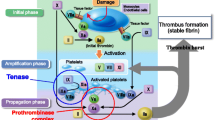
Abstract
Utilizing the previously discussed in vitro and in vivo models for the evaluation of new anticoagulants resulted in recent advances in the development of novel oral anticoagulants (NOA). Major advances in the development of NOA progressed well, with the goal of developing safe and effective oral anticoagulants that do not require frequent monitoring or dose adjustment along with minimal food/drug interactions. Indirect inhibitors such as low-molecular-weight heparin (LMWH) and the pentasaccharide fondaparinux represent improvements over traditional drugs such as unfractionated heparin for acute treatment of venous thromboembolism (VTE) with more targeted anticoagulant approaches, predictable pharmacokinetic profiles, and lack of need for monitoring. Vitamin K antagonist, with its inherent limitations of multiple food and drug interactions and frequent need for monitoring, remains the only oral anticoagulant approved for long-term secondary thromboprophylaxis in VTE. The oral direct thrombin inhibitor ximelagatran was withdrawn from the world market due to safety concerns. Newer anticoagulant drugs such as injectables (fondaparinux, idraparinux), oral direct thrombin inhibitors (dabigatran), oral direct factor Xa inhibitors (rivaroxaban, apixaban, and others), and tissue factor/factor VIIa complex inhibitors are “tailor-made” to target specific procoagulant complexes and have the potential to greatly expand oral antithrombotic targets for both acute and long-term treatment of VTE, acute coronary syndromes, and prevention of stroke in atrial fibrillation patients.
Similar content being viewed by others
References and Further Reading
Recent Advances in Novel Oral Anticoagulants (NOAs)
Birman-Deych E, Radford MJ, Nilasena DS, Gage BF (2006) Use and effectiveness of warfarin in Medicare beneficiaries with atrial fibrillation. Stroke 37:1070–1074
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, Pogue J, Reilly PA, Themeles E, Varrone J, Wang S, Alings M, Xavier D, Zhu J, Diaz R, Lewis BS, Darius H, Diener HC, Joyner CD, Wallentin L, RE-LY Steering Committee and Investigators (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361:1139–1151
Granger CB, Alexander JH, McMurray JJ, Lopes RD, Hylek EM, Hanna M, Al-Khalidi HR, Ansell J, Atar D, Avezum A, Bahit MC, Diaz R, Easton JD, Ezekowitz JA, Flaker G, Garcia D, Geraldes M, Gersh BJ, Golitsyn S, Goto S, Hermosillo AG, Hohnloser SH, Horowitz J, Mohan P, Jansky P, Lewis BS, Lopez-Sendon JL, Pais P, Parkhomenko A, Verheugt FW, Zhu J, Wallentin L, ARISTOTLE Committees and Investigators (2011) Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 365:981–992
Hylek EM, Evans-Molina C, Shea C, Henault LE, Regan S (2007) Major hemorrhage and tolerability of warfarin in the first year of therapy among elderly patients with atrial fibrillation. Circulation 115:2689–2696
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, Breithardt G, Halperin JL, Hankey GJ, Piccini JP, Becker RC, Nessel CC, Paolini JF, Berkowitz SD, Fox KA, Califf RM, ROCKET AF Investigators (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365:883–891
Dabigatran
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, Pogue J, Reilly PA, Themeles E, Varrone J, Wang S, Alings M, Xavier D, Zhu J, Diaz R, Lewis BS, Darius H, Diener HC, Joyner CD, Wallentin L, RE-LY Steering Committee and Investigators (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361:1139–1151
Connolly SJ, Ezekowitz MD, Yusuf S, Reilly PA, Wallentin L, Randomized Evaluation of Long-Term Anticoagulation Therapy Investigators (2010) Newly identified events in the RE-LY trial. N Engl J Med 363:1875–1876
