Abstract
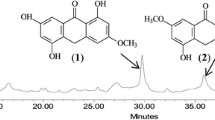
Hoppea fastigiata (Griseb.) C.B. Clarke, an annual medicinal herb belonging to Gentianaceae, is mostly found in South-Asian countries. The genus possesses a unique class of compounds called xanthones, which are known for their potential against Alzheimer’s and Parkinson’s diseases. Three major xanthones were isolated and structurally confirmed as 1,5,7-trihydroxy-3-methoxyxanthone, 1,5-dihydroxy-3,7-dimethoxyxanthone, and 1,3,5-trihydroxy-8-methoxyxanthone from the in vitro shoot cultures which showed potential inhibitions against acetylcholinesterase, monoamine oxidase A, and monoamine oxidase B enzymes. Upon treatment with different elicitors, yeast extract (YE) was found to be most effective which led to a 20-fold increase of 1,3,5-trihydroxy-8-methoxy xanthone. YE treatment caused a rapid burst of reactive oxygen species (ROS) and the subsequent increase in xanthone contents. Phenylalanine ammonia lyase activity remained suppressed and 4-coumarate: CoA ligase activity remained unaffected after elicitation. However, shikimate dehydrogenase and shikimate kinase activities increased after elicitation. This suggested phenylalanine-independent biosynthesis of xanthones. Subsequent treatment of shoots cultures with different inhibitors of superoxide radicals (O2−), hydrogen peroxide (H2O2) generation, and calcium channel was found to suppress accumulation of xanthones. Thus, calcium mediated generation of H2O2 followed by the activation of shikimate pathway enzymes is the key early step of xanthone biosynthesis in H. fastigiata.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- 4CL:
-
4-Coumarate CoA ligase
- AChE:
-
Acetylcholinesterase enzyme
- AD:
-
Alzheimer’s disease
- BA:
-
6-benzylaminopurine
- BPS:
-
Benzophenone synthase
- Ca2+:
-
Calcium ion
- DHLA:
-
Dihydrolipoic acid
- DIECA:
-
Diethyldithiocarbamic acid
- DPI:
-
Diphenylene iodide
- EGTA:
-
Ethylene glycol-bis-(β-aminoethyl)
- FDA:
-
Food and Drug Administration
- FW:
-
Fresh weight
- H2O2:
-
Hydrogen peroxide
- HPLC:
-
High pressure liquid chromatography
- La:
-
Lanthanum chloride
- MAO:
-
Monoamine oxidase enzyme
- O2−:
-
Superoxide radical
- PAL:
-
Phenylalanine ammonia-lyase
- PD:
-
Parkinson’s disease
- ROS:
-
Reactive oxygen species
- SK:
-
Shikimate kinase
- SKDH:
-
Shikimate dehydrogenase
- SOD:
-
Superoxide dismutase
- YE:
-
Yeast extract
References
GBD 2016 Neurology Collaborators (2019) Global, regional, and national burden of neurological disorders, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol 18:459–480
Houghton PJ, Howes MJ (2005) Natural products and derivatives affecting neurotransmission relevant to Alzheimer’s and Parkinson’s disease. Neurosignals 14:6–22
Mattson MP (2004) Pathways towards and away from Alzheimer’s disease. Nature 430:631–639
Heinrich N, Teoh HL (2004) Galanthamine of snowdrop-the development of a modern drug against Alzheimer’s disease from local Caucasian knowledge. J Ethnopharmacol 92:147–162
Serrano-Duen˜as M, Cardozo-Pelaez F, Sanchez-Ramos JR (2001) Effect of Banisteriopsis caapi extract on Parkinson’s disease. Sci Rev Altern Med 5:127–132
Schulz V (2003) Ginkgo extract or cholinesterase inhibitors in patients with dementia: what clinical trials and guidelines fail to consider. Phytomedicine 10:74–79
Urbain A, Marston A, Queiroz EF, Ndjoko K, Hostettmann K (2004) Xanthones fromGentiana campestris as new acetylcholinesterase inhibitors. Planta Med 70:1011–1014
Bennett GJ, Lee HH (1989) Xanthones from Guttiferae. Phytochemistry 28:967–998
Struwe L, Albert VA (eds) (2002) Gentianaceae: systematics and natural history. Cambridge University Press, Cambridge, UK
Ambasta SP (1999) The useful plants of India. National Institute of Science Communication, New Delhi
Wungsintaweekul J, Choo-malee J, Charoonratana T, Keawpradub N (2012) Methyl jasmonate and yeast extract stimulate mitragynine production in Mitragyna speciosa (Roxb.) Korth.shoot culture. Biotechnol Lett 34:1945–1950
Cardona ML, Fernandez MI, Pedro JR, Serrano A (1990) Xanthones from Hypericum reflexum. Phytochemistry 29:3003–3006
Beerhues L, Berger U (1995) Differential accumulation of xanthones in methyl-jasmonate and yeast-extract treated cell cultures of Centaurium erythraea and Centaurium littorale. Planta 197:608–612
Negi JS, Bisht VK, Singh P, Rawat MSM, Joshi GP (2013) Naturally occurring xanthones: chemistry and biology. J Appl Chem 2013:621459
Oldenburg TBP, Wilkes H, Horsfield B, Van Duin ACT, Stoddart D, Wilhelms A (2002) Xanthones-novel aromatic oxygen-containing compounds in crude oils. Org Geochem 33:595–609
Chen Y, Wang GK, Wu C, Qin MJ (2013) Chemical constituents of Gentiana rhodantha. Zhongguo Zhong Yao Za Zhi 38:362–365
Xue QC, Li CJ, Zuo L, Yang JZ, Zhang DM (2009) Three new xanthones from the roots of Polygala japonica houtt. J Asian Nat Prod Res 11:465–469
Krstic D, Jankovic T, Savikin-Fodulovic K, Menkovic N, Grubisic A (2003) Secoiridoids and xanthones in the shoots and roots of Centaurium pulchellum cultures in-vitro. In Vitro Cell Dev Biol 39:203–207
Iseda S (1957) Isolation of 1,3,6,7-tetrahydroxyxanthone and the skeletal structure of Mangiferin. Bull Chem Soc Jpn 30:625–629
Aritomi M, Kawasaki T (1970) A new xanthone c-glucoside, position isomer of mangiferin, from Anemarrhena asphodeloides Bunge. Chem Pharm Bull 18:2327–2333
Plouvier V, Massicot J, Rivaille P (1967) On gentiacauleine, a new tetra-substituted xanthone, aglycone of gentiacauloside of Gentiana acaulis L. C R Acad Sci Hebd Seances, Acad Sci 264:1219–1222
Suksamrarn S, Komutiban O, Ratananukul P, Chimnoi N, Lartpornmatulee N, Suksamrarn A (2006) Cytotoxic prenylated xanthones from the young fruit of Garcinia mangostana. Chem Pharm Bull 54:301–305
Castelão JF Jr, Gottlieb OR, De Lima RA, Mesquita HE, Gottlieb HE, Wenkert E (1977) Xantholignoids from Kielmeyera and Caraipa species-13C NMR spectroscopy of xanthones. Phytochemistry 16:735–740
Nielsen H, Arends P (1978) Structure of the xantholignoid kielcorin. Phytochemistry 17:2040–2041
Ishiguro K, Nagata S, Oku H, Masae Y (2002) Bisxanthones from Hypericum japonicum: inhibitors of PAF-induced hypotension. Planta Med 68:258–261
Nkengfack AE, Mkounga P, Meyer M, Fomum ZT, Bodo B (2002) Globulixanthones C, D and E: three prenylated xanthones with antimicrobial properties from the root bark of Symphonia globulifera. Phytochemistry 61:181–187
Kumagai K, Hosotani N, Kikuchi K, Kimura T, Saji I (2003) Xanthofulvin, a novel semaphoring inhibitor produced by a strain of Penicillium. J Antibiot 56:610–616
Don MJ, Huang YJ, Huang RL, Lin YL (2004) New phenolic principles from Hypericum sampsonii. Chem Pharm Bull 52:866–869
Guo A, Li J, Fu H, Lin W (2003) Xanthone derivatives from medicinal plant Swertia mileensis. Zhongcaoyao 34:107–109
Inuma M, Tosa H, Tanaka T, Asai F, Kobayashi Y, Shimano R, Miyauchi K (1996) Antibacterial activity of xanthones from guttiferaeous plants against methicillin resistant Staphylococcus aureus. J Pharm Pharmacol 48:861–865
Dall’Acquaa S, Violaa G, Mariella C, Innocentia G (2004) Xanthones from Polygala alpestris (Rchb.). Z Naturforsch 59c:335–338
Nickel P (1994) Chemotherapie und Prophylaxe der Malaria: neuere Entwicklungen. Pharamzeutische Zeitung 49:4307–4313
Ray S, Majumder HK, Chakravarty AK, Mukhopadhyay S, Gil RR, Cordell GA (1996) Amarogentin, a naturally occurring secoiridoid glycoside and a newly recognized inhibitor of topoisomerase I from Leishmania donouani. J Nat Prod 59:27–29
Niiho Y, Yamazaki T, Yamamota T, Ando H, Hirai Y (2006) Gastroprotective effects of bitter principles isolated from gentian root and Swertia herb on experimentally-induced gastric lesions in rats. J Nat Med 60:82–88
