Abstract
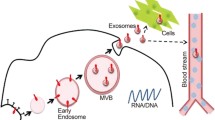
Extracellular membrane vesicles derived from the endosomal compartments and released by the fusion of the multivesicular bodies with the cell membrane are referred as exosomes (Exo) [Van Niel et al., J Biochem 140:13–21, 2006]. They function as mediators of intercellular communication and are employed by the organism in the regulation of systemic and local processes. Meantime, Exo are recognized as an indispensable entity of physiological fluids [Caby et al., Int Immunol 17:879–887, 2005; Lasser et al., J Transl Med 9:9, 2011; Lasser et al., Am J Rhinol Allergy 25:89–93, 2011]. Exo and other types of extracellular vesicles, e.g., exosome-like vesicles [van Niel et al., Gastroenterology 121:337–349, 2001] and microvesicles (MV) [Daveloose et al., Thromb Res 22:195–201, 1981], contain multiple functional molecules including lipids [Vidal et al., J Cell Physiol 140:455–462, 1989]; proteins [Simpson et al., Expert Rev Proteomics 6:267–283, 2009]; mRNA [Valadi et al., Nat Cell Biol 9:654–659, 2007]; DNA [Waldenstrom et al., PLoS One 7:e34653, 2012]; noncoding RNA, e.g., miRNA [Simpson et al., Expert Rev Proteomics 6:267–283, 2009]; and retrotransposon elements [Balaj et al., Nat Commun 2:180, 2011]. Assessment of the biological functions of Exo showed that they deliver specifically their cargo from the donor to recipient cells. Albeit the molecular mechanisms of this process are not fully understood, approaches for the application of Exo and MV as a tool for a cell-specific delivery of signalling molecules were successfully tested in in vitro and in vivo models [Maguire et al., Mol Ther 20:960–971, 2012]. Ovarian cancer cells release Exo, which bind stroma cells as well as donor cancer cells [Escrevente et al., BMC Cancer 11:108, 2011].
Here we describe an experimental approach for the assessment of Exo interaction and uptake by target cells. Methods for the isolation and purification of Exo from cell culture supernatants are included. To allow visualization of vesicle uptake, labelling of Exo with different fluorescent dyes, such as CFSE, PKH, DHPE, and DiOC18, is presented. Finally, we explain qualitative and quantitative analysis of Exo uptake by immunofluorescence and flow cytometry, respectively.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Van Niel G, Porto-Carreiro I, Simoes S, Raposo G (2006) Exosomes: a common pathway for a specialized function. J Biochem 140:13–21
Caby MP, Lankar D, Vincendeau-Scherrer C, Raposo G, Bonnerot C (2005) Exosomal-like vesicles are present in human blood plasma. Int Immunol 17:879–887
Lasser C et al (2011) Human saliva, plasma and breast milk exosomes contain RNA: uptake by macrophages. J Transl Med 9:9
Lasser C et al (2011) RNA-containing exosomes in human nasal secretions. Am J Rhinol Allergy 25:89–93
van Niel G et al (2001) Intestinal epithelial cells secrete exosome-like vesicles. Gastroenterology 121:337–349
Daveloose D et al (1981) Inhibitory effect of microvesicles collected from stored blood on platelet aggregation. Thromb Res 22:195–201
Vidal M, Sainte-Marie J, Philippot JR, Bienvenue A (1989) Asymmetric distribution of phospholipids in the membrane of vesicles released during in vitro maturation of guinea pig reticulocytes: evidence precluding a role for “aminophospholipid translocase”. J Cell Physiol 140:455–462
Simpson RJ, Lim JW, Moritz RL, Mathivanan S (2009) Exosomes: proteomic insights and diagnostic potential. Expert Rev Proteomics 6:267–283
Valadi H et al (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659
Waldenstrom A, Genneback N, Hellman U, Ronquist G (2012) Cardiomyocyte microvesicles contain DNA/RNA and convey biological messages to target cells. PLoS One 7:e34653
Balaj L et al (2011) Tumour microvesicles contain retrotransposon elements and amplified oncogene sequences. Nat Commun 2:180
Maguire CA et al (2012) Microvesicle-associated AAV Vector as a Novel Gene Delivery System. Mol Ther 20:960–971
Escrevente C, Keller S, Altevogt P, Costa J (2011) Interaction and uptake of exosomes by ovarian cancer cells. BMC Cancer 11:108
Rak J, Yu JL, Klement G, Kerbel RS (2000) Oncogenes and angiogenesis: signaling three-dimensional tumor growth. J Investig Dermatol Symp Proc 5:24–33
Psaila B, Lyden D (2009) The metastatic niche: adapting the foreign soil. Nat Rev Cancer 9:285–293
Valenti R et al (2007) Tumor-released microvesicles as vehicles of immunosuppression. Cancer Res 67:2912–2915
Van Niel G, Porto-Carreiro I, Simoes S, Raposo G (2006) Exosomes: a common pathway for a specialized function. J Biochem 140:13–21
Stoorvogel W, Kleijmeer MJ, Geuze HJ, Raposo G (2002) The biogenesis and functions of exosomes. Traffic 3:321–330
Simons M, Raposo G (2009) Exosomes–vesicular carriers for intercellular communication. Curr Opin Cell Biol 21:575–581
van Niel G, Porto-Carreiro I, Simoes S, Raposo G (2006) Exosomes: a common pathway for a specialized function. J Biochem 140:13–21
Babst M (2011) MVB vesicle formation: ESCRT-dependent, ESCRT-independent and everything in between. Curr Opin Cell Biol 23:6
Bobrie A, Colombo M, Raposo G, Thery C (2011) Exosome secretion: molecular mechanisms and roles in immune responses. Traffic 12:1659–1668
Nazarenko I et al (2010) Cell surface tetraspanin Tspan8 contributes to molecular pathways of exosome-induced endothelial cell activation. Cancer Res 70:1668–1678
Fevrier B, Raposo G (2004) Exosomes: endosomal-derived vesicles shipping extracellular messages. Curr Opin Cell Biol 16:415–421
Fevrier B, Vilette D, Laude H, Raposo G (2005) Exosomes: a bubble ride for prions? Traffic 6:10–17
Keller S, Sanderson MP, Stoeck A, Altevogt P (2006) Exosomes: from biogenesis and secretion to biological function. Immunol Lett 107:102–108
Rana S, Claas C, Kretz CC, Nazarenko I, Zoeller M (2011) Activation-induced internalization differs for the tetraspanins CD9 and Tspan8: Impact on tumor cell motility. Int J Biochem Cell Biol 43:106–119
Runz S et al (2007) Malignant ascites-derived exosomes of ovarian carcinoma patients contain CD24 and EpCAM. Gynecol Oncol 107:563–571
Taylor DD, Gercel-Taylor C, Parker LP (2009) Patient-derived tumor-reactive antibodies as diagnostic markers for ovarian cancer. Gynecol Oncol 115:112–120
Taylor DD, Gercel-Taylor C (2008) MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol Oncol 110:13–21
Li J et al (2009) Claudin-containing exosomes in the peripheral circulation of women with ovarian cancer. BMC Cancer 9:244
Cho JA et al (2011) Exosomes from ovarian cancer cells induce adipose tissue-derived mesenchymal stem cells to acquire the physical and functional characteristics of tumor-supporting myofibroblasts. Gynecol Oncol 123:379–386
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, New York
About this protocol
Cite this protocol
Nazarenko, I., Rupp, AK., Altevogt, P. (2013). Exosomes as a Potential Tool for a Specific Delivery of Functional Molecules. In: Malek, A., Tchernitsa, O. (eds) Ovarian Cancer. Methods in Molecular Biology, vol 1049. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-547-7_37
Download citation
DOI: https://doi.org/10.1007/978-1-62703-547-7_37
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-546-0
Online ISBN: 978-1-62703-547-7
eBook Packages: Springer Protocols