Abstract
Critical illness is associated with dramatic changes in metabolism driven by immune, endocrine, and adrenergic mediators. These changes involve early activation of catabolic processes leading to increased energetic substrate availability; later on, they are followed by a hypometabolic phase characterized by deranged mitochondrial function. In sepsis and ARDS, these rapid clinical changes are reflected in metabolomic profiles of plasma and other fluids, suggesting that metabolomics could one day be used to assist in the diagnosis and prognostication of these syndromes. Some metabolites, such as lactate, are already in clinical use and define patients with septic shock, a high-mortality subtype of sepsis. Larger-scale metabolomic profiling may ultimately offer a tool to identify subgroups of critically ill patients who may respond to therapy, but further work is needed before this type of precision medicine is readily employed in the clinical setting.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction: A Potential Role for Metabolomics in Understanding Critical Illness
Each year, over four million critically ill Americans are admitted into intensive care units (ICUs) around the country, a number that has only increased since the start of the COVID-19 pandemic (ICU Outcomes 2022). Despite advances in medicine, ICU mortality remains high, ranging between 8 and 20%, with approximately 500,000 American deaths per year. In particular, sepsis – a systemic inflammatory illness caused by infection with a dysregulated host immune response – and the acute respiratory distress syndrome (ARDS) – a syndrome of respiratory failure with a diverse range of etiologies – are particularly challenging illnesses in the ICU, with mortality upward of 40% (Fleischmann et al. 2016; Gaieski et al. 2013; Rudd et al. 2020).
Part of the challenge in treating these syndromes lies in their significant biological heterogeneity. The majority of clinical trials in sepsis and ARDS have been negative, and available treatment remains largely supportive and directed at treating underlying infections, while not specifically addressing the inflammatory state. Recent re-analyses of a number of negative trials have revealed that subgroups of patients appear to respond to the intended therapy (Calfee et al. 2014; Puskarich et al. 2021), underscoring the heterogeneity of these syndromes and the need for better diagnostics in identifying high-risk patients and matching them with potential treatments.
Given the dramatic and rapid metabolic alterations that are observed during critical illness, metabolomics offers a powerful potential tool for better identifying subphenotypes of critically ill patients who may differentially respond to treatments and also identifying novel treatment pathways.
2 Metabolic Alterations in Critical Illness
In health, energetic needs are largely met by nutrient intake through food, rather than catabolism of the body’s macromolecules. ATP, the main source of cellular energy, is derived by aerobic metabolism, and anabolic and catabolic processes are in balance.
In critical illness, however, dramatic changes in the endocrine and autonomic nervous system alter these metabolic processes, shifting them toward a catabolic state in which the body’s carbohydrates, lipids, and protein stores are used to meet increased energetic needs. This imbalance is often further driven by reduced caloric intake, as critically ill patients frequently suffer from symptoms like encephalopathy and anorexia that curb eating. Clinically, such catabolism leads to skeletal muscle wasting, weakness, and prolonged recovery (Casaer and Van den Berghe 2014).
3 Endocrine Drivers of Altered Metabolism
Initially, acute stress such as that caused by sepsis and other forms of critical illness leads to an inflammatory cascade which includes the release of cytokines, including IL-1, IL-2, and IL-6 that can activate the hypothalamic-pituitary-adrenal axis. This can occur through direct cytokine production by glial cells in the brain (Khardori and Castillo 2012; Englert and Rogers 2016), and through free diffusion into the pituitary, which is outside the blood brain barrier. This cytokine-derived activation results in the release of corticotropin (ACTH) from the pituitary, which stimulates production and secretion of cortisol from the adrenal glands (Englert and Rogers 2016; Cooper 2003). In addition to its increased production by the adrenal gland, decreases in corticosteroid binding globulin during critical illness and impaired hepatic metabolism of the hormone contribute to increased levels of free, biologically active cortisol (Wasyluk et al. 2021; Marik et al. 2008).
Cortisol acts as the primary endocrine mediator in sepsis, leading to a variety of changes in metabolism, immune function, and cardiovascular function critical to maintaining homeostasis (Fig. 1). Metabolically, it leads to increased blood glucose through a variety of mechanisms: by activation of enzymes involved in gluconeogenesis, increasing insulin resistance, and decreasing glucose uptake by skeletal muscle (Marik et al. 2008). It activates lipolysis in adipose tissue, leading the release of free fatty acids (Marik et al. 2008).
Direct metabolic effects of cortisol. Figure created with biorender.com
In addition to increasing energy availability by increasing glucose and fatty acid concentrations, cortisol has a major role in increasing blood pressure. In vascular smooth muscle, it increases transcription of surface receptors for catecholamines and angiotensin II, resulting in increased sensitivity to these vasoactive hormones. Cortisol also plays a complex role in regulating and abrogating the immune response, acting in a negative feedback loop that both reduces the number of circulating immune cells and decreases their production of cytokines.
While increased cortisol synthesis and secretion is part of the body’s typical response to stress, this response can be attenuated or halted in a variety of conditions in the setting of critical illness. Head injuries, central nervous system depressants, and pituitary infarction can inhibit the CRH and corticotropin driven portion of the hypothalamic-pituitary-adrenal axis (Cooper 2003). At the level of the adrenal glands, drugs such as the induction agent etomidate – frequently used during intubation – can impair cortisol synthesis, and adrenal hemorrhage, though rare, can occur in septic patients with coagulopathies (Cooper 2003). More commonly, chronic intake of exogenous corticosteroids – e.g., in patients with rheumatologic disease – can lead to adrenal atrophy and a sluggish or absent response to corticotropin.
