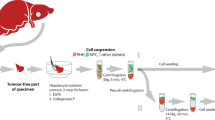
Abstract
The use of isolated human liver cells in research and development has gained increasing interest during the past years. The possible application may vary between elucidation of new biochemical pathways in liver diseases, drug development, safety issues, and new therapeutic strategies up to direct clinical translation for liver support. However, the isolation of human liver cells requires a well-developed logistic network among surgeons, biologists, and technicians to obtain a high quality of cells. Our laboratories have been involved in various applications of human liver cells and we have long-lasting experiences in human liver cell isolation and their application in R&D. We here summarize the present protocol of our laboratories for cell isolation from normal resected liver tissue, the most common tissue available. In addition, we discuss the necessary network in the clinic and quality controls to maintain human liver cells in culture and the effect of 3D extracellular matrix in cultured cells which results in preservation of hepatocyte epithelial polarity in the form of bile canaliculi and repression of epithelial to mesenchymal transitions occurring in 2D cultures.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Branster, M. V., and Morton, R. K. (1957) Isolation of intact liver cells. Nature. 180, 1283–1284.
de Boer, J., and van Bavel, B. (2009) European “REACH” (Registration, Evaluation, Authorisation and Restriction of Chemicals) program. J Chromatogr A. 1216, 301.
Alexandre, E., Cahn, M., Abadie-Viollon, C., Meyer, N., Heyd, B., Mantion, G., et al. (2002) Influence of pre-, intra- and post-operative parameters of donor liver on the outcome of isolated human hepatocytes. Cell Tissue Bank. 3, 223–233.
Lloyd, T. D., Orr, S., Patel, R., Crees, G., Chavda, S., Vadyar, H., et al. (2004) Effect of patient, operative and isolation factors on subsequent yield and viability of human hepatocytes for research use. Cell Tissue Bank. 5, 81–87.
Seglen, P. O. (1976) Preparation of isolated rat liver cells. Methods Cell Biol. 13, 29–83.
Dorko, K., Freeswick, P. D., Bartoli, F., Cicalese, L., Bardsley, B. A., Tzakis, A., et al. (1994) A new technique for isolating and culturing human hepatocytes from whole or split livers not used for transplantation. Cell Transplant. 3, 387–395.
Sharma, A. D., Cantz, T., Richter, R., Eckert, K., Henschler, R., Wilkens, L., et al. (2005) Human cord blood stem cells generate human cytokeratin 18-negative hepatocyte-like cells in injured mouse liver. Am J Pathol. 167, 555–564.
Beerheide, W., von Mach, M. A., Ringel, M., Fleckenstein, C., Schumann, S., Renzing, N., et al. (2002) Downregulation of beta2-microglobulin in human cord blood somatic stem cells after transplantation into livers of SCID-mice: an escape mechanism of stem cells? Biochem Biophys Res Commun. 294, 1052–1063.
Hengstler, J. G., Brulport, M., Schormann, W., Bauer, A., Hermes, M., Nussler, A. K., et al. (2005) Generation of human hepatocytes by stem cell technology: definition of the hepatocyte. Expert Opin Drug Metab Toxicol. 1, 61–74.
Williams, R. T. (1959) Detoxification Mechanisms. New York: John Wiley & Sons.
Parkinson, A. (1996) Biotransformation of xenobiotics. In Casarett and Doull’s Toxicology: the basic Science of Poinsons, ed. Klaassen, C. D. New York: McGraw-Hill. 113–186.
Nelson, D. R., Zeldin, D. C., Hoffman, S. M., Maltais, L. J., Wain, H. M., and Nebert, D. W. (2004) Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants. Pharmacogenetics. 14, 1–18.
Guengerich, F. P., and MacDonald, T. L. (1990) Mechanisms of cytochrome P-450 catalysis. FASEB J. 4, 2453–2459.
Pelkonen, O., Maenpaa, J., Taavitsainen, P., Rautio, A., and Raunio, H. (1998) Inhibition and induction of human cytochrome P450 (CYP) enzymes. Xenobiotica. 28, 1203–1253.
Donato, M. T., Jimenez, N., Castell, J. V., and Gomez-Lechon, M. J. (2004) Fluorescence-based assays for screening nine cytochrome P450 (P450) activities in intact cells expressing individual human P450 enzymes. Drug Metab Dispos. 32, 699–706.
Lin, J. H., and Lu, A. Y. (1997) Role of pharmacokinetics and metabolism in drug discovery and development. Pharmacol Rev. 49, 403–449.
Hewitt, N. J., Lechon, M. J., Houston, J. B., Hallifax, D., Brown, H. S., Maurel, P., et al. (2007) Primary hepatocytes: current understanding of the regulation of metabolic enzymes and transporter proteins, and pharmaceutical practice for the use of hepatocytes in metabolism, enzyme induction, transporter, clearance, and hepatotoxicity studies. Drug Metab Rev. 39, 159–234.
Guillouzo, A., Morel, F., Fardel, O., and Meunier, B. (1993) Use of human hepatocyte cultures for drug metabolism studies. Toxicology. 82, 209–219.
Rajan, N., Habermehl, J., Cote, M. F., Doillon, C. J., and Mantovani, D. (2006) Preparation of ready-to-use, storable and reconstituted type I collagen from rat tail tendon for tissue engineering applications. Nat Protoc. 1, 2753–2758.
Decaens, C., Durand, M., Grosse, B., and Cassio, D. (2008) Which in vitro models could be best used to study hepatocyte polarity? Biol Cell. 100, 387–398.
Rennert, B., and Melzig, M. F. (2002) Free fatty acids inhibit the activity of Clostridium histolyticum collagenase and human neutrophil elastase. Planta Med. 68, 767–769.
Acknowledgments
This work was supported by the German Ministry of Research FKZ-BMBF 0315741 (Virtual Liver) and FZK-BMBF 01 GG 0732 (3-D TOX).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Knobeloch, D. et al. (2012). Human Hepatocytes: Isolation, Culture, and Quality Procedures. In: Mitry, R., Hughes, R. (eds) Human Cell Culture Protocols. Methods in Molecular Biology, vol 806. Humana Press. https://doi.org/10.1007/978-1-61779-367-7_8
Download citation
DOI: https://doi.org/10.1007/978-1-61779-367-7_8
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-366-0
Online ISBN: 978-1-61779-367-7
eBook Packages: Springer Protocols