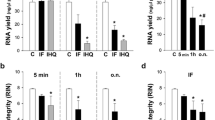
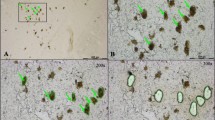
Abstract
Cell specificity of gene expression analysis is essential to avoid tissue sample related artifacts, in particular when the relative number of target cells present in the compared tissues varies dramatically, e.g., when comparing dopamine neurons in midbrain tissues from control subjects with those from Parkinson’s disease (PD) cases. Here, we describe a detailed protocol that combines contact-free UV-laser microdissection and quantitative PCR of reverse-transcribed RNA of individual neurons from postmortem human midbrain tissue from PD patients and unaffected controls. Among expression changes in a variety of dopamine neuron marker, maintenance, and cell-metabolism genes, we found that α-synuclein mRNA levels were significantly elevated in individual neuromelanin-positive dopamine midbrain neurons from PD brains when compared to those from matched controls.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Grundemann J, Schlaudraff F, Haeckel O et al (2008) Elevated alpha-synuclein mRNA levels in individual UV-laser-microdissected dopaminergic substantia nigra neurons in idiopathic Parkinson’s disease. Nucleic Acids Res 36:e38
Simunovic F, Yi M, Wang Y et al (2009) Gene expression profiling of substantia nigra dopamine neurons: further insights into Parkinson’s disease pathology. Brain 132:1795–1809
Huggett J, Dheda K, Bustin S et al (2005) Real-time RT-PCR normalisation; strategies and considerations. Genes Immun 6:279–284
Bustin SA, Nolan T (2004) Pitfalls of quantitative real-time reverse-transcription polymerase chain reaction. J Biomol Tech 15:155–166
Damier P, Hirsch EC, Agid Y et al (1999) The substantia nigra of the human brain. II. Patterns of loss of dopamine-containing neurons in Parkinson’s disease. Brain 122:1437–1448
Damier P, Hirsch EC, Agid Y et al (1999) The substantia nigra of the human brain. I. Nigrosomes and the nigral matrix, a compartmental organization based on calbindin D(28K) immunohistochemistry. Brain 122:1421–1436
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson’s disease: a target for neuroprotection?. Lancet Neurol 8:382–397
Dachsel JC, Lincoln SJ, Gonzalez J et al (2007) The ups and downs of alpha-synuclein mRNA expression. Mov Disord 22:293–295
Cookson MR (2009) alpha-Synuclein and neuronal cell death. Mol Neurodegener 4:9
Ramirez A, Heimbach A, Grundemann J et al (2006) Hereditary parkinsonism with dementia is caused by mutations in ATP13A2, encoding a lysosomal type 5 P-type ATPase. Nat Genet 38:1184–1191
Ibanez P, Bonnet AM, Debarges B et al (2004) Causal relation between alpha-synuclein gene duplication and familial Parkinson’s disease. Lancet 364:1169–1171
Eriksen JL, Przedborski S, Petrucelli L (2005) Gene dosage and pathogenesis of Parkinson’s disease. Trends Mol Med 11:91–96
Polymeropoulos MH, Lavedan C, Leroy E et al (1997) Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science 276:2045–2047
McCormack AL, Di Monte DA (2009) Enhanced alpha-synuclein expression in human neurodegenerative diseases: pathogenetic and therapeutic implications. Curr Protein Pept Sci 10:476–482
Simon-Sanchez J, Schulte C, Bras JM et al (2009) Genome-wide association study reveals genetic risk underlying Parkinson’s disease. Nat Genet 41:1308–1312
Satake W, Nakabayashi Y, Mizuta I et al (2009) Genome-wide association study identifies common variants at four loci as genetic risk factors for Parkinson’s disease. Nat Genet 41:1303–1307
Liss B (2002) Improved quantitative real-time RT-PCR for expression profiling of individual cells. Nucleic Acids Res 30:e89
Liss B, Franz O, Sewing S et al (2001) Tuning pacemaker frequency of individual dopaminergic neurons by Kv4.3L and KChip3.1 transcription. EMBO J 20:5715–5724
Acknowledgments
We are particularly grateful to the brain donors and the support by the German BrainNet (GA28). We thank Leica Microsystems for providing the LMD6000 and Jochen Roeper for critical reading the manuscript. This work was supported by the BMBF (NGFN-Net), the DFG (SFB497), the Gemeinnützige Hertiestiftung, the Parkinson’s Disease society, and the Royal Society, UK. J.G. is supported by a PhD Studentship of the Wellcome Trust; B.L. is supported by the Alfried Krupp prize for young university teachers.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Gründemann, J., Schlaudraff, F., Liss, B. (2011). UV-Laser Microdissection and mRNA Expression Analysis of Individual Neurons from Postmortem Parkinson’s Disease Brains. In: Murray, G. (eds) Laser Capture Microdissection. Methods in Molecular Biology, vol 755. Humana Press. https://doi.org/10.1007/978-1-61779-163-5_30
Download citation
DOI: https://doi.org/10.1007/978-1-61779-163-5_30
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-162-8
Online ISBN: 978-1-61779-163-5
eBook Packages: Springer Protocols