While absolute adrenal insufficiency is rare in critically ill patients, studies have shown that relative adrenal insufficiency – as defined by inadequate increase in cortisol level in response to a corticotropin challenge – is common in patients with septic shock (Annane et al. 2002). In 2007, a consensus statement between the Society of Critical Care Medicine and the European Society of Intensive Care Medicine first coined the term “CIRCI” (critical illness-related corticosteroid insufficiency) to recognize that many critically ill patients have an impaired hypothalamic-pituitary-adrenal response.
Despite this, it is unclear whether such patients benefit from hormone replacement (Annane et al. 2002; Sprung et al. 2008), as a number of large randomized controlled trials studying the use of steroids in sepsis have had conflicting results (Annane et al. 2002, 2018; Sprung et al. 2008; Venkatesh et al. 2018). At the minimum, however, these studies suggest that steroid administration in critically ill patients is safe and should be considered, particularly for patients with refractory shock. As in the sepsis literature, the benefits of steroids are unclear in ARDS, but recent high-quality studies (Villar et al. 2020) – as well as the evidence in severe COVID-19 (RECOVERY Collaborative Group et al. 2021; Tomazini et al. 2020) – suggest that they may be beneficial.
4 Adrenergic Mediators and Their Changes in Critical Illness
The sympathetic nervous system is rapidly activated in the setting of critical illness. These stress signals can occur at the level of the peripheral nerves (e.g., in the case of trauma), by chemoreceptors (in the case of hypoxemia or hypercapnia), or by baroreceptors (in the setting of shock). These events all result in the release of norepinephrine from post-ganglionic neurons, and secretion of epinephrine and norepinephrine from chromaffin cells in the adrenal medulla (Preiser et al. 2014).
Along with exogenously delivered catecholamines that may be administered therapeutically, endogenous dopamine, norepinephrine, and epinephrine play major roles in activating catabolic pathways and increasing nutrient availability. Like cortisol, these hormones work to increase plasma glucose concentrations by upregulating hepatic gluconeogenesis and glycogenolysis and increasing insulin resistance.
5 Inflammatory Mediators (Cytokines) and Their Contribution to Metabolism
In addition to the endocrine and sympathetic nervous systems, cytokines produced by immune cells play important roles in mediating metabolic changes during critical illness. Most heavily implicated are tumor necrosis factor α – previously known in the cancer literature as “cachectin” for its upregulation of lipolysis – interleukin-1β (IL-1β), and interleukin-6 (Il-6). TNFα is a potent inducer of the other two, and together, they work to increase insulin resistance, proteolysis, and lipolysis.
6 Metabolic Phases During Critical Illness
Sepsis and other forms of critical illness are characterized by an early stress response, as described above, associated with increased catabolism and energy availability. This early phase is often followed by a period of decreased metabolic activity and organ dysfunction (Singer et al. 2004; Pool et al. 2018). Initially, the organ dysfunction and elevations in lactate that are frequently seen in sepsis were thought to result from impaired tissue perfusion, but studies have since shown that although malperfusion may play a role in the early phase of critical illness, tissue oxygen delivery is adequate in the later phases of sepsis (Sair et al. 2001). Instead, cellular changes in metabolism – decreased oxidative phosphorylation and greater reliance on glycolytic pathways – termed cytopathic hypoxia – likely underly this organ dysfunction. These changes are hypothesized to be adaptive mechanisms to preserve energy for critical functions, prevent DNA damage, and limit additional injury.
In the heart, for example, sepsis and critical illness can result in reduced cardiac myocyte contractility through a process called myocardial hibernation, which preserves myocyte ATP levels at the cost of reduced function. In animal models, this change in contractility has been linked to inhibition of the oxidative phosphorylation enzyme cytochrome oxygenase, and this phenotype can be rescued by delivery of exogenous cytochrome c (Piel et al. 2007). Similar mitochondrial changes have also been implicated in animal models of sepsis-induced dysfunction of the lung, kidney, and liver (Pool et al. 2018). In the kidney, the presence of pro-inflammatory cytokines or lipopolysaccharide from bacteria leads to downregulation of energy-intensive sodium and chloride channels (Schmidt et al. 2007). Despite these observations, the full spectrum of metabolic changes during the phases of critical illness is not known. Metabolomics provides a potential avenue through which changes in multiple metabolic pathways can be better described.
7 Autophagy and Mitophagy
The mitochondrial dysfunction and electron transport chain inhibition described above likely result in increased autophagy, a cellular housekeeping mechanism that results in the lysosomal degradation of damaged and dysfunctional organelles and proteins. In brief, autophagy is upregulated by cellular/organelle damage – e.g., the mitochondrial membrane depolarization that might occur due to uncoupled respiration from an impaired electron transport chain – and inhibited by increased nutrients (Vanhorebeek et al. 2011).
Despite being a mechanism for intracellular protein catabolism, autophagy has been shown to play an important role in maintaining the health of myocytes in skeletal muscle, and inhibition of autophagy has been linked to accelerated muscle loss during fasting and muscle denervation (Masiero et al. 2009). For critically ill patients, who are in a persistent catabolic state and are at very high risk of muscle wasting and prolonged weakness (termed critical illness myopathy), the relationship between fasting and autophagy has been of increasing interest. In the 4,600+ patient EPaNIC trial (Early Parenteral Nutrition Completing Enteral Nutrition in Adult Critically Ill Patients), the effect of initiating parenteral nutrition (PN) on an early time scale (within 48 h of ICU admission) versus a late one (within 8 days) was compared. Late parenteral nutrition initiation was associated with faster recovery (as measured by ICU and hospital discharge) and fewer complications (Casaer et al. 2011). Significantly, in a prospectively planned subanalysis of the EPaNIC trial, in which a subset of patients underwent skeletal muscle biopsies 8 days after randomization and sequential strength testing through 100 days, patients randomized to the late PN group were found to have faster recovery of weakness and greater evidence of autophagosome formation (Hermans et al. 2013). Taken together, this finding suggests that a tempered approach to early nutrition in the critically ill patient may allow for more autophagy and faster recovery of skeletal muscle strength.
8 Nutrient Changes During Critical Illness
Critical illness leads to significant changes in the metabolism of carbohydrates, protein, and lipids. Most notably, increased glycogenolysis, gluconeogenesis, and reduced sensitivity to insulin lead to increased plasma glucose concentrations through the endocrine, adrenergic, and inflammatory mechanisms described above (Table 1).
Catabolism of proteins and lipids is also upregulated, particularly in early sepsis. Amino acids generated in proteolytic processes are shuttled to the liver for synthesis of acute phase reactants. Protein breakdown and a net negative nitrogen balance lead to the wasting of skeletal muscles, which will contribute to deconditioning and prolong patients’ recoveries. In adipose tissue, increased lipolysis leads to increased serum triglycerides and free fatty acids and decreased circulating lipoproteins.
Micronutrient levels also fluctuate during critical illness. Of these, changes in selenium and zinc levels in sepsis have been particularly well-described. Selenium and zinc are both trace elements with important anti-inflammatory and antioxidant properties, and levels of both minerals have been found to be lower in septic patients than in healthy controls (Allingstrup and Afshari 2015; Besecker et al. 2011). Supplementation of these micronutrients in the critically ill has been investigated with promising initial results (Manzanares et al. 2012), but due to small study sizes, further work is needed before their use in the ICU becomes widespread.
8.1 Lactate: The Original Metabolomic Biomarker
Plasma lactate is one of the most validated and frequently used biomarkers in the intensive care unit. Lactate is a by-product of anerobic metabolic pathways, in which glucose is broken down into two pyruvate molecules and then fermented to 2 lactate molecules in a net reaction that produces 2 molecules of adenosine triphosphate (ATP) and regenerates the NAD+ cofactor used in glycolysis (Fig. 2). Accordingly, rising blood lactate levels can indicate inadequate tissue perfusion and oxygenation or a perfusion-independent shift toward anerobic pathways as can occur in malignancy (the Warburg effect) or in the later phases of sepsis.
Glycolysis and lactate fermentation. Adapted from “Warburg Effect,” from Biorender.com (2022). Retrieved from https://app.biorender.com/biorender-templates and created in biorender.com
In the ICU, lactate can be used for prognostication and also to guide resuscitation during critical illness. From a risk stratification perspective, hospitalized patients with infection-related admissions had a significantly higher risk of death if their serum lactate was >4 mmol/L (as compared to those with normal concentrations), and even modestly elevated lactates of 2.5–4 mmol were associated with increased mortality (Shapiro et al. 2005). Similarly, in a study of septic patients in the emergency department, intermediate and high lactate levels (2.5–3.9 mmol/L and >4 mmol/L) were associated with increased mortality even in patients with initially normal blood pressures and without evidence of organ dysfunction (Mikkelsen et al. 2009).
Lactate levels have also been applied to the goal-directed paradigm of sepsis management and have been used to guide resuscitation, though its role in this context has been less well-established. In the EMShockNet trial which compared the use of ScvO2 to serial lactates in the goal-directed resuscitation of patients (in which both groups also had MAP and CVP goals), there was a trend toward reduced mortality in the lactate group, but this difference was not statistically significant (Jones et al. 2010). Similarly, in another trial comparing serial lactate-guided therapy to traditional early goal-directed therapy as described by Rivers et al., mortality trended lower in the lactate group, but was just outside of the predefined alpha for statistical significance (p = 0.067) (Jansen et al. 2010).
9 Metabolomics in Sepsis
Sepsis, or life-threatening organ dysfunction in response to infection (Singer et al. 2016), is one of the leading causes of death worldwide, accounting for an estimated five million deaths each year and many more hospitalizations (Fleischmann et al. 2016). The current treatment strategy is based on the delivery of early, broad-spectrum antibiotics and is otherwise supportive. Because of the disease’s heterogeneity and the limited treatments available, precision medicine approaches – in which subgroups of patients with different mechanistic drivers are identified, leading to different treatment approaches – are appealing; these strategies have been effectively applied to a range of diseases, from cancer to asthma. While broader plasma metabolomic profiling studies have yet to impact care of septic patients to date, we will discuss overarching trends in the field with selected examples from larger studies and their clinical implications below.
9.1 Diagnosis of Sepsis
Noninfectious inflammatory responses – as might occur in pancreatitis or trauma – can resemble sepsis, delaying the diagnosis until culture data are finalized and potentially contributing to the administration of unnecessary antibiotics and increasing the development of drug-resistant bacteria. Metabolomics has been used to differentiate sepsis from sterile inflammation (Mickiewicz et al. 2013, 2014; Langley et al. 2014). In these studies, plasma samples from septic patients (or in one study, infected primates) were compared to those of critically ill uninfected controls with evidence of systemic inflammation, key metabolic differences were identified, then validated in an independent cohort of critically ill patients.
In one such study, Mickiewicz et al. (2013) used supervised analysis to investigate 58 metabolites in the serum of 60 children with early septic shock and compared profiles to those of 40 noninfected pediatric ICU patients with systemic inflammatory response syndrome (SIRS) and 40 healthy children. They used principal component analysis and partial least squares-discriminant analysis to identify 18 metabolites that differed in those who survived from those who did not. Interestingly, the responses of infants and toddlers differed from those of school-aged children, suggesting that the metabolic changes associated with sepsis may differ depending on life stage. Three metabolites (2-hydroxybutyrate, 2-hydroxyisovalerate, and lactate) involved in fat breakdown and anerobic metabolism were elevated in septic patients as compared to controls regardless of age. Other studies reinforced these differences in lipid metabolism (Langley et al. 2013; Rogers et al. 2021) and also found differences in amino acid metabolism, mitochondrial metabolism, and the citric acid cycle and pentose phosphate pathway in septic patients versus noninfected inflamed controls (Eckerle et al. 2017).
More recently, Rydzak et al. used a metabolomic approach for rapid identification of blood stream infection pathogens and their antimicrobial susceptibility patterns (Rydzak et al. 2022), a highly valuable tool in sepsis, where blood stream infections contribute to significant morbidity and mortality. They used a metabolic preference assay that used the patterns of consumed and excreted metabolites of ex vivo microbial cultures to identify pathogens and their antibiotic susceptibilities, monitoring for changes in metabolite production in the presence of toxic antibiotics. They found that just seven metabolites were sufficient to differentiate between the seven most prevalent pathogens responsible for blood stream infections and identify their antimicrobial susceptibilities in half the time of traditional microbial identification and antimicrobial susceptibility testing protocols.
9.2 Prognostication in Sepsis
Sepsis has a wide range of outcomes, from full recovery to long-term disability or death (Singer et al. 2016). As such, it is not surprising that many studies in critical care metabolomics are focused on prognostication. In these studies, septic patients with worse outcomes (death or multiorgan failure) are compared to survivors or individuals with less organ failure.
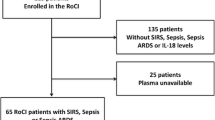
Using targeted metabolomic profiling of more than 300 metabolites, Langley et al. evaluated the plasma metabolomes and proteomes in two independent cohorts (Langley et al. 2014). The group used 150 adult patients from the Community Acquired Pneumonia and Sepsis Outcome Diagnostics (CAPSOD) study, which enrolled patients who presented to the emergency department of three US hospitals with symptoms of sepsis (Langley et al. 2013) as a discovery cohort, and used patients with sepsis from the Brigham and Women’s Hospital Registry of Critical Illness (RoCI) cohort as a validation set. They found that the metabolic signatures of survivors differed markedly from those that died. In particular, levels of proteins involved in fatty acid transport and β-oxidation, gluconeogenesis, and the citric acid cycle were significantly higher in those that died. The group used Support Vector Machine analysis to create an algorithm using two clinical features and five metabolite levels to predict survival in sepsis, using the RoCI data as a validation set. Though the RoCI cohort had higher mortality and more cancer as compared to the discovery cohort, the metabolic changes identified and the sepsis prognostication model were validated in the independent cohort.
Rogers et al. also used the CAPSOD and RoCI cohorts to perform targeted metabolic profiling on sepsis survivors and nonsurvivors (Rogers et al. 2014). Although the Langley and Rogers groups identified many of the same metabolites that differed in survivors and nonsurvivors, the Bayesian model developed by the Rogers group using RoCI as the training cohort was very different, using seven completely different metabolites than those used in the Langley algorithm. This may reflect a redundancy of metabolites found in the same metabolic pathways, but also illustrates the evolving nature of analytic methodology in the field, in which the optimal statistical methods for dealing with highly correlated data are not yet known.
9.3 Future Directions: Using Metabolomics to Guide Treatment
One of the most promising applications for the use of metabolomics in critical care is in the identification of patients who might differentially respond to therapies. Sepsis treatment is currently focused on early antimicrobial administration and is otherwise supportive, as no mechanism-targeted therapies are approved for use in the syndrome. The failure of many studies to identify effective, mechanism-based treatments has been attributed to sepsis’s significant heterogeneity, which may mask individual or subgroup responses to therapies in clinical trials.
Along these lines, Puskarich et al. used metabolomics to further phenotype patients in a reanalysis of a phase II study of L-carnitine infusion for the treatment of septic shock (Puskarich et al. 2015). The group performed metabolomics on existing serum samples from septic patients in the study and found that three metabolites involved in the synthesis and depredation of ketones differed in the pre-intervention samples of L-carnitine-treated survivors and nonsurvivors. In later work, they went on to identify acetylcarnitine as a particularly powerful biomarker for predicting L-carnitine mortality benefit (Puskarich et al. 2021). While the findings of retrospective reanalysis of a randomized controlled trials are likely insufficient to change clinical practice, the hope is that metabolomics can be used in the future to identify potential drug responders prior to their inclusion into prospective clinical trials.
10 Metabolomics Studies in ARDS
Like sepsis, ARDS is a highly heterogeneous and clinically important disease, defined by acute respiratory failure in the setting of a risk factor (such as sepsis, pneumonia, trauma, aspiration), bilateral infiltrates on imaging and low levels of blood oxygen (Ferguson et al. 2012; Thompson et al. 2017). It is estimated to affect >100,000 Americans per year with a mortality rate over 25% despite clinical improvements in recent decades (Bellani et al. 2016; Rubenfeld et al. 2005); those numbers predate the COVID epidemic, with ARDS and hypoxemic respiratory failure leading to the deaths of millions worldwide.
The heterogeneity of ARDS has no doubt contributed to a lack of effective therapies. ARDS mortality has fallen because of improvements in approaches to mechanical ventilation which decrease the pressure and volumes delivered to the lung, preventing further lung injury. But despite more than 50 years of research and substantial improvements in our understanding of the pathogenic underpinnings of lung injury and inflammation, not a single drug had been approved to treat ARDS prior to COVID-19 (Matthay et al. 2019). In COVID-19, numerous drug trials have proved effective for the syndrome, likely because of increased homogeneity in the patient population: all cases share an underlying etiology of SARS-CoV2 infection, a typical time course of disease, and a substantial inflammatory component. Multiple anti-inflammatory drug therapies are now available for COVID ARDS patients (RECOVERY Collaborative Group et al. 2021; Investigators et al. 2021; Kalil et al. 2021).
Metabolomics studies in ARDS are compelling because of the dynamic metabolomic changes in the blood and lungs, with the potential for providing a real-time snapshot of disease and identifying high-risk subjects who could be targeted for trials (Millet, “Prospective metabolomic studies in precision medicine. The AKRIBEA project.”; Turano, “NMR-based metabolomics to evaluate individual response to treatments”). Multiple studies have examined metabolomics of ARDS; major studies are summarized below and in a thorough recent review (Metwaly and Winston 2020).
10.1 Blood Metabolomics of ARDS
Several groups have performed metabolomic profiling of blood in ARDS patients and controls. As noted, there is substantial variability between studies, including (1) size of cohort, (2) how the control cohort is defined (healthy vs critically ill but without ARDS), and (3) the metabolomic profiling methodology used. Given these disparate methodologies and relatively small cohort sample sizes, it is perhaps unsurprising that the metabolites and potential pathways identified vary substantially.
Metwaly et al. examined 108 patients with ARDS, matched by age and gender with 27 mechanically ventilated controls (Metwaly and Winston 2020). They identified numerous pathways that differed in ARDS vs controls that were significant in both a training and testing cohort, with a particular focus on serine-glycine metabolism. As in prior studies, the mechanically ventilated controls were systemically much less sick than subjects with ARDS (e.g., less frequently required vasopressors, less kidney dysfunction, and markedly lower mortality).
Rogers et al. examined a large cohort of 78 ARDS cases and compared them to 75 critically ill controls with sepsis (Rogers et al. 2021). While there were numerous individual metabolites that differed, partial least squares-discriminant analysis (PLS-DA) could not separate ARDS from critically ill controls, who frequently had concomitant organ dysfunction (e.g., renal failure, liver failure, or GI malperfusion). In fact, in this cohort, many of the previously identified “ARDS metabolites” were associated with the Simplified Acute Physiology (SAPS) score, which is predictive of general ICU mortality, and were not ARDS-specific. This work suggests that many of the metabolic changes previously identified in ARDS may reflect systemic illness and inflammation rather than ARDS per se. As these studies demonstrate, how to adjust for concomitant systemic organ dysfunction is an active area of investigation in the metabolomics of ARDS.
10.2 Using Blood Metabolomics to Identify High-Risk Subsets of ARDS Cohorts
Some of the most high-profile advances in ARDS pathogenesis in the past decade have involved identifying high-risk subsets of patients who are at increased risk of bad outcomes. Most of these risk markers have involved cytokines and proteins, for example angiopoietin 2 (Calfee et al. 2012; Zinter et al. 2016), IL-18 (Rogers et al. 2019; Dolinay et al. 2012), and a hypoinflammatory/hyperinflammatory latent class analysis (LCA) designation which can be assigned using a parsimonious model of 3 blood biomarkers (IL-6, TNF receptor alpha, and bicarbonate) (Calfee et al. 2014; Sinha et al. 2020). The latent class hyperinflammatory subset has been particularly well-studied and shown to be present in more than six ARDS populations (both clinical trials and clinical cohorts) and associated with differential treatment response in numerous ARDS clinical trials, including approach to positive end-expiratory pressure (PEEP) level, fluid management, and statin therapy (Calfee et al. 2014, 2018; Famous et al. 2017).
Fewer studies have focused on using metabolomics to identify high-risk subsets of ARDS. Metwaly et al. tested whether orthogonal projections to latent structures discriminant analysis (OPLS-DA) could differentiate previously identified subsets of ARDS, including direct pulmonary risk factor for developing ARDS (e.g., pneumonia) vs indirect risk factors (e.g., pancreatitis or non-pulmonary sepsis), and hyperinflammatory subphenotype using latent class (Metwaly et al. 2021). Indeed, using metabolites identified via high-VIP score for OPLS-DA, these subsets could be separated in both testing and training sets; metabolomics outperformed protein analyses in separating the direct and indirect classes.
The Rogers et al. study of 197 patients included 75 patients with ARDS (Rogers et al. 2021). As discussed above, PLS-DA was not able to differentiate patients with sepsis from ARDS. However, unbiased metabolic clustering of the entire cohort did reveal three subsets of patients, with differential metabolites marked by lipid classes driving the subclasses, with markedly different risks of mortality, regardless of ARDS status.
10.3 Pulmonary Specific Samples for Metabolomic Profiling
Perhaps one of the most promising aspects of metabolomic profiling is the ability to examine trace amounts of a lung-specific sample which could be captured without need for a tissue biopsy to use for metabolic profiling (e.g., bronchoalveolar lavage fluid, expired breath, or free-flowing pulmonary edema fluid). Such non-blood samples may overcome the non-specific metabolic changes that occur with critical illness and allow for a more lung injury-specific signature to emerge.
To date, lung-specific metabolomics studies have been limited by similar challenges that have been seen in blood cohorts: (1) fairly small sample size, particularly given the inherent heterogeneity of ARDS, (2) varying control population, and (3) differing lung sample fluids. Not surprisingly, given such disparities in sampling, there are no common metabolites emerging that consistently differentiate ARDS from non-ARDS fluid.
Only two studies to date examined the unbiased identification of high-risk subsets within ARDS using non-blood samples. In a very small study by Rogers et al. investigating the metabolome of pulmonary edema fluid in ARDS versus congestive heart failure, ARDS edema could not be reliably distinguished from cardiogenic edema via PLS-DA, but hierarchical clustering did separate a metabolically distinct subset of one-third of the sample which was at markedly increased risk of mortality (Rogers et al. 2017).
Viswan and colleagues performed a large study, examining both blood and BAL fluid in >150 ARDS patients to examine metabolites associated with three definitions of high-risk subsets of ARDS: those defined by (1) ARDS severity by PaO2:FIO2 ratio, (2) direct vs indirect ARDS, and (3) survivors and nonsurvivors (Viswan et al. 2019). They did identify numerous associations between subsets, including overlap between the BAL and blood metabolites identified in high-risk subsets, though some methodology is unclear (Rogers et al. 2021; Investigators et al. 2021).
ARDS is a clinically-defined syndrome which carries substantial risk of mortality. It is marked by both clinical and temporal heterogeneity, suggesting that a focus on metabolomics of blood and lung biomarkers could be fruitful. Though numerous metabolomics studies have been published in the last 2 decades, more work in large cohorts is needed to identify reproducible signatures that provide targetable metabolic subgroups of ARDS. Similarly, to what extent readily available plasma samples provide an adequate metabolic sample for lung biology, versus reflect systemic illness regardless of ARDS status requires ongoing study.
11 Metabolomics of Critical Illness: Future Directions
Both sepsis and ARDS are heterogenous and life-threatening clinical syndromes in which a dysregulated immune response triggers dramatic changes in metabolism. Identifying targeted and effective treatments for these syndromes remains a challenge. The current therapeutic paradigm hinges on treating underlying infections and otherwise providing supportive care; no effective therapies have been found to treat the metabolic and inflammatory derangements that drive these syndromes.
Metabolomics offers a potential tool for better identifying the biological mechanisms underlying these syndromes and has already shown promise in prognostication of both sepsis and ARDS. Further work will be needed to more cleanly differentiate the lung-specific injury that occurs in ARDS from the frequently overlapping changes seen in sepsis. Ultimately, metabolomics offers the opportunity to uncover important biologic pathways that may serve as therapeutic targets and identify subgroups of patients in both sepsis and ARDS who may respond differentially to current and future therapeutics.
References
Allingstrup M, Afshari A (2015) Selenium supplementation for critically ill adults. Cochrane Database Syst Rev (7):CD003703. https://doi.org/10.1002/14651858.CD003703.pub3
Annane D, Sébille V, Charpentier C et al (2002) Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 288(7):862–871. https://doi.org/10.1001/jama.288.7.862
Annane D, Renault A, Brun-Buisson C et al (2018) Hydrocortisone plus fludrocortisone for adults with septic shock. N Engl J Med 378(9):809–818. https://doi.org/10.1056/NEJMoa1705716
Bellani G, Laffey JG, Pham T et al (2016) Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 315(8):788–800. https://doi.org/10.1001/jama.2016.0291
Besecker BY, Exline MC, Hollyfield J et al (2011) A comparison of zinc metabolism, inflammation, and disease severity in critically ill infected and noninfected adults early after intensive care unit admission. Am J Clin Nutr 93(6):1356–1364. https://doi.org/10.3945/ajcn.110.008417
Calfee CS, Gallagher D, Abbott J, Thompson BT, Matthay MA, Network NA (2012) Plasma angiopoietin-2 in clinical acute lung injury: prognostic and pathogenetic significance. Crit Care Med 40(6):1731–1737. https://doi.org/10.1097/CCM.0b013e3182451c87
Calfee CS, Delucchi K, Parsons PE et al (2014) Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med 2(8):611–620. https://doi.org/10.1016/S2213-2600(14)70097-9
Calfee CS, Delucchi KL, Sinha P et al (2018) Acute respiratory distress syndrome subphenotypes and differential response to simvastatin: secondary analysis of a randomised controlled trial. Lancet Respir Med 6(9):691–698. https://doi.org/10.1016/S2213-2600(18)30177-2
Casaer MP, Van den Berghe G (2014) Nutrition in the acute phase of critical illness. N Engl J Med 370(25):2450–2451. https://doi.org/10.1056/NEJMc1404896
Casaer MP, Mesotten D, Hermans G et al (2011) Early versus late parenteral nutrition in critically ill adults. N Engl J Med 365(6):506–517. https://doi.org/10.1056/NEJMoa1102662
Cooper MS (2003) Corticosteroid Insufficiency in acutely Ill patients. N Engl J Med:8
Dolinay T, Kim YS, Howrylak J et al (2012) Inflammasome-regulated cytokines are critical mediators of acute lung injury. Am J Respir Crit Care Med 185(11):1225–1234. https://doi.org/10.1164/rccm.201201-0003OC
Eckerle M, Ambroggio L, Puskarich MA et al (2017) Metabolomics as a driver in advancing precision medicine in sepsis. Pharmacotherapy 37(9):1023–1032. https://doi.org/10.1002/phar.1974
Englert JA, Rogers AJ (2016) Metabolism, metabolomics, and nutritional support of patients with sepsis. Clin Chest Med 37(2):321–331. https://doi.org/10.1016/j.ccm.2016.01.011
Famous KR, Delucchi K, Ware LB et al (2017) Acute respiratory distress syndrome subphenotypes respond differently to randomized fluid management strategy. Am J Respir Crit Care Med 195(3):331–338. https://doi.org/10.1164/rccm.201603-0645OC
Ferguson ND, Fan E, Camporota L et al (2012) The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 38(10):1573–1582. https://doi.org/10.1007/s00134-012-2682-1
Fleischmann C, Scherag A, Adhikari NKJ et al (2016) Assessment of global incidence and mortality of hospital-treated sepsis. Current estimates and limitations. Am J Respir Crit Care Med 193(3):259–272. https://doi.org/10.1164/rccm.201504-0781OC
Gaieski DF, Edwards JM, Kallan MJ, Carr BG (2013) Benchmarking the incidence and mortality of severe sepsis in the United States. Crit Care Med 41(5):1167–1174. https://doi.org/10.1097/CCM.0b013e31827c09f8
