Abstract
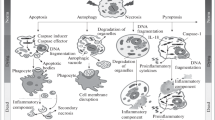
In eukaryotic organisms facing terminal stress, activation of genetically encoded cell death pathways underlies fundamental changes in core cellular processes and functional modification of critical biomolecules. These physiological alterations manifest themselves as phenotypic hallmarks during programmed cell death, and are markers of the particular mode of death initiated. A growing volume of work has illustrated that prokaryotes too are capable of exhibiting hallmarks of programmed cell death, albeit without the multiple, tight regulatory layers which control these events in higher order organisms.
This chapter describes how methods and materials which have been used to assay for hallmarks of programmed cell death in eukaryotic models are transferrable to prokaryotic models. In particular, we describe the applicability of these methods to the study of post-antibiotic effects on bacteria, notably the biochemical changes induced by the interaction of drug molecules and targets, including oxidative stress, that accompany and ensure cell death. Specifically we discuss techniques for detecting DNA fragmentation, chromosomal condensation, phosphatidylserine exposure, membrane depolarization, and caspase substrate peptide binding, thereby providing a launchpoint for the study of the evolution of these physiological events in bacteria.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26(4):239–257
Wyllie AH, Kerr JF, Currie AR (1980) Cell death: the significance of apoptosis. Int Rev Cytol 68:251–306
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116(2):205–219
Edinger AL, Thompson CB (2004) Death by design: apoptosis, necrosis and autophagy. Curr Opin Cell Biol 16(6):663–669
Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri ES, Baehrecke EH, Blagosklonny MV, El-Deiry WS, Golstein P, Green DR et al (2009) Classification of cell death: recommendations of the Nomenclature Committee on cell death 2009. Cell Death Differ 16(1):3–11
Nicholson DW, Thornberry NA (1997) Caspases: killer proteases. Trends Biochem Sci 22(8):299–306
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407(6805):770–776
Strasser A, O’Connor L, Dixit VM (2000) Apoptosis signaling. Annu Rev Biochem 69:217–245
Peterson JS, Barkett M, McCall K (2003) Stage-specific regulation of caspase activity in drosophila oogenesis. Dev Biol 260(1):113–123
Broker LE, Kruyt FA, Giaccone G (2005) Cell death independent of caspases: a review. Clin Cancer Res 11(9):3155–3162
Reddien PW, Horvitz HR (2004) The engulfment process of programmed cell death in caenorhabditis elegans. Annu Rev Cell Dev Biol 20:193–221
Ren Y, Savill J (1998) Apoptosis: the importance of being eaten. Cell Death Differ 5(7):563–568
Festjens N, Vanden Berghe T, Vandenabeele P (2006) Necrosis, a well-orchestrated form of cell demise: signalling cascades, important mediators and concomitant immune response. Biochim Biophys Acta 1757(9–10):1371–1387
Golstein P, Kroemer G (2007) Cell death by necrosis: towards a molecular definition. Trends Biochem Sci 32(1):37–43
Wyllie AH (1980) Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 284(5756):555–556
Fadok VA, Voelker DR, Campbell PA, Cohen JJ, Bratton DL, Henson PM (1992) Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J Immunol 148(7):2207–2216
Martin SJ, Reutelingsperger CP, McGahon AJ, Rader JA, van Schie RC, LaFace DM, Green DR (1995) Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: inhibition by overexpression of Bcl-2 and Abl. J Exp Med 182(5):1545–1556
Buttke TM, Sandstrom PA (1994) Oxidative stress as a mediator of apoptosis. Immunol Today 15(1):7–10
Jacobson MD (1996) Reactive oxygen species and programmed cell death. Trends Biochem Sci 21(3):83–86
Green DR, Reed JC (1998) Mitochondria and apoptosis. Science 281(5381):1309–1312
Chandra J, Samali A, Orrenius S (2000) Triggering and modulation of apoptosis by oxidative stress. Free Radic Biol Med 29(3–4):323–333
Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82(1):47–95
Cadenas E, Davies KJ (2000) Mitochondrial free radical generation, oxidative stress, and aging. Free Radic Biol Med 29(3–4):222–230
Skulachev VP (2006) Bioenergetic aspects of apoptosis, necrosis and mitoptosis. Apoptosis 11(4):473–485
Tan S, Sagara Y, Liu Y, Maher P, Schubert D (1998) The regulation of reactive oxygen species production during programmed cell death. J Cell Biol 141(6):1423–1432
Dwyer DJ, Kohanski MA, Hayete B, Collins JJ (2007) Gyrase inhibitors induce an oxidative damage cellular death pathway in Escherichia coli. Mol Syst Biol 3:91
Kohanski MA, Dwyer DJ, Hayete B, Lawrence CA, Collins JJ (2007) A common mechanism of cellular death induced by bactericidal antibiotics. Cell 130(5):797–810
Dwyer DJ, Camacho DM, Kohanski MA, Callura JM, Collins JJ (2012) Antibiotic-induced bacterial cell death exhibits physiological and biochemical hallmarks of apoptosis. Mol Cell 46(5):561–572
Madeo F, Frohlich E, Frohlich KU (1997) A yeast mutant showing diagnostic markers of early and late apoptosis. J Cell Biol 139(3):729–734
Madeo F, Frohlich E, Ligr M, Grey M, Sigrist SJ, Wolf DH, Frohlich KU (1999) Oxygen stress: a regulator of apoptosis in yeast. J Cell Biol 145(4):757–767
Vermes I, Haanen C, Steffens-Nakken H, Reutelingsperger C (1995) A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J Immunol Methods 184(1):39–51
Kroemer G, Petit P, Zamzami N, Vayssiere JL, Mignotte B (1995) The biochemistry of programmed cell death. FASEB J 9(13):1277–1287
Foti JJ, Devadoss B, Winkler JA, Collins JJ, Walker GC (2012) Oxidation of the guanine nucleotide pool underlies cell death by bactericidal antibiotics. Science 336(6079):315–319
Rohwer F, Azam F (2000) Detection of DNA damage in prokaryotes by terminal deoxyribonucleotide transferase-mediated dUTP nick end labeling. Appl Environ Microbiol 66(3):1001–1006
Gavrieli Y, Sherman Y, Ben-Sasson SA (1992) Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119(3):493–501
Darzynkiewicz Z, Crissman H, Jacobberger JW (2004) Cytometry of the cell cycle: cycling through history. Cytometry A 58(1):21–32
Galluzzi L, Zamzami N, de La Motte RT, Lemaire C, Brenner C, Kroemer G (2007) Methods for the assessment of mitochondrial membrane permeabilization in apoptosis. Apoptosis 12(5):803–813
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91(4):479–489
Rottenberg H, Wu S (1998) Quantitative assay by flow cytometry of the mitochondrial membrane potential in intact cells. Biochim Biophys Acta 1404(3):393–404
Epps DE, Wolfe ML, Groppi V (1994) Characterization of the steady-state and dynamic fluorescence properties of the potential-sensitive dye bis-(1,3-dibutylbarbituric acid)trimethine oxonol (Dibac4(3)) in model systems and cells. Chem Phys Lipids 69(2):137–150
Thornberry NA, Lazebnik Y (1998) Caspases: enemies within. Science 281(5381):1312–1316
Cohen GM (1997) Caspases: the executioners of apoptosis. Biochem J 326(Pt 1):1–16
Lamkanfi M, Festjens N, Declercq W, Vanden Berghe T, Vandenabeele P (2007) Caspases in cell survival, proliferation and differentiation. Cell Death Differ 14(1):44–55
Vercammen D, Declercq W, Vandenabeele P, Van Breusegem F (2007) Are metacaspases caspases? J Cell Biol 179(3):375–380
Talanian RV, Quinlan C, Trautz S, Hackett MC, Mankovich JA, Banach D, Ghayur T, Brady KD, Wong WW (1997) Substrate specificities of caspase family proteases. J Biol Chem 272(15):9677–9682
Garcia-Calvo M, Peterson EP, Leiting B, Ruel R, Nicholson DW, Thornberry NA (1998) Inhibition of human caspases by peptide-based and macromolecular inhibitors. J Biol Chem 273(49):32608–32613
Timmer JC, Salvesen GS (2007) Caspase substrates. Cell Death Differ 14(1):66–72
Le Gac S, Vermes I, van den Berg A (2006) Quantum dots based probes conjugated to annexin V for photostable apoptosis detection and imaging. Nano Lett 6(9):1863–1869
Acknowledgments
We thank James Collins for his mentorship and support of this work through his Howard Hughes Medical Institute Investigator and NIH Director’s Pioneer Award Program awards. We also thank Jarred Callura for his assistance with UV irradiation treatment design and Ahmad Khalil for his assistance with fluorescent microscopy, as well as with the design of Qdot-based Annexin V experiments.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Dwyer, D.J., Winkler, J.A. (2013). Identification and Characterization of Programmed Cell Death Markers in Bacterial Models. In: McCall, K., Klein, C. (eds) Necrosis. Methods in Molecular Biology, vol 1004. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-383-1_11
Download citation
DOI: https://doi.org/10.1007/978-1-62703-383-1_11
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-382-4
Online ISBN: 978-1-62703-383-1
eBook Packages: Springer Protocols