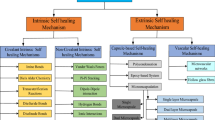
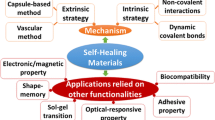
Abstract
Damage and fatigue are ever-present facts of life. Given enough time, even the most robust material, whether man-made or natural, succumbs to the deleterious effects of cracks, fissures, and defects during normal use. Traditionally, materials engineers have approached this problem by creating damage-tolerant structures, intensive quality control before use, vigilant inspection during use, and designing materials to function well below their theoretical limit. Living organisms, on the other hand, routinely produce materials that function close to their theoretical limit as a result of their remarkable ability to self-heal a range of non-catastrophic damage events. For this reason, many researchers in the last 15 years have turned to nature for inspiration for the design and development of self-healing composites and polymeric materials. However, these efforts have so far only scratched the surface of the richness of natural self-repair processes. In the present review, we provide an overview of some paradigmatic and well-studied examples of self-repair in living systems. The core of this overview takes the form of a number of case studies that provide a detailed description of the structure–function relationships defining the healing mechanism. Case studies include a number of examples dependent on cellular action in both animals (e.g., limb regeneration, antler growth, bone healing, and wound healing) and plants (e.g., latex-based healing, plant grafting, and wound closure in woody vines and succulent plants). Additionally, we examine several examples of acellular self-repair in biopolymeric materials (e.g., mussel byssus, caddisfly silks, and whelk egg capsules) that are already inspiring the development of a number of self-healing polymers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
A major future challenge in materials engineering is the production of high-performance, damage-tolerant, and renewable materials that can be used in technical and biomedical applications. In particular, the development of self-healing materials capable of repairing damage has emerged as an important and fast-growing research focus [1]. However, successfully integrating autonomic and intrinsic self-healing behavior into polymers and composites presents numerous challenges. For biological materials, on the other hand, self-repair is business as usual. For this reason, many researchers have begun to turn to biological materials for inspiration [2–8]. The field of bio-inspired and biomimetic materials is founded on the basic premise that biology has something to teach humans about efficient and economic means of designing and building versatile high-performance materials for a wide variety of functions. Self-healing, broadly defined, is a ubiquitous feature of materials made by living organisms. Familiar examples in everyday life include healing of cuts and scrapes, as well as mending of broken bones. However, as becomes clear in this review, the complexity and diversity of healing mechanisms in living nature is expansive.
It is important at the outset to define what is meant by self-healing in a biological context. Assuming a strong evolutionary pressure on the development of self-repair functions, independent evolution of mechanisms to cope with wounds and other damage can be expected. Indeed, an enormous range and variety of self-repair mechanisms can be observed throughout the various kingdoms of biology. Here, we define self-repair as the recovery toward native material properties following a damage event. Healing does not have to result in complete restoration of the pre-damaged structure and properties; however, at least the function of the material must be regained. Damage, in this sense, includes the formation of microcracks or macroscopic cracks, plastic deformation, and loss of mechanical function or even loss of tissue, organs, or entire limbs. For each of these categories, there are examples in nature where partial or complete self-repair occurs. In this review, a number of case studies are presented that provide an overview of the current understanding of specific model systems representing a range of different healing mechanisms. In addition, recent efforts to develop bio-inspired or biomimetic materials with self-repair functions are highlighted where relevant.
It is pertinent at this point to introduce a crucial distinction in biological healing – that between cellular and acellular healing. Typically, healing processes in living organisms are dependent on cellular metabolism to function. For example, during bone mending, wound closure, or healing of tissues such as tendon in vertebrates, the intervention of living cells is required to initiate the healing process [9, 10]. Here, the cells are active players in breaking down old tissue and rebuilding new, living tissue. On the other hand, there are several less prominent, but equally impressive examples of intrinsic and autonomic self-repair responses that occur in non-living biological materials functioning outside the confines of a living body (i.e., extracorporeally) in the absence of an active metabolism. Examples include the mussel byssus, caddisfly silk, and whelk egg capsule material [11–13], which are discussed in detail later (see Sect. 2.9). The differences between acellular and cellular biological healing are especially relevant when attempting to gain inspiration from a system, because they necessarily entail starkly different levels of inherent complexity.
In spite of the broad range of healing processes observed throughout living nature, self-repair mechanisms in many animals and plants can in general be subdivided into two phases: an initial self-sealing phase and a subsequent self-healing phase [9, 10, 14, 15]. Using “self-repair” as an umbrella term, the two phases are characterized by anatomical and biochemical modifications and changes in biomechanical properties (Table 1) [16]. Interestingly, these definitions also hold true for (most) bio-inspired technical solutions, where self-sealing and self-healing can be found either successively or individually. Self-sealing mechanisms rapidly close fissures and protect organisms against invasion of pathogenic germs and loss of precious body fluids (e.g., water, sap, blood) but, in general, the mechanical properties of the tissue are not recovered at this point. Self-sealing can be seen as an initial means of quickly stabilizing the wound following a damage event, and typically function by deposition/coagulation of healing agents that seal the wound (e.g., latex in plants [17] and blood clots in animals [9]) or by using existing surrounding tissue to create a physical barrier (e.g., Delosperma cooperi [14, 15, 18]). The self-healing phase, on the other hand, is characterized in general by disappearance of the fissure and at least partial recovery of the native material structure and properties. As such, this phase requires much longer time spans to develop and involves more complex and metabolically intensive mechanisms for replacing damaged tissue, such as cell proliferation, callus formation, synthesis of biomolecules (e.g., cellulose, lignin, or collagen) and even biomineralization [9, 10]. Further details of these processes are discussed in the individual case studies.
Before proceeding to the case studies, it is important to mention some crucial differences between biological materials and man-made materials that are vital when considering technological transfer based on a biological archetype. First, biological materials, in contrast to engineering materials, are assembled in a bottom-up process from biomolecular building blocks that are synthesized in living cells. They are often self-assembled into complex hierarchical assemblies with highly intricate architectures at the nanoscale and possess carefully crafted interfaces between components at multiple length scales [19, 20]. Thus, self-repair is typically an emergent function arising from multiple mechanisms cooperating at several length scales. Second, although it is often stated that biological materials are “optimized” for a particular function, biological materials have in fact evolved to be multifunctional. For engineering applications, there are usually one or more functions that the material is expected to perform. In the case of biological materials, a battery of selective pressures encountered over the evolutionary history of the organism influence the final product. In fact, as pointed out by Gould and Lewontin in their classic paper [21], the characteristics of biological organisms (and the materials they produce) could simply be a byproduct of evolution (i.e., evolutionary baggage) rather than being shaped by adaptive selection. For this reason, biological materials are almost always multifunctional, and some design considerations might be superfluous for the materials engineer hoping to replicate just a single function (e.g., self-healing). For example, although bone is a structural material that exhibits impressive self-healing behavior, it is also a factory for production of white blood cells and a store of calcium for the body, which might have competing design considerations.
These inherent differences between natural and man-made materials highlight the challenges of successful transfer of biological self-healing into technical applications. On one hand, although it is necessary to take a reductionist approach when faced with the complexity of natural material design, one must be cautious not to reduce too much and lose the desired emergent behavior. On the other hand, the fact that biological materials are inherently multifunctional means that a large portion of the design features are not essential for the function of interest, in this case self-healing. Thus, it is important to elucidate the essential parameters contributing to the healing behavior, while eliminating redundant or superfluous features. This of course requires a thorough understanding of the composition–structure–function relationships defining the material. Hence, the primary goal of this review is to provide a range of case studies of well-investigated examples of biological self-healing.
2 Case Studies
The following subsections provide case studies for a range of self-repair mechanisms observed in biological archetypes. This is not meant to be an exhaustive list of self-healing in living nature, but rather an overview that highlights the inherent diversity of damage that can be healed and current understanding of the mechanisms at play. Space restrictions necessitate that the level of detail for each case study is condensed; thus, we recommend that the reader refers to the cited references for further details. The review is primarily aimed at chemical or physical material scientists, who are probably less familiar with biology and molecular biology. Hence, general terminology is used and little detail is given for the signaling pathways and molecules involved in the regulation of biological healing responses.
2.1 Limb Regrowth
Regeneration refers to the replacement of lost body parts and the restoration of both their structure and function. Regeneration occurs in simple organisms such as planarians (a type of flatworm) and Hydra (simple freshwater polyps), which are able to regenerate their whole body from only small parts, as well as in more complex organisms such as urodele amphibians (salamander, newt, and axolotl), frogs (Xenopus spp.), and zebrafish [22]. It is not understood why some animals are able to regenerate and others, including humans, largely lack this ability [22]; however, the general view is that regenerative capacities were lost rather than gained during evolution. In this case study, we focus on limb regrowth in salamanders as it represents a well-studied and striking example of regeneration [23].
In addition to limbs and tails, salamanders are able to regenerate the upper and lower jaw, certain parts of the eye, the intestine, and small sections of the heart [24]. Of these, the complete regrowth of a limb (including its complex tissue patterning, full vascularization, and innervation) stands out as the most remarkable. Cutting the limb of a salamander at any place along the proximodistal axis (i.e., somewhere between shoulder and wrist) initiates a regeneration process that results in regrowth of the missing part of the limb (Fig. 1). Remarkably, this procedure can be repeated a number of times with the same result. Limb regeneration can be subdivided into three phases, which are neither temporally nor spatially distinct [26]. During the initial sealing phase, the wound surface becomes covered by a specialized epithelium (the so-called wound epithelium) within a few hours after amputation. Over time, this epithelium transforms into a specialized secretory epithelia, which is thought to be the source of signals crucial in organizing the regenerative response. During the second phase, the wound epithelium prompts the cells in the underlying tissues to de-differentiate (i.e., to return to a state of less-specialized cell characteristics) and to proliferate, forming the so-called blastema at the end of the amputated limb. The blastema is a collection of cells with the potential to form specific organs or body parts (Fig. 1a). In contrast to previous theories, this blastema does not consist of a homogeneous mass of pluripotent cells, but rather of a more heterogeneous aggregation of progenitor cells with clear restrictions in their differentiation potential [23]. During the final phase, which occurs after about 2 weeks, the cells in the blastema re-differentiate and organize to build an exact replica of the amputated part of the limb (Fig. 1). At this stage, the blastema behaves similarly to a developing embryonic limb bud, such that the pathways of the two processes – limb regeneration and development – are thought to converge [26].
Amphibian limb regeneration. (a) Limb regrowth in salamanders: after amputating the limb, a blastema (in brown) forms at the wound surface. The cells of the blastema are the source for the newly forming tissue of the regenerated limb. (b) Progression over time of limb regeneration following amputation of distal (left) and proximal (right) regions of the forelimb in a red spotted newt. For comparison, the original limb is shown at the top (Adapted from [24, 25] with permission)
Diverse and creative transplantation experiments with amputated salamander limbs have clearly demonstrated the considerable autonomy of the limb blastema [23, 24] and its positional memory of its original environment (i.e., the blastema carries the information to grow the limb from which it was excised). The rule of distal transformation states that only limb elements that are more distal to its positional memory are regenerated. As a consequence, when an upper arm blastema is transplanted onto a lower arm stump, the blastema re-forms all the distal structures starting from its position (i.e., the regenerated limb includes parts of a second lower arm). The rule of distal transformation was convincingly demonstrated by an experiment in which the orientation of the limb was inverted [27]. A salamander forelimb was amputated through the hand and the hand end was sutured to the side of the body. After successful healing, a second cut was performed through the upper limb. Because both stumps carried the positional memory of an upper limb, two complete limbs were regenerated. Blastemal autonomy extends further to limb identity. The transplantation of a forelimb blastema onto a hindlimb stump gives rise to regeneration of a forelimb. Furthermore, positional memory is not restricted just to the proximodistal axis of the limb, but extends to circumferential information in the amputation plane. For example, in a transplantation experiment in which a left upper arm blastema was transplanted to a right upper arm stump, positional discontinuities in the circumferential order of tissues was unavoidable, with the result that two extra limbs were formed [28].
Although clear parallels can be drawn between the process of limb regeneration and embryonic development of limbs from limb buds, it is still an open question how similar the underlying mechanisms actually are. Along these lines, although the cellular composition and cell signaling of the blastema and limb bud were found to be largely similar, significant differences are present regarding the starting point of each process and the relative length scales. In contrast to a developing embryo, the multiple tissues in an amputated limb of an adult salamander represent an enormously complex initial condition for regeneration [23]. Additionally, the size of a limb amputation plane is approximately tenfold larger than a limb bud. Although some have postulated that regeneration is controlled by diffusion of signaling molecules, the large length scale of the amputation plane calls this assertion into question [24]. Additional differences between regeneration and development concern the role of innervation for the regenerative response. In fact, in some cases, surgical intervention to deviate the nerve endings to a lateral wound on the limb induced the growth of a fully patterned limb out of this lateral wound in adult animals [23, 26]. Under the same experimental conditions, the regenerative outcome was significantly improved by an additional skin graft, which is thought to promote regeneration by its diverse positional information [29].
2.2 Antler Growth
In a process not dissimilar to limb regeneration, antlers grow on the skull in most species of the deer family. Antlers are branched structures made out of bone that regrow each year and should not be confused with horns, which are unbranched permanent structures that consist of keratinized tissue at their surface [30–32]. In practice, antler growth is a quite impressive feat: antler can grow to a length exceeding 1.5 m in a time span of less than 5 months, with peak growth rates of nearly 1.75 cm/day [30]. The enormous demand for minerals to build up an antler of 30 kg is satisfied by an increase in bone turnover of the skeleton, resulting in osteoporotic conditions, particularly in non-weight-bearing bones (e.g., ribs) [32].
Notably, deer antler growth is the only example of regeneration of lost organs in mammals. The growth of antler does not start directly from the skull, but rather from bony protuberances termed pedicles, which are formed when the animal approaches puberty. The tissue from which pedicles and antlers develop is the so-called antlerogenic periosteum, which covers specific parts of the deer’s frontal bone [33]. The antlerogenic potential of this early tissue was demonstrated in grafting experiments, in which the growth of antler was induced on different underlying bones and even on the skulls of nude mice [34]. The annual cycle of antler regeneration is controlled by sex hormones and begins in red deer with the casting (loss) of the old antler in spring. Sealing of the breakage plane occurs via formation of a blood clot followed by a scab. Within hours after shedding the old antler, formation of a wound epithelium over the distal ends of the pedicles begins, similar to the second stage of limb regeneration in salamander (see Sect. 2.1). It was argued that wound healing with minimal scar formation is a necessary prerequisite for successful appendage regeneration [31, 35]. In contrast to limb regeneration, full-thickness skin, including a dermis, is formed that develops later into the so-called velvet, which is a hairy surface that covers the antler during growth and is primarily responsible for the blood supply. At the distal ends of the pedicles below the skin, a mass of undifferentiated cells begins to accumulate. Unlike limb regeneration, however, this mesenchymal growth zone is based on stem cells without strong de-differentiation of cells and, therefore, is not considered a blastema [36].
Longitudinal growth of the antler occurs via endochondral ossification (i.e., ossification via a cartilage precursor). Within the growth zone, cell proliferation and differentiation into cartilage cells occurs. These chondroblasts and chondrocytes organize themselves into vertical columns with vascular space between them. At this point, remodeling occurs, by which cartilage is gradually replaced by bone (Fig. 2). Additionally, the antler grows laterally, but at a much slower rate and via a direct bone formation process termed intramembranous ossification. The final result – the mature antler – consists of a core of trabecular bone surrounded by a sleeve of compact bone. After growth has ceased and mineralization of the antler is complete, the velvet (together with the periosteum) is removed. With the bare bone exposed, the structure is then referred to as “hard antler.” The general view is that the hard antler consists of dead material because velvet shedding results in interruption of the blood supply [31].
Antler growth. Longitudinal antler growth proceeds via the initial construction of a tubular scaffold, which is then filled with cylindrical osteons (for a more detailed description see text) (From [37] with permission)
The morphology of antlers typically changes with age. In red deer, the peak in antler complexity (possessing a maximum number of tines) is reached in middle-aged adult animals. It is not known how information about previously cast antler morphology is stored in the animal’s organism. Even more puzzling is the phenomenon referred to as trophic memory. An injury of the antler during growth resulting in malformation of its morphology is not restricted to the afflicted antler. Rather, the morphological abnormality can reappear in the antler of the successive year and can be observed over several cycles of antler regrowth [38]. A crucial role of the nervous system was hypothesized because morphological abnormalities were not conserved when the trauma occurred with the animal under anesthesia [39]. To explain the phenomena of trophic memory, a model was proposed based on an encoded target morphology in the organism [40]; however, this is still an open question.
From a materials science perspective, with regards to the development of self-healing materials, antler provides two further notable features: (1) Antler achieves its remarkable growth rates by first constructing a porous framework. During longitudinal growth, for example, cartilage is transformed into bone with a honeycomb structure possessing cylindrical pores aligned along the antler axis (Fig. 2). In a second step, this scaffold is filled with bone with a collagen matrix that is well organized along the pores, leading to rapid assembly of a high-performance composite [37]. (2) Antlers are damage-tolerant structures with mechanical properties that are specifically adapted to withstand the high impacts occurring during combat. Compared with bone, antler is protected against fracture by high impact energy absorption [41] and high fracture toughness as a result of crack deflection mechanisms at the crack tip and bridging mechanisms in its wake [42]. Assessment of the deformation behavior showed that sliding between mineral and collagen in antler can dissipate energy on different length scales and, thereby, increase toughness [43].
2.3 Bone Healing
Bone is a complex hierarchically structured material (organ) possessing self-healing abilities on multiple length scales, ranging from repairing macroscopic fractures to nanoscale re-formation of reversible bonds [10]. Importantly, bone healing is an entirely regenerative process, resulting in the restoration of the pre-fractured state without scar tissue formation. At the micron scale, bone is constantly freed from damaged material by bone remodeling, and at the molecular scale, plastic deformation of bone is enabled by constant exchange of noncovalent molecular bonds.
Loading bones beyond their ultimate strength results not only in fracture of the bone, but is usually accompanied by significant damage to the surrounding soft tissue. In a typical situation following mechanical fixation of the fractured region, the bone fragments are able to move slightly against one another, leading to so-called secondary bone healing via the formation of extra tissue in the form of a callus. The process of secondary bone healing is subdivided into three overlapping phases [44, 45] (Fig. 3). During the initial reactive phase, a blood clot is formed and immune cells clean the site of dead material in an inflammatory response. At this point, the hematoma is infiltrated by small blood vessels and fibroblasts and is transformed into so-called granulation tissue. In the second reparative phase, mesenchymal progenitor cells migrate into the callus. Differentiation of these cells enables them to produce fibrous tissue, cartilage, fibro-cartilage, or bone. This crucial cell differentiation step is thought to be at least partly mechanically controlled, such that low mechanical stimulation results in direct bone formation, whereas cartilage is formed in locations of higher mechanical stimulation (e.g., around the fracture gap) [46]. Similarly to antler growth, bone formation during bone healing can occur directly via intramembranous ossification or by first forming cartilage in the process of endochondral ossification [47]. Also similar to antler growth, the fast bone growth required for rapid mechanical stabilization of the fracture site is obtained by first providing a scaffold of low bone quality, which is then filled by bone of improved quality [48]. In the final remodeling phase, dispensable excess bone material is removed, resulting in bone that is virtually indistinguishable in structure and function from the pre-fractured initial configuration.
The different phases of secondary bone healing of a long bone. The longitudinal sections show, from left to right, the broken cortex (purple) with the hematoma (light blue), followed by the succession of different soft and hard tissues within the fracture callus: fibrous tissue and cartilage (green) and bone (yellow) (Adapted from [44] with permission)
Remodeling occurs not only in the final phase of healing, but continuously during the lifetime of organisms, during which bone material is constantly renewed and microdamage is removed [20]. During remodeling, specialized bone cells called osteoclasts and osteoblasts resorb and deposit small packets of bone material. In the spongy type of bone called trabecular bone, remodeling events occur at the surface. Inside the less porous cortical bone, the surface for remodeling has first to be created, which occurs when osteoclasts dig a tunnel through cortical bone with osteoblasts in their wake to close the tunnel. This results in the formation of a cylindrical structure of new bone, termed an osteon. Historically, the process of remodeling by replacing old damaged bone by new bone (bone material maintenance) is distinguished from modeling in which the uncoupled formation and resorption of bone allows an adaptive structural response of bone to an altered mechanical environment. Here, we use the term “remodeling” to refer to both processes [49].
To allow for such structural adaptation, it has been argued that bone remodeling must be a mechanically regulated process. Computer algorithms, with the implemented remodeling rule stating that bone is formed preferentially at sites of high mechanical load and resorbed where mechanical stimulation is low, could successfully describe the adaptive response of trabecular bone to changes in loading [50, 51]. A new experimental view on bone remodeling is offered by the possibility of obtaining multiple three-dimensional microcomputed tomography (μCT) images from the same bone, at least in small animals. Comparison of two such images taken with a time interval of about 1 week allows one to determine each remodeling event that occurs during this interval. In combination with mechanical assessment performed by finite element analysis (FEA), sites of remodeling can be correlated with the local mechanical loading, providing a quantitative description of the mechano-regulation [52] and its change with age [53]. The nature of the mechanical stimulus that triggers remodeling and the manner in which the stimulus is sensed by bone is still an open question. However, it is generally accepted that osteocytes, cells that are organized into a network-like architecture within the mineralized bone matrix, are involved in mechano-sensation [54]. One hypothesis that is gaining traction is that remodeling is stimulated when microcracks in bone result in a local interruption in the osteocyte network connecting two cells, resulting in cell death [10]. In bone, remodeling is a “housekeeping” process that protects the material against fatigue failure by using occurring microdamage as a signal for initiating the repair mechanism. From a materials science perspective, remodeling is a highly attractive mechanism that can be used in designing a material that not only responds to potentially catastrophic damage events such as fractures, but that also preemptively addresses minor damage events before they can escalate into larger issues. Nonetheless, the frequent occurrence of stress fractures in the bones of military recruits reveals one potential drawback of a remodeling response that is too active. For example, if the microdamage caused by an intense training regime results in increased bone resorption to remove the damage [55], this can lead to further mechanical weakening of the bone, making stress fractures even more likely. This highlights the delicate balance that must be achieved between material breakdown and buildup.
At the molecular level, repair mechanisms contribute to the plastic behavior of bone and its characteristic high toughness. Similarly to antler, bone also utilizes deformation by sliding of structural elements against each other to increase material toughness. A staggered arrangement between the stiff mineralized fibrils embedded in a softer, but ductile, matrix results in tensile loading of the fibrils, while the extrafibrillar matrix is loaded under shear [20]. The matrix was previously described as a glue layer, in which a continuous breaking and re-formation of molecular bonds (so-called sacrificial bonds) [56] enables fibrils to slide against each other while dissipating energy. Mechanical tests with different strain rates and temperatures provided clues that sacrificial bonds in bone consist of calcium-mediated ionic bonds [57]. The concept of reversible sacrificial bonds is discussed extensively in the case studies of self-healing in extracorporeal biopolymers (Sect. 2.9).
2.4 Cutaneous Wound Healing
For most people, the healing of wounds to our skin (e.g., cuts and scrapes) is probably the most familiar biological repair mechanism. Even for a small cut, however, the resulting cascade of events is tremendously complex and warrants a whole review on its own [58, 59]. Consequently, we choose here to limit our focus to the classical description of the phases of cutaneous wound healing, with an additional focus on scar formation [60]. Many aspects of cutaneous wound healing are relevant to the design of self-healing materials, especially the ability to rapidly transport essential healing agents via a complex network of vasculature to the damage site in order to quickly seal the wound.
Analogous to bone healing, cutaneous wound healing in mammals has been divided into three distinct, but overlapping, phases (Fig. 4) [59, 61]: inflammation, new tissue formation, and remodeling. As with many of the other case studies presented, the first step in the repair process is to seal the wound with a temporary covering to staunch loss of blood and fluid. This is accomplished by formation of a clot consisting of aggregates of platelets embedded in a loose fibrin matrix. The clot serves as a scaffold for infiltrating cells and as a reservoir for signaling molecules. In parallel, diverse inflammatory leucocytes are recruited to the site of tissue injury to prevent infection by ridding the site of bacteria and removing dead tissue. At the onset of the second phase, the barrier function of the epithelium is re-established. Keratinocytes migrate from the wound edge over the injured dermis separating the viable tissue from the desiccated eschar, which contains necrotic tissue as well as dried blood. As in bone healing, the formation of new small blood vessels (angiogenesis) establishes reliable blood supply to the injured tissue and transforms the fibrin matrix into granulation tissue, which consists of a dense population of cells in a mesh of collagen, fibronectin, and hyaluronic acid. At this point, some fibroblasts differentiate into myofibroblasts, which use their contractile abilities to compact the surrounding tissue and contract the wound. Extracellular matrix (ECM), mainly in the form of collagen, is generated by fibroblasts and myofibroblasts. During the final remodeling phase, most of the cells activated after the injury undergo controlled cell death (apoptosis) or leave the wound. Over a time period of roughly a year the remaining acellular collagen matrix is modestly remodeled, with a resulting scar that predominantly consists of type I collagen. The attained mechanical properties of the scar tissue are inferior to uninjured skin, with an ultimate tensile strength of only 70% of its original value [59, 61]. Although not as grave in its consequences as the life-threatening myocardial scar formed after a heart attack [61], excessive scarring of the skin, particularly after burns, is a major clinical problem [62].
Three phases of wound healing of skin: (a) inflammation, (b) new tissue formation, and (c) remodeling. Healing in skin with its two main layers, the upper epidermis and the lower dermis, is illustrated (for a detailed description of the process see text). Note that the region of the newly formed scar is free of the normal skin appendages (e.g., hairs, sweat glands) (From [61] with permission)
Comparison between regenerative scar-free healing (as in the initial phase of limb regeneration) and non-regenerative healing with a fibrotic healing outcome reveals several important differences between the two processes that may open new possibilities for improving wound healing [60]. First, closure of the wound by a new epithelium is much more rapid in regenerative healing. For example, in axolotl (a type of salamander), wound closure occurs in less than half a day, whereas this usually requires a few days during cutaneous wound healing in mammals. Second, the composition and mechanical properties of the extracellular matrix differ for regenerative and non-regenerative healing. Myofibroblasts are also largely absent in regenerative healing and, thus, much less collagen is produced. There is general agreement that an attenuated immune response promotes scar-free healing [61, 63]. Two observations fuel hope for improving the fibrotic result of tissue repair: (1) during early gestation most mammalian fetuses (including humans) show scarless healing outcomes [64], and (2) a mouse strain was serendipitously discovered that is able to fully regenerate ear punches as large as 2 mm [65].
2.5 Biochemical Self-Repair: Latex-Bearing Plants
We now shift focus from animal systems to plant systems for the next four case studies. Although specific details of the healing mechanisms naturally differ between these two biological kingdoms, many similarities are obvious [66]. Plants differ from many (higher) animals by their modular construction, that is, they are composed of many (self-similar) modules. This modular construction is one reason why plants can survive severe damage caused by storms, but also by pruning or mowing. In plants, self-repair can be found at all modular and hierarchical levels from the molecular scale to the entire organ, including cellular and acelluar self-repair mechanisms. In the following four case studies, we highlight specific examples of plant-based self-repair relevant to the development of self-healing materials.
Latex is found in more than 20,000 plant species coming from over 40 different plant families [67, 68]. This indicates that latex has evolved many times independently in different lineages of higher plants. One can therefore hypothesize that not only the physiological pathways of latex formation vary within these lineages, but also that the function and coagulation mechanisms differ widely regarding the biochemical structure of the latex and main selective pressures acting on latex formation. In plants, latex is typically found in cellular microtubes (laticifers), where the characteristically white to yellow milky exudate is often stored under high internal pressure (up to 15 bar). Latex contains, among other substances, phenolics, alkaloids, proteases, chitinases, and other proteins. In ca. 300 plant genera, rubber particles (cis-1.4-polyisoprene) are also found, often in high concentrations. As a result of the complex biochemistry, many functions for the sticky and often poisonous latex have been suggested and studied (e.g., as defense against herbivores or germs or as a transport system or reservoir for water, nutrients, and/or waste substances) [69, 70]. Surprisingly, the potential function of latex as a self-repairing substance was hardly studied in living plants [14]. This contrasts with the intensive studies of the chemistry and coagulation mechanisms of natural rubber, harvested mainly from the Pará rubber tree (Hevea brasiliensis), which were performed for more than 150 years, mostly for commercial reasons. Comparative analyses of latex-mediated self-repair have primarily focused on the genera Ficus (Moraceae, fig family), Euphorbia (Euphobiaceae, spurge family), and Campanula (Campanulaceae, bellflower family) [15, 71] (Fig. 5).
Latex coagulation after injuring the bark of a weeping fig tree (Ficus benjamina). The fresh latex droplet directly after injury is white as a result of total light reflection by the latex emulsion. The droplet becomes more and more transparent with increasing coagulation, which mirrors the chemical alterations in the latex. From [72]
Rubber-based self-sealing is easily observed after injuring an organ of a latex-bearing plant (Fig. 5). Directly after injury, a white drop of fluid latex leaks out, typically becoming solid and translucent and sealing the wound after a time span of 20–30 min in tested Ficus and Hevea species, and after ca. 60 min in Euphorbia species. In contrast, the two bellflower species tested (Campanula glomerata and Campanula latifolia) exhibit coagulation in just a few seconds [14, 15, 72, 73]. The latex coagulation mechanism of the Pará rubber tree (H. brasiliensis) is one of the best understood from a chemical perspective. The latex of this species contains, among other substances, rubber particles and vacuolar structures (lutoids) that contain the protein hevein. The lutoids burst following injury as a result of the sudden pressure drop from over 7 bar in the intact laticifers to ambient pressure of 1 bar. This causes the release of hevein monomers, which form dimers in the presence of Ca2+ ions. Binding sites for hevein on the surface of the rubber particles induce latex coagulation, causing autonomous self-sealing via hevein-mediated crosslinking of the rubber particles [74–76]. Recent studies suggest that the latex coagulation mechanism in the weeping fig (Ficus benjamina) is very similar to that described for H. brasiliensis and is also based on protein binding between latex particles after pressure-drop-driven bursting of lutoids [15, 72]. In addition to the very efficient self-sealing of injuries, a self-healing effect of the coagulated latex has also been observed in the weeping fig. In tension experiments, it was observed that, directly after injury, the tension strength of cortex strips is reduced to 42% of that of the uninjured control sample and remains low for the next 20–25 min. After ca. 30 min (i.e., after coagulation), a significant increase in tension strength to 55% of the uninjured sample occurs. This increase is not found if the latex is removed directly after injury and confirms the self-healing function of latex. After this (mainly) physicochemical phase of self-repair, the tensile strength does not change for several hours or days until, as a result of cell proliferation and growth processes, the biological phase of self-healing begins and the tensile strength further increases [15, 17] (Fig. 6).
Self-healing efficiency as percentage recovery of tensile strength in bark strips of the weeping fig (Ficus benjamina) at different time intervals after injury. White bars indicate barks samples in which the latex was removed directly after injury and therefore cannot contribute to self-healing. Copyright Plant Biomechanics Group Freiburg
Related studies have demonstrated that alternative mechanisms of latex coagulation and wound sealing exist in other latex-bearing plants [71, 73]. In addition to the weeping fig and Pará rubber tree, three spurge species (Euphorbia amygdaloides, Euphorbia characias, Euphorbia myrsinites) and two bellflower species (C. glomerata, C. latifolia) were studied and characterized concerning the time span between latex discharge and coagulation, size distribution of latex particles, and the rheology and wettability of latices. In addition to drastic differences in coagulation time, the latex of the two bellflowers coagulated much faster than that of the other species (see above). In addition, significant differences in the other parameters were found. In tested Euphorbia species, the latex particles were more densely packed and the size distribution (only one type of small latex particles) markedly differed from the bimodal particle distribution (as a result of latex particles and vacuolar structures) occurring in F. benjamina and H. brasiliensis. Additionally, the rheological behavior during latex coagulation and wettability varied between the species. These findings suggest that the coagulation mechanism in the tested Euphorbia species differs from the biochemically initiated mechanism found in Ficus and Hevea and is mainly caused by physical changes (i.e., simply by water evaporation). In addition to the physicochemically interesting differences, these findings suggest different evolutionary scenarios in the tested species concerning the main function of the latex. As a result of the prolonged liquid phase in Euphorbia spp., the anti-herbivory compounds remain dissolved longer and the main function is probably defense against herbivores, whereas the (much) faster coagulation found in F. benjamina, H. brasiliensis, and the two Campanula species suggests that the main function of latex is self-sealing and self-healing of wounds [14, 15, 71, 73].
The wide variety of physical and chemical properties found in plant latices and the differences in coagulation mechanisms make plant latices very promising role models for the development of bio-inspired self-repairing materials. Different ideas for bio-inspired self-repair have been tested and include three basic approaches: (1) The first approach was inspired by the role of Ca2+ ions during latex coagulation in Hevea and was implemented by the development of technical ionomeric elastomers. The best results were found for carboxylated nitrile butadiene rubber (NBR). After rejoining rectangular strips (cut in half and then annealed for 24 h at 55°C), restoration of tensile strength to 50% of the undamaged strips was found for unvulcanized material, whereas in vulcanized strips the restoration dropped to 15% [14, 15, 77]. (2) The second approach was inspired by the bursting lutoid vesicles found in the latex of Ficus and Hevea and aimed for the development of technical elastomers equipped with microcapsules filled with healing agents. This approach has not been successful because of problems with the stability of the microcapsules [14, 77–79]. (3) An advancement of the idea of microcapsules was successful, in which microphase separation of the healing agent in the elastomer was used. This approach avoids a microcapsule wall by “chemically mimicking” micro-encapsulation. The concept led to the development of bio-inspired multiphase NBR blends with liquid polymers as self-sealing and self-healing agent. High internal pressure could be built up in their microphase-separated domains as a result of the different thermal expansions of NBR and polymer. Microcracks reaching such a domain are sealed and (at least partly) healed by the self-healing agent filling the microfissure. Hyperbranched polyethyleneimine (PEI) proved to be the most suitable self-healing agent for NBR and showed the best self-repairing effect. After re-joining rectangular NBR/PEI strips (cut in half, annealed in a heated sample holder for 12 h at 100°C, and then stored for another 12 h at room temperature), restoration of tensile strength to 44% of the values for undamaged strips was found [7, 14].
2.6 Cellular Self-Repair: Aristolochia macrophylla
In many climbing lianas (woody vines), the young axes are stiff in bending and torsion and act as searching twigs, exploring the space for new supporting structures on other branches of the current supporting tree or on new supporting trees up to several meters away. These searching twigs are anatomically characterized by a dense and stiff central wood cylinder and/or peripheral stabilizing tissue layers consisting of thick-walled collenchyma and/or sclerenchyma fibers. After being attached onto the new support, the liana stems develop a cylinder of secondary wood with large vessels and huge wood rays. This wood type is very flexible in bending and torsion. The resulting increased flexibility in bending and torsion is advantageous because it enables attached liana stems to passively follow wind-induced movements of the supporting tree or deformations caused by climbing animals as a result of elastic deformation. It may even enable the plant to survive collapse of a supporting branch or the entire tree [80–82].
In species with closed outer cylinders of stabilizing tissues, as in twining pipe-vine species (Aristolochia spp.), these internal growth processes cause increasing radial and tangential stresses and strains in the stiff peripheral cylinders comprising collenchyma and/or sclerenchyma. As a result, the highly lignified and stiff peripheral sclerenchyma cylinders often fail and (micro)fissures occur. Although these fissures further increase the bending and torsional flexibility of the liana stems, fissures running outward to the stem are perfect entrance gates for fungus spores and bacteria that can infect the liana stem. Thus, high selective pressure for the evolution of efficient self-repair mechanisms that close the fissures quickly can be hypothesized (Fig. 7). Studies of different Aristolochia species showed that, following internal microfissure formation, parenchymatous cells of the outer cortex tissue swell into this fissure and seal it as a result of their internal cell pressure (turgor) of, typically, 2–3 bar. This is caused by relaxation of pressurized tissue into a newly formed opening and represents the fast self-sealing phase of wound repair. This is based (mainly) on physicochemical processes and involves neither cell wall biosynthesis nor cell division. In the further course of ontogeny, the vascular cambium produces more secondary phloem and especially secondary xylem, which causes a further increase in diameter of the central vascular tissues and provokes enlargement of the fissures in the peripheral stiffening rings. The repair cells react to this with cell divisions in tangential and radial directions (metabolic processes including significant cell wall biosynthesis) (Fig. 8). This stage of self-repair in Aristolochia spp. can be interpreted as the starting point of the prolonged phase of self-healing. During this self-healing phase, repair cells can even develop thickened and lignified cell walls, which (partly) restore the mechanical properties of the peripheral stiffening tissues [15, 83, 84].
Self-repair in stems of the pipe-vine (Aristolochia macrophylla). (a) Cross-section of a 1-year-old stem with closed peripheral cylinder of stiffening sclerenchymatous tissue. (b) Cross-section of a 10-year-old stem with repaired fissures in the peripheral cylinder of stiffening sclerenchymatous tissue. Copyright Plant Biomechanics Group Freiburg
Different phases of self-repair in (a–e) Aristolochia macrophylla and (f–j) Aristolochia ringens. (a, f) Initial self-sealing phase; a fissure in the sclerenchymatous cylinder (stained in blue) is sealed from a parenchyma cell. In subsequent phases, the fissure increases in size as a result of increasing formation of central vascular tissues. The fissure increase is compensated by cell division of thin-walled, irregularly shaped parenchymatous cells, which first occurs in the radial direction (b, g) and later – in increasingly broad lesions – also in the tangential direction (c, h), which represents the beginning of the self-healing phase of self-repair. In even later phases of repair the cell walls of the most peripheral repairing parenchyma cells become thickened and lignified (d, e, i, j). This allows, at least partly, restoration of the mechanical properties of the sclerenchymatous cylinder. From [83] with permission
Detailed and quantitative descriptions of the fast self-sealing processes for stems of the winding lianas Aristolochia macrophylla and Aristolochia ringens were used as concept generators for the production of a novel self-sealing foam. Pneumatic structures (e.g., rubber boats, tires, and airbeds) are especially “vulnerable” technical products that typically lose their functionality entirely following air loss as a result of puncturing. Joint efforts between academia and industry have resulted in the development and patenting of a self-sealing foam coating for membranes of pneumatic structures [6, 14]. Especially good results were achieved with the foam Raku-PUR 33-1024-3 (Rampf Polymer Solutions, Germany). Tensairity® technology has been utilized for applications such as ultralight bridges and roof construction. These structures consist of a combination of compression struts and tension cables, with a central stiffening pneumatic cylinder equipped with a self-repair function [85, 86]. The self-sealing phase is especially well suited for biomimetic transfer because (mainly) physicochemical processes are involved. The best results were found for closed-pore polyurethane foams cured under an overpressure of 1–2 bar, causing a pre-strained repairing layer on the inside of the membrane. The layer is only a few millimeters thick and does not add much to the weight of the membrane. As a result of pre-straining and the compression strains evoked in the foam layer on the inside of the membrane by the toroidal form typical for all pneumatic structures, the foam layer relaxes after puncturing and seals the fissure very efficiently. By this process, air leakage through a hole in the membrane can be entirely stopped or at least markedly reduced. In the case of holes of up to 5 mm diameter in foam-coated fiber-reinforced PVC membranes (as used for Tensairity® structures) the air leakage could be reduced in all tests by a factor of at least 1,000. In over 80% of the tested samples the punctured membranes were entirely sealed [6, 14, 87, 88].
2.7 Self-Sealing by Deformation
Plants growing under extreme ecological conditions are under especially high selective pressure regarding the development of effective self-repair mechanisms. Over the course of evolution, succulent plants, which evolved independently in various systematic groups, have developed the ability to store water in their tissues as an adaptation for survival in arid environments. After external damage, which can lead to exceptional drought stress, rapid self-sealing protects the plant from dehydration and is therefore of dominant selective advantage [14, 15, 18]. Highly effective self-sealing mechanisms were found in the succulent leaves of Delosperma cooperi belonging to the Aizoaceae family. The species is native to South Africa, where it grows in semi-arid regions with cold winters. This perennial forms dense plant stocks with striking pink flowers, responsible for the common name Pink Carpet [14, 15, 18, 89]. In cross-section, the almost cylindrical leaves reveal a centripetal arrangement of five tissue types consisting of an outer layer of epidermis with window cells, a peripheral ring of chlorenchyma, a thin net comprising vascular bundles, an inner ring of parenchyma, and a strand of vascular bundles in the leaf center (Fig. 9) [14, 15, 18, 89].
After an artificial incision, wound sealing in leaves of D. cooperi takes place by deformation and leaf movement. Two mechanisms are involved, the effectiveness of which are dependent on the air humidity: (1) rolling in of the wound edges within a few minutes, and (2) bending or contraction of the entire leaf within approximately 1 h. Rolling in of the fringes probably results from the different shrinkage properties of the epidermis with its huge window cells and cuticula. Occasionally, the wound edges form a hook-like structure that helps to seal the wound cavity and prevent dehydration. This feature can be found in the leaves and stems of D. cooperi [14, 15, 18, 89]. Depending on the wound type, the entire leaf can deform in such a way that the incision becomes closed within a maximum of 60 min. Three types of external injuries along the leaf were studied. Lateral cuts in both transversal and longitudinal directions lead to bending of the entire leaf until the wound edges are close enough together to seal the wound. Leaves with a circular cut do not bend; rather, they contract until the wound margins close the incision [14, 15, 18, 89]. Subsequent wound healing leads to wound tissue that appears white to the naked eye, and occasionally results in permanent curvature or circular necking of the leaf.
For better understanding, the mechanical properties of the entire leaf and dissected single tissue layers were studied. Tensile tests were carried out with a microtensile tester by fixing either the entire leaf or single tissues, such as the central vascular strand or the epidermis, on two sample holders. The force-displacement diagram of an entire leaf showed a linear elastic portion of the curve (Fig. 10a, b). At maximum tensile strength the leaf seems to be fully intact (Fig. 10c). Failure occurred stepwise, showing several pre-failure events, whereby crack propagation was observed after the first visible crack (Fig. 10e) on the surface of the leaf.
Force-displacement diagram as a result of a tensile test with an entire leaf of Delosperma cooperi: (a, b) linear-elastic range, (c) maximum tensile strength, (c–g) stepwise failure with crack propagation after the first visible crack (e) on the surface of the leaf, and (h) total failure. Copyright Plant Biomechanics Group Freiburg
On the basis of geometric data obtained from anatomic studies, combined with mechanical properties found as a result of tensile tests and pressure probe measurements, an analytical model for intact leaves and wounded leaves with regard to elastic and viscoelastic behavior was developed. The plant model was defined after considering the elastic modulus, the radius, and the Poisson’s number of all five tissue layers [89, 90]. The equations are capable of describing the self-sealing process of D. cooperi after longitudinal, transversal, and circular damage of the leaves in terms of elastic and viscoelastic behavior. The analytical model provides a universal language in order to better understand the biological template and will help in developing a range of technical applications (e.g., multilayer materials with self-sealing function) [89].
2.8 Grafting in Plants
The seamless fusion of plant individuals or organs, known as grafting, provides another pertinent example for research on self-healing materials. Numerous descriptions of “natural grafts,” the incidental fusion of plant parts, can be found in the literature [91–95]. Botanists have been understandably fascinated by this behavior for centuries, because a successful, compatible graft can lead to tissue fusion, vascular continuity, and a single physiologically and mechanically functioning unit [96]. Grafting is possible within an individual (autograft), between individuals of the same species (homograft), or even between different species (heterograft) [96, 97]. Natural grafting only rarely occurs above ground, whereas root fusion among forest trees seems to be a common phenomenon, probably facilitated by the dense network of roots and the surrounding soil matrix [96, 98–100]. The resulting network between roots of one individual or several individuals raises questions about the individuality of a single tree, as well as interplant relationships and communication within a whole forest [98] and, thus, about the evolutionary and ecological relevance of natural grafting.
Natural grafting (Fig. 11a) probably inspired the application of grafting in agriculture and horticulture thousands of years ago, which is currently applied for vegetative propagation and the creation of unusual growth forms in ornamental plants [100]. Several studies have shown that the tensile strength of successful autografts reaches and sometimes even exceeds the tensile strength measured for intact control plants [97, 101–105]. The connection of different tissues into one functionally united structure has clear implications for future and advanced biomimetic materials research, especially when focusing on adaptive and self-healing materials. Thus, understanding the detailed mechanisms underlying a successful graft is of utmost importance.
Grafting behavior and mechanism in plants. (a) Strangler fig aerial roots fusing to a network around the host. (b) Diagram showing the position of meristematic tissues phellogen (p) and vascular cambium (v) in the plant stem’s transverse section; m mark, c cortex. (c) Steps 1–5 toward the fusion of plant parts; white mainly parenchyma, orange vascular cambium and derived cells, brown phellogen and derived cells
Grafting always requires tight contact between the partners, as well as pressure and/or injury at the points of contact [92–96, 100, 106]. Lesion formation in the closely contacting organs of both grafting partners activates wound healing processes on both sides [106, 107] by stimulating meristematic tissues (e.g., phellogen and vascular cambium) located very close beneath the stem or root surface (Fig. 11b). The vascular cambium consists of cell initials, which produce daughter cells mainly by tangential division. The descendants differentiate into the secondary conductive tissues phloem and xylem, as well as into fibers and parenchyma, taking over water and nutrient conductivity, storage, and mechanical stability [108]. The vascular cambium is a dynamic tissue and the daughter cells vary in form, function, and rate of production, which are dependent on the age of the plant or season of the year [109]. The phellogen is a secondary meristem that develops de novo in the peripheral region of the stem. Living cells convert to meristematic active cells and produce the periderm, a secondary protective tissue, also by tangential division [108, 109]. Both meristems are sensitive to internal and external signals and stimuli such as wounding or mechanical influences. Stimulation of meristematic tissues and initiation of wound-healing processes are always accompanied by the formation of a callus (i.e., a mass of uninjured, rapidly dividing parenchyma cells) [96]. However, instead of sealing each wound individually, the activated meristematic tissues of both partners fuse to form a joint callus and coordinated healing of the wounds of each grafting partner [94].
The sequence of structural events during the grafting process (Fig. 11c) has been defined by several studies [96, 106, 110–112]. If both partners are in close contact after physical injury, deposits and residues of killed cells form a material compound between the freshly exposed surfaces, consisting of cellulosic cell wall remnants, resins, and other cell content (Fig. 11c, step 2). Adjacent parenchyma cells enlarge and extend into the interface and subsequently start to divide (i.e., callus formation), filling up the spaces between partners (Fig. 11c, step3). Formation of the callus facilitates temporary by-pass of the damaged vascular tissues. Callus cells then differentiate into wound-repair xylem and phloem, followed by formation of a new vascular cambium layer (Fig. 11c, step 4), leading to a continuous cambial connection between the two grafting partners [96, 111, 113] (Fig. 11c, step 5). Finally, the vascular connectivity between both grafting partners is established by the regular production of conductive tissue [96, 106, 113]. The early steps of grafting are supported by adhesion and cohesion between the adjacent cells, resulting from the secretion of insoluble carbohydrates and pectin polysaccharides [113]. The interactions between grafting partners during formation of a callus and the vascular connectivity are regulated and controlled by hormones such as auxin, which are released by both grafting partners and stimulate vascular tissue differentiation [107, 114–116]. Recent studies suggest that small RNA or DNA fragments and even complete chloroplast and nuclear genomes can be exchanged between the grafting partners, directing DNA methylation and allowing horizontal gene or even genome transfer between sexually incompatible species [117–120].
The fascinating subject of uniting different plant individuals or parts includes events such as cell elongation and callus formation, which are also observed elsewhere [111]. These are common plant responses and are related to a naturally much more important process, the healing of wounds. Furthermore, parallels are also seen in the response of plants to infection, such as cancer-like tissue formation in club root disease. The fusion of natural tissues and structures is fascinating and might serve as stimulation for the development of artificial adaptive and self-healing materials. In particular, the processes of sealing, gap filling, and subsequent formation of new tissue, although difficult to mimic, may open new routes for materials synthesis and functionality.
2.9 Healing in Acellular Biopolymers: Reversible Bonds
Extracorporeal biological materials such as spider and insect silks are synthesized from biomolecular building blocks (e.g., proteins) and function outside the confines of the body of the organism. Such materials are acellular and, thus, their material properties must arise from the intrinsic chemical and physical features of the building blocks themselves. For this reason, extracorporeal materials provide ideal biological archetypes for inspiration of polymeric materials. Although self-healing is typically regarded as a cell-driven process, there are several examples in nature of self-healing extracorporeal biopolymers (e.g., mussel byssus, caddisfly silks, and egg capsules from marine snails), which achieve these behaviors through the use of reversible bonds.
2.9.1 Mussel Byssal Threads
Marine mussels of the species Mytilus anchor securely to wave-battered surfaces in rocky seashore habitats using protein-based holdfast fibers known as mussel byssal threads (MBTs) (Fig. 12a) [121]. The distal region of MBTs is initially stiff (500–800 MPa), but yields at a critical stress, dissipating up to 70% of applied energy in the process (Fig. 12b) [12, 122]. Yield, however, results in apparent damage in subsequent loading cycles, with stiffness and strain energy reduced by as much at 65% (Fig. 12b) [12, 122]. Initial properties are largely recovered when threads are rested for several hours in water, indicating an intrinsic and autonomic self-healing of material damage [122].
Self-healing in mussel byssal threads. (a) Mussels anchor on hard surfaces with the byssus. (b) The distal region of byssal threads experiences damage following cyclic loading beyond the yield point (red curve); however, recovery toward native behavior is possible over time (blue curve). (c) Distal byssal threads are hierarchically structured semicrystalline fibers composed primarily of preCol protein. PreCols are collagenous protein with N- and C-terminal domains rich in histidine residues, which are believed to coordinate divalent transition metal ions. (d) The hard and extensible outer cuticle of byssal threads is composed of hard micron-sized granules that are centers of high DOPA–metal crosslink density surrounded by low crosslink dense matrix material
Structurally, the thread core is composed principally of a protein family known as the preCols [123], which are elongated block copolymer-like proteins with several distinctive structural domains, including a central collagen domain, flanking domains surrounding the collagen domain, and terminal histidine-rich domains (HRD) at both ends (Fig. 12c). The collagen domain forms a rigid triple helical trimer [12, 124], whereas the flanking domains of some preCol variants form a compact β-sheet structure [125, 126]. The N- and C-terminal HRDs contain at least 20 mol% of the amino acid histidine, which forms metal coordination bonds with Zn2+ and Cu2+ ions in threads [127, 128].
Recent X-ray diffraction (XRD) studies reveal that preCols are organized serially into a nano-architectured semicrystalline framework (Fig. 12c), which is highly deformable and recovers elastically immediately when relaxed through reversible unfolding of the flanking domains and HRDs [12, 124]. Slower healing of mechanical properties thus indicates a second healing stage, involving recovery of reversible bonds [124]. Histidine–metal coordination crosslinks localized in the HRDs are thought to function as load-bearing reversible bonds that provide high strength, but also high lability [123, 129]. The reduction of byssal thread stiffness and loss of healing behavior in response to low pH and metal chelation treatments aimed at disrupting metal coordination provides important support for this model [12, 129, 130]. Indeed, recent evidence from X-ray absorption spectroscopic (XAS) studies confirms the presence of mechanically active protein–Zn2+ crosslinks in the distal thread core, which are disrupted during the post-yield stress plateau and reorganize toward the native state during thread healing [128]. The ability of histidine–metal coordination bonds to behave as strong and reversible crosslinks was also demonstrated in vitro with micromechanical adhesion studies using HRD-mimicking peptides attached to soft-colloidal probes [127].
In addition to the protein–metal complexes in the thread core, labile crosslinks between 3,4-dihydroxyphenylalanine (DOPA) amino acid residues of mussel foot protein-1 (mfp-1) and metal ions such as Fe, V, and Al have been detected in the hard and extensible outer cuticle of MBTs (Fig. 12d) [131–133]. In this case, the coordination crosslinks are organized into submicron-sized crosslink dense regions (granules) with stiff mechanical behavior and lower crosslink density regions (matrix) that are less stiff (Fig. 12d) [134]. Microcracking in the matrix caused by rupture of DOPA–metal bonds is thought to provide extensibility in this stiff and hard biopolymer, which could facilitate resealing and healing of cracks as a result of the reversible nature of the bonds [131, 134]. Reversibility was demonstrated by metal chelation experiments in which cuticle metals could be removed with EDTA (lowering the stiffness/hardness by more than 85%) and later reintroduced by soaking in metal chloride solutions, leading to complete mechanical recovery [132]. Notably, Fe, V, and Al could all be reintegrated into the mfp-1 DOPA crosslink network without causing a pronounced difference in mechanical performance compared with the native state, underlining the inherent adaptability and versatility of this reversible crosslink network [132].
2.9.2 Caddisfly Silks
Larvae of certain caddisfly species live in aqueous environments and sheath themselves in protective composite cases made of pebbles and stones that are glued together by adhesive silk fibers [135] (Fig. 13a). The silk fibers were recently discovered to exhibit tensile mechanical behaviors similar to those of byssal threads, with an initially stiff behavior, a yield event at low strain followed by a stress plateau, and a large hysteresis in cyclic loading (Fig. 13b) [11, 135]. Native material properties are similarly reduced significantly during subsequent loading, but are recovered over time to near native values [11].
Self-healing in caddisfly silk. (a) Caddisfly larvae build composite cases from small stones and an adhesive silk. (b) Silks undergo damage upon yielding, but can recover initial properties within ~2 h. (c) Caddisfly silk has a complex hierarchical structure in which protein β-crystallites are embedded in an amorphous protein matrix. The β-sheets are stabilized by interactions between phosphorylated serine residues and Ca2+ ions. Adapted with permission from [135]. Copyright (2013) American Chemical Society
Caddisfly silk is composed primarily of H-fibroin, a large protein with tandemly repeated domains [136], but also contains PEVK-like proteins that may function in material elasticity [137]. Serine residues in prevalent H-fibroin [S-X]n repeat domains are post-translationally modified to phosphoserine, a highly acidic amino acid side chain. These domains are thought to form β-crystalline nanodomains that are stabilized by inter- and intramolecular interactions between phosphate moieties and Ca/Mg ions (Fig. 13c) [11, 135, 138, 139]. Removal of ions with EDTA treatment revealed a large reduction in β-sheet structure and a marked loss of mechanical performance – an effect that was completely reversed by reintroducing calcium ions [135]. Based on these findings, a molecular model was proposed involving a multinetwork system with two different ionomeric clusters, which, like the proposed metal coordination sacrificial bonding network in the byssus, is able to reversibly rupture and re-form, leading to self-healing behavior [11].
2.9.3 Whelk Egg Capsules
Whelks (Busycotypus canaliculatus) are marine snails (gastropods) that lay their eggs encapsulated within a tough and compliant leathery egg case consisting of individual disk-shaped capsules linked into chains of up to meter in length(Fig. 14a) [140]. Whelk egg capsule (WEC) material is a proteinaceous biopolymer that, like mussel byssus and caddisfly silk, functions to dissipate energy in aqueous environments. The WEC biopolymer is less stiff than MBTs or caddisfly silk, but similarly undergoes yield at low strain and exhibits a large hysteresis (~50%) during cyclic tensile loading (Fig. 14b) [13, 140]. However, unlike MBTs and caddisfly silk, WEC biopolymer recovers initial material properties almost instantaneously [13, 140] (Fig. 14b), which is behavior reminiscent of pseudoelastic/shape memory polymers and alloys.
Whelk egg capsule. (a) Whelks are marine gastropods that lay eggs in elaborate egg cases. (b) Whelk egg case (WEC) material exhibits yield and hysteresis behaviors similar to those of MBTs and caddisfly silk, but recovers initial behavior immediately through multiple cycles. (c) Shape memory behavior of WEC is attributed to reversible unfolding and refolding of α-helical protein structure into a β-sheet-like structure
XRD and vibrational spectroscopy studies clearly identify a dominant α-helical protein structure in the egg capsules aligned in orthogonal layers parallel to the capsule surface [13, 141]. Three α-helical proteins, named capsule protein (CP)-1, CP-2, and CP-3, are thought to be the primary load-bearing elements [142]. Beyond the yield point, the α-helical proteins oriented in the stretching direction undergo reversible conversion to a β-sheet-like structure, in which the hydrogen bond network stabilizing the α-helical structure is disrupted (Fig. 14c) [13, 141]. The conversion from extended β-sheet-like structure back to compact α-helical structure is associated with a notable hysteresis at the molecular level, probably arising from re-formation of the reversible hydrogen bond network along the helical axis [141, 143]. The interconversion between the two conformational protein structures was modeled successfully as a classical phase transformation, comparable to the martensitic transformation observed in shape memory metal alloys (e.g., NiTi) [141, 143].
2.9.4 Extracted Concepts and Biomimetic Materials
From these three examples of biopolymeric self-healing, several common design principles can be extracted for design of self-healing polymeric materials: (1) The use of reversible sacrificial bonds combined with folded protein lengths leads to material yield and energy dissipation, but also to molecular level damage in the crosslinking network. Time-dependent recovery toward the native (healed) network appears to depend on transient and labile bond dissociation kinetics. The reversible bonds used in nature range from hydrogen bonds to ionic interactions and metal coordination bonds, which apparently tune material performance (e.g., stiffness and yield stress). (2) Elastic recoil of the unfolded (damaged) structure is necessary to bring ruptured reversible bonds back into register so they can re-form, somewhat analogous to the self-sealing phase observed in cellular materials. (3) Hierarchical structure at numerous length scales (e.g., protein secondary structure, higher order assemblies) localizes damage, and thus healing, to specific regions of the protein (polymer) network, probably facilitating reversible bond recovery and healing. This last point is perhaps the least well understood aspect of biological healing via reversible bonding and is being actively pursued for these materials.
Efforts to mimic these self-healing biopolymers by introducing reversible bonds into polymer networks is an active area of research that is currently based on both recombinant biopolymers [144, 145] and traditional polymers presenting metal-binding amino acid side chain moieties [4, 146–149]. In particular, a number of mussel-inspired polymers based on DOPA– and histidine–metal binding have been developed that exhibit self-healing performance as well as other industrially relevant properties such as wet adhesion and actuation [4, 146–150]. Additionally, polymeric hydrogels inspired by the phosphate-based chemistry observed in caddisfly silk have recently been produced, exhibiting similar self-recovery behavior to the natural material [151]. Efforts to mimic these materials via recombinant technology have also shown some success. For example, the rubbery insect protein resilin was recently bio-engineered to contain histidine–metal binding groups that enabled mechanical tunability with metal addition [144]. Proteins from the WEC have also been recombinantly expressed and found to self-organize into coiled-coil α-helical fibrils [145]. Although these early findings are encouraging, these materials are largely still lacking the structural and mechanical complexity of their natural counterparts (e.g., hierarchical structure and organization). Nonetheless, further progress is certainly expected in this very active area of research.
3 Conclusions
Our goal in providing these specific case studies has been, on the one hand, to bring attention to the broad range of mechanisms that living organisms employ to accomplish self-healing behavior and, on the other hand, to address the potential challenges that researchers face in hoping to emulate these behaviors in man-made systems. If the aim is to directly copy natural self-healing behaviors, such as tissue regeneration, in all their complexity, the challenge is significant [152]. However, if the goal is to abstract and integrate into materials specific features and design principles of natural healing mechanisms, such as vascularization or reversible sacrificial bonding, then the literature is already rife with examples of progress in this direction [2–8]. Several examples of successful bio-inspired design based on latex-based healing of plants (Sect. 2.5), cell-based healing in pipe vines (Sect. 2.6), and reversible sacrificial bonding in extracorporeal biopolymers such as the mussel byssus (Sect. 2.9.4) have been discussed. The concluding remarks of this review are dedicated to highlighting and discussing the most promising design paradigms presented in the specific case studies.
For the purposes of this review, we have provided only a superficial glimpse of cell-initiated healing and regeneration in animals and plants because of the inherent underlying complexities of such processes. Deeper examination of such mechanisms reveals that biomolecular cell–cell interactions, cell–matrix interactions [153, 154], vascularization, and innervation are crucial to the self-healing response. Furthermore, there probably exists a carefully coordinated synergy of local and non-local interactions that gives rise to the memory effects observed during tissue regeneration. Faithfully emulating such a complex network of interactions and sub-processes as present in regeneration and cellular healing, even roughly, seems unlikely in our lifetime.
In spite of these clear challenges, certain concepts and design paradigms observed in cellular healing and regeneration processes (e.g., vascularization, callus (scaffold) formation, remodeling, stimuli responsive growth, and programmed growth) are interesting for technology transfer. For example, as emphasized in the introductory section, the concept of “sealing-and-healing” (Table 1) found in various forms across the plant and animal kingdoms, is already inspiring development of self-healing materials [6, 7, 14, 15]. In examples from nature, biological systems clearly follow a “first things first” approach that is aimed at avoiding further damage and stabilizing the damaged site. In latex-based healing (Sect. 2.5) and cutaneous wound healing (Sect. 2.4), the rapid mobilization of healing agents is a crucial factor in quickly shoring up the damage so that the subsequent healing stage can bring the material back toward the native state. In both cases, this is facilitated through an extensive vascular transport network. Inspired by these and other examples, there are numerous reports in the literature of self-healing composites possessing a vascular network of channels for the transport of chemical reagents [2, 3, 5, 8]. In such systems, healing agents are conveyed to impact sites within the composite through capillaries comprising one-, two-, or three-dimensional networks in the material. Once there, the various healing agents polymerize and seal/heal the damage, resulting in significant recovery of initial material properties.
Once the damaged area is sealed and the general integrity restored, many of the biological examples in the presented case studies exhibit a subsequent, slower self-healing phase to restore the initial properties and function of the damaged site. However, in general, the current generation of bio-inspired vascular composite systems combine both steps, which limits the damage volume that can be healed to microscopic cracks and defects [3, 155]. From this perspective, the examples of antler growth (Sect. 2.2), bone healing (Sect. 2.3), and plant grafting (Sect. 2.8) reveal how the use of relatively quickly synthesized, but mechanically inferior, scaffolds enables the eventual complete healing, regeneration, or fusion of tissues on the macroscopic scale. In this respect, there is at least one example of a man-made vascular composite capable of healing larger damage volumes that employs a two-stage healing mechanism [155]. In this case, control of the polymerization reaction kinetics of the healing mixture allows highly viscous gel-like behavior at early stages of self-repair that is capable of filling holes of more than 35 mm, while still resulting in a slow polymerization over hours that recovers more than 60% of native material properties.
In addition to healing in response to specific damage events, the process of remodeling and stimuli-responsive healing/growth, as observed in bone, suggests that it is possible to be more proactive in the design of synthetic self-repair mechanisms. For example, the idea of using remodeling to preemptively heal microcracking in a material as a result of fatigue before it can develop into a more serious fracture is highly appealing and perhaps not so far from actualization. One possibility already present in the self-healing literature is the use of dynamic chemical bonds that are able to reorganize under relevant environmental conditions and timescales [156, 157]. Notably, as discussed in Sect. 2.9.4, dynamic reversible bonding in acellular biopolymers (e.g., mussel byssus) perhaps possesses the best possibility of directly transferring design principles from nature to technical and biomedical materials [148]. This stems from the fact that the protein biomolecules comprising these materials are essentially polymeric in nature and are constructed of linear chains of relatively simple amino acid building blocks. Although the concept of reversible bonding has been present in self-healing literature for some time [156, 158], the recent successes of mussel-inspired polymers based on DOPA– and histidine–metal crosslinking suggest that biological systems have something to offer in this regard [4, 148].
Additionally, as we have seen in these highly dynamic examples of biological healing, external physical stimuli can potentially be harnessed to guide healing in a “smart” way. In bone healing, mechanical loading provides information – in terms of the local strain – that dictates what material with which properties should be formed where and when. This has further implications during healing, because new tissue formation results in local stiffening of the tissue. Consequently, an intricate feedback loop between the effect of global mechanical stimulation and local mechanical properties is responsible for the control of bone healing. Inspired by bone healing, recent computer simulations of a generic self-healing material demonstrated that small changes in this local response of the material can grossly change the global healing outcome – the broken ends either reconnected by directly closing the fracture gap or the two segments were first reconnected by a material bridge at the outside of the fracture gap, as observed in bone healing [159]. Although such complex feedback mechanisms are not currently found in any man-made self-healing systems, mechanically responsive crosslinks in polymers (so-called mechanophores) could conceivably provide a pathway for the development of materials that sense mechanical loading in the damaged state and respond by localizing healing and recovery of specific structures [160]. It remains to be seen whether the potential of biological materials research to inspire development of a new generation of bio-inspired self-healing materials will be fully realized; however, current progress seems quite encouraging.
