block and analgesics. In one case, neuropathic pain required reoperation with removal of the implant. In addition to proper postoperative treatment, intraoperative local analgesia may be an important preventive measure against pain. Also, the use of a shorter implant, with slight retraction and bent ends, prevents a later compression of periosteal ribs, which may be a source of pain.
A failure to obtain the satisfactory cosmetic appeasement does not belong to complications, but it may determine the overall deterioration of surgery effects. The assessment of the chest appearance after correction is subjective, most often independent of the dimension of the changes made, and it is related to the preferences and psychic characteristics of patients. Therefore, in the present study, attention was paid only to the most disadvantageous cosmetic effects that prompted the patient and surgeon for reoperation or for other corrective measures. We qualified patients for the Nuss surgery mostly those who had symmetric deformities. The creator and promoter of the method has reported very good results in 71%, good in 21%, and a relapse in 7.8% of patients (Nuss et al. 2002). Fonkalsrud (2003), comparing the long-term results of the Nuss surgery with other methods, has reported that the need for reoperation occurred after minimally invasive surgery in six patients, and in none operated with the modified Rawitch surgery. Hosie et al. (2002) have reported that the early results are very good or good in 81.5% of cases, but distant results have not been provided. In several cases, we also qualified for surgery patients with asymmetry in the sagittal axis of the sternum. However, in these severely distorted cases, such as a fusion of the sternum cavity with the manubrium bulge, twisting of the sternum over 45°, or horns at the sterno-costal joint, we employed the Rawitch method of surgery in line with Cartoski et al.’s (2006) recommendation. That may have a bearing on a relatively low number of failures and recurrences we encountered. Park et al. (2004) have suggested the presence of a ‘grand canyon’ type of deformation, along with an inadequate initial experience of the surgical team in treating this kind of deformation, as a cause of the high 16% rate of complications. However, another reason for the complications reported in that study could be the high 43% rate of asymmetric distortions qualified for the Nuss surgery.
In the present study, the need for reoperation occurred in eight patients, accounting for 20% of all complications. We found no difference in the incidence of complications between adolescents and adults. Likewise, there were no age-related differences in the frequency of deformity recurrence. Such findings have also been reported, albeit in a lower age range, in other studies. Nakagawa et al. (2008) have set the age threshold for comparison at 10 years of age. The computed tomographic preoperative/postoperative index fell, on average, from 6.20 ± 3.58 to 2.93 ± 0.49 in the group below 10 years of age and from 5.50 ± 2.64 to 3.40 ± 0.79 in the group over 10 years of age. The difference was statistically insignificant between the two age-groups, therefore, the authors conclude that the Nuss procedure may be used independent of the patient’s age. That is somehow at variance with the findings of Johnson et al. (2014) who have shown more complications in persons over 17 years of age and of Pawlak et al. (2016) who have shown a greater perioperative morbidity in young patients, but a greater relapse rate in adults.
In the present study, the rate of postoperative complications was 17.2%, in adults and 16.1% in the group of children and adolescents. Kelly et al. (2010) and Lawson et al. (2011) have reported that surgery with one implant has similar effects in adults and children. Two implants are required in 24% of adult patients (Zhang et al. 2015). However, the use of two implants is essentially dictated by anatomical conditions rather than age (Johnson et al. 2014). In the present study, two implants were required in 11 patients; in four up to 15 years of age, and in seven older patients. The patients of the older group suffered from significantly more overall complications (Table 2).
The limitation of the present study is that we focused only on serious complications of the Nuss surgery, requiring surgical interventions. Another imperfection is the lack of a comparative spirometry assessment before and after surgery. There was no computed tomography performed for the accurate assessment of a degree of deformity on the Haller scale in patients treated before 2008. An additional limitation is a narrowing of the Nuss treatment to patients mainly with symmetric chest deformities confined to the sternum body.
