Abstract
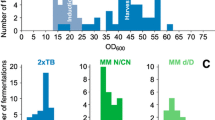
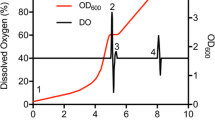
The Gram-negative bacterium Escherichia coli offer a means for rapid, high-yield, and economical production of recombinant proteins. However, when preparing protein samples for NMR, high-level production of functional isotopically labeled proteins can be quite challenging. This is especially true for the preparation of triple-labeled protein samples in D2O (2H/13C/15N). The large expense and time-consuming nature of triple-labeled protein production for NMR led us to revisit the current bacterial protein expression protocols. Our goal was to develop an efficient bacterial expression method for very high-level production of triple-labeled proteins that could be routinely utilized in every NMR lab without changing expression vectors or requiring fermentation. We developed a novel high cell-density IPTG-induction bacterial expression method that combines tightly controlled traditional IPTG-induction expression with the high cell-density of auto-induction expression. In addition, we optimize several key experimental protocols and parameters to ensure that our new high cell-density bacterial expression method routinely produces 14–25 mg of triple-labeled proteins and 15–35 mg of unlabeled proteins from 50-mL bacterial cell cultures.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Swartz, J.R. (2001) Advances in Escherichia coli production of therapeutic proteins. Curr. Opin. Biotechnol. 12, 195–201.
Hewitt, L., and McDonnell, J.M. (2004) Screening and optimizing protein production in E. coli methods. Methods Mol. Biol. 278, 1–16.
McIntosh, L.P. and Dahlquist, F.W. (1990) Biosynthetic incorporation of 15N and 13C for assignment and interpretation of nuclear magnetic resonance spectra of proteins. Q Rev. Biophys. 23, 1–38.
Studier, F.W. (2005) Protein production by auto-induction in high density shaking cultures. Protein Expr. Purif. 41, 207–234.
Sivashanmugam, A., Murray, V., Cui, C., Zhang, Y., Wang, J., and Li, Q. (2009) Practical protocols for production of very high yields of recombinant proteins using Escherichia coli. Protein Sci. 18, 936–948.
Chen, H.C., Hwang, C.F., and Mou, D.G. (1992) High-density Escherichia coli cultivation process for hyperexpression of recombinant porcine growth hormone. Enzyme Microb. Technol. 14, 321–326.
Baneyx, F. (1999) Recombinant protein expression in Escherichia coli. Curr. Opin. Biotechnol. 10, 411–421.
Sambrook, J. and Russell, D. (2001) Molecular Cloning: A Laboratory Manual (3rd ed.). Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York (ISBN 978-087969577-4).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Murray, V., Huang, Y., Chen, J., Wang, J., Li, Q. (2012). A Novel Bacterial Expression Method with Optimized Parameters for Very High Yield Production of Triple-Labeled Proteins. In: Shekhtman, A., Burz, D. (eds) Protein NMR Techniques. Methods in Molecular Biology, vol 831. Humana Press. https://doi.org/10.1007/978-1-61779-480-3_1
Download citation
DOI: https://doi.org/10.1007/978-1-61779-480-3_1
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-479-7
Online ISBN: 978-1-61779-480-3
eBook Packages: Springer Protocols