Oztürk N, Korkmaz S, Oztürk Y, Baser KH (2006) Effects of gentiopicroside, sweroside and swertiamarine, secoiridoids from gentian (Gentiana lutea ssp. symphyandra), on cultures chicken embryonic fibroblasts. Planta Med 72:289–294
Ariňo A, Arberas I, Leiton MJ, de Renobales M, Dominguez JB (1997) The extraction of yellow gentian root (Gentiana lutea L.). Z Lebensm Unters Forsch A 205:295–299
Ya BQ, Nian LC, Li C, Gen XP (1999) Protective effect of swerchirin on hematopoiesis in 60 co-irradiated mice. Phytomedicine 6:85–88
Tosa H, Iinuma M, Tanaka T, Nozaki H, Ikeda S, Tsutsui K, Yamada M, Fujimori S (1997) Inhibitory activity of xanthone derivatives isolated from some guttiferaeous plants against DNA topoisomerases I and II. Chem Pharm Bull 42:418–420
Urbain A, Marston A, Queiroz EF, Ndjoko K, Hostettmann K (2004) Xanthones from Gentiana campestris as new acetylcholinesterase inhibitors. Planta Med 70:1011–1014
Urbain A, Marston A, Sintra GL, Bravo J, Purev O, Purevsuren B, Batsuren D, Reist M, Carrupt PA, Hostettmann K (2008) Xanthones from Gentiana amarella ssp. acuta with acetylcholinesterase and monoamine oxidase inhibitory activities. J Nat Prod 71:895–897
Moon UR, Sen SK, Mitra A (2014) Antioxidant capacities and acetylcholinesterase-inhibitory activity of Hoppea fastigiata. Int J Geogr Inf Syst 20:115–123
Schaufelberger D, Hostettmann K (1988) Chemistry and pharmacology of Gentiana lactea. Planta Med 54:219–221
Rocha L, Marston A, Kaplan MAC, Stoeckli-Evans H, Thull U, Testa B, Hostettmann K (1994) An antifungal 7-pyrone and xanthones with monoamine oxidase inhibitory activity from Hypericum brasiliense. Phytochemistry 36:1381–1385
Moon UR, Sircar D, Barthwal R, Sen SK, Beuerle T, Beerhues L, Mitra A (2015) Shoot cultures of Hoppea fastigiata (Griseb.) C.B. Clarke as a potential source of neuroprotective xanthones. J Nat Med 69:375–386
Chopra RN, Nayar SL, Chopra IC (1956) Glossary of Indian medicinal plants. CSIR Publication, New Delhi, p 161
Mukherjee KS, Chakraborty CK, Chatterjee TP, Bhattacharjee D, Laha S (1991) 1,5,7-Trihydroxy-3-methoxyxanthone from Hoppea fastigiata. Phytochemistry 30:1036–1037
Mukherjee KS, Manna TK, Laha S, Chakraborty CK (1994) A new xanthone from Hoppea fastigiata. Pharm Biol 32:201–203
Mukherjee KS, Laha S, Manna TK, Roy SC (1995) Further work on Limnophilla rugosa and Hoppea fastigiata (Gentianaceae). J Indian Chem Soc 72:63–64
Brahmachari G, Gorai D, Mondal S, Gangopadhay A, Chatterjee D (2003) A new naturally occurring xanthone bearing rare oxygenation pattern from Hoppea fastigiata. J Chem Res 6:362–363
Eugster PJ, Guillarme D, Rudaz S, Veuthey JL, Carrupt PA, Wolfender JL (2011) Ultra high pressure liquid chromatography for crude plant extract profiling. J AOAC Int 94:51–70
Moon UR, Durge AA, Kumar M (2020) Value addition of khus (Vetiveria zizanioides L. Nash) by elicitor and heat treatment. Ind Crop Prod 144:112037
Wang CZ, Maier UH, Keil M, Zenk MH, Bacher A, Rohdich F, Eisenreich W (2003) Phenylalanine-independent biosynthesis of 1,3,5,8-tetrahydroxyxanthone, a retro biosynthetic NMR study with root cultures of Swertia chirata. Eur J Biochem 270:2950–2958
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this entry
Cite this entry
Moon, U.R., Dey, P.K., Mitra, A. (2021). Neuroprotective Xanthones and Their Biosynthesis in Shoot Cultures of Hoppea fastigiata (Griseb.) C.B. Clarke. In: Ramawat, K.G., Ekiert, H.M., Goyal, S. (eds) Plant Cell and Tissue Differentiation and Secondary Metabolites. Reference Series in Phytochemistry. Springer, Cham. https://doi.org/10.1007/978-3-030-30185-9_17
Download citation
DOI: https://doi.org/10.1007/978-3-030-30185-9_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-30184-2
Online ISBN: 978-3-030-30185-9
eBook Packages: Chemistry and Materials ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics