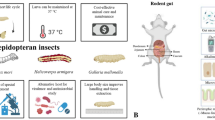
Abstract
Insect models, such as Galleria mellonella and Drosophila melanogaster have significant ethical, logistical, and economic advantages over mammalian models for the studies of infectious diseases. Using these models, various pathogenic microbes have been studied and many novel virulence genes have been identified. Notably, because insects are susceptible to a wide variety of human pathogens and have immune responses similar to those of mammals, they offer the opportunity to understand innate immune responses against human pathogens better. It is important to note that insect pathosystems have also offered a simple strategy to evaluate the efficacy and toxicity of many antimicrobial agents. Overall, insect models provide a rapid, inexpensive, and reliable way as complementary hosts to conventional vertebrate animal models to study pathogenesis and antimicrobial agents.

Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Pathogenic microbes cause a variety of infectious diseases in human hosts, and the threat from these pathogens has never faded in human history [1–6]. For example, methicillin-resistant Staphylococcus aureus (MRSA) alone infects more than 94,000 people and kills nearly 19,000 in the United States every year, more deaths than those caused by HIV/AIDS, Parkinson’s disease, emphysema, and homicide combined [7, 8]. Also, in addition to bacterial infections, the frequency, spectrum, and associated cost of opportunistic invasive fungal infections have significantly increased over the past two decades and accounted for the rapidly growing populations of immunosuppressed and debilitated patients [9–11]. The substantial disease burden of infectious diseases in humans underscores the need for better understanding of the pathogenicity and virulence of human pathogens.
Pathogenesis, immunology, and pharmacology research have traditionally relied on mammalian models such as mice, rats, rabbits, and guinea pigs, but such experiments are costly, time consuming, and require full ethical consideration. Hence, cheaper and ethically more acceptable insect models of infection have been introduced, including the larvae of the greater wax moth Galleria mellonella, Drosophila melanogaster, and other insects. Accumulating data indicate that the virulence of many human pathogens is comparable in insects and mammals, and often identical virulence factors are used by human pathogenic microbes to infect insects and mammals. Moreover, insects have an immune system that is functionally similar to the innate immune system of mammals, which offers a simple model to understand innate immunity better. Furthermore, the insect infection models provide a rapid, inexpensive, and reliable evaluation of the efficacy and toxicity of new antimicrobial agents in vivo. In this chapter, we discuss how the insects G. mellonella, D. melanogaster, and other insects can be employed to study various human pathogens and to evaluate new antimicrobial agents.
2 Use of Insects for Studying Human Pathogens
Galleria mellonella and Drosophila melanogaster have emerged at the forefront of host–pathogen interaction research and show promise for identification of novel virulence genes and deciphering conserved innate immunity mechanisms. Insects have both cellular and humoral immune response to infection [12–14], making them attractive models to study pathogen–host interactions. Although adaptive immunity is unique to vertebrates, the innate immune response seems to be well conserved between vertebrates and invertebrates [15–18]. Moreover, insects have immune responses mediated by antimicrobial peptides [14], which also play a crucial role in human immunity. Assays using insects as the infection host are usually inexpensive, simple to perform, and yield results within a short timeframe [19, 20]. These advantages make insects attractive hosts for studying human pathogens.
2.1 Galleria mellonella Infection Model
The greater wax moth G. mellonella (Lepidoptera: Pyralidae) is found in most of the world [19] and has many advantages over other invertebrate hosts [18, 19, 21]. Firstly, G. mellonella is easy to work with and the larvae are inexpensive [19]. The larvae do not require any specialized equipment; they are housed in petri dishes and can be kept in an incubator or at room temperature [22, 23]. Also, G. mellonella larvae are a convenient size (2–3 cm in length) to work with and a large number of larvae may be inoculated in a short period of time [20]. Second, the G. mellonella larvae can be maintained at temperatures between 15 and 37 °C [22, 24]. This makes the larvae well suited to study pathogens at human body temperature. Other invertebrate hosts, such as D. melanogaster, cannot be maintained at temperatures over 30 °C [18, 19, 25]. This is significant because it affects the pathogenicity of organisms inasmuch as virulence factors are known to be regulated by temperature [19, 22, 25–27]. Third, quantifying the infecting inoculum is accurate in G. mellonella. Precise infection inoculum can be delivered to G. mellonella by injection into the larva’s hemocoel [22]. This is notable because an accurate dose of inoculated pathogen partly contributes to the reproducible disease progression and survival outcomes [19].
The G. mellonella pathosystem has been widely used in the virulence study of pathogenic microbes, especially in the identification of novel virulence genes through comparing the virulence of the mutant and the wildtype strains. Interestingly, this model was previously mainly used to characterize virulence factors in Bacillus cereus and Bacillus thuringiensis [28–30]. More recently, G. mellonella has been employed to investigate a variety of pathogens (Table 1), including Gram-negative bacteria such as Acinetobacter baumannii [24, 31–38], Burkholderia spp. [39–56], Listeria monocytogenes [57–61], Pseudomonas aeruginosa [58, 62–67], Yersinia spp. [68–71], Campylobacter jejuni [49, 72, 73], Proteus mirabilis [74–77], Escherichia coli [78], and Legionella pneumophila [79], Gram-positive bacteria such as Enterococcus faecalis [58, 80–86] and Staphylococcus aureus [13, 87–89], and fungi such as Candida albicans [22, 90–95], Cryptococcus neoformans [22, 96–100], and Aspergillus spp. [97, 101–111]. Infections caused by A. baumannii [24, 31–38], Burkholderia spp. [39–56], L. monocytogenes [57–61], C. albicans [22, 90–95], and C. neoformans [22, 96–100] have been particularly well characterized in the G. mellonella infection model; the G. mellonella pathosystem was efficient especially in Gram-negative bacteria and in fungi. It is important to note that the virulence determinants of most pathogens are similar in the G. mellonella larvae and mammals [20, 39, 42, 50, 65, 68, 98, 107, 112–114]. Nevertheless, the G. mellonella model is not appropriate for studying infections caused by Pneumocystis murina [115] or Dermatophytes [116], as the G. mellonella larva is resistant to these fungi.
Many studies have characterized the immune defense responses of the G. mellonella larvae [59, 68, 72, 90, 96, 98, 114, 115, 117–119]. There are considerable similarities between the systemic cellular and humoral immune responses of the G. mellonella larvae and the innate immune responses of mammals [12, 120–122]. Both the G. mellonella and mammals have many immune recognition proteins [12, 121, 122]. After pathogen recognition, both the insect and mammalian immune defenses rely on phagocytosis, the production of reactive oxygen species, the expression of antimicrobial peptides, and clotting cascades to combat invasive pathogens [12, 25, 123]. However, the G. mellonella larvae also form melanin during infection, but this process does not occur in mammals. In addition, the G. mellonella larvae form pathogen microaggregates that ultimately lead to hemocyte nodule or capsule formation [12], which is also different from mammalian immune responses. Compared with D. melanogaster, a notable disadvantage of the G. mellonella model is that the Galleria genome sequence has not been completed yet, although a huge array of genetic tools has been used in the D. melanogaster model. The recent characterization of the Galleria immune gene repertoire and transcriptome by next generation sequencing and traditional Sanger sequencing has led to the design of gene microarrays and paves the way for further use of Galleria for elucidation of innate antimicrobial immune mechanisms [124].
2.2 Drosophila melanogaster Infection Model
Drosophila melanogaster is a species of Diptera in the Drosophilidae family. The species is known generally as the common fruit fly. Beginning with Charles W. Woodworth, this species has been a widely used model organism for biological research in studies of genetics, physiology, and microbial pathogenesis. It is typically used because it is an animal species that is easy to handle and breed. The most important advantage of D. melanogaster as a mini-host is that the fruit fly is amenable to forward and reverse genetics and large collections of Drosophila mutants and transgenic cell lines are commercially available (http://flybase.org). The Drosophila genome sequence has been completed and is among the most fully annotated eukaryotic genomes. Thus, gene microarrays have been generated, double-stranded RNA has been synthesized for all genes (www.flyrnai.org), and RNA interference technology is commercially available for conditional inactivation of any gene at the whole-animal or tissue levels (http://stockcenter.vdrc.at/control/main) [124].
Drosophila melanogaster requires a more significant commitment than the G. mellonella model. Working with D. melanogaster as a host to study human pathogens requires considerable experience and specialized equipment such as microinjectors [125–131] to infect it with a certain infecting inoculum. In addition, because wildtype D. melanogaster are resistant to infection with some pathogens, Imd or Toll pathway-deficient flies need to be used, which in some cases requires a fly genetic cross [125, 126, 128–130]. Nevertheless, the D. melanogaster pathosystem is still among the simplest infection models.
The D. melanogaster pathosystem has also been widely used to identify virulence determinants of pathogenic microbes, including P. aeruginosa [15, 63, 132–137], Streptococcus pneumoniae [138, 139], Serratia marcescens [140–154], L. monocytogenes [138, 139, 155], C. albicans [92, 125, 128, 131, 156–158], C. neoformans [99, 159, 160], and Aspergillus fumigatus [126, 129, 161] (Table 1), and this pathosystem is promising for large-scale studies. Note that there is also a significant concordance for virulence of most pathogens in D. melanogaster and mammals. Three infection assays have been used for assessment of fungal virulence in insects: injection, rolling, and ingestion assays. Although quantification of the infecting inoculum is feasible only in the injection assay, the availability of different routes of infection permits comparative analyses of virulence and host–pathogen interactions between an acute infection introduced directly into the hemolymph (injection assay) versus more protracted infections originating from epithelial surfaces [i.e., skin (rolling assay) or gastrointestinal mucosa (ingestion assay)]. Interestingly, the alb1-deficient A. fumigatus mutant was found to be hypovirulent in D. melanogaster when introduced via epithelial surfaces but not by injection [130].
Drosophila melanogaster has been a major tool for studying innate immunity [124] inasmuch as they mount a highly efficient innate immune defense, the first line of which consists of epithelial responses that prevent infections. When physical barriers are breached and pathogenic microbes invade within the insect body, insects induce a highly coordinated immune response that has both cellular and humoral constituents. In the case of fungal infection, the immune responses at the epithelial level are Toll-independent, which is opposed to the requirement of intact Toll signaling for defense against systemic fungal infection. Consistently, the epithelial antifungal immune responses in the fruit fly are mediated by the dual oxidase (DUOX), JAK-STAT, and immune deficiency (imd) pathways [147, 162, 163] instead of Toll. Notably, the epithelial immune responses are conserved in the fruit fly and mammals, and the similarity in the intestinal epithelium anatomy and regeneration time between flies and mammals [164] supports the utility of Drosophila for examining immunological mechanisms of mucosal infection.
Some Drosophila strains have been employed to study cellular and humoral immunity. With regard to cellular immunity, a phagocytosis-defective eater-null Drosophila strain was used to reveal that phagocytosis is indispensable for fly survival against zygomycosis [165]. A Drosophila S2 phagocytic cell line was used to describe a macroglobulin complement-related protein; the protein bound specifically on the surface of C. albicans and enhanced phagocytosis. Also by using the S2 phagocytic cell line, some autophagy host factors (e.g., Atg2, Atg5, Atg9, Pi3K59F) were identified; the autophagy molecules were induced after exposure to C. neoformans in the fly and were also required for cryptococcal intracellular trafficking and replication within murine phagocytes [166]. With regard to humoral immunity, the Toll signaling cascade in D. melanogaster is crucial for host defense against systemic infection via induction of antifungal peptide [167, 168], the role of which is similar to the Toll/IL-1β receptor signaling in mammals.
Of note, comparative analyses of immune responses using more than one insect host and more than one inoculation assay could be enlightening for dissecting pathogen- and tissue-specific innate immune mechanisms [169], because insects have differential susceptibility to some infections (e.g., wildtype Galleria is susceptible to Candida or Cryptococcus injection whereas wildtype Drosophila is not) [20, 98, 125, 159], and because an insect may exhibit differential susceptibility to a specific pathogen depending on the route of inoculation (e.g., Cryptococcus ingestion but not injection kills wildtype Drosophila [98], and Candida injection but not ingestion kills adult Toll-deficient flies [125]).
2.3 Other Insect Infection Models
In addition to D. melanogaster and G. mellonella, the red flour beetle Tribolium castaneum (Coleoptera, Tenebrionidae) has previously been used to investigate host–pathogen interactions with a wide array of pathogenic bacteria, sporozoa, cestoda, nematoda, mites, and hymenopterous parasites [170]. The Tribolium genome has been sequenced by the Human Genome Sequencing Center, Baylor College of Medicine, USA (Tribolium Genome Sequencing Consortium 2008). Similar to D. melanogaster, Tribolium is also amenable to systemic RNAi-mediated gene silencing and other genetic tools for functional gene analyses [171–177]. Moreover, the soil-living amoebas Acanthamoeba castellanii and Dictyostelium discoideum, the lepidopteran insect silkworm Bombyx mori, the mosquito Culex quinquefasciatus, and the German cockroach Blattella germanica have attracted interest due to their potential as good model systems for the screening of virulence factors of pathogenic microbes (Table 1).
3 Use of Insects for Evaluating New Antimicrobial Agents
Host-based antimicrobial drug discovery is important because efficacy of potential antimicrobial agents might be altered by in vivo factors such as pH, enzymatic degradation, or binding to molecular components within the host [19, 212]. Moreover, some compounds without in vitro activity may be antimicrobially effective because of their immune regulating role or the production of effective metabolites in vivo. With regard to the in vivo studies, animal infection models not only provide data on effectiveness of antimicrobial agents in vivo and their toxicity, but also enable dose and medication schedule recommendations for use in the first human dose. Using insect models for initial toxicity and efficacy screening is financially and ethically more acceptable in the early stages of antimicrobial discovery and development [19]. As a consequence, interest in using insect models to evaluate compounds before testing in mammalian species has increased [213].
3.1 Galleria mellonella Infection Model for Evaluating New Antimicrobial Agents
Infection of G. mellonella is amenable to antimicrobial treatment [19, 98, 117]. This makes the G. mellonella model highly suitable for evaluating the efficacy and toxicity of potential antimicrobial agents in vivo prior to testing in mammalian species.
A key benefit of using G. mellonella larvae to assess antimicrobial efficacy is that, as with administration of the infectious inoculum, an accurate dose of antimicrobial can be delivered directly into the hemocoel by injection [19, 22]. This is not always possible in other invertebrate models [23]. Moreover, this model simulates the intravenous systemic administration of antimicrobial agents in a relative dose and schedule that would be used in patients. The G. mellonella model is useful for testing different treatment regimens. Furthermore, the experimental course of therapy can be varied according to the dose of antimicrobial agent administered, the number of doses, and the timing of the first and successive administrations [19].
The G. mellonella model has been used to study the efficacy of many antimicrobial agents against a multitude of bacterial and fungal pathogens (Table 2). For example, streptomycin, ciprofloxacin, and levofloxacin were evaluated for treating Francisella tularensis live vaccine strain (LVS) [117]; gentamicin, meropenem, tetracycline, and cefotaxime were evaluated for treating A. baumannii infection [24]; amphotericin B and other antifungal agents were evaluated for treating C. neoformans infection in this G. mellonella model [98].
Note that the antimicrobial drug susceptibility profiles of pathogens in G. mellonella larvae are largely the same as those in vitro studies [5, 15, 41–44]. Peleg et al. infected G. mellonella larvae with a lethal dose of a reference strain of A. baumannii, which was susceptible to gentamicin and meropenem but resistant to tetracycline and cefotaxime in vitro [24]. Survival was significantly higher in the groups receiving gentamicin and meropenem (p < 0.001) compared to the infected untreated control group. Treatment with tetracycline and cefotaxime had no effect on survival. Also, Hornsey et al. demonstrated the same phenomenon using a strain of A. baumannii susceptible to gentamicin and colistin but resistant to teicoplanin [35]. Another notable example is focused on the treatment of S. aureus infection using this G. mellonella model. When the larvae were infected with a penicillin-susceptible strain, larvae could be protected significantly by penicillin; when the larvae were infected with a penicillin-resistant strain, larvae could not be protected by penicillin. Of note, many studies have showed that effective weight-based doses of antibiotics used in G. mellonella larvae are similar to recommended doses used in human subjects [24, 35, 98, 117, 214]. Dose data from this model are likely to provide a more precise estimate of doses in subsequent mammalian studies than in vitro data based on minimum inhibitory concentration values [19].
Nowadays combination antibiotic therapy is widely used to prevent the emergence of resistant strains of pathogens [215]. The G. mellonella model is well placed to test the efficacy of combination antimicrobial therapy and drug interactions in vitro (Table 3). Mylonakis et al. demonstrated the benefits of combination antifungal therapy in larvae infected with C. neoformans [98]. Larvae treated with combination amphotericin B (1.5 mg/kg) and flucytosine (20 mg/kg) had significantly higher survival rates than those treated with amphotericin B alone. Vu and Gelli showed significantly higher survival in C. neoformans-infected larvae treated with combination flucytosine (53 mg/kg) and astemizole (53 mg/kg), an antihistamine, or an astemizole homologue compared to monotherapy with these agents alone [166, 216]. Similarly, Cowen et al. demonstrated higher survival in treating C. albicans and A. fumigatus infections in this model using a combination of Hsp90 inhibitors with fluconazole or caspofungin, respectively, than with monotherapy with these agents [217]. These data imply that insects are promising hosts for assessing the efficacy of innovative therapeutic strategies such as a combination of antifungal agents with immune- or virulence-modulating drugs.
Although larval survival is the most common used measurable endpoint to assess antimicrobial efficacy, the microbial burden in larvae can also be used to quantify antimicrobial efficacy [39, 50, 65, 68, 96, 98, 117]. The microbial burden may be a better indicator of the more subtle effects of antimicrobial agents where larval survival is less indicative of antimicrobial efficacy [19]. It can also be used to confirm the complete treatment of infection after eradication of pathogens or to study the dynamics of infection during treatment [19, 39, 65]. There is generally an inverse relationship between the microbial burden and larval survival, although this is not always the case. The treatment of infection and efficacy of antimicrobial agents can also be assessed using hemocyte counts in hemolymph or changes in larval gene expression [93, 118, 119, 218]. Hemocyte counts should be interpreted with caution because counts may be lower in more virulent infections or remain unchanged if the interaction is nonpathogenic [118]. This depends upon the pathogen under investigation.
