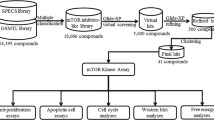
Abstract
The mammalian Target of Rapamycin (mTOR)-mediated signaling transduction pathway has been observed to be deregulated in a wide variety of cancer and metabolic diseases. Despite extensive clinical development efforts, the well-known allosteric mTOR inhibitor rapamycin and structurally related rapalogs have failed to show significant single-agent antitumor efficacy in most types of cancer. This limited clinical success may be due to the inability of the rapalogs to maintain a complete blockade mTOR-mediated signaling. Therefore, numerous efforts have been initiated to develop ATP-competitive mTOR inhibitors that would block both mTORC1 and mTORC2 complex activity. Here, we describe our experimental approaches to develop Torin1 using a medium throughput cell-based screening assay and structure-guided drug design.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Sarbassov, D. D., Ali, S. M., Sabatini, D. M. (2005) Growing roles for the mTOR pathway. Curr. Opin. Cell Biol. 17, 596–603.
Sehgal, S. N.; Baker, H.; Vezina, C. (1975) Rapamycin (AY-22989), a new antifungal antibiotic. II. Fermentation, isolation and characterization. J. antibiotics (Tokyo), 28, 727–732.
Rao, R. D.; Buckner, J. C.; Sarkaria, J. N. (2004) mammalian Target of Rapamycin (mTOR) Inhibitors as Anti-Cancer Agents. Curr. Cancer Drug. Targets. 4, 621–635.
Guertin, D. A., Sabatini, D.M. (2007) Defining the role of mTOR in cancer. Cancer cell, 12, 9–22.
Molinolo, A. A., Hewitt, S. M., Amornphimoltham, P., Keelawat, S., Rangdaeng, S., Meneses, A. et al (2007) Dissecting the Akt/ mammalian target of rapamycin signaling network: emerging results from the head and neck cancer tissue array initiative. Clin Cancer Res, 13, 4964–4973.
Karbowniczek, M., Spittle, C.S., Morrison, T., Wu, H., Henske, E.P. (2008) mTOR is activated in the majority of malignant melanomas. J Invest Dermatol 128, 980–987.
Meric-Bernstam, F., Gonzalez-Angulo, A. M. (2009) Targeting the mTOR signaling network for cancer therapy. J Clin Oncol, 27, 2278–2287.
Thoreen, C. C., Kang, S. A., Chang, J. W., Liu, Q., Zhang, J., Gao, Y., et al. (2009) An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-insensitive functions of mTORC1. J Biol Chem. 284, 8023–8032.
Wan, X., Harkavy, B., Shen, N., Grohar, P., Helman, L. J. (2007) Rapamycin induces feedback activation of Akt signaling through an IGF-1R-dependent mechanism. Oncogene, 26, 1932–1940.
Walker, E. H., Pacold, M. E., Perisic, O., Stephens, L., Hawkins, P. T., Wymann, M. P., et al. (2000) Structural determinants of phosphoinositide 3-kinase inhibition by wortmannin, LY294002, quercetin, myricetin, and staurosporine. Mol. Cell, 6, 909–919.
Hayakawa, M., Kaizawa, H., Moritomo, H., Koizumi, T., Ohishi, T., Yamano, M., et al. (2007) Synthesis and biological evaluation of pyrido[3’,2’:4,5]furo[3,2-d]pyrimidine derivatives as novel PI3 kinase p110alpha inhibitors. Bioorg. Med. Chem. Lett., 17, 2438–2442.
Park, S., Chapuis, N., Bardet, V., Tamburini, J., Gallay, N., Willems, L., et al. (2008) PI-103, a dual inhibitor of class IA phosphatidylinositide 3-kinase and mTOR, has antileukemic activity in AML. Leukemia, 22, 1698–1706.
García-Martínez, J. M., Moran, J., Clarke, R. G., Gray, A., Cosulich, S. C., Chresta, C. M., et al. (2009) Ku-0063794 is a specific inhibitor of the mammalian target of rapamycin (mTOR). Biochem J, 421, 29–42.
Yu, K., Toral-Barza, L., Shi, C., Zhang, W. G., Lucas, J., Shor, B., et al. (2009) Biochemical, cellular, and in vivo activity of novel ATP-competitive and selective inhibitors of the mammalian target of rapamycin. Cancer Res. 69, 6232–6240.
Liu, Q., Thoreen, C. C., Wang, J., Sabatini, D. M., Gray, N. S. (2009) mTOR medicated anti-cancer drug discovery. Drug. Discov. Today: Therapeutic Strategies. 6, 47–55.
Ding, S., Gray, N. S., Wu, X., Ding, Q., Schultz, P. G. (2002) A combinatorial scaf-fold approach toward kinase-directed heterocycle libraries. J. Am. Chem. Soc., 124, 1594–1596.
Liu, Q., Chang, J., Wang, J., Kang, S. A., Thoreen, C.C., Markhard, A., Hur, W., Zhang, J., Sim, T., Sabatini, D. M., Gray, N. S. (2010) Discovery of 1-(4-(4-propionylpiperazin-1-yl)-3-(trifluoromethyl)phenyl)-9-(quinolin-3-yl)benzo[h][1,6]naphthyridin-2(1 H)-one as a highly potent, selective mTOR inhibitor for the treatment of cancer. J. Med. Chem. DOI: 10.1021/jm101144f
Guertin, D. A., Stevens, D. M., Thoreen, C. C., Burds, A. A., Kalaany, N. Y., Moffat, J., Brown, M., Fitzgerald, K. J., Sabatini, D. M. (2006) Ablation in mice of the mTORC components raptor, rictor, or mLST8 reveals that mTORC2 is required for signaling to Akt-FOXO and PKCalpha, but not S6K1. Dev. Cell, 11, 859–871
Trott, O., Olson, A. J. (2010) Autodock vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem., 31, 455–461.
Acknowledgements
We thank Life Technologies Corporation for SelectScreen® Kinase Profiling Service and Ambit Bioscience for performing KinomeScanTM profiling. We also thank SAI Advantium Pharma Limited Inc. (India) for the pharmacokinetic study.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Liu, Q. et al. (2012). Development of ATP-Competitive mTOR Inhibitors. In: Weichhart, T. (eds) mTOR. Methods in Molecular Biology, vol 821. Humana Press. https://doi.org/10.1007/978-1-61779-430-8_29
Download citation
DOI: https://doi.org/10.1007/978-1-61779-430-8_29
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-429-2
Online ISBN: 978-1-61779-430-8
eBook Packages: Springer Protocols