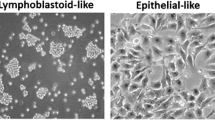
Abstract
Studies of the same cell lines by different laboratories are common in the literature and often show different results with the same methodology. Use of best cell culture practices is essential to ensure consistent and reproducible results. Assay outcomes are easily influenced by many factors including changes in functionality, morphology, doubling time of cells, passage numbers, microbial contamination, and misidentification of cells. Simple observation, monitoring, and documentation of cell morphology and behavior, including growth rates, provide early warning and should be standard practice. Changes may indicate microbial contamination, genotypic drift due to high passage number, or cross-contamination with another cell line. Rapid molecular methods allow the identification of microbial and cross-contamination. Increasingly, authentication of cell lines is a prerequisite for scientific publication to avoid erroneous results entering the literature.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hartung, T., Balls, M., Bardouille, C., Blanck, O., Coecke, S., Gstraunthaler, G., and Lewis, D. (2002) Good cell culture practice: ECVAM good cell culture practice task force report 1, ATLA 30, 407–14.
Freshney, R.I. Culture of glioma of the brain. (1980) In: Thomas, D.G.T., and Graham, D.I. Eds. Brain tumours: Scientific basic, clinical investigation and current therapy. London Butterworths, pp. 21–50.
Bayreuther, K., Rodemann, H.P., Hommel, R., Dittmann, K., Albiez, M., and Francz, P.I. (1988) Human skin fibroblasts in vitro differentiate along a terminal cell lineage. PNAS USA, 85, 5112–6.
Freshney, R.I. (2005) Analysis of Growth Curve, In: Culture of Animal Cells: A manual of basic techniques 5th Ed. New York: John Wiley & Sons Inc., pp. 351.
Freshney, R.I. (2000) Growth Curve. In: Masters JRW, ed. Animal Cell culture A Practical Approach, 3rd ed. Oxford University press, pp.12–13.
Hay, R.J. (1992) Continuous Cell lines - An International Workshop on Current Issues. Develop Biol Standard, 76, 25–379.
Beary, S.J., and Walczak, E.M. (1983) Cytopathic Effect of Whole Cells and Purified Membranes of Mycoplasma hyopneumoniae. Infect Immun, 41, 132–6.
Macy, M.L. (1979) Tests for mycoplasma contamination of cultured cells as applied at the ATCC. TCA Manual 5, 1151–6.
Chen, T.R. (1977) In situ detection of mycoplasma contamination in cell culture by fluorescent Hoechst 33258 strain. Exp Cell Res 104, 255–262.
Tang, J., Hu, M., Lee, S., and Roblin, R. (2000) A polymerase chain reaction based method for detecting Mycoplasma/Acholeplasma contaminants in cell culture. J Microbiol Methods 39, 121–6.
Hay, R.J. (1998) Cell Line Banking and Authentication. Dev Biol Stand 93, 15–19.
Briske-Anderson, M.J., Finley, J.W., and Newman, S.M. (1997) Influence of culture time and passage number on morphological and physiological development of Caco-2 cells. Proc Soc Exp Biol Med. 214; 3, 248–57.
Chang-Liu, C-M., and Woloschak, G.E. (1997) Effect of passage number on cellular response to DNA-damaging agents: cell survival and gene expression. Cancer Lett 113, 77–86.
Esquenet, M., Swinnen, J.V., Heyns, W., and Verhoeven, G. (1997) LNCaP prostatic adenocarcinoma cells derived from low and high passage numbers display divergent responses not only to androgens but also to retinoids. J Steroid Biochem Mol Biol 62, 391–9.
Langeler, E.G., van Uffelen, C.J., Blankenstein, M.A., van Steenbrugge, G.J., and Mulder, E. (1993) Effects of culture conditions on androgen sensitivity of the human prostate cancer cell line LNCaP. Prostate 23, 213–23.
Sambuy, Y., De Angelis, I., Ranaldi, G., Scarino, M.L., Stammati, A., and Zucco, F. (2005) The Caco-2 cell line as a model of the intestinal barrier: influence of cell and culture-related factors on Caco-2 cell functional characteristics. Cell Biol Toxicol 21, 1–26.
Yu, H. (1997) Evidence for diminished functional expression of intestinal transporters in Caco-2 Cell monolayers at high passages. Pharm Res 14, 757–62.
Matsumura, T. (1980) Multinucleation and polyploidization of aging human cells in culture. Adv Exp Med Biol, 129, 31–38.
Dumont, P., Burton, M., Chen, Q.M., Gonos, E.S., Frippiat, C., Mazarati, J.B., Eliaers, F., Remacle, J., and Toussaint, O. (2000) Induction of replicative senescence biomarkers by sublethal oxidative stresses in normal human fibroblast. Free Radical Biol Med, 28, 361–73.
Hughes, P., Marshall, D., Reid, Y., Parkes, H., and Gelber, C. (2007) The costs of using unauthenticated, over-passaged cell lines: how much more data do we need? Biotechniques 43, 575–86.
Mossberg, P.l. (2005) Wise Marketing Consultancy. Study Report: Cell Lines and Their Use in Research.
Lu, S., Gough, A.W., Babrowski, W.F., and Stewart, B.H. (1996) Transport properties are not altered across Caco-2 cells with heightened TEER despite underlying physiological and ultrastructural changes. J Pharm Sci, 85, 270–273.
Ranaldi, G., Consalvo, R., Sambuy, Y., and Scarino, M.I. (2003) Permeability characteristics of parental and clonal human intestinal Caco-2 cell lines differentiated in serum-supplemented and serum-free media. Toxicol In Vitro 17, 761–767.
Wenger, S.L., Senft, J.R., Sargent, L.M., Bamezai, R., Bairwa, N., and Grant, S.G. (2004) Comparison of established cell lines at different passages by karyotype and comparative genomic hybridization. Biosci Rep 24, 631–9.
Draper, J.S., Smith, K., Gokhale, H.D., Moore, E., Maltby, J., Johnson, L., Meisner, T., Zwakea, T.P., Thomson, J.A., and Andrews, P.W. (2004) Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nat Biotechnol 22, 53–4.
McCulloch, E.A., and Parker, R.C. (1957) In Begg, R.W. Ed Canadian cancer conference. Vol.2 New York: Academic Press.
Reid, Y.A., O’Neill, K., Chen, T.R. (1995) Cell cross-contamination of U-937. J Leukoc Biol 57, 804.
Masters, J.R., Thomson, J.A., Daly-Burns, B., Reid, Y.A., Dirks, W.G., Packer, P., Toji, L.H., Ohno, T., Tanabe, H., Arlett, C.F., Kelland, L.R., Harrison, M., Virmani, A., Ward, T.H., Ayers, K.L., and Debenham, P.G. (2001) Short tandem repeat profiling provides an international reference standard for human cell lines. PNAS 98, 8012–7.
Gilbert, D.A., Reid, Y.A., Gail, M.H., Pee, D., White, C., Hay, R.J., and O’Brien, S.J. (1990) Applications of DNA fingerprints for cell line individualization. Am J Hum Genet 47, 499–514.
Durkin, A.S., Cedrone, E., Sykes, G., Boyles, D., Reid, Y.A. (2000) Utility of gender determination in cell line identity. In Vitro Cell Dev Biol Anim 36, 344–7.
Markovic, O., Markovic, N. (1998) Cell cross-contamination in cell culture: the silent and neglected danger. In Vitro Cell Dev Biol Anim 34, 1–8.
Gold, M.A. (1986) Conspiracy Of Cells, One Woman’s Immortal Legacy and The Medical Scandal It Caused. Albany, NY: State Univ. of New York Press.
Buehring, G.C., Eby, E.A., and Eby, M.J. (2004) Cell Line cross-contamination: how aware are mammalian cell culturists of the problem and how to monitor it? In Vitro Cell Dev Biol Anim 40, 211–5.
Nelson-Rees WA, Flandermeyer RR. HeLa cultures defined. Science, 191(4222):96–98, 1976.
Nelson-Rees, W.A., Daniels, D.W., and Flandermeyer, R.R. (1981) Cross-contamination of cells in culture. Science 212, 446–52.
Drexler, H.G., Dirks, W.G., Matsuo, Y., and MacLeod, R.A.F. (2003) False leukemia-lymphoma cell lines: an update on over 500 cell lines. Leukemia 17, 416–26.
Liscovitch, M., and Ravid, D. (2006) A case of misidentification of cancer cell lines: MCF-7/AdrR cells (re-designated NCI/ADR-RES) are derived from OVCAR-8 human ovarian carcinoma cells. Cancer Lett 245, 350–2.
Ross, D.T., Scherf, U., Eisen, M.B., Perou, C.M., Rees, C., Spellman, P., Iyer, V., Jeffrey, S.S., Van de Rijn, M., Walthan, M., Pergamenschikov, A., Lee, J.C., Lashkari, D., Shalon, D., Myers, T.G., Weinstein, J.N., Botstein, D., and Brown, P.O. (2000) Systematic variation in gene expression patterns in human cancer cell lines. Nat Genet 24, 227–35.
Rae, J.M., Creighton, C.J., Meck, J.M., Haddad, B.R., and Johnson, M.D. (2007) MDA-MB-435 is derived from M14 melanoma cells - a loss for breast cancer but a boon for melanoma research. Breast Cancer Res Treat 104, 13–19.
Parodi, B., Aresu, O., Bini, D., Lorenzini, R., Schena, F., Visconti, P., Cesaro, M., Ferrera, D., Andreotti, V., and Ruzzon, T. (2002) Species identification and confirmation of human and animal cell lines: a PCR-based method. BioTechniques 32, 432–4, 436, and 438–440.
Cooper, J.K., Sykes, G., King, S., Cottrill, K., Inanova, N.V., Hanner, R., and Ikonomi, P. (2007) Species identification in cell culture: a two-pronged molecular approach. In Vitro Cell Dev Biol Anim 43, 344–51.
Demichelis, F., Greulich, H., Macoska, J.A., Beroukhim, R., Sellers, W.R., Garraway, L., and Rubin, M.A. (2008) SNP panel identification assay (SPIA): a genetic-based assay for the identification. Nucleic Acids Res 36, 2446–56.
Reid, Y.A., Gilbert, D.A., and O’Brien, S.J. (1990) The use of DNA hypervariable probes for human cell line identification. American Type Culture Collection Newsletter 10, 1–3.
Nardone, R.M. (2007) Eradication of cross-contaminated cell lines: a call for action. Cell Biol Toxicol 23, 367–72.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Reid, Y.A. (2011). Characterization and Authentication of Cancer Cell Lines: An Overview. In: Cree, I. (eds) Cancer Cell Culture. Methods in Molecular Biology, vol 731. Humana Press. https://doi.org/10.1007/978-1-61779-080-5_4
Download citation
DOI: https://doi.org/10.1007/978-1-61779-080-5_4
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-079-9
Online ISBN: 978-1-61779-080-5
eBook Packages: Springer Protocols