Hermans G, Casaer MP, Clerckx B et al (2013) Effect of tolerating macronutrient deficit on the development of intensive-care unit acquired weakness: a subanalysis of the EPaNIC trial. Lancet Respir Med 1(8):621–629. https://doi.org/10.1016/S2213-2600(13)70183-8
ICU Outcomes. Philip R. Lee Institute for Health Policy Studies Accessed 24 Apr 2022. https://healthpolicy.ucsf.edu/icu-outcomes
Investigators RC, Gordon AC, Mouncey PR et al (2021) Interleukin-6 receptor antagonists in critically ill patients with Covid-19. N Engl J Med 384(16):1491–1502. https://doi.org/10.1056/NEJMoa2100433
Jansen TC, van Bommel J, Schoonderbeek FJ et al (2010) Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial. Am J Respir Crit Care Med 182(6):752–761. https://doi.org/10.1164/rccm.200912-1918OC
Jones AE, Shapiro NI, Trzeciak S et al (2010) Lactate clearance vs central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial. JAMA 303(8):739–746. https://doi.org/10.1001/jama.2010.158
Kalil AC, Patterson TF, Mehta AK et al (2021) Baricitinib plus Remdesivir for hospitalized adults with Covid-19. N Engl J Med 384(9):795–807. https://doi.org/10.1056/NEJMoa2031994
Khardori R, Castillo D (2012) Endocrine and metabolic changes during sepsis. Med Clin North Am 96(6):1095–1105. https://doi.org/10.1016/j.mcna.2012.09.005
Langley RJ, Tsalik EL, van Velkinburgh JC et al (2013) An integrated clinico-metabolomic model improves prediction of death in sepsis. Sci Transl Med 5(195):195ra95. https://doi.org/10.1126/scitranslmed.3005893
Langley RJ, Tipper JL, Bruse S et al (2014) Integrative “omic” analysis of experimental bacteremia identifies a metabolic signature that distinguishes human sepsis from systemic inflammatory response syndromes. Am J Respir Crit Care Med 190(4):445–455. https://doi.org/10.1164/rccm.201404-0624OC
Manzanares W, Dhaliwal R, Jiang X, Murch L, Heyland DK (2012) Antioxidant micronutrients in the critically ill: a systematic review and meta-analysis. Crit Care Lond Engl 16(2):R66. https://doi.org/10.1186/cc11316
Marik PE, Pastores SM, Annane D et al (2008) Recommendations for the diagnosis and management of corticosteroid insufficiency in critically ill adult patients: consensus statements from an international task force by the American College of Critical Care Medicine. Crit Care Med 36(6):1937–1949. https://doi.org/10.1097/CCM.0b013e31817603ba
Masiero E, Agatea L, Mammucari C et al (2009) Autophagy is required to maintain muscle mass. Cell Metab 10(6):507–515. https://doi.org/10.1016/j.cmet.2009.10.008
Matthay MA, Zemans RL, Zimmerman GA et al (2019) Acute respiratory distress syndrome. Nat Rev Primer 5(1):18. https://doi.org/10.1038/s41572-019-0069-0
Metwaly SM, Winston BW (2020) Systems biology ARDS research with a focus on metabolomics. Metabolites 10(5). https://doi.org/10.3390/metabo10050207
Metwaly S, Cote A, Donnelly SJ et al (2021) ARDS metabolic fingerprints: characterization, benchmarking, and potential mechanistic interpretation. Am J Physiol Lung Cell Mol Physiol 321(1):L79–L90. https://doi.org/10.1152/ajplung.00077.2021
Mickiewicz B, Vogel HJ, Wong HR, Winston BW (2013) Metabolomics as a novel approach for early diagnosis of pediatric septic shock and its mortality. Am J Respir Crit Care Med 187(9):967–976. https://doi.org/10.1164/rccm.201209-1726OC
Mickiewicz B, Duggan GE, Winston BW et al (2014) Metabolic profiling of serum samples by 1H nuclear magnetic resonance spectroscopy as a potential diagnostic approach for septic shock. Crit Care Med 42(5):1140–1149. https://doi.org/10.1097/CCM.0000000000000142
Mikkelsen ME, Miltiades AN, Gaieski DF et al (2009) Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock. Crit Care Med 37(5):1670–1677. https://doi.org/10.1097/CCM.0b013e31819fcf68
Piel DA, Gruber PJ, Weinheimer CJ et al (2007) Mitochondrial resuscitation with exogenous cytochrome c in the septic heart. Crit Care Med 35(9):2120–2127. https://doi.org/10.1097/01.ccm.0000278914.85340.fe
Pool R, Gomez H, Kellum JA (2018) Mechanisms of organ dysfunction in sepsis. Crit Care Clin 34(1):63–80. https://doi.org/10.1016/j.ccc.2017.08.003
Preiser JC, Ichai C, Orban JC, Groeneveld ABJ (2014) Metabolic response to the stress of critical illness. Br J Anaesth 113(6):945–954. https://doi.org/10.1093/bja/aeu187
Puskarich MA, Finkel MA, Karnovsky A et al (2015) Pharmacometabolomics of l-carnitine treatment response phenotypes in patients with septic shock. Ann Am Thorac Soc 12(1):46–56. https://doi.org/10.1513/AnnalsATS.201409-415OC
Puskarich MA, Jennaro TS, Gillies CE et al (2021) Pharmacometabolomics identifies candidate predictor metabolites of an L-carnitine treatment mortality benefit in septic shock. Clin Transl Sci. https://doi.org/10.1111/cts.13088
RECOVERY Collaborative Group, Horby P, Lim WS et al (2021) Dexamethasone in hospitalized patients with Covid-19. N Engl J Med 384(8):693–704. https://doi.org/10.1056/NEJMoa2021436
Rogers AJ, McGeachie M, Baron RM et al (2014) Metabolomic derangements are associated with mortality in critically ill adult patients. PLoS One 9(1):e87538. https://doi.org/10.1371/journal.pone.0087538