References
Hager MD et al (2010) Self-healing materials. Adv Mater 22(47):5424–5430
Bond I et al (2008) Self healing fibre-reinforced polymer composites: an overview. In: Zwaag S (ed) Self healing materials: an alternative approach to 20 centuries of materials science. Springer, Dordrecht, pp 115–138
Diesendruck CE et al (2015) Biomimetic self-healing. Angew Chem Int Ed. doi:10.1002/anie.201500484
Holten-Andersen N et al (2011) pH-induced metal-ligand cross-links inspired by mussel yield self-healing polymer networks with near-covalent elastic moduli. Proc Natl Acad Sci USA 108(7):2651–2655
Norris CJ et al (2011) Self-healing fibre reinforced composites via a bioinspired vasculature. Adv Funct Mater 21(19):3624–3633
Rampf M et al (2013) Investigation of a fast mechanical self-repair mechanism for inflatable structures. Int J Eng Sci 63:61–70
Schuessele AC et al (2012) Self-healing rubbers based on NBR blends with hyperbranched polyethylenimines. Macromol Mater Eng 297(5):411–419
Toohey KS et al (2007) Self-healing materials with microvascular networks. Nat Mater 6(8):581–585
Clark RAF (1988) Overview and general considerations of wound repair. In: Clark RAF, Henson PM (eds) The molecular and cellular biology of wound repair. Springer, New York
Fratzl P, Weinkamer R (2007) Hierarchical structure and repair of bone: deformation, remodelling, healing. In: Self healing materials: an alternative approach to 20 centuries of materials science, vol 100. Springer, Dordrecht, pp 323–335
Ashton NN, Stewart RJ (2015) Self-recovering caddisfly silk: energy dissipating, Ca2+-dependent, double dynamic network fibers. Soft Matter 11(9):1667–1676
Harrington MJ et al (2009) Collagen insulated from tensile damage by domains that unfold reversibly: in situ X-ray investigation of mechanical yield and damage repair in the mussel byssus. J Struct Biol 167(1):47–54
Miserez A et al (2009) Non-entropic and reversible long-range deformation of an encapsulating bioelastomer. Nat Mater 8(11):910–916
Speck T et al (2013) Bio-inspired self-healing materials. In: Fratzl P, Dunlop JWC, Weinkamer R (eds) Materials design inspired by nature: function through inner architecture. Royal Society of Chemistry, Cambridge, pp 359–389
Speck T, Muelhaupt R, Speck O (2013) Self-healing in plants as bio-inspiration for self-repairing polymers. In: Self-healing polymers: from principles to applications. Wiley, Weinheim, pp 61–89
Speck O et al (2014) Selbstreparatur in Natur und Technik. Konstruktion 9:72–75
Bauer G, Speck T (2012) Restoration of tensile strength in bark samples of Ficus benjamina due to coagulation of latex during fast self-healing of fissures. Ann Bot 109(4):807–811
Speck O, Speck T (2015) Selbstreparatur in Natur und Technik – Versiegeln, heilen, reparieren. Biologie in unserer Zeit 45:44–51
Dunlop JWC, Weinkamer R, Fratzl P (2011) Artful interfaces within biological materials. Mater Today 14(3):70–78
Fratzl P, Weinkamer R (2007) Nature’s hierarchical materials. Prog Mater Sci 52(8):1263–1334
Gould SJ, Lewontin RC (1979) Spandrels of San Marco and the Panglossian paradigm – a critique of the adaptationist program. Proc R Soc Lond B Biol Sci 205(1161):581–598
Alvarado AS (2000) Regeneration in the metazoans: why does it happen? Bioessays 22(6):578–590
Nacu E, Tanaka EM (2011) Limb regeneration: a new development? Annu Rev Cell Dev Biol 27:409–440
Brockes JP, Kumar A (2005) Appendage regeneration in adult vertebrates and implications for regenerative medicine. Science 310(5756):1919–1923
Brockes JP (1997) Amphibian limb regeneration: rebuilding a complex structure. Science 276(5309):81–87
Bryant SV, Endo T, Gardiner DM (2002) Vertebrate limb regeneration and the origin of limb stem cells. Int J Dev Biol 46(7):887–896
Butler EG (1955) Regeneration of the urodele forelimb following after reversal of its proximo-distal axis. J Morphol 96(4):265–281
French V, Bryant PJ, Bryant SV (1976) Pattern regulation in epimorphic fields. Science 193(4257):969–981
Maden M, Holder N (1984) Axial characteristics of nerve induced supernumerary limbs in the axolotl. Rouxs Arch Dev Biol 193(6):394–401
Goss RJ (2012) Deer antlers: regeneration, function and evolution. Academic, New York
Kierdorf U, Kierdorf H (2011) Deer antlers - a model of mammalian appendage regeneration: an extensive review. Gerontology 57(1):53–65
Price JS et al (2005) Deer antlers: a zoological curiosity or the key to understanding organ regeneration in mammals? J Anat 207(5):603–618
Li CY, Suttie JM (2001) Deer antlerogenic periosteum: a piece of postnatally retained embryonic tissue? Anat Embryol 204(5):375–388
Li CY, Harris AJ, Suttie JM (2001) Tissue interactions and antlerogenesis: new findings revealed by a xenograft approach. J Exp Zool 290(1):18–30
Goss RJ (1995) Future-directions in antler research. Anat Rec 241(3):291–302
Li CY, Suttie JM, Clark DE (2005) Histological examination of antler regeneration in red deer (Cervus elaphus). Anat Rec A Discov Mol Cell Evol Biol 282A(2):163–174
Krauss S et al (2011) Tubular frameworks guiding orderly bone formation in the antler of the red deer (Cervus elaphus). J Struct Biol 175(3):457–464
Bubenik AB, Pavlansk R (1965) Trophic responses to trauma in growing antlers. J Exp Zool 159(3):289–302
Bubenik GA (1990) The role of the nervous system in the growth of antlers. In: Bubenik GA, Bubenik AB (eds) Horns, pronghorns, and antlers. Springer, New York, pp 339–358
Lobo D, Solano M, Bubenik GA, Levin M (2014) A linear-encoding model explains the variability of the target morphology in regeneration. J R Soc Interface 11(92):20130918. doi:10.1098/rsif.2013.0918
Currey JD et al (2009) The mechanical properties of red deer antler bone when used in fighting. J Exp Biol 212(24):3985–3993
Launey ME et al (2010) Mechanistic aspects of the fracture toughness of elk antler bone. Acta Biomater 6(4):1505–1514
Gupta HS et al (2013) Intrafibrillar plasticity through mineral/collagen sliding is the dominant mechanism for the extreme toughness of antler bone. J Mech Behav Biomed Mater 28:366–382
Sfeir C et al (2005) Fracture repair. In: Bone regeneration and repair. Humana Press, Totowa, New Jersey, pp 21–44
Betts DC, Muller R (2014) Mechanical regulation of bone regeneration: theories, models, and experiments. Front Endocrinol (Lausanne) 5:211
Vetter A et al (2012) The spatio-temporal arrangement of different tissues during bone healing as a result of simple mechanobiological rules. Biomech Model Mechanobiol 11(1–2):147–160
Gerstenfeld LC et al (2003) Fracture healing as a post-natal developmental process: molecular, spatial, and temporal aspects of its regulation. J Cell Biochem 88(5):873–884
Liu Y et al (2010) Size and habit of mineral particles in bone and mineralized callus during bone healing in sheep. J Bone Miner Res 25(9):2029–2038
Huiskes R et al (2000) Effects of mechanical forces on maintenance and adaptation of form in trabecular bone. Nature 405(6787):704–706
Ruimerman R et al (2005) A theoretical framework for strain-related trabecular bone maintenance and adaptation. J Biomech 38(4):931–941
Dunlop JWC et al (2009) New suggestions for the mechanical control of bone remodeling. Calcif Tissue Int 85(1):45–54
Schulte FA et al (2013) Local mechanical stimuli regulate bone formation and resorption in mice at the tissue level. PLoS One 8(4):e62172
Razi H et al (2015) Aging leads to a dysregulation in mechanically driven bone formation and resorption. J Bone Miner Res. doi:10.1002/jbmr.2528
Bonewald LF (2011) The amazing osteocyte. J Bone Miner Res 26(2):229–238
Evans RK et al (2008) Effects of a 4-month recruit training program on markers of bone metabolism. Med Sci Sports Exerc 40(11):S660–S670
Fantner GE et al (2005) Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat Mater 4(8):612–616
Gupta HS et al (2007) Evidence for an elementary process in bone plasticity with an activation enthalpy of 1 eV. J R Soc Interface 4(13):277–282
Martin P (1997) Wound healing--aiming for perfect skin regeneration. Science 276(5309):75–81
Singer AJ, Clark RA (1999) Cutaneous wound healing. N Engl J Med 341(10):738–746
Murawala P, Tanaka EM, Currie JD (2012) Regeneration: the ultimate example of wound healing. Semin Cell Dev Biol 23(9):954–962
Gurtner GC et al (2008) Wound repair and regeneration. Nature 453(7193):314–321
Aarabi S, Longaker MT, Gurtner GC (2007) Hypertrophic scar formation following burns and trauma: new approaches to treatment. PLoS Med 4(9), e234
Harty M et al (2003) Regeneration or scarring: an immunologic perspective. Dev Dyn 226(2):268–279
Larson BJ, Longaker MT, Lorenz HP (2010) Scarless fetal wound healing: a basic science review. Plast Reconstr Surg 126(4):1172–1180
Clark LD, Clark RK, Heber-Katz E (1998) A new murine model for mammalian wound repair and regeneration. Clin Immunol Immunopathol 88(1):35–45
Birnbaum KD, Alvarado AS (2008) Slicing across kingdoms: regeneration in plants and animals. Cell 132(4):697–710
Lewinsohn TM (1991) The geographical distribution of plant latex. Chemoecology 2:64–68
Metcalfe CR (1967) Distribution of latex in plant kingdom. Econ Bot 21(2):115–127
Agrawal AA, Konno K (2009) Latex: a model for understanding mechanisms, ecology, and evolution of plant defense against herbivory. Annu Rev Ecol Evol Syst 40:311–331
Hunter JR (1994) Reconsidering the functions of latex. Trees Struct Funct 9(1):1–5
Bauer G et al (2014) Comparative study on plant latex particles and latex coagulation in Ficus benjamina, Campanula glomerata and three Euphorbia species. PLoS One 9(11):e113336
Bauer G, Nellesen A, Speck T (2010) Biological lattices in fast self-repair mechanisms in plants and the development of bio-inspired self-healing polymers. In: Brebbia C, Carpi A (eds) Design and nature V: comparing design in nature with science and engineering. WIT, Southampton, pp 453–459
Bauer G, Freidrich C, Gillig C, Vollrath F, Speck T, Holland C (2014) Investigating the rheological properties of native plant latex. J R Soc Interface 11(90):20130847. doi:10.1098/rsif.2013.0847
D’Auzac J, Prevot JC, Jacob JL (1995) What’s new about lutoids – a vacuolar system model from Hevea latex. Plant Physiol Biochem 33(6):765–777
Gidrol X et al (1994) Hevein, a lectin-like protein from Hevea brasiliensis (rubber tree) is involved in the coagulation of latex. J Biol Chem 269(12):9278–9283
Wititsuwannakul R et al (2008) Hevea latex lectin binding protein in C-serum as an anti-latex coagulating factor and its role in a proposed new model for latex coagulation. Phytochemistry 69(3):656–662
Nellesen A et al (2011) Self-healing in plants as a model for self-repairing elastomer materials. Int Polym Sci Technol 38:T/1–T/4
Binder W (2013) Self-healing polymers. Wiley, Weinheim
Jin H et al (2013) Self-healing epoxies and their composites. In: Self-healing polymers: from principles to applications. Wiley, Weinheim, pp 361–380
Rowe NP et al (2006) Diversity of mechanical architectures in climbing plants: an ecological perspective. In: Herrel A, Speck T, Rowe NP (eds) Ecology and biomechanics: a mechanical approach to the ecology of animals and plants. CRC, Boca Raton, pp 35–59
Rowe NP, Speck T (2004) Hydraulics and mechanics of plants: novelty, innovation and evolution. In: Hemsley AR, Poole I (eds) The evolution of plant physiology. Academic, London, pp 301–329
Rowe NP, Speck T (2015) Stem biomechanics, strength of attachment, and developmental plasticity of vines and lianas. In: Schnitzer S et al (eds) The ecology of lianas. Wiley-Blackwell, Chichester, pp 323–341
Busch S et al (2010) Morphological aspects of self-repair of lesions caused by internal growth stresses in stems of Aristolochia macrophylla and Aristolochia ringens. Proc R Soc B Biol Sci 277(1691):2113–2120
Speck T et al (2004) The potential of plant biomechanics in functional biology and systematics. In: Stuessey T, Hörandl F, Mayer V (eds) Deep morphology: toward a renaissance of morphology in plant systematics. Koeltz, Königstein, pp 241–271
Luchsinger RH, Pedretti M, Reinhard A (2004) Pressure induced stability: from pneumatic structures to Tensairity. J Bionic Eng 1:141–148
Speck T et al (2006) Self-healing processes in nature and engineering: self-repairing biomimetic membranes for pneumatic structures. In: Brebbia CA (ed) Design and nature III: comparing design in nature with science and engineering. WIT, Southampton, pp 105–114
Rampf M et al (2011) Self-repairing membranes for inflatable structures inspired by a rapid wound sealing process of climbing plants. J Bionic Eng 8(3):242–250
Rampf M et al (2012) Structural and mechanical properties of flexible polyurethane foams cured under pressure. J Cell Plast 48(1):53–69
Konrad W et al (2013) An analytic model of the self-sealing mechanism of the succulent plant Delosperma cooperi. J Theor Biol 336:96–109
Caliaro M et al (2013) Novel method for measuring tissue pressure in herbaceous plants. Int J Plant Sci 174(2):161–170
Beddie AD (1941) Natural root grafts in New Zealand trees. Trans R Soc New Zealand 71(3):199–203
Dallimore W (1917) Natural grafting of branches and roots. Bull Misc Inf (Royal Botanical Gardens Kew) 1917:303–306
Küster E (1899) Über Stammverwachsungen. Jahrbücher für Wissenschaftliche Botanik 33:487–512
Millner ME (1932) Natural grafting in Hedera helix. New Phytol 31(1):2–25
Seidel CF (1879) Über Verwachsungen von Stämmen und Zweigen von Holzgewächsen und ihren Einfluss auf das Dickenwachsthum der betreffenden Theile. Isis Sitzber, Dresden, pp 161–168
Hartmann HT et al (2002) Hartmann and Kesters’s plant propagation: principles and practices. Prentice-Hall, New Jersey
Yeoman MM, Brown R (1976) Implication of formation of graft union for organization in intact plant. Ann Bot 40(170):1265–1276
Graham BF, Bormann FH (1966) Natural root grafts. Bot Rev 32(3):255–292
La Rue CD (1934) Root grafting in trees. Am J Bot 21(3):121–126
Mudge K et al (2009) A history of grafting. Hortic Rev 35:437–493
Moore R (1982) Graft formation in Kalanchoe blossfeldiana. J Exp Bot 33(134):533–540
Moore R (1983) Studies of vegetative compatibility-incompatibility in higher plants. IV. The development of tensile strength in a compatible and an incompatible graft. Am J Bot 70(2):226–231
Moore R (1984) Graft formation in Solanum pennellii (Solanaceae). Plant Cell Rep 3(5):172–175
Moore R, Walker DB (1983) Studies of vegetative compatibility-incompatibility in higher plants. VI. Grafting of Sedum and Solanum callus tissue in vitro. Protoplasma 115(2–3):114–121
Pedersen BH (2005) Development of tensile strength in compatible and incompatible sweet cherry graftings. Can J Bot 83(2):202–210
Pina A, Errea P (2005) A review of new advances in mechanism of graft compatibility-incompatibility. Sci Hortic 106(1):1–11
Yin H et al (2012) Graft-union development: a delicate process that involves cell-cell communication between scion and stock for local auxin accumulation. J Exp Bot 63(11):4219–4232
Esau K, Evert RF, Eichhorn SE (2006) Esau’s plant anatomy: meristems, cells, and tissues of the plant body: their structure, function, and development. Wiley, Hoboken
Larson PR (1994) The vascular cambium: development and structure. Springer, Berlin
Dormling I (1963) Anatomical and histological examination of the union of scion and stock in grafts of scots pine (Pinus silvestris L.) and Norway spruce (Picea abies (L.) Karit). Stud For Suec 13:1–136
McCulley ME (1983) In: Moore R (ed) Vegetative compatibility responses in plants. Baylor University Press, Waco, pp 71–88
Moore R (1983) In: Moore R (ed) Vegetative compatibility responses in plants. Baylor University Press, Waco, pp 89–105
Pina A, Errea P, Martens HJ (2012) Graft union formation and cell-to-cell communication via plasmodesmata in compatible and incompatible stem unions of Prunus spp. Sci Hortic 143:144–150
Aloni B et al (2010) Hormonal signaling in rootstock-scion interactions. Sci Hortic 127(2):119–126
Aloni R (1987) Differentiation of vascular tissues. Annu Rev Plant Physiol Plant Mol Biol 38:179–204
Mattsson J, Ckurshumova W, Berleth T (2003) Auxin signaling in Arabidopsis leaf vascular development. Plant Physiol 131(3):1327–1339
Fuentes I et al (2014) Horizontal genome transfer as an asexual path to the formation of new species. Nature 511(7508):232–235
Molnar A et al (2010) Small silencing RNAs in plants are mobile and direct epigenetic modification in recipient cells. Science 328(5980):872–875
Stegemann S, Bock R (2009) Exchange of genetic material between cells in plant tissue grafts. Science 324(5927):649–651
Stegemann S et al (2012) Horizontal transfer of chloroplast genomes between plant species. Proc Natl Acad Sci USA 109(7):2434–2438
Denny MW, Gaylord B (2010) Marine ecomechanics. Ann Rev Mar Sci 2(1):89–114
Carrington E, Gosline JM (2004) Mechanical design of mussel byssus: load cycle and strain rate dependence. Am Malacol Bull 18(1/2):135–142
Waite JH, Qin X-X, Coyne KJ (1998) The peculiar collagens of mussel byssus. Matrix Biol 17(2):93–106
Krauss S et al (2013) Self-repair of a biological fiber guided by an ordered elastic framework. Biomacromolecules 14(5):1520–1528
Arnold AA et al (2013) Solid-state NMR structure determination of whole anchoring threads from the blue mussel Mytilus edulis. Biomacromolecules 14(1):132–141
Hagenau A et al (2011) Mussel collagen molecules with silk-like domains as load-bearing elements in distal byssal threads. J Struct Biol 175(3):339–347
Schmidt S et al (2014) Metal-mediated molecular self-healing in histidine-rich mussel peptides. Biomacromolecules 15(5):1644–1652
Schmitt CNZ, Politi Y, Reinecke A, Harrington MJ (2015) The role of sacrificial protein-metal bond exchange in mussel byssal thread self-healing. Biomacromolecules 16(9):2852–2861. doi:10.1021/acs.biomac.5b00803
Harrington MJ, Waite JH (2007) Holdfast heroics: comparing the molecular and mechanical properties of Mytilus californianus byssal threads. J Exp Biol 210(24):4307–4318
Vaccaro E, Waite JH (2001) Yield and post-yield behavior of mussel byssal thread: a self-healing biomolecular material. Biomacromolecules 2(3):906–911
Harrington MJ et al (2010) Iron-clad fibers: a metal-based biological strategy for hard flexible coatings. Science 328(5975):216–220
Schmitt CNZ, Winter A, Bertinetti L, Masic A, Strauch P, Harrington MJ (2015) Mechanical homeostasis of a DOPA-enriched biological coating from mussels in response to metal variation. J R Soc Interface 12(110):20150466. doi:10.1098/rsif.2015.0466
Taylor SW et al (1996) Ferric ion complexes of a DOPA-containing adhesive protein from Mytilus edulis. Inorg Chem 35(26):7572–7577
Holten-Andersen N et al (2007) Protective coatings on extensible biofibres. Nat Mater 6(9):669–672
Ashton NN et al (2013) Self-tensioning aquatic caddisfly silk: Ca2+-dependent structure, strength, and load cycle hysteresis. Biomacromolecules 14(10):3668–3681
Yonemura N et al (2009) Conservation of silk genes in Trichoptera and Lepidoptera. J Mol Evol 68(6):641–653
Wang C-S et al (2014) Peroxinectin catalyzed dityrosine crosslinking in the adhesive underwater silk of a casemaker caddisfly larvae, Hysperophylax occidentalis. Insect Biochem Mol Biol 54:69–79
Addison JB et al (2013) Beta-sheet nanocrystalline domains formed from phosphorylated serine-rich motifs in caddisfly larval silk: a solid state NMR and XRD study. Biomacromolecules 14(4):1140–1148
Addison JB et al (2014) Reversible assembly of beta-sheet nanocrystals within caddisfly silk. Biomacromolecules 15(4):1269–1275
Rapoport HS, Shadwick RE (2002) Mechanical characterization of an unusual elastic biomaterial from the egg capsules of marine snails (Busycon spp.). Biomacromolecules 3(1):42–50
Harrington MJ et al (2012) Pseudoelastic behaviour of a natural material is achieved via reversible changes in protein backbone conformation. J R Soc Interface 9(76):2911–2922
Wasko SS et al (2014) Structural proteins from whelk egg capsule with long range elasticity associated with a solid-state phase transition. Biomacromolecules 15(1):30–42
Fischer FD, Harrington MJ, Fratzl P (2013) Thermodynamic modeling of a phase transformation in protein filaments with mechanical function. New J Phys 15(6):065004
Degtyar E et al (2015) Recombinant engineering of reversible cross-links into a resilient biopolymer. Polymer 69:255–263
Fu T et al (2015) Biomimetic self-assembly of recombinant marine snail egg capsule proteins into structural coiled-coil units. J Mater Chem B 3(13):2671–2684
Fullenkamp DE et al (2013) Mussel-inspired histidine-based transient network metal coordination hydrogels. Macromolecules 46(3):1167–1174
Krogsgaard M et al (2013) Self-healing mussel-inspired multi-pH-responsive hydrogels. Biomacromolecules 14(2):297–301
Li L, Smitthipong W, Zeng H (2015) Mussel-inspired hydrogels for biomedical and environmental applications. Polym Chem 6(3):353–358
Enke M et al (2015) Self-healing response in supramolecular polymers based on reversible zinc–histidine interactions. Polymer 69:274–282
Lee BP, Konst S (2014) Novel hydrogel actuator inspired by reversible mussel adhesive protein chemistry. Adv Mater 26(21):3415–3419
Lane DD et al (2015) Toughened hydrogels inspired by aquatic caddisworm silk. Soft Matter. doi:10.1039/C5SM01297J
van der Zwaag S et al (2009) Self-healing behaviour in man-made engineering materials: bioinspired but taking into account their intrinsic character. Philos Trans R Soc A Math Phys Eng Sci 367(1894):1689–1704
Discher DE, Janmey P, Wang YL (2005) Tissue cells feel and respond to the stiffness of their substrate. Science 310(5751):1139–1143
Kollmannsberger P et al (2011) The physics of tissue patterning and extracellular matrix organisation: how cells join forces. Soft Matter 7(20):9549–9560
White SR et al (2014) Restoration of large damage volumes in polymers. Science 344(6184):620–623
Wojtecki RJ, Meador MA, Rowan SJ (2011) Using the dynamic bond to access macroscopically responsive structurally dynamic polymers. Nat Mater 10(1):14–27
Ying H, Zhang Y, Cheng J (2014) Dynamic urea bond for the design of reversible and self-healing polymers. Nat Commun 5:3218. doi: 10.1038/ncomms4218
Cordier P et al (2008) Self-healing and thermoreversible rubber from supramolecular assembly. Nature 451(7181):977–980
Vetter A et al (2013) Healing of a mechano-responsive material. Europhys Lett 104(6):68005
Brown CL, Craig SL (2015) Molecular engineering of mechanophore activity for stress-responsive polymeric materials. Chem Sci 6(4):2158–2165
Acknowledgement
The authors thank A. Miserez and R. Stewart for providing images for figures, and C. Neinhuis for helpful input. Part of the work on mussel byssal thread healing was funded by the DFG priority program 1568 on “Design and Generic Principles of Self-Healing Materials” (HA6369/1-1 and HA6369/1-2). Financial support from the DFG for research within the Cluster of Excellence: “Image Knowledge Gestaltung: An Interdisciplinary Laboratory” is acknowledged. Several of the projects on self-repair mechanisms in plants were funded by the German Federal Ministry of Education and Research in the frameworks of the funding programme BIONA (project ‘Self-healing polymers “OSIRIS”’) and of the Ideenwettbewerb “Bionik – Innovationen aus der Natur” (FKZ0313778A, together with Empa Dübendorf). Two other projects on self-repair mechanisms in plants are part of the European Marie Curie Initial Training Network “Self-Healing Materials: from Concepts to Market” (SHeMat).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Harrington, M.J., Speck, O., Speck, T., Wagner, S., Weinkamer, R. (2015). Biological Archetypes for Self-Healing Materials. In: Hager, M., van der Zwaag, S., Schubert, U. (eds) Self-healing Materials. Advances in Polymer Science, vol 273. Springer, Cham. https://doi.org/10.1007/12_2015_334
Download citation
DOI: https://doi.org/10.1007/12_2015_334
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-32776-1
Online ISBN: 978-3-319-32778-5
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)