5 Conclusions
Although the Nuss surgery is more commonly performed in children and adolescents than in adults, anatomical conditions rather than age, are a limitation of the method. The operation of the funnel chest deformity using the Nuss procedure is feasible in adults and it is more likely to require two implants. At this point, there is a greater chance of complications and failures. A careful selection of patients, with exclusion of almost all extensive and asymmetric deformation, is important for achieving good results in older patients. The best clinical effect is achieved in patients with symmetric distortions. When there is asymmetry, cosmetic results are sometimes worse, although usually acceptable by patients. The presence of asymmetric deformations is an indication to consider another surgical approach, for instance, the Rawitch method.
References
Antonoff M, Alexandra E, Erickson A, Hess D, Robert D, Acton R (2009) When patients choose: comparison of Nuss, Rawitch, and Leonard procedures for primary repair of pectus excavatum. J Pediatr Surg 44:1113–1119
Bilgi Z, Ermerak NO, Çetinkaya Ç, Laçin T, Yüksel M (2017) Risk of serious perioperative complications with removal of double bars following the Nuss procedure. Interact Cardiovasc Thorac Surg 24(2):257–259
Cartoski MJ, Nuss D, Goretsky MJ, Proud VK, Croitoru DP, Gustin T, Mitchell K, Vasser E, Kelly RE Jr (2006) Classification of the dysmorphology of pectus excavatum. J Pediatr Surg 41(9):1573–1581
Cobben JM, Oostra RJ, van Dijk FS (2014) Pectus excavatum and carinatum. Eur J Med Genet 57(8):414–417
Dzielicki J, Korlacki W, Janicka I, Dzielicka E (2006) Difficulties and limitations in minimally invasive repair of pectus excavatum – 6 years experiences with Nuss technique. Eur J Cardiothorac Surg 30(5):801–804
Engum S, Rescorla F, West K, Rouse T, Scherer LR, Grosfeld J (2000) Is the grass greener? Early results of the Nuss procedure. J Pediatr Surg 35(2):246–251
Fonkalsrud EW (2003) Current management of pectus excavatum. World J Surg 27(5):502–508
Hebra A, Gauderer MW, Tagge EP, Adamson WT, Othersen HB Jr (2001) A simple technique for preventing bar displacement with the Nuss repair of pectus excavatum. J Pediatr Surg 36(8):1266–1268
Hosie S, Sitkiewicz T, Petersen C, Gobel P (2002) Minimally invasive repair of pectus excavatum – the Nuss procedure. A European multicentre experience. Eur J Pediatr Surg 12(4):235–238
Jacobs JP, Quintessenza JA, Morell VO, Botero LM, van Gelder HM, Tchervenkov CI (2002) Minimally invasive endoscopic repair of pectus excavatum. Eur J Cardiothorac Surg 21(5):869–873
Jo WM, Choi YH, Sohn YS, Kim HJ, Hwang JJ, Cho SJ (2003) Surgical treatment for pectus excavatum. J Korean Med Sci 18(3):360–364
Johnson WR, Fedor D, Singhal S (2014) Systematic review of surgical treatment techniques for adult and pediatric patients with pectus excavatum. J Cardiothorac Surg 9:25
Kelly RE Jr, Mellins RB, Shamberger RC, Mitchell KK, Lawson ML, Oldham KT, Azizkhan RG, Hebra AV, Nuss D, Goretsky MJ, Sharp RJ, Holcomb GW, Shim WK, Megison SM, Moss RL, Fecteau AH, Colombani PM, Cooper D, Bagley T, Quinn A, Moskowitz AB, Paulson JF (2013) Multicenter study of pectus excavatum, final report: complications, static/exercise pulmonary function, and anatomic outcomes. J Am Coll Surg 217(6):1080–1089
Kelly RE, Goretsky MJ, Obermeyer R, Kuhn MA, Redlinger R, Haney TS, Moskowitz A, Nuss D (2010) Twenty-one years of experience with minimally invasive repair of pectus excavatum by the Nuss procedure in 1215 patients. Ann Surg 252(6):1072–1081