Rogers AJ, Contrepois K, Wu M et al (2017) Profiling of ARDS pulmonary edema fluid identifies a metabolically distinct subset. Am J Physiol Lung Cell Mol Physiol 312(5):L703–L709. https://doi.org/10.1152/ajplung.00438.2016
Rogers AJ, Guan J, Trtchounian A et al (2019) Association of elevated plasma interleukin-18 level with increased mortality in a clinical trial of statin treatment for acute respiratory distress syndrome. Crit Care Med 47(8):1089–1096. https://doi.org/10.1097/CCM.0000000000003816
Rogers AJ, Leligdowicz A, Contrepois K et al (2021) Plasma metabolites in early sepsis identify distinct clusters defined by plasma lipids. Crit Care Explor 3(8):e0478. https://doi.org/10.1097/CCE.0000000000000478
Rubenfeld GD, Caldwell E, Peabody E et al (2005) Incidence and outcomes of acute lung injury. N Engl J Med 353(16):1685–1693. https://doi.org/10.1056/NEJMoa050333
Rudd KE, Johnson SC, Agesa KM et al (2020) Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the global burden of disease study. Lancet Lond Engl 395(10219):200–211. https://doi.org/10.1016/S0140-6736(19)32989-7
Rydzak T, Groves RA, Zhang R et al (2022) Metabolic preference assay for rapid diagnosis of bloodstream infections. Nat Commun 13:2332. https://doi.org/10.1038/s41467-022-30048-6
Sair M, Etherington PJ, Peter Winlove C, Evans TW (2001) Tissue oxygenation and perfusion in patients with systemic sepsis. Crit Care Med 29(7):1343–1349. https://doi.org/10.1097/00003246-200107000-00008
Schmidt C, Höcherl K, Schweda F, Bucher M (2007) Proinflammatory cytokines cause down-regulation of renal chloride entry pathways during sepsis. Crit Care Med 35(9):2110–2119. https://doi.org/10.1097/01.ccm.0000281447.22966.8b
Shapiro NI, Howell MD, Talmor D et al (2005) Serum lactate as a predictor of mortality in emergency department patients with infection. Ann Emerg Med 45(5):524–528. https://doi.org/10.1016/j.annemergmed.2004.12.006
Singer M, De Santis V, Vitale D, Jeffcoate W (2004) Multiorgan failure is an adaptive, endocrine-mediated, metabolic response to overwhelming systemic inflammation. Lancet 364(9433):545–548. https://doi.org/10.1016/S0140-6736(04)16815-3
Singer M, Deutschman CS, Seymour CW et al (2016) The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 315(8):801–810. https://doi.org/10.1001/jama.2016.0287
Sinha P, Delucchi KL, McAuley DF, O’Kane CM, Matthay MA, Calfee CS (2020) Development and validation of parsimonious algorithms to classify acute respiratory distress syndrome phenotypes: a secondary analysis of randomised controlled trials. Lancet Respir Med 8(3):247–257. https://doi.org/10.1016/S2213-2600(19)30369-8
Sprung CL, Annane D, Keh D et al (2008) Hydrocortisone therapy for patients with septic shock. N Engl J Med 358(2):111–124. https://doi.org/10.1056/NEJMoa071366
Thompson BT, Chambers RC, Liu KD (2017) Acute respiratory distress syndrome. N Engl J Med 377(6):562–572. https://doi.org/10.1056/NEJMra1608077
Tomazini BM, Maia IS, Cavalcanti AB et al (2020) Effect of dexamethasone on days alive and ventilator-free in patients with moderate or severe acute respiratory distress syndrome and COVID-19: the CoDEX randomized clinical trial. JAMA 324(13):1307–1316. https://doi.org/10.1001/jama.2020.17021
Vanhorebeek I, Gunst J, Derde S et al (2011) Insufficient activation of autophagy allows cellular damage to accumulate in critically ill patients. J Clin Endocrinol Metab 96(4):E633–E645. https://doi.org/10.1210/jc.2010-2563
Venkatesh B, Finfer S, Cohen J et al (2018) Adjunctive glucocorticoid therapy in patients with septic shock. N Engl J Med 378:797. https://doi.org/10.1056/NEJMoa1705835
Villar J, Ferrando C, Martínez D et al (2020) Dexamethasone treatment for the acute respiratory distress syndrome: a multicentre, randomised controlled trial. Lancet Respir Med 8(3):267–276. https://doi.org/10.1016/S2213-2600(19)30417-5
Viswan A, Ghosh P, Gupta D, Azim A, Sinha N (2019) Distinct metabolic endotype mirroring acute respiratory distress syndrome (ARDS) subphenotype and its heterogeneous biology. Sci Rep 9(1):2108. https://doi.org/10.1038/s41598-019-39017-4
Wasyluk W, Wasyluk M, Zwolak A (2021) Sepsis as a pan-endocrine illness-endocrine disorders in septic patients. J Clin Med 10(10):2075. https://doi.org/10.3390/jcm10102075
Zinter MS, Spicer A, Orwoll BO et al (2016) Plasma angiopoietin-2 outperforms other markers of endothelial injury in prognosticating pediatric ARDS mortality. Am J Physiol Lung Cell Mol Physiol 310(3):L224–L231. https://doi.org/10.1152/ajplung.00336.2015
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pacheco-Navarro, A.E., Rogers, A.J. (2022). The Metabolomics of Critical Illness. In: Ghini, V., Stringer, K.A., Luchinat, C. (eds) Metabolomics and Its Impact on Health and Diseases. Handbook of Experimental Pharmacology, vol 277. Springer, Cham. https://doi.org/10.1007/164_2022_622
Download citation
DOI: https://doi.org/10.1007/164_2022_622
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-26858-8
Online ISBN: 978-3-031-26859-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)