Also, the G. mellonella model provides a unique opportunity to identify toxic effects of antimicrobial agents during an infection, which is not possible with in vitro testing [19]. In standard in vivo testing, the median lethal dose (LD50) is established by administering various doses to otherwise healthy animals. It is possible, however, that toxicity of some antimicrobial agents may only become apparent once administered to sick animals. As reviewed by Desalermos et al., certain doses of antimicrobial agents that were nontoxic in standard in vivo testing in healthy animals can cause greater or faster mortality in infected animals compared to a control group of infected animals administered carrier solvent only [219]. The G. mellonella model is ideal for this type of study, which can only be achieved using an in vivo infection model. Prescreening the promising antimicrobial agents using the G. mellonella model before progressing to the mammals will save time, money, and needless experimentation in mammals [19].
3.2 Drosophila melanogaster Infection Model for Evaluating New Antimicrobial Agents
The D. melanogaster infection model is not often used to evaluate antimicrobial agents (Table 2). Some studies have used the D. melanogaster infection model to study the efficacy of some licensed antifungal agents and shown remarkable correlation between in vitro susceptibility testing results and in vivo drug efficacy in both insects and mammals [125, 130]. Notably, the synergy between voriconazole and terbinafine against A. fumigatus was demonstrated in this model [130], which is consistent with the synergistic effect in vitro and in mammals, thus further providing evidence that the D. melanogaster model may be used as a complementary assay to evaluate antimicrobial agents.
Of note, pharmacology studies in insects also have limitations despite their potential. Although both Drosophila and Galleria can be used for testing orally absorbable compounds, the exact ingested drug dose per insect is difficult to quantify precisely. Testing of parenteral antimicrobial compounds also has constraints as repeated drug injections lead to injury, especially in Drosophila.
3.3 Other Insect Infection Models for Evaluating New Antimicrobial Agents
The silkworm B. mori infection model is useful for evaluating the efficacy, pharmacokinetics, and toxicity of antifungal drugs, similar to the G. mellonella model [210]. Antifungal drugs, amphotericin B, flucytosine, fluconazole, and ketoconazole showed therapeutic effects in silkworms infected with C. neoformans [210]. However, amphotericin B was not therapeutically effective when injected into the B. mori intestine, comparable to the fact that amphotericin B is not absorbed by the intestine in mammals [210].
Despite the potential of insect models, pharmacokinetic analyses are problematic in insects and it is technically challenging to measure drug levels inside the insects. It is important to note that it is difficult to rely on insect models for critical pharmacological parameters such as drug absorption, distribution, metabolism, excretion, and drug–drug interactions, and therefore testing is necessary in mammalian hosts that are phylogenetically closer to humans.
4 Conclusion
Drosophila melanogaster and G. mellonella have emerged at the forefront of host–fungal interaction research and show promise for evaluating antimicrobial agents. Because no single nonvertebrate organism fully reproduces all aspects of mammalian infection, comparative research in these hosts is required and should be complemented by studies in mammalian models of infection.
Abbreviations
- MRSA:
-
Methicillin-resistant Staphylococcus aureus
- RNAi:
-
RNA interference
References
Parkins MD, Gregson DB, Pitout JD, Ross T, Laupland KB (2010) Population-based study of the epidemiology and the risk factors for Pseudomonas aeruginosa bloodstream infection. Infection 38(1):25–32
Roberts RR, Hota B, Ahmad I, Scott RD 2nd, Foster SD, Abbasi F, Schabowski S, Kampe LM, Ciavarella GG, Supino M, Naples J, Cordell R, Levy SB, Weinstein RA (2009) Hospital and societal costs of antimicrobial-resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship. Clin Infect Dis 49(8):1175–1184
Filice GA, Nyman JA, Lexau C, Lees CH, Bockstedt LA, Como-Sabetti K, Lesher LJ, Lynfield R (2010) Excess costs and utilization associated with methicillin resistance for patients with Staphylococcus aureus infection. Infect Control Hosp Epidemiol 31(4):365–373
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR (2001) Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 29(7):1303–1310
Mauldin PD, Salgado CD, Hansen IS, Durup DT, Bosso JA (2010) Attributable hospital cost and length of stay associated with health care-associated infections caused by antibiotic-resistant gram-negative bacteria. Antimicrob Agents Chemother 54(1):109–115
Davies A, Ridley S, Hutton J, Chinn C, Barber B, Angus DC (2005) Cost effectiveness of drotrecogin alfa (activated) for the treatment of severe sepsis in the United Kingdom. Anaesthesia 60(2):155–162
Vashishtha VM (2010) Growing antibiotics resistance and the need for new antibiotics. Indian Pediatr 47(6):505–506
(2009) Urgently needed: new antibiotics. Lancet 374(9705):1868
Miller LG, Hajjeh RA, Edwards JE Jr (2001) Estimating the cost of nosocomial candidemia in the United States. Clin Infect Dis 32(7):1110
Pfaller MA, Diekema DJ (2007) Epidemiology of invasive candidiasis: a persistent public health problem. Clin Microbiol Rev 20(1):133–163
Paulitsch A, Weger W, Ginter-Hanselmayer G, Marth E, Buzina W (2006) A 5-year (2000–2004) epidemiological survey of Candida and non-Candida yeast species causing vulvovaginal candidiasis in Graz. Austria Mycoses 49(6):471–475
Kavanagh K, Reeves EP (2004) Exploiting the potential of insects for in vivo pathogenicity testing of microbial pathogens. FEMS Microbiol Rev 28(1):101–112
Peleg AY, Monga D, Pillai S, Mylonakis E, Moellering RC Jr, Eliopoulos GM (2009) Reduced susceptibility to vancomycin influences pathogenicity in Staphylococcus aureus infection. J Infect Dis 199(4):532–536
Wiesner J, Vilcinskas A (2010) Antimicrobial peptides: the ancient arm of the human immune system. Virulence 1(5):440–464
Limmer S, Quintin J, Hetru C, Ferrandon D (2011) Virulence on the fly: Drosophila melanogaster as a model genetic organism to decipher host-pathogen interactions. Curr Drug Targets 12(7):978–999
Chamilos G, Samonis G, Kontoyiannis DP (2011) Drosophila melanogaster as a model host for the study of microbial pathogenicity and the discovery of novel antimicrobial compounds. Curr Pharm Des 17(13):1246–1253
Glavis-Bloom J, Muhammed M, Mylonakis E (2012) Of model hosts and man: using Caenorhabditis elegans, Drosophila melanogaster and Galleria mellonella as model hosts for infectious disease research. Adv Exp Med Biol 710:11–17
Mylonakis E, Casadevall A, Ausubel FM (2007) Exploiting amoeboid and non-vertebrate animal model systems to study the virulence of human pathogenic fungi. PLoS Pathog 3(7):e101
Desbois AP, Coote PJ (2012) Utility of greater wax moth larva (Galleria mellonella) for evaluating the toxicity and efficacy of new antimicrobial agents. Adv Appl Microbiol 78:25–53
Cotter G, Doyle S, Kavanagh K (2000) Development of an insect model for the in vivo pathogenicity testing of yeasts. FEMS Immunol Med Microbiol 27(2):163–169
Junqueira JC (2012) Galleria mellonella as a model host for human pathogens: Recent studies and new perspectives. Virulence 3(6):474–476
Fuchs BB, O’Brien E, Khoury JB, Mylonakis E (2010) Methods for using Galleria mellonella as a model host to study fungal pathogenesis. Virulence 1(6):475–482
Chamilos G, Lionakis MS, Lewis RE, Kontoyiannis DP (2007) Role of mini-host models in the study of medically important fungi. Lancet Infect Dis 7(1):42–55
Peleg AY, Jara S, Monga D, Eliopoulos GM, Moellering RC Jr, Mylonakis E (2009) Galleria mellonella as a model system to study Acinetobacter baumannii pathogenesis and therapeutics. Antimicrob Agents Chemother 53(6):2605–2609
Garcia-Lara J, Needham AJ, Foster SJ (2005) Invertebrates as animal models for Staphylococcus aureus pathogenesis: a window into host-pathogen interaction. FEMS Immunol Med Microbiol 43(3):311–323
Konkel ME, Tilly K (2000) Temperature-regulated expression of bacterial virulence genes. Microbes Infect/Inst Pasteur 2(2):157–166
Smoot LM, Smoot JC, Graham MR, Somerville GA, Sturdevant DE, Migliaccio CA, Sylva GL, Musser JM (2001) Global differential gene expression in response to growth temperature alteration in group A Streptococcus. Proce Natl Acad Sci USA 98(18):10416–10421
Fedhila S, Daou N, Lereclus D, Nielsen-LeRoux C (2006) Identification of Bacillus cereus internalin and other candidate virulence genes specifically induced during oral infection in insects. Mol Microbiol 62(2):339–355
Salamitou S, Ramisse F, Brehelin M, Bourguet D, Gilois N, Gominet M, Hernandez E, Lereclus D (2000) The plcR regulon is involved in the opportunistic properties of Bacillus thuringiensis and Bacillus cereus in mice and insects. Microbiology (Reading, England) 146(Pt 11):2825–2832
Bouillaut L, Ramarao N, Buisson C, Gilois N, Gohar M, Lereclus D, Nielsen-Leroux C (2005) FlhA influences Bacillus thuringiensis PlcR-regulated gene transcription, protein production, and virulence. Appl Environ Microbiol 71(12):8903–8910
Wand ME, Bock LJ, Turton JF, Nugent PG, Sutton JM (2012) Acinetobacter baumannii virulence is enhanced in Galleria mellonella following biofilm adaptation. J Med Microbiol 61(Pt 4):470–477
Antunes LC, Imperi F, Carattoli A, Visca P (2011) Deciphering the multifactorial nature of Acinetobacter baumannii pathogenicity. PloS One 6(8):e22674
Iwashkiw JA, Seper A, Weber BS, Scott NE, Vinogradov E, Stratilo C, Reiz B, Cordwell SJ, Whittal R, Schild S, Feldman MF (2012) Identification of a general O-linked protein glycosylation system in Acinetobacter baumannii and its role in virulence and biofilm formation. PLoS Pathog 8(6):e1002758
Antunes LC, Imperi F, Minandri F, Visca P (2012) In vitro and in vivo antimicrobial activities of gallium nitrate against multidrug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother 56(11):5961–5970
Hornsey M, Wareham DW (2011) In vivo efficacy of glycopeptide-colistin combination therapies in a Galleria mellonella model of Acinetobacter baumannii infection. Antimicrob Agents Chemother 55(7):3534–3537
Gaddy JA, Arivett BA, McConnell MJ, Lopez-Rojas R, Pachon J, Actis LA (2012) Role of acinetobactin-mediated iron acquisition functions in the interaction of Acinetobacter baumannii strain ATCC 19606T with human lung epithelial cells, Galleria mellonella caterpillars, and mice. Infect Immun 80(3):1015–1024
Zimbler DL, Park TM, Arivett BA, Penwell WF, Greer SM, Woodruff TM, Tierney DL, Actis LA (2012) Stress response and virulence functions of the Acinetobacter baumannii NfuA Fe-S scaffold protein. J Bacteriol 194(11):2884–2893
Skiebe E, de Berardinis V, Morczinek P, Kerrinnes T, Faber F, Lepka D, Hammer B, Zimmermann O, Ziesing S, Wichelhaus TA, Hunfeld KP, Borgmann S, Grobner S, Higgins PG, Seifert H, Busse HJ, Witte W, Pfeifer Y, Wilharm G (2012) Surface-associated motility, a common trait of clinical isolates of Acinetobacter baumannii, depends on 1,3-diaminopropane. Int J Med Microbiol 302(3):117–128
Seed KD, Dennis JJ (2008) Development of Galleria mellonella as an alternative infection model for the Burkholderia cepacia complex. Infect Immun 76(3):1267–1275
Mil-Homens D, Fialho AM (2012) A BCAM0223 mutant of Burkholderia cenocepacia is deficient in hemagglutination, serum resistance, adhesion to epithelial cells and virulence. PloS One 7(7):e41747
Thomson EL, Dennis JJ (2012) A Burkholderia cepacia complex non-ribosomal peptide-synthesized toxin is hemolytic and required for full virulence. Virulence 3(3):286–298
Schell MA, Lipscomb L, DeShazer D (2008) Comparative genomics and an insect model rapidly identify novel virulence genes of Burkholderia mallei. J Bacteriol 190(7):2306–2313
Tegos GP, Haynes MK, Schweizer HP (2012) Dissecting novel virulent determinants in the Burkholderia cepacia complex. Virulence 3(3):234–237
Ibrahim M, Tang Q, Shi Y, Almoneafy A, Fang Y, Xu L, Li W, Li B, Xie GL (2012) Diversity of potential pathogenicity and biofilm formation among Burkholderia cepacia complex water, clinical, and agricultural isolates in China. World J Microbiol Biotechnol 28(5):2113–2123
Seed KD, Dennis JJ (2009) Experimental bacteriophage therapy increases survival of Galleria mellonella larvae infected with clinically relevant strains of the Burkholderia cepacia complex. Antimicrob Agents Chemother 53(5):2205–2208
Agnoli K, Schwager S, Uehlinger S, Vergunst A, Viteri DF, Nguyen DT, Sokol PA, Carlier A, Eberl L (2012) Exposing the third chromosome of Burkholderia cepacia complex strains as a virulence plasmid. Mol Microbiol 83(2):362–378
Mil-Homens D, Rocha EP, Fialho AM (2010) Genome-wide analysis of DNA repeats in Burkholderia cenocepacia J2315 identifies a novel adhesin-like gene unique to epidemic-associated strains of the ET-12 lineage. Microbiology (Reading, England) 156(Pt 4):1084–1096
Uehlinger S, Schwager S, Bernier SP, Riedel K, Nguyen DT, Sokol PA, Eberl L (2009) Identification of specific and universal virulence factors in Burkholderia cenocepacia strains by using multiple infection hosts. Infect Immun 77(9):4102–4110
Champion OL, Karlyshev AV, Senior NJ, Woodward M, La Ragione R, Howard SL, Wren BW, Titball RW (2010) Insect infection model for Campylobacter jejuni reveals that O-methyl phosphoramidate has insecticidal activity. J Infect Dis 201(5):776–782
Wand ME, Muller CM, Titball RW, Michell SL (2011) Macrophage and Galleria mellonella infection models reflect the virulence of naturally occurring isolates of B. pseudomallei, B. thailandensis and B. oklahomensis. BMC Microbiol 11(1):11
Silva IN, Ferreira AS, Becker JD, Zlosnik JE, Speert DP, He J, Mil-Homens D, Moreira LM (2011) Mucoid morphotype variation of Burkholderia multivorans during chronic cystic fibrosis lung infection is correlated with changes in metabolism, motility, biofilm formation and virulence. Microbiology (Reading, England) 157(Pt 11):3124–3137
Vial L, Groleau MC, Lamarche MG, Filion G, Castonguay-Vanier J, Dekimpe V, Daigle F, Charette SJ, Deziel E (2010) Phase variation has a role in Burkholderia ambifaria niche adaptation. ISME J 4(1):49–60
Muller CM, Conejero L, Spink N, Wand ME, Bancroft GJ, Titball RW (2012) Role of RelA and SpoT in Burkholderia pseudomallei virulence and immunity. Infect Immun 80(9):3247–3255
Mil-Homens D, Bernardes N, Fialho AM (2012) The antibacterial properties of docosahexaenoic omega-3 fatty acid against the cystic fibrosis multiresistant pathogen Burkholderia cenocepacia. FEMS Microbiol Lett 328(1):61–69
Fazli M, O’Connell A, Nilsson M, Niehaus K, Dow JM, Givskov M, Ryan RP, Tolker-Nielsen T (2011) The CRP/FNR family protein Bcam1349 is a c-di-GMP effector that regulates biofilm formation in the respiratory pathogen Burkholderia cenocepacia. Mol Microbiol 82(2):327–341
Costello A, Herbert G, Fabunmi L, Schaffer K, Kavanagh KA, Caraher EM, Callaghan M, McClean S (2011) Virulence of an emerging respiratory pathogen, genus Pandoraea, in vivo and its interactions with lung epithelial cells. J Med Microbiol 60(Pt 3):289–299
McLaughlin HP, Xiao Q, Rea RB, Pi H, Casey PG, Darby T, Charbit A, Sleator RD, Joyce SA, Cowart RE, Hill C, Klebba PE, Gahan CG (2012) A putative P-type ATPase required for virulence and resistance to haem toxicity in Listeria monocytogenes. PloS One 7(2):e30928
Fedhila S, Buisson C, Dussurget O, Serror P, Glomski IJ, Liehl P, Lereclus D, Nielsen-LeRoux C (2010) Comparative analysis of the virulence of invertebrate and mammalian pathogenic bacteria in the oral insect infection model Galleria mellonella. J Invertebr Pathol 103(1):24–29
Mukherjee K, Altincicek B, Hain T, Domann E, Vilcinskas A, Chakraborty T (2010) Galleria mellonella as a model system for studying Listeria pathogenesis. Appl Environ Microbiol 76(1):310–317
Joyce SA, Gahan CG (2010) Molecular pathogenesis of Listeria monocytogenes in the alternative model host Galleria mellonella. Microbiology (Reading, England) 156(Pt 11):3456–3468
Seifart Gomes C, Izar B, Pazan F, Mohamed W, Mraheil MA, Mukherjee K, Billion A, Aharonowitz Y, Chakraborty T, Hain T (2011) Universal stress proteins are important for oxidative and acid stress resistance and growth of Listeria monocytogenes EGD-e in vitro and in vivo. PloS One 6(9):e24965
McLaughlin HP, Caly DL, McCarthy Y, Ryan RP, Dow JM (2012) An orphan chemotaxis sensor regulates virulence and antibiotic tolerance in the human pathogen Pseudomonas aeruginosa. PloS One 7(8):e42205
Lore NI, Cigana C, De Fino I, Riva C, Juhas M, Schwager S, Eberl L, Bragonzi A (2012) Cystic fibrosis-niche adaptation of Pseudomonas aeruginosa reduces virulence in multiple infection hosts. PloS One 7(4):e35648
Choi JY, Sifri CD, Goumnerov BC, Rahme LG, Ausubel FM, Calderwood SB (2002) Identification of virulence genes in a pathogenic strain of Pseudomonas aeruginosa by representational difference analysis. J Bacteriol 184(4):952–961
Jander G, Rahme LG, Ausubel FM (2000) Positive correlation between virulence of Pseudomonas aeruginosa mutants in mice and insects. J Bacteriol 182(13):3843–3845
Inglis RF, Gardner A, Cornelis P, Buckling A (2009) Spite and virulence in the bacterium Pseudomonas aeruginosa. Proc Natl Acad Sci USA 106(14):5703–5707
Miyata S, Casey M, Frank DW, Ausubel FM, Drenkard E (2003) Use of the Galleria mellonella caterpillar as a model host to study the role of the type III secretion system in Pseudomonas aeruginosa pathogenesis. Infect Immun 71(5):2404–2413
Champion OL, Cooper IA, James SL, Ford D, Karlyshev A, Wren BW, Duffield M, Oyston PC, Titball RW (2009) Galleria mellonella as an alternative infection model for Yersinia pseudotuberculosis. Microbiology (Reading, England) 155(Pt 5):1516–1522
Strong PC, Hinchliffe SJ, Patrick H, Atkinson S, Champion OL, Wren BW (2011) Identification and characterisation of a novel adhesin Ifp in Yersinia pseudotuberculosis. BMC Microbiol 11:85
Erickson DL, Russell CW, Johnson KL, Hileman T, Stewart RM (2011) PhoP and OxyR transcriptional regulators contribute to Yersinia pestis virulence and survival within Galleria mellonella. Microb Pathog 51(6):389–395
Champion OL, Karlyshev A, Cooper IA, Ford DC, Wren BW, Duffield M, Oyston PC, Titball RW (2011) Yersinia pseudotuberculosis mntH functions in intracellular manganese accumulation, which is essential for virulence and survival in cells expressing functional Nramp1. Microbiology (Reading, England) 157(Pt 4):1115–1122
Senior NJ, Bagnall MC, Champion OL, Reynolds SE, La Ragione RM, Woodward MJ, Salguero FJ, Titball RW (2011) Galleria mellonella as an infection model for Campylobacter jejuni virulence. J Med Microbiol 60(Pt 5):661–669
Gundogdu O, Mills DC, Elmi A, Martin MJ, Wren BW, Dorrell N (2011) The Campylobacter jejuni transcriptional regulator Cj1556 plays a role in the oxidative and aerobic stress response and is important for bacterial survival in vivo. J Bacteriol 193(16):4238–4249
Dunphy GB, Chadwick JS (1989) Effects of selected carbohydrates and the contribution of the prophenoloxidase cascade system to the adhesion of strains of Pseudomonas aeruginosa and Proteus mirabilis to hemocytes of nonimmune larval Galleria mellonella. Can J Microbiol 35(4):524–527
Chadwick JS, Aston WP, Ricketson JR (1980) Further studies on the effect and role of cobra venom factor on protective immunity in Galleria mellonella: activity in the response against Proteus mirabilis. Dev Comp Immunol 4(2):223–231
Morton DB, Dunphy GB, Chadwick JS (1987) Reactions of hemocytes of immune and non-immune Galleria mellonella larvae to Proteus mirabilis. Dev Comp Immunol 11(1):47–55
Morton DB, Barnett RI, Chadwick JS (1984) Structural alterations to Proteus mirabilis as a result of exposure to haemolymph from the larvae of Galleria mellonella. Microbios 39(157–158):177–185
Leuko S, Raivio TL (2012) Mutations that impact the enteropathogenic Escherichia coli Cpx envelope stress response attenuate virulence in Galleria mellonella. Infect Immun 80(9):3077–3085
Harding CR, Schroeder GN, Reynolds S, Kosta A, Collins JW, Mousnier A, Frankel G (2012) Legionella pneumophila pathogenesis in the Galleria mellonella infection model. Infect Immun 80(8):2780–2790
Lebreton F, Riboulet-Bisson E, Serror P, Sanguinetti M, Posteraro B, Torelli R, Hartke A, Auffray Y, Giard JC (2009) ace, Which encodes an adhesin in Enterococcus faecalis, is regulated by Ers and is involved in virulence. Infect Immun 77(7):2832–2839
de Oliveira NE, Abranches J, Gaca AO, Laport MS, Damaso CR, Bastos Mdo C, Lemos JA, Giambiagi-deMarval M (2011) clpB, a class III heat-shock gene regulated by CtsR, is involved in thermotolerance and virulence of Enterococcus faecalis. Microbiology (Reading, England) 157(Pt 3):656–665
Lebreton F, Le Bras F, Reffuveille F, Ladjouzi R, Giard JC, Leclercq R, Cattoir V (2011) Galleria mellonella as a model for studying Enterococcus faecium host persistence. J Mol Microbiol Biotechnol 21(3–4):191–196
Zhao C, Hartke A, La Sorda M, Posteraro B, Laplace JM, Auffray Y, Sanguinetti M (2010) Role of methionine sulfoxide reductases A and B of Enterococcus faecalis in oxidative stress and virulence. Infect Immun 78(9):3889–3897
Michaux C, Sanguinetti M, Reffuveille F, Auffray Y, Posteraro B, Gilmore MS, Hartke A, Giard JC (2011) SlyA is a transcriptional regulator involved in the virulence of Enterococcus faecalis. Infect Immun 79(7):2638–2645
Yan X, Zhao C, Budin-Verneuil A, Hartke A, Rince A, Gilmore MS, Auffray Y, Pichereau V (2009) The (p)ppGpp synthetase RelA contributes to stress adaptation and virulence in Enterococcus faecalis V583. Microbiology (Reading, England) 155(Pt 10):3226–3237
Gaspar F, Teixeira N, Rigottier-Gois L, Marujo P, Nielsen-LeRoux C, Crespo MT, Lopes Mde F, Serror P (2009) Virulence of Enterococcus faecalis dairy strains in an insect model: the role of fsrB and gelE. Microbiology (Reading, England) 155(Pt 11):3564–3571
Latimer J, Forbes S, McBain AJ (2012) Attenuated virulence and biofilm formation in Staphylococcus aureus following sublethal exposure to triclosan. Antimicrob Agents Chemother 56(6):3092–3100
Purves J, Cockayne A, Moody PC, Morrissey JA (2010) Comparison of the regulation, metabolic functions, and roles in virulence of the glyceraldehyde-3-phosphate dehydrogenase homologues gapA and gapB in Staphylococcus aureus. Infect Immun 78(12):5223–5232
Gao W, Chua K, Davies JK, Newton HJ, Seemann T, Harrison PF, Holmes NE, Rhee HW, Hong JI, Hartland EL, Stinear TP, Howden BP (2010) Two novel point mutations in clinical Staphylococcus aureus reduce linezolid susceptibility and switch on the stringent response to promote persistent infection. PLoS Pathog 6(6):e1000944
Mowlds P, Kavanagh K (2008) Effect of pre-incubation temperature on susceptibility of Galleria mellonella larvae to infection by Candida albicans. Mycopathologia 165(1):5–12
Fallon J, Kelly J, Kavanagh K (2012) Galleria mellonella as a model for fungal pathogenicity testing. Methods Mol Biol 845:469–485
Junqueira JC (2012) Models hosts for the study of oral candidiasis. Adv Exp Med Biol 710:95–105
Mowlds P, Barron A, Kavanagh K (2008) Physical stress primes the immune response of Galleria mellonella larvae to infection by Candida albicans. Microbes Infect/Inst Pasteur 10(6):628–634
Fuchs BB, Eby J, Nobile CJ, El Khoury JB, Mitchell AP, Mylonakis E (2010) Role of filamentation in Galleria mellonella killing by Candida albicans. Microbes and Infect/Inst Pasteur 12(6):488–496
Dunphy GB, Oberholzer U, Whiteway M, Zakarian RJ, Boomer I (2003) Virulence of Candida albicans mutants toward larval Galleria mellonella (Insecta, Lepidoptera, Galleridae). Can J Microbiol 49(8):514–524
Garcia-Rodas R, Casadevall A, Rodriguez-Tudela JL, Cuenca-Estrella M, Zaragoza O (2011) Cryptococcus neoformans capsular enlargement and cellular gigantism during Galleria mellonella infection. PloS One 6(9):e24485
Mylonakis E (2008) Galleria mellonella and the study of fungal pathogenesis: making the case for another genetically tractable model host. Mycopathologia 165(1):1–3
Mylonakis E, Moreno R, El Khoury JB, Idnurm A, Heitman J, Calderwood SB, Ausubel FM, Diener A (2005) Galleria mellonella as a model system to study Cryptococcus neoformans pathogenesis. Infect Immun 73(7):3842–3850
London R, Orozco BS, Mylonakis E (2006) The pursuit of cryptococcal pathogenesis: heterologous hosts and the study of cryptococcal host-pathogen interactions. FEMS Yeast Res 6(4):567–573
Fuchs BB, Mylonakis E (2006) Using non-mammalian hosts to study fungal virulence and host defense. Curr Opin Microbiol 9(4):346–351
Fallon JP, Troy N, Kavanagh K (2011) Pre-exposure of Galleria mellonella larvae to different doses of Aspergillus fumigatus conidia causes differential activation of cellular and humoral immune responses. Virulence 2(5):413–421
Reeves EP, Reiber K, Neville C, Scheibner O, Kavanagh K, Doyle S (2006) A nonribosomal peptide synthetase (Pes1) confers protection against oxidative stress in Aspergillus fumigatus. FEBS J 273(13):3038–3053
Scully LR, Bidochka MJ (2009) An alternative insect pathogenic strategy in an Aspergillus flavus auxotroph. Mycol Res 113(Pt 2):230–239
Jackson JC, Higgins LA, Lin X (2009) Conidiation color mutants of Aspergillus fumigatus are highly pathogenic to the heterologous insect host Galleria mellonella. PloS One 4(1):e4224
Reeves EP, Messina CG, Doyle S, Kavanagh K (2004) Correlation between gliotoxin production and virulence of Aspergillus fumigatus in Galleria mellonella. Mycopathologia 158(1):73–79
Soukup AA, Farnoodian M, Berthier E, Keller NP (2012) NosA, a transcription factor important in Aspergillus fumigatus stress and developmental response, rescues the germination defect of a laeA deletion. Fungal Genet Biol 49(11):857–865
Slater JL, Gregson L, Denning DW, Warn PA (2011) Pathogenicity of Aspergillus fumigatus mutants assessed in Galleria mellonella matches that in mice. Med Mycol 49(Suppl 1):S107–S113
Christians JK, Cheema MS, Vergara IA, Watt CA, Pinto LJ, Chen N, Moore MM (2011) Quantitative trait locus (QTL) mapping reveals a role for unstudied genes in Aspergillus virulence. PloS One 6(4):e19325
Renwick J, Daly P, Reeves EP, Kavanagh K (2006) Susceptibility of larvae of Galleria mellonella to infection by Aspergillus fumigatus is dependent upon stage of conidial germination. Mycopathologia 161(6):377–384
Fallon JP, Reeves EP, Kavanagh K (2011) The Aspergillus fumigatus toxin fumagillin suppresses the immune response of Galleria mellonella larvae by inhibiting the action of haemocytes. Microbiology (Reading, England) 157(Pt 5):1481–1488
Cheema MS, Christians JK (2011) Virulence in an insect model differs between mating types in Aspergillus fumigatus. Med Mycol 49(2):202–207
Mukherjee K, Abu Mraheil M, Silva S, Muller D, Cemic F, Hemberger J, Hain T, Vilcinskas A, Chakraborty T (2011) Anti-Listeria activities of Galleria mellonella hemolymph proteins. Appl Environ Microbiol 77(12):4237–4240
Brennan M, Thomas DY, Whiteway M, Kavanagh K (2002) Correlation between virulence of Candida albicans mutants in mice and Galleria mellonella larvae. FEMS Immunol Medl Microbiol 34(2):153–157
Olsen RJ, Watkins ME, Cantu CC, Beres SB, Musser JM (2011) Virulence of serotype M3 Group A Streptococcus strains in wax worms (Galleria mellonella larvae). Virulence 2(2):111–119
Fuchs BB, Bishop LR, Kovacs JA, Mylonakis E (2011) Galleria mellonella are resistant to Pneumocystis murina infection. Mycopathologia 171(4):273–277
Achterman RR, Smith AR, Oliver BG, White TC (2011) Sequenced dermatophyte strains: growth rate, conidiation, drug susceptibilities, and virulence in an invertebrate model. Fungal Genet Biol 48(3):335–341
Aperis G, Fuchs BB, Anderson CA, Warner JE, Calderwood SB, Mylonakis E (2007) Galleria mellonella as a model host to study infection by the Francisella tularensis live vaccine strain. Microbes Infect/Inst Pasteur 9(6):729–734
Bergin D, Brennan M, Kavanagh K (2003) Fluctuations in haemocyte density and microbial load may be used as indicators of fungal pathogenicity in larvae of Galleria mellonella. Microbes Infect/Inst Pasteur 5(15):1389–1395
Bergin D, Murphy L, Keenan J, Clynes M, Kavanagh K (2006) Pre-exposure to yeast protects larvae of Galleria mellonella from a subsequent lethal infection by Candida albicans and is mediated by the increased expression of antimicrobial peptides. Microbes Infect/Inst Pasteur 8(8):2105–2112
Altincicek B, Linder M, Linder D, Preissner KT, Vilcinskas A (2007) Microbial metalloproteinases mediate sensing of invading pathogens and activate innate immune responses in the lepidopteran model host Galleria mellonella. Infect Immun 75(1):175–183
Bergin D, Reeves EP, Renwick J, Wientjes FB, Kavanagh K (2005) Superoxide production in Galleria mellonella hemocytes: identification of proteins homologous to the NADPH oxidase complex of human neutrophils. Infect Immun 73(7):4161–4170
Vogel H, Altincicek B, Glockner G, Vilcinskas A (2011) A comprehensive transcriptome and immune-gene repertoire of the lepidopteran model host Galleria mellonella. BMC Genom 12:308
Brown SE, Howard A, Kasprzak AB, Gordon KH, East PD (2009) A peptidomics study reveals the impressive antimicrobial peptide arsenal of the wax moth Galleria mellonella. Insect Biochem Mol Biol 39(11):792–800
Lionakis MS (2011) Drosophila and Galleria insect model hosts: new tools for the study of fungal virulence, pharmacology and immunology. Virulence 2(6):521–527
Chamilos G, Lionakis MS, Lewis RE, Lopez-Ribot JL, Saville SP, Albert ND, Halder G, Kontoyiannis DP (2006) Drosophila melanogaster as a facile model for large-scale studies of virulence mechanisms and antifungal drug efficacy in Candida species. J Infect Dis 193(7):1014–1022
Lionakis MS, Kontoyiannis DP (2012) Drosophila melanogaster as a model organism for invasive aspergillosis. Methods Mol Biol 845:455–468
Levitin A, Marcil A, Tettweiler G, Laforest MJ, Oberholzer U, Alarco AM, Thomas DY, Lasko P, Whiteway M (2007) Drosophila melanogaster thor and response to Candida albicans infection. Eukaryot Cell 6(4):658–663
Alarco AM, Marcil A, Chen J, Suter B, Thomas D, Whiteway M (2004) Immune-deficient Drosophila melanogaster: a model for the innate immune response to human fungal pathogens. J Immunol 172(9):5622–5628
Lionakis MS, Kontoyiannis DP (2010) The growing promise of Toll-deficient Drosophila melanogaster as a model for studying Aspergillus pathogenesis and treatment. Virulence 1(6):488–499
Lionakis MS, Lewis RE, May GS, Wiederhold NP, Albert ND, Halder G, Kontoyiannis DP (2005) Toll-deficient Drosophila flies as a fast, high-throughput model for the study of antifungal drug efficacy against invasive aspergillosis and Aspergillus virulence. J Infect Dis 191(7):1188–1195
Glittenberg MT, Silas S, MacCallum DM, Gow NA, Ligoxygakis P (2011) Wild-type Drosophila melanogaster as an alternative model system for investigating the pathogenicity of Candida albicans. Dis Model Mech 4(4):504–514
Apidianakis Y, Rahme LG (2009) Drosophila melanogaster as a model host for studying Pseudomonas aeruginosa infection. Nat Protoc 4(9):1285–1294
Mulcahy H, Sibley CD, Surette MG, Lewenza S (2011) Drosophila melanogaster as an animal model for the study of Pseudomonas aeruginosa biofilm infections in vivo. PLoS Pathog 7(10):e1002299
Kim SH, Park SY, Heo YJ, Cho YH (2008) Drosophila melanogaster-based screening for multihost virulence factors of Pseudomonas aeruginosa PA14 and identification of a virulence-attenuating factor, HudA. Infect Immun 76(9):4152–4162
Lutter EI, Purighalla S, Duong J, Storey DG (2012) Lethality and cooperation of Pseudomonas aeruginosa quorum-sensing mutants in Drosophila melanogaster infection models. Microbiology (Reading, England) 158(Pt 8):2125–2132
Lutter EI, Faria MM, Rabin HR, Storey DG (2008) Pseudomonas aeruginosa cystic fibrosis isolates from individual patients demonstrate a range of levels of lethality in two Drosophila melanogaster infection models. Infect Immun 76(5):1877–1888
Limmer S, Haller S, Drenkard E, Lee J, Yu S, Kocks C, Ausubel FM, Ferrandon D (2011) Pseudomonas aeruginosa RhlR is required to neutralize the cellular immune response in a Drosophila melanogaster oral infection model. Proc Natl Acad Scin USA 108(42):17378–17383
Jensen RL, Pedersen KS, Loeschcke V, Ingmer H, Leisner JJ (2007) Limitations in the use of Drosophila melanogaster as a model host for gram-positive bacterial infection. Lett Appl Microbiol 44(2):218–223
Linderman JA, Chambers MC, Gupta AS, Schneider DS (2012) Infection-Related Declines in Chill Coma Recovery and Negative Geotaxis in Drosophila melanogaster. PloS One 7(9):e41907
Nehme NT, Liegeois S, Kele B, Giammarinaro P, Pradel E, Hoffmann JA, Ewbank JJ, Ferrandon D (2007) A model of bacterial intestinal infections in Drosophila melanogaster. PLoS Pathog 3(11):e173
Mueller JL, Page JL, Wolfner MF (2007) An ectopic expression screen reveals the protective and toxic effects of Drosophila seminal fluid proteins. Genetics 175(2):777–783
Valtonen TM, Roff DA, Rantala MJ (2011) Analysis of the effects of early nutritional environment on inbreeding depression in Drosophila melanogaster. J Evol Biol 24(1):196–205
Thomas P, Yamada R, Johnson KN, Asgari S (2010) Ectopic expression of an endoparasitic wasp venom protein in Drosophila melanogaster affects immune function, larval development and oviposition. Insect Mol Biol 19(4):473–480
Kallio J, Myllymaki H, Gronholm J, Armstrong M, Vanha-aho LM, Makinen L, Silvennoinen O, Valanne S, Ramet M (2010) Eye transformer is a negative regulator of Drosophila JAK/STAT signaling. FASEB J 24(11):4467–4479
Kuraishi T, Binggeli O, Opota O, Buchon N, Lemaitre B (2011) Genetic evidence for a protective role of the peritrophic matrix against intestinal bacterial infection in Drosophila melanogaster. Proc Natl Acad Sci USA 108(38):15966–15971
Lazzaro BP, Sackton TB, Clark AG (2006) Genetic variation in Drosophila melanogaster resistance to infection: a comparison across bacteria. Genetics 174(3):1539–1554
Cronin SJ, Nehme NT, Limmer S, Liegeois S, Pospisilik JA, Schramek D, Leibbrandt A, Simoes Rde M, Gruber S, Puc U, Ebersberger I, Zoranovic T, Neely GG, von Haeseler A, Ferrandon D, Penninger JM (2009) Genome-wide RNAi screen identifies genes involved in intestinal pathogenic bacterial infection. Science 325(5938):340–343
Sackton TB, Lazzaro BP, Clark AG (2010) Genotype and gene expression associations with immune function in Drosophila. PLoS Genet 6(1):e1000797
Chang HJ, Dhanasingh I, Gou X, Rice AM, Dushay MS (2012) Loss of Hemolectin reduces the survival of Drosophila larvae after wounding. Dev Comp Immunol 36(2):274–278
Imroze K, Prasad NG (2011) Mating with large males decreases the immune defence of females in Drosophila melanogaster. J Genet 90(3):427–434
Miest TS, Bloch-Qazi M (2008) Sick of mating: sexual transmission of a pathogenic bacterium in Drosophila melanogaster. Fly (Austin) 2(4):215–219
Khan I, Prasad NG (2013) The aging of the immune response in Drosophila melanogaster. J Gerontol A Biol Sci Med Sci 68(2):129–135
Valtonen TM, Kangassalo K, Polkki M, Rantala MJ (2012) Transgenerational effects of parental larval diet on offspring development time, adult body size and pathogen resistance in Drosophila melanogaster. PloS One 7(2):e31611
Wong ZS, Hedges LM, Brownlie JC, Johnson KN (2011) Wolbachia-mediated antibacterial protection and immune gene regulation in Drosophila. PloS One 6(9):e25430
Jensen A, Thomsen LE, Jorgensen RL, Larsen MH, Roldgaard BB, Christensen BB, Vogel BF, Gram L, Ingmer H (2008) Processing plant persistent strains of Listeria monocytogenes appear to have a lower virulence potential than clinical strains in selected virulence models. Int J Food Microbiol 123(3):254–261
Chamilos G, Nobile CJ, Bruno VM, Lewis RE, Mitchell AP, Kontoyiannis DP (2009) Candida albicans Cas5, a regulator of cell wall integrity, is required for virulence in murine and toll mutant fly models. J Infect Dis 200(1):152–157
Lohse MB, Johnson AD (2008) Differential phagocytosis of white versus opaque Candida albicans by Drosophila and mouse phagocytes. PloS One 3(1):e1473
Davis MM, Alvarez FJ, Ryman K, Holm AA, Ljungdahl PO, Engstrom Y (2011) Wild-type Drosophila melanogaster as a model host to analyze nitrogen source dependent virulence of Candida albicans. PloS One 6(11):e27434
Apidianakis Y, Rahme LG, Heitman J, Ausubel FM, Calderwood SB, Mylonakis E (2004) Challenge of Drosophila melanogaster with Cryptococcus neoformans and role of the innate immune response. Eukaryot Cell 3(2):413–419
Netea MG, Van der Graaf C, Van der Meer JW, Kullberg BJ (2004) Recognition of fungal pathogens by Toll-like receptors. Eur J Clin Microbiol Infect Dis 23(9):672–676
Chamilos G, Bignell EM, Schrettl M, Lewis RE, Leventakos K, May GS, Haas H, Kontoyiannis DP (2010) Exploring the concordance of Aspergillus fumigatus pathogenicity in mice and Toll-deficient flies. Med Mycol 48(3):506–510
Ha EM, Oh CT, Bae YS, Lee WJ (2005) A direct role for dual oxidase in Drosophila gut immunity. Science 310(5749):847–850
Tzou P, Ohresser S, Ferrandon D, Capovilla M, Reichhart JM, Lemaitre B, Hoffmann JA, Imler JL (2000) Tissue-specific inducible expression of antimicrobial peptide genes in Drosophila surface epithelia. Immunity 13(5):737–748
Apidianakis Y, Rahme LG (2011) Drosophila melanogaster as a model for human intestinal infection and pathology. Dis Model Mech 4(1):21–30
Chamilos G, Lewis RE, Hu J, Xiao L, Zal T, Gilliet M, Halder G, Kontoyiannis DP (2008) Drosophila melanogaster as a model host to dissect the immunopathogenesis of zygomycosis. Proc Natl Acad Sci USA 105(27):9367–9372
Qin QM, Luo J, Lin X, Pei J, Li L, Ficht TA, de Figueiredo P (2011) Functional analysis of host factors that mediate the intracellular lifestyle of Cryptococcus neoformans. PLoS Pathog 7(6):e1002078
Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA (1996) The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 86(6):973–983
Hoffmann JA (2003) The immune response of Drosophila. Nature 426(6962):33–38
Lionakis MS, Lim JK, Lee CC, Murphy PM (2011) Organ-specific innate immune responses in a mouse model of invasive candidiasis. J Innate Immun 3(2):180–199
Altincicek B, Knorr E, Vilcinskas A (2008) Beetle immunity: Identification of immune-inducible genes from the model insect Tribolium castaneum. Dev Comp Immunol 32(5):585–595
Berghammer AJ, Klingler M, Wimmer EA (1999) A universal marker for transgenic insects. Nature 402(6760):370–371
Bucher G, Scholten J, Klingler M (2002) Parental RNAi in Tribolium (Coleoptera). Curr Biol 12(3):R85–R86
Lorenzen MD, Kimzey T, Shippy TD, Brown SJ, Denell RE, Beeman RW (2007) piggyBac-based insertional mutagenesis in Tribolium castaneum using donor/helper hybrids. Insect Mol Biol 16(3):265–275
Lorenzen MD, Berghammer AJ, Brown SJ, Denell RE, Klingler M, Beeman RW (2003) piggyBac-mediated germline transformation in the beetle Tribolium castaneum. Insect Mol Biol 12(5):433–440
Pavlopoulos A, Berghammer AJ, Averof M, Klingler M (2004) Efficient transformation of the beetle Tribolium castaneum using the Minos transposable element: quantitative and qualitative analysis of genomic integration events. Genetics 167(2):737–746
Tomoyasu Y, Denell RE (2004) Larval RNAi in Tribolium (Coleoptera) for analyzing adult development. Dev Genes Evol 214(11):575–578
Wimmer EA (2003) Innovations: applications of insect transgenesis. Nat Rev Genet 4(3):225–232
Tamang MD, Kim S, Kim SM, Kong HH, Kim J (2011) Interaction of Acinetobacter baumannii 19606 and 1656–2 with Acanthamoeba castellanii. J Microbiol 49(5):841–846
Silver AC, Williams D, Faucher J, Horneman AJ, Gogarten JP, Graf J (2011) Complex evolutionary history of the Aeromonas veronii group revealed by host interaction and DNA sequence data. PloS One 6(2):e16751
Castonguay-Vanier J, Vial L, Tremblay J, Deziel E (2010) Drosophila melanogaster as a model host for the Burkholderia cepacia complex. PloS One 5(7):e11467
Bare J, Sabbe K, Huws S, Vercauteren D, Braeckmans K, van Gremberghe I, Favoreel H, Houf K (2010) Influence of temperature, oxygen and bacterial strain identity on the association of Campylobacter jejuni with Acanthamoeba castellanii. FEMS Microbiol Ecol 74(2):371–381
Bui XT, Qvortrup K, Wolff A, Bang DD, Creuzenet C (2012) Effect of environmental stress factors on the uptake and survival of Campylobacter jejuni in Acanthamoeba castellanii. BMC Microbiol 12(1):232
Bui XT, Winding A, Qvortrup K, Wolff A, Bang DD, Creuzenet C (2012) Survival of Campylobacter jejuni in co-culture with Acanthamoeba castellanii: role of amoeba-mediated depletion of dissolved oxygen. Environ Microbiol 14(8):2034–2047
Ahlund MK, Ryden P, Sjostedt A, Stoven S (2010) Directed screen of Francisella novicida virulence determinants using Drosophila melanogaster. Infect Immun 78(7):3118–3128
Saeed A, Johansson D, Sandstrom G, Abd H (2012) Temperature depended role of Shigella flexneri invasion plasmid on the interaction with Acanthamoeba castellanii. Int J Microbiol 2012:917031
El-Etr SH, Margolis JJ, Monack D, Robison RA, Cohen M, Moore E, Rasley A (2009) Francisella tularensis type A strains cause the rapid encystment of Acanthamoeba castellanii and survive in amoebal cysts for three weeks postinfection. Appl Environ Microbiol 75(23):7488–7500
Zhou X, Elmose J, Call DR (2007) Interactions between the environmental pathogen Listeria monocytogenes and a free-living protozoan (Acanthamoeba castellanii). Environ Microbiol 9(4):913–922
Chieda Y, Iiyama K, Lee JM, Kusakabe T, Yasunaga-Aoki C, Shimizu S (2011) Virulence of an exotoxin A-deficient strain of Pseudomonas aeruginosa toward the silkworm, Bombyx mori. Microbial Pathog 51(6):407–414
Bradbury RS, Reid DW, Inglis TJ, Champion AC (2011) Decreased virulence of cystic fibrosis Pseudomonas aeruginosa in Dictyostelium discoideum. Microbiol Immunol 55(4):224–230
Nicoletti M, Iacobino A, Prosseda G, Fiscarelli E, Zarrilli R, De Carolis E, Petrucca A, Nencioni L, Colonna B, Casalino M (2011) Stenotrophomonas maltophilia strains from cystic fibrosis patients: genomic variability and molecular characterization of some virulence determinants. Int J Med Microbiol 301(1):34–43
Hamamoto H, Kurokawa K, Kaito C, Kamura K, Manitra Razanajatovo I, Kusuhara H, Santa T, Sekimizu K (2004) Quantitative evaluation of the therapeutic effects of antibiotics using silkworms infected with human pathogenic microorganisms. Antimicrob Agents Chemother 48(3):774–779
An D, Apidianakis Y, Boechat AL, Baldini RL, Goumnerov BC, Rahme LG (2009) The pathogenic properties of a novel and conserved gene product, KerV, in proteobacteria. PloS One 4(9):e7167
Vlahou G, Schmidt O, Wagner B, Uenlue H, Dersch P, Rivero F, Weissenmayer BA (2009) Yersinia outer protein YopE affects the actin cytoskeleton in Dictyostelium discoideum through targeting of multiple Rho family GTPases. BMC microbiology 9:138
Guichard A, McGillivray SM, Cruz-Moreno B, van Sorge NM, Nizet V, Bier E (2010) Anthrax toxins cooperatively inhibit endocytic recycling by the Rab11/Sec15 exocyst. Nature 467(7317):854–858
Dey R, Hoffman PS, Glomski IJ (2012) Germination and amplification of anthrax spores by soil-dwelling amoebas. Appl Environ Microbiol 78(22):8075–8081
Fournier D, Berge JB, Cardoso de Almeida ML, Bordier C (1988) Acetylcholinesterases from Musca domestica and Drosophila melanogaster brain are linked to membranes by a glycophospholipid anchor sensitive to an endogenous phospholipase. J Neurochem 50(4):1158–1163
Alseth I, Rognes T, Lindback T, Solberg I, Robertsen K, Kristiansen KI, Mainieri D, Lillehagen L, Kolsto AB, Bjoras M (2006) A new protein superfamily includes two novel 3-methyladenine DNA glycosylases from Bacillus cereus, AlkC and AlkD. Mol Microbiol 59(5):1602–1609
Pandiarajan J, Cathrin BP, Pratheep T, Krishnan M (2011) Defense role of the cocoon in the silk worm Bombyx mori L. Rapid Commun Mass Spectrom 25(21):3203–3206
Huang L, Cheng T, Xu P, Cheng D, Fang T, Xia Q (2009) A genome-wide survey for host response of silkworm, Bombyx mori during pathogen Bacillus bombyseptieus infection. PloS One 4(12):e8098
Stenfors Arnesen L, Granum PE, Buisson C, Bohlin J, Nielsen-LeRoux C (2011) Using an insect model to assess correlation between temperature and virulence in Bacillus weihenstephanensis and Bacillus cereus. FEMS Microbiol Lett 317(2):196–202
Cox CR, Gilmore MS (2007) Native microbial colonization of Drosophila melanogaster and its use as a model of Enterococcus faecalis pathogenesis. Infect Immun 75(4):1565–1576
Channaiah LH, Subramanyam B, Zurek L (2010) Survival of Enterococcus faecalis OG1RF:pCF10 in poultry and cattle feed: vector competence of the red flour beetle, Tribolium castaneum (Herbst). J Food Prot 73(3):568–573
Wang Z, Flax LA, Kemp MM, Linhardt RJ, Baron MJ (2011) Host and pathogen glycosaminoglycan-binding proteins modulate antimicrobial peptide responses in Drosophila melanogaster. Infect Immun 79(2):606–616
Shamim M, Baig M, Nataraju B, Datta RK, Gupta SK (1995) Evaluation of protein-A linked monoclonal antibody latex agglutination test for diagnosis of nuclear polyhedrosis virus (BmNPV) of silkworm Bombyx mori L. J Immunoass 16(2):155–166
Abranches J, Miller JH, Martinez AR, Simpson-Haidaris PJ, Burne RA, Lemos JA (2011) The collagen-binding protein Cnm is required for Streptococcus mutans adherence to and intracellular invasion of human coronary artery endothelial cells. Infect Immun 79(6):2277–2284
Shiratsuchi A, Mori T, Sakurai K, Nagaosa K, Sekimizu K, Lee BL, Nakanishi Y (2012) Independent recognition of Staphylococcus aureus by two receptors for phagocytosis in Drosophila. J Biol Chem 287(26):21663–21672
Miyazaki S, Matsumoto Y, Sekimizu K, Kaito C (2012) Evaluation of Staphylococcus aureus virulence factors using a silkworm model. FEMS Microbiol Lett 326(2):116–124
Hobson RP (2000) The effects of diffusates from the spores of Aspergillus fumigatus and A. terreus on human neutrophils, Naegleria gruberi and Acanthamoeba castellanii. Med Mycol 38(2):133–141
Hanaoka N, Takano Y, Shibuya K, Fugo H, Uehara Y, Niimi M (2008) Identification of the putative protein phosphatase gene PTC1 as a virulence-related gene using a silkworm model of Candida albicans infection. Eukaryot Cell 7(10):1640–1648
Matsumoto Y, Miyazaki S, Fukunaga DH, Shimizu K, Kawamoto S, Sekimizu K (2012) Quantitative evaluation of cryptococcal pathogenesis and antifungal drugs using a silkworm infection model with Cryptococcus neoformans. J Appl Microbiol 112(1):138–146
Evans SE, Leventakos K, Ben-Ami R, You D, Thakkar SG, Lewis RE, Kontoyiannis DP (2010) Toll-deficient Drosophila are resistant to infection by Pneumocystis spp.: additional evidence of specificity to mammalian hosts. Virulence 1(6):523–525
Belmatoug N, Fantin B (1997) Contribution of animal models of infection for the evaluation of the activity of antimicrobial agents. Int J Antimicrob Agents 9(2):73–82
Wilson-Sanders SE (2011) Invertebrate models for biomedical research, testing, and education. ILAR J Natl Res Counc, Inst Lab Anim Resour 52(2):126–152
Desbois AP, Coote PJ (2011) Wax moth larva (Galleria mellonella): an in vivo model for assessing the efficacy of antistaphylococcal agents. J Antimicrob Chemother 66(8):1785–1790
Spitzer M, Griffiths E, Blakely KM, Wildenhain J, Ejim L, Rossi L, De Pascale G, Curak J, Brown E, Tyers M, Wright GD (2011) Cross-species discovery of syncretic drug combinations that potentiate the antifungal fluconazole. Mol Syst Biol 7:499
Vu K, Gelli A (2010) Astemizole and an analogue promote fungicidal activity of fluconazole against Cryptococcus neoformans var. grubii and Cryptococcus gattii. Med Mycol 48(2):255–262
Cowen LE, Singh SD, Kohler JR, Collins C, Zaas AK, Schell WA, Aziz H, Mylonakis E, Perfect JR, Whitesell L, Lindquist S (2009) Harnessing Hsp90 function as a powerful, broadly effective therapeutic strategy for fungal infectious disease. Proc Natl Acad Sci USA 106(8):2818–2823
Mowlds P, Coates C, Renwick J, Kavanagh K (2010) Dose-dependent cellular and humoral responses in Galleria mellonella larvae following beta-glucan inoculation. Microbes Infect/Inst Pasteur 12(2):146–153
Desalermos A, Muhammed M, Glavis-Bloom J, Mylonakis E (2011) Using C. elegans for antimicrobial drug discovery. Expert Opin Drug Discov 6(6):645–652
Brackman G, Cos P, Maes L, Nelis HJ, Coenye T (2011) Quorum sensing inhibitors increase the susceptibility of bacterial biofilms to antibiotics in vitro and in vivo. Antimicrob Agents Chemother 55(6):2655–2661
Ahmad S, Hunter L, Qin A, Mann BJ, van Hoek ML (2010) Azithromycin effectiveness against intracellular infections of Francisella. BMC Microbiol 10:123
Barman TK, Arora P, Rao M, Bhadauriya T, Upadhyay DJ (2008) Utilization of Bombyx mori larvae as a surrogate animal model for evaluation of the anti-infective potential of oxazolidinones. J Infect Chemother 14(2):166–169
Rowan R, Moran C, McCann M, Kavanagh K (2009) Use of Galleria mellonella larvae to evaluate the in vivo anti-fungal activity of [Ag2(mal)(phen)3]. Biometals 22(3):461–467
Kelly J, Kavanagh K (2011) Caspofungin primes the immune response of the larvae of Galleria mellonella and induces a non-specific antimicrobial response. J Med Microbiol 60(Pt 2):189–196
Acknowledgments
This work was supported by the National Institutes of Health through an R01 award (AI075286) and an R21 award (AI070569) to Eleftherios Mylonakis, and National Natural Science Foundation of China (81273558, 81072678) to Yan Wang.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Wang, Y., Li, DD., Jiang, YY., Mylonakis, E. (2013). Utility of Insects for Studying Human Pathogens and Evaluating New Antimicrobial Agents. In: Vilcinskas, A. (eds) Yellow Biotechnology I. Advances in Biochemical Engineering/Biotechnology, vol 135. Springer, Berlin, Heidelberg. https://doi.org/10.1007/10_2013_194
Download citation
DOI: https://doi.org/10.1007/10_2013_194
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-39862-9
Online ISBN: 978-3-642-39863-6
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)