Lawson ML, Mellins RB, Paulson JF, Shamberger RC, Oldham K, Azizkhan RG, Hebra AV, Nuss D, Goretsky MJ, Sharp RJ, Holcomb GW, Shim WK, Megison SM, Moss RL, Fecteau AH, Colombani PM, Moskowitz AB, Hill J, Kelly RE Jr (2011) Increasing severity of pectus excavatum is associated with reduced pulmonary function. J Pediatr 159(2):256–261
Malek MH, Fonkalsrud EW, Cooper CB (2003) Ventilatory and cardiovascular responses to exercise in patients with pectus excavatum. Chest 124(3):870–882
Nakagawa Y, Uemura S, Nakaoka T, Yano T, Tanaka N (2008) Evaluation of the Nuss procedure using pre- and postoperative computed tomographic index. J Pediatr Surg 43(3):518–521
Nuss D, Kelly RE Jr, Croitoru DP, Katz ME (1998) A 10-year review of minimally invasive technique for the correction of pectus excavatum. J Pediatr Surg 33(4):545–552
Nuss D, Croitoru DP, Kelly RE, Goretsky MJ, Nuss KJ, Gustin TS (2002) Review and discussion of the complications of minimally invasive pectus excavatum repair. Eur J Pediatr Surg 12(4):230–234
Obermeyer RJ, Goretsky MJ (2012) Chest wall deformities in pediatric surgery. Surg Clin North Am 92(3):669–684
Park HJ, Lee SY, Lee CS (2004) Complications associated with the Nuss procedure: analysis of risk factors and suggested measures for prevention of complications. J Pediatr Surg 39(3):391–395
Pawlak K, Gąsiorowski Ł, Gabryel P, Gałęcki B, Zieliński P, Dyszkiewicz W (2016) Early and late results of the Nuss procedure in surgical treatment of pectus excavatum in different age groups. Ann Thorac Surg 102(5):1711–1716
Rawitch MM (1949) The operative treatment of pectus excavatum. Ann Surg 129(4):429–444
Saxena AK, Schaarschmidt K, Schleef J, Morcate JJ, Willital GH (1999) Surgical correction of pectus excavatum: the Münster experience. Langenbeck’s Arch Surg 384(2):187–193
Schaarschmidt K, Kolberg-Schwerdt A, Dimitrov G, Straubeta J (2002) Submuscular bar, multiple pericostal bar fixation, bilateral thoracoscopy: a modified Nuss repair in adolescents. J Pediatr Surg 37(9):1276–1280
Umuroglu T, Bostancı K, Thomas DT, Yuksel M, Gogus FY (2013) Perioperative anesthetic and surgical complications of the Nuss procedure. J Cardiothorac Vasc Anesth 27(3):436–440
Wu PC, Knauer EM, McGowan GE, Hight DW (2016) Repair of pectus excavatum deformities in children: a new perspective of treatment using minimal access surgical technique. Arch Surg 136(4):419–424
Zhang DK, Tang JM, Ben XS, Xie L, Zhou HY, Ye X, Zhou ZH, Shi RQ, Xiao P, Chen G (2015) Surgical correction of 639 pectus excavatum cases via the Nuss procedure. J Thorac Dis 7(9):1595–1605
Acknowledgments
This study was supported by the statutory budget of Wroclaw Medical University in Poland
Conflict of Interest
The authors declare no conflicts of interest in relation to this article.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Cierpikowski, P., Rzechonek, A., Błasiak, P., Lisowska, H., Pniewski, G., Le Pivert, P. (2018). Surgical Correction of Pectus Excavatum by the Nuss Procedure: A 15-Year Experience Study. In: Pokorski, M. (eds) Clinical Investigation. Advances in Experimental Medicine and Biology(), vol 1047. Springer, Cham. https://doi.org/10.1007/5584_2017_121
Download citation
DOI: https://doi.org/10.1007/5584_2017_121
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-74079-9
Online ISBN: 978-3-319-74080-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)