Ezekowitz MD, Connolly S, Parekh A, Reilly PA, Varrone J, Wang S, Oldgren J, Themeles E, Wallentin L, Yusuf S (2009) Rationale and design of RE-LY: randomized evaluation of long-term anticoagulant therapy, warfarin, compared with dabigatran. Am Heart J 157:805–810, 810.e1–2
Larsen TB, Rasmussen LH, Skjøth F, Due KM, Callréus T, Rosenzweig M, Lip GY (2013) Efficacy and safety of dabigatran etexilate and warfarin in “real-world” patients with atrial fibrillation: a prospective nationwide cohort study. J Am Coll Cardiol 61:2264–2273
Rivaroxaban
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, Breithardt G, Halperin JL, Hankey GJ, Piccini JP, Becker RC, Nessel CC, Paolini JF, Berkowitz SD, Fox KA, Califf RM, ROCKET AF Investigators (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365:883–891
Piccini JP, Stevens SR, Chang Y, Singer DE, Lokhnygina Y, Go AS, Patel MR, Mahaffey KW, Halperin JL, Breithardt G, Hankey GJ, Hacke W, Becker RC, Nessel CC, Fox KA, Califf RM, ROCKET AF Steering Committee and Investigators (2013) Renal dysfunction as a predictor of stroke and systemic embolism in patients with nonvalvular atrial fibrillation: validation of the R (2)CHADS(2) index in the ROCKET AF (Rivaroxaban once-daily, oral, direct factor Xa inhibition compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation) and ATRIA (AnTicoagulation and Risk factors In Atrial fibrillation) study cohorts. Circulation 127:224–232
Apixaban
Goto S, Zhu J, Liu L, Oh BH, Wojdyla DM, Aylward P, Bahit MC, Gersh BJ, Hanna M, Horowitz J, Lopes RD, Wallentin L, Xavier D, Alexander JH, ARISTOTLE Investigators (2014) Efficacy and safety of apixaban compared with warfarin for stroke prevention in patients with atrial fibrillation from East Asia: a sub-analysis of the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation (ARISTOTLE) Trial. Am Heart J 168(3):303–309
Hanna MS, Mohan P, Knabb R, Gupta E, Frost C, Lawrence JH (2014) Development of apixaban: a novel anticoagulant for prevention of stroke in patients with atrial fibrillation. Ann N Y Acad Sci 1329:93–106
Liotta EM, Levasseur-Franklin KE, Naidech AM (2015) Reversal of the novel oral anticoagulants dabigatran, rivoraxaban, and apixaban. Curr Opin Crit Care 21(2):127–133
Edoxaban
Fukuda T, Honda Y, Kamisato C, Morishima Y, Shibano T (2012) Reversal of anticoagulant effects of edoxaban, an oral, direct factor Xa inhibitor, with haemostatic agents. Thromb Haemost 107:253–259
Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, Waldo AL, Ezekowitz MD, Weitz JI, Špinar J, Ruzyllo W, Ruda M, Koretsune Y, Betcher J, Shi M, Grip LT, Patel SP, Patel I, Hanyok JJ, Mercuri M, Antman EM, ENGAGE AF-TIMI 48 Investigators (2013) Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med 369:2093–2104
Ogata K, Mendell-Harary J, Tachibana M, Masumoto H, Oguma T, Kojima M, Kunitada S (2010) Clinical safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel factor Xa inhibitor edoxaban in healthy volunteers. J Clin Pharmacol 50:743–753
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer-Verlag Berlin Heidelberg
About this entry
Cite this entry
Mousa, S.A. (2015). Advances in Anticoagulants. In: Hock, F. (eds) Drug Discovery and Evaluation: Pharmacological Assays. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-27728-3_151-1
Download citation
DOI: https://doi.org/10.1007/978-3-642-27728-3_151-1
Received:
Accepted:
Published:
Publisher Name: Springer, Berlin, Heidelberg
Online ISBN: 978-3-642-27728-3
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences