Abstract
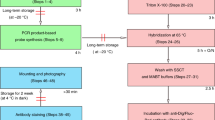
Whole-mount in situ hybridization is the preferred method for detecting transcript distributions in whole embryos, tissues, and organs. We present here a sensitive fluorescent in situ hybridization method for colocalization analysis of different transcripts in whole embryonic zebrafish brains. The method is based on simultaneous hybridization of differently hapten-labeled RNA probes followed by sequential rounds of horseradish peroxidase (POD)-based transcript detection. Sequential detection involves enhancement of fluorescent signals by tyramide signal amplification (TSA) and effective inactivation of the antibody–POD conjugate prior to the following detection round. We provide a detailed description of embryo preparation, hybridization, antibody detection, POD–TSA reaction, and mounting of embryos for imaging. To achieve high signal intensities, we optimized key steps of the method. This includes improvement of embryo permeability by hydrogen peroxide treatment and efficacy of hybridization and TSA–POD reaction by addition of the viscosity-increasing polymer dextran sulfate. The TSA–POD reaction conditions are further optimized by application of substituted phenol compounds as POD accelerators and use of highly efficient bench-made tyramide substrates. The obtained high signal intensities and cellular resolution of our method allows for co-expression analysis and generation of three-dimensional models. Our protocol is tailored to optimally work in zebrafish embryos, but can surely be modified for application in other species as well.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abellan A, Medina L (2009) Subdivisions and derivatives of the chicken subpallium based on expression of LIM and other regulatory genes and markers of neuron subpopulations during development. J Comp Neurol 515(4):465–501
Puelles L, Kuwana E, Puelles E, Bulfone A, Shimamura K, Keleher J, Smiga S, Rubenstein JL (2000) Pallial and subpallial derivatives in the embryonic chick and mouse telencephalon, traced by the expression of the genes Dlx-2, Emx-1, Nkx-2.1, Pax-6, and Tbr-1. J Comp Neurol 424(3):409–438, doi:10.1002/1096-9861(20000828)424:3<409::AID-CNE3>3.0.CO;2-7 [pii]
Diez-Roux G, Banfi S, Sultan M, Geffers L, Anand S, Rozado D, Magen A, Canidio E, Pagani M, Peluso I, Lin-Marq N, Koch M, Bilio M, Cantiello I, Verde R, De Masi C, Bianchi SA, Cicchini J, Perroud E, Mehmeti S, Dagand E, Schrinner S, Nurnberger A, Schmidt K, Metz K, Zwingmann C, Brieske N, Springer C, Hernandez AM, Herzog S, Grabbe F, Sieverding C, Fischer B, Schrader K, Brockmeyer M, Dettmer S, Helbig C, Alunni V, Battaini MA, Mura C, Henrichsen CN, Garcia-Lopez R, Echevarria D, Puelles E, Garcia-Calero E, Kruse S, Uhr M, Kauck C, Feng G, Milyaev N, Ong CK, Kumar L, Lam M, Semple CA, Gyenesei A, Mundlos S, Radelof U, Lehrach H, Sarmientos P, Reymond A, Davidson DR, Dolle P, Antonarakis SE, Yaspo ML, Martinez S, Baldock RA, Eichele G, Ballabio A (2011) A high-resolution anatomical atlas of the transcriptome in the mouse embryo. PLoS Biol 9(1):e1000582
Shimogori T, Lee DA, Miranda-Angulo A, Yang Y, Wang H, Jiang L, Yoshida AC, Kataoka A, Mashiko H, Avetisyan M, Qi L, Qian J, Blackshaw S (2010) A genomic atlas of mouse hypothalamic development. Nat Neurosci 13(6):767–775
Suzuki-Hirano A, Ogawa M, Kataoka A, Yoshida AC, Itoh D, Ueno M, Blackshaw S, Shimogori T (2011) Dynamic spatiotemporal gene expression in embryonic mouse thalamus. J Comp Neurol 519(3):528–543
Ferran JL, Sanchez-Arrones L, Sandoval JE, Puelles L (2007) A model of early molecular regionalization in the chicken embryonic pretectum. J Comp Neurol 505(4):379–403. doi:10.1002/cne.21493
Lauter G, Söll I, Hauptmann G (2013) Molecular characterization of prosomeric and intraprosomeric subdivisions of the embryonic zebrafish diencephalon. J Comp Neurol 521(5):1093–1118
Tautz D, Pfeifle C (1989) A non-radioactive in situ hybridization method for the localization of specific RNAs in Drosophila embryos reveals translational control of the segmentation gene hunchback. Chromosoma 98(2):81–85
Hauptmann G, Gerster T (1994) Two-color whole-mount in situ hybridization to vertebrate and Drosophila embryos. Trends Genet 10(8):266
Hauptmann G, Gerster T (1996) Multicolour whole-mount in situ hybridization to Drosophila embryos. Dev Genes Evol 206(4):292–295
Hauptmann G, Gerster T (2000) Multicolor whole-mount in situ hybridization. Methods Mol Biol 137:139–148, doi:1-59259-066-7-139 [pii] 10.1385/1-59259-066-7:139
Jowett T, Lettice L (1994) Whole-mount in situ hybridizations on zebrafish embryos using a mixture of digoxigenin- and fluorescein-labelled probes. Trends Genet 10(3):73–74
Hauptmann G (1999) Two-color detection of mRNA transcript localizations in fish and fly embryos using alkaline phosphatase and beta-galactosidase conjugated antibodies. Dev Genes Evol 209(5):317–321
Hauptmann G (2001) One-, two-, and three-color whole-mount in situ hybridization to Drosophila embryos. Methods 23(4):359–372, doi:10.1006/meth.2000.1148 S1046-2023(00)91148-4 [pii]
O’Neill JW, Bier E (1994) Double-label in situ hybridization using biotin and digoxigenin-tagged RNA probes. Biotechniques 17(5):870, 874, 875
Bobrow MN, Harris TD, Shaughnessy KJ, Litt GJ (1989) Catalyzed reporter deposition, a novel method of signal amplification. Application to immunoassays. J Immunol Methods 125(1–2):279–285, doi:0022-1759 (89)90104-X [pii]
van de Corput MP, Dirks RW, van Gijlswijk RP, van Binnendijk E, Hattinger CM, de Paus RA, Landegent JE, Raap AK (1998) Sensitive mRNA detection by fluorescence in situ hybridization using horseradish peroxidase-labeled oligodeoxynucleotides and tyramide signal amplification. J Histochem Cytochem 46(11):1249–1259
Kosman D, Mizutani CM, Lemons D, Cox WG, McGinnis W, Bier E (2004) Multiplex detection of RNA expression in Drosophila embryos. Science 305(5685):846, doi:10.1126/science.1099247 305/5685/846 [pii]
Tessmar-Raible K, Steinmetz PR, Snyman H, Hassel M, Arendt D (2005) Fluorescent two-color whole mount in situ hybridization in Platynereis dumerilii (Polychaeta, Annelida), an emerging marine molecular model for evolution and development. Biotechniques 39(4):460, 462, 464. doi:000112023 [pii]
Clay H, Ramakrishnan L (2005) Multiplex fluorescent in situ hybridization in zebrafish embryos using tyramide signal amplification. Zebrafish 2(2):105–111. doi:10.1089/zeb.2005.2.105
Lauter G, Söll I, Hauptmann G (2011) Two-color fluorescent in situ hybridization in the embryonic zebrafish brain using differential detection systems. BMC Dev Biol 11:43
Vize PD, McCoy KE, Zhou X (2009) Multichannel wholemount fluorescent and fluorescent/chromogenic in situ hybridization in Xenopus embryos. Nat Protoc 4(6):975–983, doi:nprot.2009.69 [pii] 10.1038/nprot.2009.69
Denkers N, Garcia-Villalba P, Rodesch CK, Nielson KR, Mauch TJ (2004) FISHing for chick genes: triple-label whole-mount fluorescence in situ hybridization detects simultaneous and overlapping gene expression in avian embryos. Dev Dyn 229(3):651–657. doi:10.1002/dvdy.20005
Lauter G, Söll I, Hauptmann G (2011) Multicolor fluorescent in situ hybridization to define abutting and overlapping gene expression in the embryonic zebrafish brain. Neural Dev 6(1):10, doi:1749-8104-6-10 [pii]10.1186/1749-8104-6-10
Fischer AH, Jacobson KA, Rose J, Zeller R (2008) Media for mounting fixed cells on microscope slides. CSH Protoc 2008:pdb ip52
Liu G, Amin S, Okuhama NN, Liao G, Mingle LA (2006) A quantitative evaluation of peroxidase inhibitors for tyramide signal amplification mediated cytochemistry and histochemistry. Histochem Cell Biol 126(2):283–291. doi:10.1007/s00418-006-0161-x
Acknowledgments
The Stockholm Greater Council and the Karolinska Institutet supported this work. Imaging was in part performed at the Live Cell Imaging Unit, Department of Biosciences and Nutrition, Karolinska Institutet, Huddinge, Sweden, supported by grants from the Knut and Alice Wallenberg Foundation, the Swedish Research Council, and the Centre for Biosciences.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Lauter, G., Söll, I., Hauptmann, G. (2014). Sensitive Whole-Mount Fluorescent In Situ Hybridization in Zebrafish Using Enhanced Tyramide Signal Amplification. In: Sprecher, S. (eds) Brain Development. Methods in Molecular Biology, vol 1082. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-655-9_12
Download citation
DOI: https://doi.org/10.1007/978-1-62703-655-9_12
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-654-2
Online ISBN: 978-1-62703-655-9
eBook Packages: Springer Protocols