Abstract
Monoamine oxidases (MAOs) are involved in the oxidative deamination of different amines and neurotransmitters. This pointed them as potential targets for several disorders and along the last 70 years a wide variety of MAO inhibitors have been developed as successful drugs for the treatment of complex diseases, being the first drugs approved for depression in the late 1950s. The discovery of two MAO isozymes (MAO-A and B) with different substrate selectivity and tissue expression patterns led to novel therapeutic approaches and to the development of new classes of inhibitors, such as selective irreversible and reversible MAO-B inhibitors and reversible MAO-A inhibitors. Significantly, MAO-B inhibitors constitute a widely studied group of compounds, some of them approved for the treatment of Parkinson’s disease. Further applications are under development for the treatment of Alzheimer’s disease, amyotrophic lateral sclerosis, and cardiovascular diseases, among others. This review summarizes the most important aspects regarding the development and clinical use of MAO inhibitors, going through mechanistic and structural details, new indications, and future perspectives.
Graphical Abstract

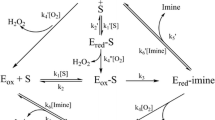
Monoamine oxidases (MAOs) catalyze the oxidative deamination of different amines and neurotransmitters. The two different isozymes, MAO-A and MAO-B, are located at the outer mitochondrial membrane in different tissues. The enzymatic reaction involves formation of the corresponding aldehyde and releasing hydrogen peroxide (H2O2) and ammonia or a substituted amine depending on the substrate. MAO’s role in neurotransmitter metabolism made them targets for major depression and Parkinson’s disease, among other neurodegenerative diseases. Currently, these compounds are being studied for other diseases such as cardiovascular ones.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Overview
Monoamine oxidases (MAOs) are part of the family of flavin adenine dinucleotide (FAD)-dependent enzymes that catalyze the oxidative deamination of different amines and neurotransmitters. There are two different isozymes that are well characterized (MAO-A and MAO-B), and they are located at the outer mitochondrial membrane in most mammalian tissues with different expression patterns. MAOs are responsible of neurotransmitters regulation by their degradation in which the enzymatic reaction involves formation of the corresponding aldehyde from the neurotransmitter to form the oxidized FAD and releasing hydrogen peroxide resulting from reduction of molecular oxygen and ammonia or a substituted amine depending on the substrate. The production of H2O2 by these enzymes might lead to the generation of free radicals that can exert cytotoxicity under pathological conditions. The relationship of MAOs with the neurotransmitter metabolism pointed them as potential target for different disorders including major depression, Parkinson’s disease (PD), Alzheimer’s disease (AD), Lewy body diseases with dementia, and amyotrophic lateral sclerosis (ALS), and currently they are used in clinics for some of these disorders.
In general, MAO inhibition has a long history since first drug was approved in the late 1950s. Initially, iproniazid was tested as a treatment for tuberculosis; although it was not effective, it showed an improvement in patient emotional state. It would be later described as a non-selective irreversible MAO inhibitor of the hydrazine group and approved in 1958 for depression. Nevertheless, those drugs showed high liver toxicity and were associated to hypertensive crisis, secondary effects that resulted in market removal of some of them. Thereafter, non-hydrazine MAO inhibitors were developed as an alternative to avoid liver toxicity (i.e., tranylcypromine, early 1960s); however, hypertensive crisis as secondary effect was still present. Therefore, some initially approved MAO inhibitors were retired as antidepressants and substituted by other drugs directed to novel targets (Shulman et al. 2013; Youdim et al. 2006; Edmondson and Binda 2018).
The discovery of two different MAO isozymes (MAO-A and B) with different substrate selectivity and expression pattern in different tissues led to novel therapeutic approaches and the development of new classes of inhibitors, such as selective irreversible and reversible MAO-B inhibitors and reversible MAO-A inhibitors. Typically, MAO-A is related to the oxidative deamination of serotonin, while MAO-B catalyzes preferentially benzylamine and 2-Phenethylamine. Dopamine, noradrenaline, adrenaline, tryptamine and tyramine exhibit similar substrate specificities for both isozymes. Hypertensive crises suffered by patients treated with MAO inhibitors were then associated with a strong MAO-A inhibition, since this enzyme metabolizes tyramine in the small intestine due to its higher expression compared to MAO-B in that organ. The toxic effect is related with the ingestion of food containing tyramine coupled to MAO-A inhibitors. This combination induces tyramine accumulation leading to high blood pressure due to the displacement of norepinephrine from neuronal storage, extremely increasing its concentration in the bloodstream (Anderson et al. 1993). The related toxic effect is known as the “cheese effect” (tyramine-induced hypertensive crisis of MAO-A inhibitors). In this sense, selective MAO-B inhibitors provide an improved pharmacological profile avoiding this effects; also reversible inhibitors of MAO-A (RIMAs) obtaining in some cases a sufficient blockade without toxicity (Da Prada et al. 1988).
MAO-B inhibitors were extensively studied after the case of intoxication with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in 1980. It was discovered that the toxicity induced by MPTP was related to its conversion to 1-methyl-4-phenylpyridinium (MPP+) by MAO-B, a metabolite that induced Parkinsonian symptoms. These symptoms were reversed by pargyline, a selective and irreversible MAO-B inhibitor (Langston et al. 1984). This correlation induced an intensive investigation of MAO-B inhibitors as potential therapeutics for the treatment PD. Nowadays, three selective MAO-B inhibitors are approved for the treatment of this disease: selegiline, rasagiline, and safinamide.
2 Structure and Binding Site of MAO
Human MAO proteins are flavoenzymes constituted by 527 and 520 amino acids for MAO-A and MAO-B isozymes, respectively. They are attached to the mitochondrial outer membrane as dimer structures (Fig. 1a) (Upadhyay et al. 2008). Both isozymes share about 70% aminoacidic sequence with a flavin adenine dinucleotide (FAD) coenzyme covalently bonded by 8α-methylene to Cys397 in MAO-B and Cys406 in MAO-A (Hubalek et al. 2003). Studies on their crystal structures suggest that the C-terminal α-helix would be related to the anchoring at the outer mitochondrial membrane of each monomer of the dimer. Substrate entry to the catalytic site of each monomer is independent from each other (Binda et al. 2002; Edmondson et al. 2009). Human MAO-A is demonstrated to be also a dimer, but it crystallizes as a monomer due to its higher instability (Edmondson and Binda 2018).
Structural details about MAO enzymes. (a) Schematic model of the human MAO-B crystal structure PDB-ID 2V5Z (Binda et al. 2007) as a dimer inserted in a phospholipid bilayer shown as sphere-stick representation. MAO-B protein is represented as dark green cartoon with FAD coenzyme of each monomer as yellow sticks and binding site cavity as light orange surface. (b) Zoom of the human MAO-B binding site with key residues as dark green sticks. (c) Detail of different inhibitor binding modes. Isatin represented as pink sticks is located in the substrate cavity near FAD coenzyme, 2-(2-benzofuranyl)-2-imidazoline represented as blue sticks is located in the entrance cavity at the other end of the binding site, and safinamide represented as light green sticks fits along the bipartite cavity. Isatin and 2-(2-benzofuranyl)-2-imidazoline come from crystal structures (PDB-ID 1OJA and 2XFN) (Bonivento et al. 2010; Binda et al. 2003) aligned to 2V5Z structure in complex with safinamide. (d) Comparison between binding site key residues of human MAO-A (PDB-ID 2Z5X) (Son et al. 2008) and MAO-B (PDB-ID 2V5Z) isozymes represented as yellow and dark green cartoon, respectively. Most of the residues are conserved and show equivalent positions of the side chains, except for Phe208/Ile199 and Ile335/Tyr326 (MAO-A/MAO-B), key residues for substrate and inhibitor specificity. All images were constructed using PyMOL software [The PyMOL Molecular Graphics System, Version 2.2 Schrödinger, LLC]
The catalytic mechanism is similar in both enzymes; however, different binding site structures revealed important aspects for substrate selectivity and inhibitors binding. One of the most important differences is a unique 550 Å3 substrate cavity present in MAO-A opposite to FAD coenzyme. MAO-B isozyme presents two cavities, an entry cavity and a reactive site cavity, with total combined volume of about 700 Å3 (Fig. 1b). MAO-B cavity exhibits an entrance cavity (290 Å3) that precedes the flat hydrophobic substrate cavity (490 Å3) (Binda et al. 2002; De Colibus et al. 2005). Open or closed conformation of Ile199 side chain in MAO-B determines the connectivity between cavities, flanked by other important residues as Tyr326 or Phe168 (Hubalek et al. 2005). In spite of the higher total pocket volume for MAO-B, narrowness and stiffness of the bipartite cavity limit size of ligands in comparison with the A isozyme, and depending on its nature, they will bind in different positions along the cavity (Fig. 1c, discussed in the next section). The main differences of both isozymes can be found in these residues: Ile199 and Tyr326 of MAO-B instead of Phe208 and Ile335 of MAO-A. In this sense, MAO-A Phe208 side chain prevents the possibility of stablishing a double cavity. The human mutant I199F MAO-B protein showed no binding for some selective MAO-B inhibitors stating the importance of Ile199 gate as a determinant for MAO-B specificity (Hubalek et al. 2005). Likewise, double I199A/Y326A human MAO-B mutation led to a protein that exhibits binding properties closer to MAO-A, highlighting the importance of Ile199 and Tyr326 side chains for selectivity (Milczek et al. 2011). In addition, the hydrophobic environment near FAD and some polar residues as Gln206 in MAO-B are important for substrate recognition and, therefore, inhibitor orientation at the coenzyme surroundings (Bonivento et al. 2010; Dasgupta et al. 2018). Furthermore, the presence of aromatic amino acid residues such as Tyr398 and Tyr435 in MAO-B oriented perpendicular to the flavin ring has been suggested as important for catalysis (Tyr407 and Tyr444 form a similar aromatic cavity in MAO-A, see Fig. 1d). Mutations in Tyr435 residue revealed differences in catalysis and function of this aromatic cage for substrate specificity (Li et al. 2006). Besides that, Y407F/Y444F mutant MAO-A enzymes also led to modified catalytic properties (Nandigama et al. 2001). Altogether, there are many structural evidences and information about what are the most influential residues in relationship with MAO activity and, therefore, crucial for inhibitor performance and design (for review, see Edmondson and Binda (2018)). Structural knowledge and binding site details are summarized in Fig. 1.
2.1 Mechanisms of MAO Catalysis and Inhibition
Compounds targeting MAO inhibition can be generally classified as irreversible or reversible inhibitors. This refers to the ability to react covalently or not with the enzyme, in particular with FAD coenzyme involved in catalysis. Irreversible inhibitors block the enzyme, and they are not released from binding site over time as a covalent bond is stablished. Conversely, reversible inhibitors are stabilized by weaker interactions in the pocket for inhibition. Depending on the therapeutic approach, reversible or irreversible inhibition will be desired. Classically, irreversible MAO inhibitors were associated with higher toxicity, although the development of specific isozyme selective compounds would reduce these complications (Youdim et al. 2006; Edmondson and Binda 2018; Anderson et al. 1993). In general, irreversible inhibitors can be classified into different classes attending to its chemical structure: hydrazines, cyclopropylamines, and propargylamines, among others. Irreversible inhibitors permanently deactivate the enzyme, and its action can only be recovered by the expression of new enzyme. The formation of the enzyme-inhibitor adduct has been related to increased secondary effects and potential immunogenicity of these adducts.
From a structural point of view, there are some important details to mention, particularly for MAO-B inhibitors. Related to the well-known architecture of MAO-B cavities, nature of the inhibitor will determine different binding site conformations. Larger compounds will occupy both, entrance and substrate cavity, inducing an open conformation of Ile199 side chain (i.e., safinamide, approved drug for the treatment of PD). In this case, this compound guides its amine moieties to flavin ring mimicking natural substrate orientation and stablishing hydrogen bonds with Gln206 (Binda et al. 2007). This situation, with both cavities engaged, is found also with other compounds (Binda et al. 2003). In turn, smaller compounds can bind in the substrate cavity with a close conformation of Ile199 gate. Thus, depending on the inhibitor size, connection between cavities will vary, regardless of nature of inhibition (Fig. 1c) (Binda et al. 2003). Considering mechanistic details, known irreversible inhibitors such as rasagiline stablish a covalent bond with N5 atom of the flavin ring (Binda et al. 2004, 2005). Other mechanisms have been described for other compounds as tranylcypromine, one of the first antidepressants, consisting in covalent linkage in this case with C4A atom of the flavin ring (Bonivento et al. 2010).
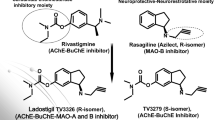
Considering selectivity, compounds can exhibit different levels of inhibition towards A or B isozyme. Apart from this, it is important to mention brain selectivity. Some compounds, regardless its affinity for A or B enzymes, can exhibit preferential brain activity and no peripheral MAO activities. Ladostigil, a compound that has completed phase II clinical trials for mild cognitive impairment as dual neuroprotective agent with cholinesterase and brain-selective MAO activity (Schneider et al. 2019) is a good example of this situation. This compound did not show intestinal MAO-A inhibition upon oral administration in rabbits, exhibiting brain selectivity for MAO inhibition (Weinreb et al. 2012).
3 Therapeutic Value of MAO Inhibitors
Several chemical structures are described as MAO inhibitors, and many new compounds are being developed. An extensive review about privileged scaffolds as MAO inhibitors is described in Tripathi et al. (Tripathi et al. 2018). Here we focused on preclinical and clinical studies with MAO inhibitors targeting several diseases from classical to new therapeutic approaches.
3.1 Affective Diseases
Classically, the effectiveness of MAO inhibitors as antidepressants raised the hypothesis of a potential overexpression of MAO enzymes as the cause of some forms of depression, and on the opposite, reduced MAO activity has been related to violent behavior (Alia-Klein et al. 2008). MAO inhibitors have been typically used for the treatment of depression and related disorders, and they were the first antidepressant drugs developed (Ramachandraih et al. 2011). The therapeutic value for this and other related affective diseases is, in general, related to MAO-A inhibition in the central nervous system (CNS), leading to increased levels of serotonin among other neurotransmitters such as noradrenaline (Youdim et al. 2006; Finberg 2014; Finberg and Rabey 2016). In addition, the selective MAO-A inhibitor 11C-harmine was used for brain imaging and gave evidence of increased MAO-A levels in striatal, midbrain, and cortical locations of major depressive disorder patients (Meyer et al. 2006). In this line, MAO-A activity is potentiated upon chronic glucocorticoid treatment in several experimental models, thus indicating relationship between stress and increased MAO-A activity (Soliman et al. 2012). MAO-A overactivity results in a decrease in monoamine neurotransmitters supporting the idea of using MAO inhibitors for the treatment of this kind of diseases, in line with classic biogenic amine hypothesis of depression (Ramachandraih et al. 2011; Finberg 2014). In addition, different MAO-A gene polymorphisms have been connected with behavioral traits, associating reduced MAO-A expression to aggressive mood and overexpression to depression (Alia-Klein et al. 2008). Most relevant MAO inhibitors for the treatment of affective disorders are summarized in Table 1.
Some of the first non-selective irreversible MAO inhibitors are still being used in clinic (i.e., phenelzine and tranylcypromine). Nevertheless, the abovemention medical complications lead to new compound profiles. In this sense, reversible and selective MAO-A inhibitors (RIMAs) appeared from 1980s to deal with “cheese effect” among other complications. In relation with it, the safety of these compounds was assessed by several clinical studies with tyramine combination (Finberg 2014; Finberg and Rabey 2016). Also, a potential secondary effect is related to the combination of irreversible MAO inhibitors and drugs with the ability to elevate 5-HT, such us serotonin reuptake inhibitors (SSRIs), leading to the serotonin toxicity syndrome (Gillman 2006). Thus, in the case of needing a serotonin reuptake inhibitor, it is necessary a washout period between 7 and 10 days for complete recovery of MAO activity. The reversible mechanism of action facilitates the competition of concentrated substrate with the inhibitor; thus, in case of high inhibition, the substrate is able to displace the inhibitor from the catalytic site limiting the secondary effects.
Moclobemide is the most important RIMA available for clinical use (approved in several western countries such as United Kingdom) also pirlindole (available in Russia) (Fasipe 2019; Lotufo-Neto et al. 1999). Other RIMAs were marketed; however, most of them were rejected. They are valuable in the treatment of depression associated to aged people and also other affective disorders such as bulimia or hypersomnia (Zisook 1985). In this line, compound CX157 was developed to achieve higher levels of brain MAO-A inhibition, and it is the first RIMA with documented reversible brain MAO-A inhibition which correlates to its plasma concentration (Fowler et al. 2010). It ended phase II clinical trials for major depressive disorder (NCT00739908); however, no more clinical studies have been documented to date. Novel compounds targeting MAO-A are still being developed, i.e., pyrazoline and hydrazone derivatives, that led to more potent MAO-A inhibitors than moclobemide that exerted antidepressant properties in behavioral in vivo tests (Evranos-Aksoz et al. 2017).
Development of selective MAO-B inhibitors, accelerated for their use in PD, leads to their evaluation in depression, considering that they do not induce hypertensive crisis (“cheese effect”). A prominent example was selegiline, a selective irreversible MAO-B inhibitor, approved for major depressive disorder (Finberg and Rabey 2016; Lee and Chen 2007). In order to increase CNS levels, it was administrated as transdermal patch, a formulation that increased its brain distribution. Its high brain concentration inhibited both isozymes (A and B), since it is a selective MAO-B inhibitor only at low concentrations. Therefore, under this administration regime, MAO-A inhibition was observed at CNS level without hepatic or intestinal effects (Mawhinney et al. 2003). Seleginine was further evaluated clinically for the treatment of attention deficit hyperactivity disorder (ADHD) in children where it showed increased levels of attention similar to those achieved with methylphenidate (first-line treatment) (Akhondzadeh et al. 2003; Rubinstein et al. 2006). Rasagiline and safinamide, other selective irreversible and reversible MAO-B inhibitors, respectively, were also found to be effective in the treatment of depression associated to PD patients, as shown in different clinical trials (Barone et al. 2015; Korchounov et al. 2012; Ryan et al. 2019; Smith et al. 2015) (Table 1).
3.2 Parkinson’s Disease
PD is a chronic and fatal neurodegenerative disease that affects mainly the nigrostriatal dopaminergic neurons, resulting in tremor, muscle rigidity, bradykinesia, and instability. Following the development of MAO inhibitors as treatments for depression and other affective disorders, and considering the implication of MAO enzymes in the metabolism of dopamine, their application for the treatment of PD started to be clinically tested. Furthermore, the target in this case was MAO-B inhibition that also led to reduced toxicity risk. In general, MAO-B inhibitors are indicated for patients showing mild motor deficits as results in clinical trials in early PD demonstrated mobility scores improvement compared to levodopa. These compounds showed benefits in the rate of motor fluctuations with reduced adverse effects compared to other dopaminergic agents (Caslake et al. 2009). In that sense, the application of MAO-B inhibitors for PD relies on several observations: (1) both MAO-A and MAO-B isozymes show similar rates for dopamine metabolism (Youdim et al. 2006); (2) levels of MAO-B are increased in PD conditions as consequence of gliosis, considering that this enzyme is mainly present in glial cells (Nagatsu and Sawada 2006); (3) human basal ganglia dysfunction is closely related to PD, and these structures contain higher levels of MAO-B isozyme. In this sense, MAO-B activity has been found to be increased in PD affected areas as substantia nigra (Youdim et al. 2006; Mallajosyula et al. 2008); (4) aging is the most important PD risk factor, and MAO-B levels increase with age, as observed in human brain postmortem studies. Thus, MAO-B overactivity might be related to neurodegeneration as a consequence increased ROS levels (Mallajosyula et al. 2008; Fowler et al. 1997).
ROS are considered to play a central role on the onset and progression of the disease. In PD there is a selective loss of dopaminergic neurons in the substantia nigra pars compacta, and this neuronal death is considered to be linked to oxidative stress due to dopamine metabolism (Segura-Aguilar et al. 2014). In brief, tyrosine is converted into L-DOPA by tyrosine hydroxylase (TH) and then decarboxylated by the DOPA-decarboxylase to generate dopamine in the brain (Meiser et al. 2013). The metabolism of dopamine by MAO enzymes, or its auto-oxidation, leads to the production of reactive oxygen species (ROS) (Blesa et al. 2015). On the one hand, the auto-oxidized form of dopamine, a quinone-related structure, is a powerful electrophile that exerts high toxicity to the cell. On the other hand, MAO metabolism of dopamine releases hydrogen peroxide that decomposes to generate ROS, thus, exacerbating cellular damage. In this line, dopaminergic neurons present in substantia nigra are surrounded by astrocytes containing high levels of MAO-B enzyme; thus, they are exposed to increased oxidative stress, as previously described. In general, neurons contain lower levels of glutathione (GSH) than astrocytes to control oxidative stress; therefore, they are highly susceptible to this toxic stimulus (Mallajosyula et al. 2008; Sian et al. 1994). In fact, decreased levels of GSH increase neuronal vulnerability to free radicals that induce mitochondrial dysfunction and maintain α-synuclein in a protofibril state. α-Synuclein protofibril finally aggregates to form Lewy bodies observed in postmortem brain of PD patients (Croisier et al. 2005; Poewe et al. 2017). Most relevant MAO inhibitors for PD treatment are summarized in Table 2.
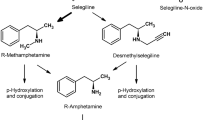
The first MAO inhibitor approved for the treatment of PD was selegiline (L-deprenyl), a selective irreversible MAO-B inhibitor. This compound was marketed on the 1980s and used in combination with L-DOPA or levodopa, a dopamine precursor that was the main treatment for PD symptoms. This inhibitor belongs to propargylamine class of compounds, in general, acting through covalent bonding to N5 atom of flavin ring of FAD coenzyme, as previously mentioned. It suffers first-pass metabolism to generate amphetamine and methamphetamine (Yasar et al. 2006), related with potential CNS and cardiovascular secondary effects (Gal et al. 2005). Nevertheless, there is no reported toxicity derived from selegiline metabolites probably due to the safer clinical doses used, although it could contribute to sleep disturbances (Muller et al. 2013; Yasar et al. 1996). Conversely, it has been reported a psychostimulant-like behavioral effects related with production of selegiline-derived amphetamine metabolites at higher doses (Yasar et al. 2006). Although it can be used in monotherapy (Youdim et al. 2006), clinical trials demonstrated that levodopa dosing can be reduced 30–40% when combined with selegiline (Myllyla et al. 1997); additionally, the DATATOP study demonstrated that only 26% of selegiline treated patients needed levodopa combination after 12 months compared to 47% of placebo group (Parkinson Study 1989). Thereafter, the SELEDO study demonstrated that the mean time delay for an increase of 50% of levodopa dosage in selegiline treated patients was 4.9 years, compared to 2.6 years of placebo patients (Przuntek et al. 1999). This is important for reducing levodopa-induced dyskinesia observed in levodopa-treated PD patients.
Another propargylamine potent selective irreversible MAO-B inhibitor, rasagiline, was then developed on the 2000s and approved for the treatment of PD. This compound is not metabolized into amphetamine as selegiline, offering an improved profile in terms of potential neurotoxicity (Edmondson and Binda 2018). Rasagiline can improve motor behavior, motor complications, mood, and sleep disorders due to its additional glutamate antagonizing properties (Muller et al. 2013; Dong et al. 2016). After satisfactory safety clinical trials, rasagiline showed reduced PD advance (Rabey et al. 2000), and this observation was further evaluated in the TEMPO study. Results from this study suggested a potential disease modifying activity of rasagiline that reduced the disease progression and increased patient quality of life (Parkinson Study 2002, 2004). Considering these encouraging results, a new clinical trial was designed to evaluate the effectiveness of rasagiline in combination with levodopa in advanced PD patients, the LARGO study (Rascol et al. 2005). In this case, rasagiline showed reduced off-time duration intraday and improved clinical global improvement and motor function on patients. More recently, a new double-blind clinical trial was conducted to further evaluate the potential disease modifying ability of rasagiline in PD, the ADAGIO study (Olanow et al. 2009). The results showed a possible disease modifying effect of rasagiline given at 1 mg daily dosage; however, it did not show this effect at 2 mg daily dosage; thus, these divergent results are not conclusive (Rascol et al. 2011; Jankovic et al. 2014).
As previously depicted, irreversible inhibitors led to strong and long-lasting inhibition, and new enzyme has to be synthesized. In that sense, reversible inhibition appears to be more manageable, maintaining selectivity to the B isozyme. Safinamide, a reversible and highly selective MAO-B inhibitor, was then developed following this idea and has been recently approved as an adjunctive treatment to L-DOPA for mild- to late-stage PD (Borgohain et al. 2014). Separately from selective reversible MAO-B inhibition, this drug acts through multiple mechanisms of action. It is able to block sodium and calcium channels and reduce excessive glutamate release (Stocchi et al. 2006). It has shown a safer profile, and there is evidence of improved motor symptoms in early PD patients (Edmondson and Binda 2018; Bette et al. 2018; Fabbri et al. 2015). Initial clinical studies (study 015 and continued in study 017) showed improved motor scores in safinamide treated patients as well as improved quality of life (Schapira et al. 2013). Thereafter, the MOTION study also demonstrated benefits in the ADL score, heath-related quality of life, better cognition, and increased rate of responders (Stocchi and Torti 2016). Thereafter, the SETTLE trial (in combination with levodopa) demonstrated a significant increase in the on-time and improved motor symptoms (Schapira et al. 2013) (For a critical review of safinamide clinical evidence in PD, see Bette et al. (2018)). In addition, a recent post hoc analysis of the 2-year study 018 revealed a reduction on chronic pain suffered by PD patients with a reduction on pain drugs usage (Cattaneo et al. 2018). Finally, the recent SIN-DEP-PAR and SELEDO clinical studies further confirm the beneficial effects of seleginine for the management of PD (Muller and Mohr 2019).
Apart from MAO-B inhibition-related aspects, this kind of inhibitors exhibit MAO-independent activities, also responsible of its beneficial effects as mentioned for safinamide. It has been demonstrated that selegiline exerts neuroprotective activities against MPTP toxicity also in cell lines lacking MAO-B (Le et al. 1997). In this sense, rasagiline and selegiline have been effective in several neuroprotection models of disease, and they have demonstrated antiapoptotic properties, an effect attributed to the propargylamine moiety (Szoko et al. 2018).
During the last years, new multitarget drugs derived from rasagiline have been developed for the treatment of PD among other neurodegenerative diseases. Ladostigil, an example of rasagiline multitarget derivative, inhibits brain acetylcholinesterase, butyrylcholinesterase, and brain MAO-A and MAO-B isozymes after chronic treatment in rats. Furthermore, it exerts neuroprotection against MPTP toxicity in mice, among other activities (Youdim 2013). Related to its neuroprotective profile in preclinical models, ladostigil has completed phase IIb clinical trials for mild cognitive impairment and Alzheimer’s disease (AD) (NCT01354691, NCT01429623) (discussed below). M30, another rasagiline derivative, is a multitarget iron chelator that exhibits brain-selective MAO inhibition and neuroprotection against MPTP, lacatcystin, and 6-hydroxydopamine in animal models of PD. The protective properties of M30 were also dependent to the ability of the drug to activate the hypoxia-induced factor (HIF) and derived induction of neurotrophins as brain-derived neurotrophic factor (BDNF), vascular endothelial growth factor (VEGF), erythropoietin, and glia-derived neurotrophic factor (GDNF) (Youdim 2013). Finally, it was demonstrated that M30 elevated dopaminergic and transferrin receptor cell count in the substantia nigra of MPTP-treated mice (Youdim 2012). Apart from the previously mentioned increase in MAO activity with PD and ageing, iron was also observed to increase in such conditions stating the promising future of multitarget iron chelators (Youdim et al. 2014).
Last years, repurposing programs are being used for the development of new treatments for complex diseases, due to their interesting cost reduction. In that sense, glitazones, used as antidiabetic drugs, were evaluated for PD as they showed MAO-B inhibition properties. This is the case of pioglitazone and rosiglitazone. Crystal structure of pioglitazone in complex with MAO-B explained binding mode through both the entrance and substrate cavities (Binda et al. 2011a). Several studies position this compound for potential PD treatment: (1) acute treatment of MPTP mice leaded to the absence of striatal dopamine reduction and improved motor behaviors (Barbiero et al. 2011); (2) beneficial effects on rotenone model of PD in rats related to recovery of striatal dopamine levels and locomotion (Ulusoy et al. 2011); (3) improvements in the 6-OHDA model in rats: reduced mortality rate, attenuated microglial activation, exerted antidepressant-like effects, induction of hippocampal neurogenesis (often associated with depression in PD patients), and protection against hypolocomotion (Bonato et al. 2018; Machado et al. 2019); and (4) decreased neuroinflammation in the midbrain and striatum and improved motor phenotype in Cox10/DAT-cre mice (dopaminergic neuron knockout of Cox10 essential for maturation of COXI, catalytic subunit of Complex IV of mitochondrial respiratory chain) (Pinto et al. 2016). Phase II clinical trials in early PD patients was carried out although pioglitazone was not found to modify progression of the disease (NCT01280123) (Neurol 2015). On the other hand, rosiglitazone showed low selectivity for MAO-B inhibition (Binda et al. 2011a) and also reported properties of this drug in PD models: (1) pre-treatment with rosiglitazone prior to 6-OHDA insult in rats leads to protection against striatal dopaminergic death and decrease in some pro-inflammatory markers as TNF-α (Lee et al. 2012) and (2) partial recovery of striatal dopamine content and dopamine neuron degeneration in substantia nigra after rosiglitazone treatment in MPTP mice (Carta et al. 2011). The protective effects of this kind of compounds may be MAO-independent, at least in part, as demonstrated by importance of peroxisome proliferator-activated receptor γ (PPARγ) activation by glitazones for neuroprotection in substantia nigra and its related anti-inflammatory actions (Martin et al. 2012).
Another important example of repurposing is the case of the antiepileptic drug zonisamide, compound approved for the treatment of PD in Japan. Its binding mode was elucidated in the substrate cavity in complex with MAO-B (Binda et al. 2011b). Among several results in PD-related models, it has been demonstrated implication of zonisamide as MAO inhibitor, showing an attenuation of striatal dopamine and tyrosine hydroxylase reduction in MPTP mice (Sonsalla et al. 2010). Zonisamide has also showed a neuroprotective profile in a genetic mouse model of PD, reducing motor symptoms (Sano et al. 2015), enhancing L-DOPA treatment (Nishijima et al. 2018), and exhibiting a reduction in associated dyskinesia in 6-OHDA rats (Oki et al. 2017). In this line, zonisamide has been evaluated in several clinical trials, most of them in Japan (Murata et al. 2015, 2016, 2018), and there is one recent open study in advanced PD in Egyptian population (NCT04182399). As previously mentioned, it was approved in Japan for use as anti-PD agent in 2009, in patients treated with other drugs in combination with L-DOPA that show inadequate responses (Grover et al. 2013) (Table 2).
3.3 Other Neurodegenerative Disorders
Given the neuroprotective profile of several MAO inhibitors, they could be useful for treatment of other neurodegenerative diseases. These complex diseases share many physiopathological mechanisms, in which MAO enzymes are involved, such as oxidative stress and neuroinflammation.
In this sense, several MAO-B inhibitors have been evaluated as potential treatments of AD. This is the most prevalent neurodegenerative disease, and it is characterized by progressive memory loss and incapacitation for daily task. AD major symptoms are accompanied by depression, agitation, delusions, and hallucinations. AD patients showed higher levels of MAO-B activity, and this may lead to an exacerbated oxidative stress condition in this disease (Kennedy et al. 2003). MAO activation has also been related to cognitive impairment, altering balance of neurotransmitters, and the formation of amyloid plaques through the modulation of amyloid precursor protein (APP) processing by MAO (for review, see Cai (2014)). These evidences prompted the application of MAO inhibitors as potential treatments of AD. In this line, selegiline and rasagiline have been evaluated in clinical trials for AD (NCT01701089, NCT00104273, NCT02359552). Initial clinical trials with seleginine pointed to a potential cognitive and behavioral improvement after 3–6 months treatment (Campi et al. 1990; Filip and Kolibas 1999); however, these results were not corroborated in other cases (Tariot et al. 1987; Burke et al. 1993). Further post hoc analysis determined that selegiline might have a short-term beneficial effect with no clinical relevance.
Given the potential protective effects of these compounds, also novel rasagiline derivatives have been tested. This is the case of M30 and ladostigil, previously introduced in this review for the treatment of PD. These compounds were developed as multitarget drugs combining interesting activities for the treatment of neurodegenerative diseases (see Sect. 3.2). M30 has been observed to be involved in regulation of amyloid β (Aβ), inhibiting its accumulation in APP/presenilin 1 mice, neurogenesis, suppression of oxidative stress, pro-cognitive, and anti-inflammatory effects (improvement in a rat model of AD with amyloid pathology McGill-R-Thy1-APP transgenic rats) (Cai 2014; Kupershmidt et al. 2012; Pimentel et al. 2015; Zheng et al. 2010). Ladostigil was shown to be related with APP translation and processing (Yogev-Falach et al. 2006) and neuroinflammation in a model of aged rats (Panarsky et al. 2012). These among other properties such as its acetylcholinesterase inhibition point out ladostigil as a new candidate for AD treatment, and phase II clinical trials has been conducted (NCT01354691, NCT01429623). Although it was safe and well tolerated, ladostigil did not delay progression to dementia. In spite of its failure, ladostigil treatment was associated with reduced brain and hippocampus volume loss (Schneider et al. 2019).
Apart from the abovementioned compounds, many others have shown potential therapeutic value for AD: (1) harmine improved memory and learning in animal models and exerted anti-inflammatory properties in LPS-mouse model (Dos Santos and Hallak 2017; Liu et al. 2017b); (2) methylene blue relationship between serotonin levels and psychological symptoms of AD could be an approach for this compound. Apart from this, methylene blue has shown attenuation of the formation of amyloid plaques, neurofibrillary tangles of tau protein, and involvement in mitochondrial function restoration, among others (Oz et al. 2009). It is currently under clinical trials (NCT02380573); (3) KDS2010 is a novel compound highly potent and selective reversible MAO-B inhibitor (Fig. 2). It has demonstrated learning and memory improvements, promotion of synaptic transmission, and reduction of astrogliosis and astrocytic GABA levels in APP/presenilin 1 mice (Park et al. 2019). Importantly, abundant GABA production by MAO-B in reactive astrocytes has been linked to AD-like pathology in animal models of AD (Jo et al. 2014); (4) ASS234 is a multitarget compound that inhibits acetylcholinesterase, butyrylcholinesterase, and MAO enzymes (Fig. 2). From crystal structure in complex with MAO-B, it is known that it acts as irreversible inhibitor binding to the FAD coenzyme (Esteban et al. 2014). It has been shown that ASS234 is able to restore scopolamine-induced cognitive impairment and prevent Aβ aggregation in animal models of AD (Marco-Contelles et al. 2016); (5) sembragiline has shown potent and reversible MAO-B inhibition with an improved safety profile, regarding serotonin syndrome and the “cheese effect” observed with other MAO inhibitors (Fig. 2). Interestingly, this compound was protective against oxidative stress and astrogliosis in transgenic mice overexpressing MAO-B in astroglia (Borroni et al. 2017). In accordance with these promising results and after phase I trials, sembragiline was evaluated in phase II clinical trials for AD (MAyflOwer RoAD study) suggesting potential benefits on behavioral symptoms in some subpopulations (NCT01677754) (Nave et al. 2017). No differences were found between treated groups and placebo in ADAS-Cog11 scale performance, missing the primary endpoint. Positron emission tomography measurements have demonstrated that same dose used in phase II clinical trial (NCT01677754) achieved near-complete brain MAO-B inhibition in AD patients daily treated with sembragiline (NCT01701089) (Sturm et al. 2017).
Amyotrophic lateral sclerosis (ALS) and Huntington’s disease (HD) share many of the pathophysiological characteristics of PD and AD. Although selegiline was shown to be ineffective in clinical trials for ALS (Lange et al. 1998), other MAO inhibitors have demonstrated potential properties. Previously mentioned iron chelator M30 was evaluated in SOD1-G93A transgenic mouse model of ALS in combination with high-calorie energy supplemented diet, and it has been related with protective effects on motor performance and increased survival, with brain MAO inhibition and reduction in dopamine metabolism (Golko-Perez et al. 2016). Other MAO inhibitors such as rasagiline have been evaluated in clinical trials for ALS. This compound was safe in ALS patients, also suggesting potential disease modifying properties in some subpopulations of the study when co-administered with riluzole (NCT01879241) (Ludolph et al. 2018). However, when administered alone it did not show any alteration of the disease progression when compared to control after 12 months (Statland et al. 2019).
Regarding dopamine imbalance present in HD (Chen et al. 2013), MAO inhibitors have been evaluated for this condition. MAO activity is increased in brain tissue from HD patients and mouse striatal neural cells expressing huntingtin. Treatment with MAO inhibitors (clorgyline, phenelzine, selegiline) improved oxidative stress condition and survival of these cells (Ooi et al. 2015). In this line, clorgyline treatment of YAC128 HD mice restored dopamine, serotonin, and norepinephrine levels in the striatum (Garcia-Miralles et al. 2016).
Other related diseases such as cerebral ischemia have been postulated for MAO inhibition treatment. Probably related to its neuroprotective activities, selegeline treatment within 48 h after stroke improved patient recovery in a phase II study (Sivenius et al. 2001; Bartolo et al. 2015). Recently, the MAO inhibitor nialamide has shown to be protective in terms of improved functional recovery and survival after post-ischemic administration in a stroke mouse model (Liu et al. 2019) (Fig. 2).
3.4 Cardiac Diseases
Apart from the deeply discussed role of oxidative stress in neurodegenerative diseases, it also appears as an important factor for cardiovascular diseases. Serotonin and norepinephrine, which are MAO-A substrates, are related with cardiac function. On the one hand, variations in serotonin levels can lead to cardiovascular dysfunction, being important regulator of heart function (Cote et al. 2004). On the other hand, norepinephrine metabolism by MAO-A is increased in mice subjected to hemodynamic stress leading to enhanced oxidative stress, hypertrophy, chamber dilation, and reduced systolic function; clorgyline treatment of these animals showed beneficial effects on cardiac function (Kaludercic et al. 2010). Levels of MAO-A are increased in several models of cardiomyopathies, and cardiac-specific MAO-A overexpression in transgenic mice was related with oxidative mitochondrial damage, cardiomyocyte necrosis, and chronic ventricular dysfunction (Villeneuve et al. 2013). Recent studies with reversible MAO-A inhibitor moclobemide exhibited myocardium protection in rats submitted to acute volume overload (Huuskonen et al. 2019). There are some studies in the same way with MAO-B demonstrating a role of enhance activity of this isozyme in cardiac injuries after chronic hemodynamic stress (Kaludercic et al. 2014). All this shows the potential for the future clinical use of MAO inhibitors for the treatment of heart disease.
4 Summary and Future Prospects
MAO inhibitors have provided a number of successful drugs for the treatment of complex diseases, being the first drugs approved for depression. During the last 70 years, the advances on the knowledge of the crystal structures of both isozymes, MAO-A and MAO-B, tissue distribution, and substrate selectivity have led to the development of a plethora of highly selective reversible and irreversible inhibitors with clinical application. These advances overcome the deleterious side effects improving the efficacy and security of these classes of advanced drugs. Novel therapeutics as MAO inhibitors included classical applications such as major depressive disorder, transient resistant depression, bipolar depression, etc., and it is expected as exponential growth of their application in this therapeutic area. Nonetheless, novel therapeutic areas are under use due to the development of selective MAO-B inhibitors such us the treatment of neurodegenerative diseases. In particular, the treatment of Parkinson’s disease is a prominent example with four compounds in clinical use. Further applications are under development for the treatment of Alzheimer’s disease, amyotrophic lateral sclerosis, and cardiovascular diseases. The ubiquitous distribution of MAO enzymes ensures future exciting development of novel therapeutics including the development of novel and safer formulations.
References
Aguiar LM et al (2008) CSC, an adenosine A(2A) receptor antagonist and MAO B inhibitor, reverses behavior, monoamine neurotransmission, and amino acid alterations in the 6-OHDA-lesioned rats. Brain Res 1191:192–199
Akhondzadeh S et al (2003) Selegiline in the treatment of attention deficit hyperactivity disorder in children: a double blind and randomized trial. Prog Neuropsychopharmacol Biol Psychiatry 27(5):841–845
Alda M et al (2017) Methylene blue treatment for residual symptoms of bipolar disorder: randomised crossover study. Br J Psychiatry 210(1):54–60
Alia-Klein N et al (2008) Brain monoamine oxidase A activity predicts trait aggression. J Neurosci 28(19):5099–5104
Anderson MC et al (1993) Monoamine oxidase inhibitors and the cheese effect. Neurochem Res 18(11):1145–1149
Bar-Am O et al (2015) Neuroprotective and neurorestorative activities of a novel iron chelator-brain selective monoamine oxidase-A/monoamine oxidase-B inhibitor in animal models of Parkinson’s disease and aging. Neurobiol Aging 36(3):1529–1542
Barbiero JK et al (2011) Acute but not chronic administration of pioglitazone promoted behavioral and neurochemical protective effects in the MPTP model of Parkinson’s disease. Behav Brain Res 216(1):186–192
Barone P et al (2015) A randomized clinical trial to evaluate the effects of rasagiline on depressive symptoms in non-demented Parkinson’s disease patients. Eur J Neurol 22(8):1184–1191
Bartolo M et al (2015) An explorative study regarding the effect of l-deprenyl on cognitive and functional recovery in patients after stroke. J Neurol Sci 349(1-2):117–123
Berlin I et al (1995) A reversible monoamine oxidase A inhibitor (moclobemide) facilitates smoking cessation and abstinence in heavy, dependent smokers. Clin Pharmacol Ther 58(4):444–452
Berry MD (1999) R-2HMP: an orally active agent combining independent antiapoptotic and MAO-B-inhibitory activities. CNS Drug Rev 5(2):105–124
Bette S et al (2018) Safinamide in the management of patients with Parkinson’s disease not stabilized on levodopa: a review of the current clinical evidence. Ther Clin Risk Manag 14:1737–1745
Binda C et al (2002) Structure of human monoamine oxidase B, a drug target for the treatment of neurological disorders. Nat Struct Biol 9(1):22–26
Binda C et al (2003) Insights into the mode of inhibition of human mitochondrial monoamine oxidase B from high-resolution crystal structures. Proc Natl Acad Sci U S A 100(17):9750–9755
Binda C et al (2004) Crystal structures of monoamine oxidase B in complex with four inhibitors of the N-propargylaminoindan class. J Med Chem 47(7):1767–1774
Binda C et al (2005) Binding of rasagiline-related inhibitors to human monoamine oxidases: a kinetic and crystallographic analysis. J Med Chem 48(26):8148–8154
Binda C et al (2007) Structures of human monoamine oxidase B complexes with selective noncovalent inhibitors: safinamide and coumarin analogs. J Med Chem 50(23):5848–5852
Binda C et al (2011a) Molecular insights into human monoamine oxidase B inhibition by the glitazone anti-diabetes drugs. ACS Med Chem Lett 3(1):39–42
Binda C et al (2011b) Interactions of monoamine oxidases with the antiepileptic drug zonisamide: specificity of inhibition and structure of the human monoamine oxidase B complex. J Med Chem 54(3):909–912
Blesa J et al (2015) Oxidative stress and Parkinson’s disease. Front Neuroanat 9:91
Bonato JM et al (2018) Pioglitazone reduces mortality, prevents depressive-like behavior, and impacts hippocampal neurogenesis in the 6-OHDA model of Parkinson’s disease in rats. Exp Neurol 300:188–200
Bonivento D et al (2010) Potentiation of ligand binding through cooperative effects in monoamine oxidase B. J Biol Chem 285(47):36849–36856
Borgohain R et al (2014) Two-year, randomized, controlled study of safinamide as add-on to levodopa in mid to late Parkinson’s disease. Mov Disord 29(10):1273–1280
Borroni E et al (2017) Sembragiline: a novel, selective monoamine oxidase type B inhibitor for the treatment of Alzheimer’s disease. J Pharmacol Exp Ther 362(3):413–423
Buneeva O et al (2018) The effect of neurotoxin MPTP and neuroprotector isatin on the profile of ubiquitinated brain mitochondrial proteins. Cell 7(8):91
Burke WJ et al (1993) L-deprenyl in the treatment of mild dementia of the Alzheimer type: results of a 15-month trial. J Am Geriatr Soc 41(11):1219–1225
Cai Z (2014) Monoamine oxidase inhibitors: promising therapeutic agents for Alzheimer’s disease (Review). Mol Med Rep 9(5):1533–1541
Campi N, Todeschini GP, Scarzella L (1990) Selegiline versus L-acetylcarnitine in the treatment of Alzheimer-type dementia. Clin Ther 12(4):306–314
Carta AR et al (2011) Rosiglitazone decreases peroxisome proliferator receptor-gamma levels in microglia and inhibits TNF-alpha production: new evidences on neuroprotection in a progressive Parkinson’s disease model. Neuroscience 194:250–261
Caslake R et al (2009) Monoamine oxidase B inhibitors versus other dopaminergic agents in early Parkinson’s disease. Cochrane Database Syst Rev 4:CD006661
Castagnoli K et al (2003) Inhibition of human MAO-A and MAO-B by a compound isolated from flue-cured tobacco leaves and its neuroprotective properties in the MPTP mouse model of neurodegeneration. Inflammopharmacology 11(2):183–188
Cattaneo C et al (2018) Long-term efficacy of safinamide on Parkinson’s disease chronic pain. Adv Ther 35(4):515–522
Chen JY et al (2013) Dopamine imbalance in Huntington’s disease: a mechanism for the lack of behavioral flexibility. Front Neurosci 7:114
Chouinard G et al (1993) Brofaromine in depression: a Canadian multicenter placebo trial and a review of standard drug comparative studies. Clin Neuropharmacol 16(Suppl 2):S51–S54
Coelho Cerqueira E et al (2011) Molecular insights into human monoamine oxidase (MAO) inhibition by 1,4-naphthoquinone: evidences for menadione (vitamin K3) acting as a competitive and reversible inhibitor of MAO. Bioorg Med Chem 19(24):7416–7424
Cote F et al (2004) Recent advances in understanding serotonin regulation of cardiovascular function. Trends Mol Med 10(5):232–238
Croisier E et al (2005) Microglial inflammation in the Parkinsonian substantia nigra: relationship to alpha-synuclein deposition. J Neuroinflammation 2:14
Curet O et al (1996) Befloxatone, a new reversible and selective monoamine oxidase-A inhibitor. I. Biochemical profile. J Pharmacol Exp Ther 277(1):253–264
Cutillas B, Ambrosio S, Unzeta M (2002) Neuroprotective effect of the monoamine oxidase inhibitor PF 9601N [N-(2-propynyl)-2-(5-benzyloxy-indolyl) methylamine] on rat nigral neurons after 6-hydroxydopamine-striatal lesion. Neurosci Lett 329(2):165–168
Da Prada M et al (1988) On tyramine, food, beverages and the reversible MAO inhibitor moclobemide. J Neural Transm Suppl 26:31–56
Dakic V et al (2016) Harmine stimulates proliferation of human neural progenitors. PeerJ 4:e2727
Dasgupta S et al (2018) Recognition dynamics of dopamine to human monoamine oxidase B: role of Leu171/Gln206 and conserved water molecules in the active site cavity. J Biomol Struct Dyn 36(6):1439–1462
De Colibus L et al (2005) Three-dimensional structure of human monoamine oxidase A (MAO A): relation to the structures of rat MAO A and human MAO B. Proc Natl Acad Sci U S A 102(36):12684–12689
Dong J et al (2016) Current pharmaceutical treatments and alternative therapies of Parkinson’s disease. Curr Neuropharmacol 14(4):339–355
Dos Santos RG, Hallak JE (2017) Effects of the natural beta-carboline alkaloid harmine, a main constituent of Ayahuasca, in memory and in the hippocampus: a systematic literature review of preclinical studies. J Psychoactive Drugs 49(1):1–10
Edmondson DE, Binda C (2018) Monoamine oxidases. Subcell Biochem 87:117–139
Edmondson DE et al (2009) Molecular and mechanistic properties of the membrane-bound mitochondrial monoamine oxidases. Biochemistry 48(20):4220–4230
Esteban G et al (2014) Kinetic and structural analysis of the irreversible inhibition of human monoamine oxidases by ASS234, a multi-target compound designed for use in Alzheimer’s disease. Biochim Biophys Acta 1844(6):1104–1110
Evranos-Aksoz B et al (2017) New human monoamine oxidase A inhibitors with potential anti-depressant activity: design, synthesis, biological screening and evaluation of pharmacological activity. Comb Chem High Throughput Screen 20(6):461–473
Fabbri M et al (2015) Clinical pharmacology review of safinamide for the treatment of Parkinson’s disease. Neurodegener Dis Manag 5(6):481–496
Fasipe OJ (2019) The emergence of new antidepressants for clinical use: agomelatine paradox versus other novel agents. IBRO Rep 6:95–110
Filip V, Kolibas E (1999) Selegiline in the treatment of Alzheimer’s disease: a long-term randomized placebo-controlled trial. Czech and Slovak Senile Dementia of Alzheimer Type Study Group. J Psychiatry Neurosci 24(3):234–243
Finberg JP (2014) Update on the pharmacology of selective inhibitors of MAO-A and MAO-B: focus on modulation of CNS monoamine neurotransmitter release. Pharmacol Ther 143(2):133–152
Finberg JP, Rabey JM (2016) Inhibitors of MAO-A and MAO-B in psychiatry and neurology. Front Pharmacol 7:340
Fowler JS et al (1997) Age-related increases in brain monoamine oxidase B in living healthy human subjects. Neurobiol Aging 18(4):431–435
Fowler JS et al (2010) Reversible inhibitors of monoamine oxidase-A (RIMAs): robust, reversible inhibition of human brain MAO-A by CX157. Neuropsychopharmacology 35(3):623–631
Gal S et al (2005) Novel multifunctional neuroprotective iron chelator-monoamine oxidase inhibitor drugs for neurodegenerative diseases. In vivo selective brain monoamine oxidase inhibition and prevention of MPTP-induced striatal dopamine depletion. J Neurochem 95(1):79–88
Gal S, Abassi ZA, Youdim MB (2010) Limited potentiation of blood pressure in response to oral tyramine by the anti-Parkinson brain selective multifunctional monoamine oxidase-AB inhibitor, M30. Neurotox Res 18(2):143–150
Garcia-Miralles M et al (2016) Treatment with the MAO-A inhibitor clorgyline elevates monoamine neurotransmitter levels and improves affective phenotypes in a mouse model of Huntington disease. Exp Neurol 278:4–10
Gillman PK (2006) A review of serotonin toxicity data: implications for the mechanisms of antidepressant drug action. Biol Psychiatry 59(11):1046–1051
Golko-Perez S et al (2016) Additive neuroprotective effects of the multifunctional iron chelator M30 with enriched diet in a mouse model of amyotrophic lateral sclerosis. Neurotox Res 29(2):208–217
Grover ND et al (2013) Zonisamide: a review of the clinical and experimental evidence for its use in Parkinson’s disease. Indian J Pharmacol 45(6):547–555
Henriot S et al (1994) Lazabemide (Ro 19-6327), a reversible and highly sensitive MAO-B inhibitor: preclinical and clinical findings. J Neural Transm Suppl 41:321–325
Hoon M et al (2017) The design and evaluation of an l-dopa-lazabemide prodrug for the treatment of Parkinson’s disease. Molecules 22(12):2076
Hubalek F, Pohl J, Edmondson DE (2003) Structural comparison of human monoamine oxidases A and B: mass spectrometry monitoring of cysteine reactivities. J Biol Chem 278(31):28612–28618
Hubalek F et al (2005) Demonstration of isoleucine 199 as a structural determinant for the selective inhibition of human monoamine oxidase B by specific reversible inhibitors. J Biol Chem 280(16):15761–15766
Huuskonen C et al (2019) Monoamine oxidase A inhibition protects the myocardium after experimental acute volume overload. Anatol J Cardiol 21(1):39–45
Jankovic J et al (2014) Symptomatic efficacy of rasagiline monotherapy in early Parkinson’s disease: post-hoc analyses from the ADAGIO trial. Parkinsonism Relat Disord 20(6):640–643
Jo S et al (2014) GABA from reactive astrocytes impairs memory in mouse models of Alzheimer’s disease. Nat Med 20(8):886–896
Justo LA et al (2016) Effects and mechanism of action of isatin, a MAO inhibitor, on in vivo striatal dopamine release. Neurochem Int 99:147–157
Kaludercic N et al (2010) Monoamine oxidase A-mediated enhanced catabolism of norepinephrine contributes to adverse remodeling and pump failure in hearts with pressure overload. Circ Res 106(1):193–202
Kaludercic N et al (2014) Monoamine oxidase B prompts mitochondrial and cardiac dysfunction in pressure overloaded hearts. Antioxid Redox Signal 20(2):267–280
Kennedy BP et al (2003) Early and persistent alterations in prefrontal cortex MAO A and B in Alzheimer’s disease. J Neural Transm 110(7):789–801
Khalil AA, Davies B, Castagnoli N Jr (2006) Isolation and characterization of a monoamine oxidase B selective inhibitor from tobacco smoke. Bioorg Med Chem 14(10):3392–3398
Korchounov A, Winter Y, Rossy W (2012) Combined beneficial effect of rasagiline on motor function and depression in de novo PD. Clin Neuropharmacol 35(3):121–124
Kupershmidt L et al (2012) Multi-target, neuroprotective and neurorestorative M30 improves cognitive impairment and reduces Alzheimer’s-like neuropathology and age-related alterations in mice. Mol Neurobiol 46(1):217–220
Lange DJ et al (1998) Selegiline is ineffective in a collaborative double-blind, placebo-controlled trial for treatment of amyotrophic lateral sclerosis. Arch Neurol 55(1):93–96
Langston JW et al (1984) Pargyline prevents MPTP-induced parkinsonism in primates. Science 225(4669):1480–1482
Le W et al (1997) (−)-Deprenyl protection of 1-methyl-4 phenylpyridinium ion (MPP+)-induced apoptosis independent of MAO-B inhibition. Neurosci Lett 224(3):197–200
Lee KC, Chen JJ (2007) Transdermal selegiline for the treatment of major depressive disorder. Neuropsychiatr Dis Treat 3(5):527–537
Lee EY et al (2012) Rosiglitazone, a PPAR-gamma agonist, protects against striatal dopaminergic neurodegeneration induced by 6-OHDA lesions in the substantia nigra of rats. Toxicol Lett 213(3):332–344
Li M et al (2006) Functional role of the “aromatic cage” in human monoamine oxidase B: structures and catalytic properties of Tyr435 mutant proteins. Biochemistry 45(15):4775–4784
Liu F et al (2017a) Harmine produces antidepressant-like effects via restoration of astrocytic functions. Prog Neuro-Psychopharmacol Biol Psychiatry 79(Pt B):258–267
Liu X et al (2017b) Harmine is an inflammatory inhibitor through the suppression of NF-kappaB signaling. Biochem Biophys Res Commun 489(3):332–338
Liu Y et al (2019) Attenuation of ischemic stroke-caused brain injury by a monoamine oxidase inhibitor involves improved proteostasis and reduced neuroinflammation. Mol Neurobiol 57(2):937–948
Lotufo-Neto F, Trivedi M, Thase ME (1999) Meta-analysis of the reversible inhibitors of monoamine oxidase type A moclobemide and brofaromine for the treatment of depression. Neuropsychopharmacology 20(3):226–247
Ludolph AC et al (2018) Safety and efficacy of rasagiline as an add-on therapy to riluzole in patients with amyotrophic lateral sclerosis: a randomised, double-blind, parallel-group, placebo-controlled, phase 2 trial. Lancet Neurol 17(8):681–688
Machado MMF et al (2019) PPAR-gamma agonist pioglitazone reduces microglial proliferation and NF-kappaB activation in the substantia nigra in the 6-hydroxydopamine model of Parkinson’s disease. Pharmacol Rep 71(4):556–564
Mallajosyula JK et al (2008) MAO-B elevation in mouse brain astrocytes results in Parkinson’s pathology. PLoS One 3(2):e1616
Marco-Contelles J et al (2016) ASS234, as a new multi-target directed propargylamine for Alzheimer’s disease therapy. Front Neurosci 10:294
Martin HL et al (2012) Pharmacological manipulation of peroxisome proliferator-activated receptor gamma (PPARgamma) reveals a role for anti-oxidant protection in a model of Parkinson’s disease. Exp Neurol 235(2):528–538
Mawhinney M, Cole D, Azzaro AJ (2003) Daily transdermal administration of selegiline to guinea-pigs preferentially inhibits monoamine oxidase activity in brain when compared with intestinal and hepatic tissues. J Pharm Pharmacol 55(1):27–34
Meiser J, Weindl D, Hiller K (2013) Complexity of dopamine metabolism. Cell Commun Signal 11(1):34
Meyer JH et al (2006) Elevated monoamine oxidase a levels in the brain: an explanation for the monoamine imbalance of major depression. Arch Gen Psychiatry 63(11):1209–1216
Milczek EM et al (2011) The ‘gating’ residues Ile199 and Tyr326 in human monoamine oxidase B function in substrate and inhibitor recognition. FEBS J 278(24):4860–4869
Morais M et al (2014) The effects of chronic stress on hippocampal adult neurogenesis and dendritic plasticity are reversed by selective MAO-A inhibition. J Psychopharmacol 28(12):1178–1183
Muller T, Mohr JD (2019) Pharmacokinetics of monoamine oxidase B inhibitors in Parkinson’s disease: current status. Expert Opin Drug Metab Toxicol 15(5):429–435
Muller T et al (2013) Switch from selegiline to rasagiline is beneficial in patients with Parkinson’s disease. J Neural Transm 120(5):761–765
Murata M et al (2015) Zonisamide improves wearing-off in Parkinson’s disease: a randomized, double-blind study. Mov Disord 30(10):1343–1350
Murata M et al (2016) Randomized placebo-controlled trial of zonisamide in patients with Parkinson’s disease. Neurol Clin Neurosci 4(1):10–15
Murata M et al (2018) Adjunct zonisamide to levodopa for DLB parkinsonism: a randomized double-blind phase 2 study. Neurology 90(8):e664–e672
Myllyla VV et al (1997) Selegiline as the primary treatment of Parkinson’s disease--a long-term double-blind study. Acta Neurol Scand 95(4):211–218
Nagatsu T, Sawada M (2006) Molecular mechanism of the relation of monoamine oxidase B and its inhibitors to Parkinson’s disease: possible implications of glial cells. J Neural Transm Suppl 71:53–65
Nandigama RK, Miller JR, Edmondson DE (2001) Loss of serotonin oxidation as a component of the altered substrate specificity in the Y444F mutant of recombinant human liver MAO A. Biochemistry 40(49):14839–14846
Nave S et al (2017) Sembragiline in moderate Alzheimer’s disease: results of a randomized, double-blind, placebo-controlled phase II trial (MAyflOwer RoAD). J Alzheimers Dis 58(4):1217–1228
Neurol L (2015) Pioglitazone in early Parkinson’s disease: a phase 2, multicentre, double-blind, randomised trial. Lancet Neurol 14(8):795–803
Nishijima H et al (2018) Zonisamide enhances motor effects of levodopa, not of apomorphine, in a rat model of Parkinson’s disease. Parkinsons Dis 2018:8626783
Ogata A et al (2003) Isatin, an endogenous MAO inhibitor, improves bradykinesia and dopamine levels in a rat model of Parkinson’s disease induced by Japanese encephalitis virus. J Neurol Sci 206(1):79–83
Oki M et al (2017) Zonisamide ameliorates levodopa-induced dyskinesia and reduces expression of striatal genes in Parkinson model rats. Neurosci Res 122:45–50
Olanow CW et al (2009) A double-blind, delayed-start trial of rasagiline in Parkinson’s disease. N Engl J Med 361(13):1268–1278
Ooi J, Hayden MR, Pouladi MA (2015) Inhibition of excessive monoamine oxidase A/B activity protects against stress-induced neuronal death in Huntington disease. Mol Neurobiol 52(3):1850–1861
Oz M, Lorke DE, Petroianu GA (2009) Methylene blue and Alzheimer’s disease. Biochem Pharmacol 78(8):927–932
Panarsky R, Luques L, Weinstock M (2012) Anti-inflammatory effects of ladostigil and its metabolites in aged rat brain and in microglial cells. J Neuroimmune Pharmacol 7(2):488–498
Park JH et al (2019) Newly developed reversible MAO-B inhibitor circumvents the shortcomings of irreversible inhibitors in Alzheimer’s disease. Sci Adv 5(3):eaav0316
Parkinson Study G (1989) Effect of deprenyl on the progression of disability in early Parkinson’s disease. N Engl J Med 321(20):1364–1371
Parkinson Study G (1994) A controlled trial of lazabemide (Ro 19-6327) in levodopa-treated Parkinson’s disease. Arch Neurol 51(4):342–347
Parkinson Study G (2002) A controlled trial of rasagiline in early Parkinson disease: the TEMPO study. Arch Neurol 59(12):1937–1943
Parkinson Study G (2004) A controlled, randomized, delayed-start study of rasagiline in early Parkinson disease. Arch Neurol 61(4):561–566
Perez V, Unzeta M (2003) PF 9601N [N-(2-propynyl)-2-(5-benzyloxy-indolyl) methylamine], a new MAO-B inhibitor, attenuates MPTP-induced depletion of striatal dopamine levels in C57/BL6 mice. Neurochem Int 42(3):221–229
Pérez V et al (1999) Relevance of benzyloxy group in 2-indolyl methylamines in the selective MAO-B inhibition. Br J Pharmacol 127(4):869–876
Pimentel LS et al (2015) The multi-target drug m30 shows pro-cognitive and anti-inflammatory effects in a rat model of Alzheimer’s disease. J Alzheimers Dis 47(2):373–383
Pinto M et al (2016) Pioglitazone ameliorates the phenotype of a novel Parkinson’s disease mouse model by reducing neuroinflammation. Mol Neurodegener 11:25
Poewe W et al (2017) Parkinson disease. Nat Rev Dis Primers 3:17013
Przuntek H et al (1999) SELEDO: a 5-year long-term trial on the effect of selegiline in early Parkinsonian patients treated with levodopa. Eur J Neurol 6(2):141–150
Rabey JM et al (2000) Rasagiline mesylate, a new MAO-B inhibitor for the treatment of Parkinson’s disease: a double-blind study as adjunctive therapy to levodopa. Clin Neuropharmacol 23(6):324–330
Ramachandraih CT et al (2011) Antidepressants: from MAOIs to SSRIs and more. Indian J Psychiatry 53(2):180–182
Rascol O et al (2005) Rasagiline as an adjunct to levodopa in patients with Parkinson’s disease and motor fluctuations (LARGO, Lasting effect in Adjunct therapy with Rasagiline Given Once daily, study): a randomised, double-blind, parallel-group trial. Lancet 365(9463):947–954
Rascol O et al (2011) A double-blind, delayed-start trial of rasagiline in Parkinson’s disease (the ADAGIO study): prespecified and post-hoc analyses of the need for additional therapies, changes in UPDRS scores, and non-motor outcomes. Lancet Neurol 10(5):415–423
Rubinstein S et al (2006) Placebo-controlled study examining effects of selegiline in children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol 16(4):404–415
Ryan M, Eatmon CV, Slevin JT (2019) Drug treatment strategies for depression in Parkinson disease. Expert Opin Pharmacother 20(11):1351–1363
Sagi Y, Drigues N, Youdim MB (2005) The neurochemical and behavioral effects of the novel cholinesterase-monoamine oxidase inhibitor, ladostigil, in response to L-dopa and L-tryptophan, in rats. Br J Pharmacol 146(4):553–560
Sanches RF et al (2016) Antidepressant effects of a single dose of ayahuasca in patients with recurrent depression: a SPECT study. J Clin Psychopharmacol 36(1):77–81
Sano H, Murata M, Nambu A (2015) Zonisamide reduces nigrostriatal dopaminergic neurodegeneration in a mouse genetic model of Parkinson’s disease. J Neurochem 134(2):371–381
Santhanasabapathy R, Sudhandiran G (2015) Farnesol attenuates lipopolysaccharide-induced neurodegeneration in Swiss albino mice by regulating intrinsic apoptotic cascade. Brain Res 1620:42–56
Sanz E et al (2008) Anti-apoptotic effect of Mao-B inhibitor PF9601N [N-(2-propynyl)-2-(5-benzyloxy-indolyl) methylamine] is mediated by p53 pathway inhibition in MPP+-treated SH-SY5Y human dopaminergic cells. J Neurochem 105(6):2404–2417
Schapira AH et al (2013) Long-term efficacy and safety of safinamide as add-on therapy in early Parkinson’s disease. Eur J Neurol 20(2):271–280
Schneider LS et al (2019) Low-dose ladostigil for mild cognitive impairment: a phase 2 placebo-controlled clinical trial. Neurology 93(15):e1474–e1484
Segura-Aguilar J et al (2014) Protective and toxic roles of dopamine in Parkinson’s disease. J Neurochem 129(6):898–915
Shulman KI, Herrmann N, Walker SE (2013) Current place of monoamine oxidase inhibitors in the treatment of depression. CNS Drugs 27(10):789–797
Sian J et al (1994) Alterations in glutathione levels in Parkinson’s disease and other neurodegenerative disorders affecting basal ganglia. Ann Neurol 36(3):348–355
Sivenius J et al (2001) Selegiline treatment facilitates recovery after stroke. Neurorehabil Neural Repair 15(3):183–190
Smith KM, Eyal E, Weintraub D (2015) Combined rasagiline and antidepressant use in Parkinson disease in the ADAGIO study: effects on nonmotor symptoms and tolerability. JAMA Neurol 72(1):88–95
Soliman A et al (2012) Convergent effects of acute stress and glucocorticoid exposure upon MAO-A in humans. J Neurosci 32(48):17120–17127
Son SY et al (2008) Structure of human monoamine oxidase A at 2.2-Å resolution: the control of opening the entry for substrates/inhibitors. Proc Natl Acad Sci U S A 105(15):5739–5744
Sonsalla PK et al (2010) The antiepileptic drug zonisamide inhibits MAO-B and attenuates MPTP toxicity in mice: clinical relevance. Exp Neurol 221(2):329–334
Statland JM et al (2019) Rasagiline for amyotrophic lateral sclerosis: a randomized, controlled trial. Muscle Nerve 59(2):201–207
Stocchi F, Torti M (2016) Adjuvant therapies for Parkinson’s disease: critical evaluation of safinamide. Drug Des Devel Ther 10:609–618
Stocchi F et al (2006) Symptom relief in Parkinson disease by safinamide: biochemical and clinical evidence of efficacy beyond MAO-B inhibition. Neurology 67(7 Suppl 2):S24–S29
Sturm S et al (2017) Positron emission tomography measurement of brain MAO-B inhibition in patients with Alzheimer’s disease and elderly controls after oral administration of sembragiline. Eur J Nucl Med Mol Imaging 44(3):382–391
Szoko E et al (2018) Pharmacological aspects of the neuroprotective effects of irreversible MAO-B inhibitors, selegiline and rasagiline, in Parkinson’s disease. J Neural Transm (Vienna) 125(11):1735–1749
Tariot PN et al (1987) Cognitive effects of L-deprenyl in Alzheimer’s disease. Psychopharmacology 91(4):489–495
Tripathi AC et al (2018) Privileged scaffolds as MAO inhibitors: retrospect and prospects. Eur J Med Chem 145:445–497
Ulusoy GK et al (2011) Effects of pioglitazone and retinoic acid in a rotenone model of Parkinson’s disease. Brain Res Bull 85(6):380–384
Upadhyay AK et al (2008) Determination of the oligomeric states of human and rat monoamine oxidases in the outer mitochondrial membrane and octyl beta-D-glucopyranoside micelles using pulsed dipolar electron spin resonance spectroscopy. Biochemistry 47(6):1554–1566
Villeneuve C et al (2013) p53-PGC-1alpha pathway mediates oxidative mitochondrial damage and cardiomyocyte necrosis induced by monoamine oxidase-A upregulation: role in chronic left ventricular dysfunction in mice. Antioxid Redox Signal 18(1):5–18
Weinreb O et al (2012) Ladostigil: a novel multimodal neuroprotective drug with cholinesterase and brain-selective monoamine oxidase inhibitory activities for Alzheimer’s disease treatment. Curr Drug Targets 13(4):483–494
Yasar S, Goldberg JP, Goldberg SR (1996) Are metabolites of l-deprenyl (selegiline) useful or harmful? Indications from preclinical research. J Neural Transm Suppl 48:61–73
Yasar S et al (2006) Metabolic transformation plays a primary role in the psychostimulant-like discriminative-stimulus effects of selegiline [(R)-(−)-deprenyl]. J Pharmacol Exp Ther 317(1):387–394
Yogev-Falach M et al (2006) A multifunctional, neuroprotective drug, ladostigil (TV3326), regulates holo-APP translation and processing. FASEB J 20(12):2177–2179
Youdim MB (2012) M30, a brain permeable multitarget neurorestorative drug in post nigrostriatal dopamine neuron lesion of parkinsonism animal models. Parkinsonism Relat Disord 18(Suppl 1):S151–S154
Youdim MB (2013) Multi target neuroprotective and neurorestorative anti-Parkinson and anti-Alzheimer drugs ladostigil and m30 derived from rasagiline. Exp Neurobiol 22(1):1–10
Youdim MB, Edmondson D, Tipton KF (2006) The therapeutic potential of monoamine oxidase inhibitors. Nat Rev Neurosci 7(4):295–309
Youdim MB et al (2014) Promises of novel multi-target neuroprotective and neurorestorative drugs for Parkinson’s disease. Parkinsonism Relat Disord 20(Suppl 1):S132–S136
Zanderigo F et al (2018) [(11)C]Harmine binding to brain monoamine oxidase A: test-retest properties and noninvasive quantification. Mol Imaging Biol 20(4):667–681
Zanotti-Fregonara P, Bottlaender M (2014) [11C]befloxatone distribution is well correlated to monoamine oxidase A protein levels in the human brain. J Cereb Blood Flow Metab 34(12):1951–1952
Zanotti-Fregonara P et al (2014) Imaging of monoamine oxidase-A in the human brain with [11C]befloxatone: quantification strategies and correlation with mRNA transcription maps. Nucl Med Commun 35(12):1254–1261
Zheng H et al (2005) Novel multifunctional neuroprotective iron chelator-monoamine oxidase inhibitor drugs for neurodegenerative diseases: in vitro studies on antioxidant activity, prevention of lipid peroxide formation and monoamine oxidase inhibition. J Neurochem 95(1):68–78
Zheng H, Fridkin M, Youdim MB (2010) Site-activated chelators derived from anti-Parkinson drug rasagiline as a potential safer and more effective approach to the treatment of Alzheimer’s disease. Neurochem Res 35(12):2117–2123
Zhou Y, Zhao ZQ, Xie JX (2001) Effects of isatin on rotational behavior and DA levels in caudate putamen in Parkinsonian rats. Brain Res 917(1):127–132
Zhou S, Chen G, Huang G (2018) Design, synthesis and biological evaluation of lazabemide derivatives as inhibitors of monoamine oxidase. Bioorg Med Chem 26(17):4863–4870
Zisook S (1985) A clinical overview of monoamine oxidase inhibitors. Psychosomatics 26(3):240–251
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Duarte, P., Cuadrado, A., León, R. (2020). Monoamine Oxidase Inhibitors: From Classic to New Clinical Approaches. In: Schmidt, H.H.H.W., Ghezzi, P., Cuadrado, A. (eds) Reactive Oxygen Species . Handbook of Experimental Pharmacology, vol 264. Springer, Cham. https://doi.org/10.1007/164_2020_384
Download citation
DOI: https://doi.org/10.1007/164_2020_384
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-68509-6
Online ISBN: 978-3-030-68510-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)