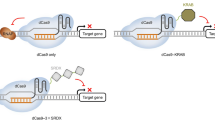
Abstract
RNAi (RNA interference, RNA silencing) is a powerful tool in functional genomics. We report here the use of transient RNAi to isolate regulatory factor genes involved in isoquinoline alkaloid biosynthesis in Coptis japonica protoplasts. Double-stranded (ds) RNAs prepared against candidate regulatory factors, which were predicted from an EST library, were introduced into C. japonica protoplasts by polyethylene glycol (PEG)-mediated transformation, and their effects were monitored by real-time reverse transcription (RT)-polymerase chain reaction (PCR). The potential of this transient RNAi system to characterize the functions of regulatory factor genes in alkaloid research is discussed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- MES:
-
2-morpholinoethanesulfonic acid;
- PEG:
-
polyethylene glycol;
- RNAi:
-
RNA interference;
- RT:
-
reverse transcription;
- TF:
-
transcription factor.
References
Van der Fits, L., and Memelink, J. (2000) ORCA3, a jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism. Science 289, 295–297.
Rischer, H., Oresic, M., Seppanen-Laakso, T., Katajamaa, M., Lammertyn, F., Ardiles-Diaz, W., Van Montagu, M. C, Inze, D., Oksman-Caldentey, K. M., and Goossens, A. (2006) Gene-to-metabolite networks for terpenoid indole alkaloid biosynthesis in Catharanthus roseus cells. Proc. Natl. Acad. Sci. USA 103, 5614–5619.
Kato, N., Dubouzet, E., Kokabu, Y., Yoshida, S., Taniguchi, Y., Dubouzet, J. G., Yazaki, K., and Sato, F. (2007) Identification of a WRKY protein as a transcriptional regulator of benzylisoquinoline alkaloid biosynthesis in Coptis japonica. Plant Cell Physiol. 48, 8–18.
Kakimoto, T. (1996) CKI1, a histidine kinase homolog implicated in cytokinin signal transduction. Science 274, 982–985.
Oksman-Caldentey, K. M., and Inze, D. (2004) Plant cell factories in the post-genomic era: new ways to produce designer secondary metabolites. Trends Plant Sci. 9, 433–440.
Apuya, N. R., Park, J-H., Zhang, L., Ahyow, M., Davidow, P., Van Fleet, J., Rarang, J. C., Hippley, M., Johnson, T. W., Yoo, H-D., Trieu, A., Krueger, S., Wu, C-Y., Yu-ping Lu, Y-p., Flavell, R. B., and Bobzin, S. C. (2008) Enhancement of alkaloid production in opium and California poppy by transactivation using heterologous regulatory factors. Plant Biotech. J. 6, 160–175.
Pauw, B., Hilliou, F. A., Martin, V. S., Chatel, G., de Wolf, C. J., Champion, A., Pre, M., van Duijn, B., Kijne, J .W., van der Fits, L., and Memelink, J. (2004) Zinc finger proteins act as transcriptional repressors of alkaloid biosynthesis genes in Catharanthus roseus. J. Biol. Chem. 279, 52940–52948.
Dubouzet, J. G., Morishige, T., Fujii, N., An, C. I., Fukusaki, E., Ifuku, K., and Sato, F. (2005) Transient RNA silencing of scoulerine 9-O-methyltransferase expression by double stranded RNA in Coptis japonica protoplasts. Biosci. Biotechnol. Biochem. 69, 63–70.
Park, S.-U., Yu, M., and Facchini, P. J. (2002) Antisense RNA-mediated suppression of benzophenanthridine alkaloid biosynthesis in transgenic cell cultures of California poppy. Plant Physiol. 128, 696–706.
Ifuku, K., Yamamoto, Y., and Sato, F. (2003) Specific RNA interference in psbP genes encoded by a multigene family in Nicotiana tabacum with a short 3'-untranslated sequence. Biosci. Biotechnol. Biochem. 67, 107–113.
Sato, F., and Yamada, Y. (1984) High berberine-producing cultures of Coptis japonica cells. Phytochem. 23, 281–285.
Sato, F., Hashimoto, T., Hachiya, A., Tamura, K., Choi, K-B., Morishige, T., Fujimoto, H., and Yamada, Y. (2001) Metabolic engineering of plant alkaloid biosynthesis. Proc. Natl. Acad. Sci. USA 98, 367–372.
Sheen, J. A transient expression assay using Arabidopsis mesophyll protoplasts. http://genetics.mgh.harvard.edu/sheenweb/protocols_reg.html (accessed August 31, 2009).
Carthew, R., and Kennerdell, J. RNAi in Drosophila embryos, http://www.bioexchange.com/tools/protocol_detail.cfm?protocol_id=31#MMB101 (accessed August 31, 2009).
Chiu, W.-l., Niwa, Y., Zeng, W., Hirano, T., Kobayashi, H., and Sheen, J. (1996) Engineered GFP as a vital reporter in plants. Curr. Biol. 6, 325–330.
Ifuku , K., Yamamoto, Y., Ono, T., Ishihara, S., and Sato, F. (2005) PsbP protein, but not PsbQ protein, is essential for the regulation and stabilization of Photosystem II in higher plants. Plant Physiol. 139, 1175–1184.
Zhai, Z., Sooksa-nguan, T., and Vatamaniuk, O. K. (2009) Establishing RNA interference as a reverse-genetic approach for gene functional analysis in protoplasts. Plant Physiol. 149, 642–652.
Maniatis, T., Fritsch, E. F., and Sambrook, J. (1982) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, USA.
Pawlowski, K., Kunze, R., De vries, S., and Bisseling, T. (1994) Isolation of total, poly (A) and polysomal RNA from plant tissues, in Plant molecular biology manual, 2nd ed., Gelvin, S.B, and Schilperoort, R.A., eds., Kluwer Academic Publishers, Dordrecht, pp. 1–13.
Linsmaier, E. M., and Skoog, F. (1965) Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 18, 100–127.
Kao, K. N., and Michayluk, M. R. (1975) Nutritional requirements for growth of Vicia hajastana cells and protoplasts at very low population density in liquid media. Planta 126, 105–110.
Acknowledgements
We thank Dr. Kentaro Ifuku for his critical reading of this manuscript. This work was supported in part by a grant from the Ministry of Education, Culture, Sports, and Science and Technology of Japan (21248013 to F. S.).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Yamada, Y., Kato, N., Kokabu, Y., Luo, Q., Dubouzet, J.G., Sato, F. (2010). Identification of Regulatory Protein Genes Involved in Alkaloid Biosynthesis Using a Transient RNAi System. In: Fett-Neto, A. (eds) Plant Secondary Metabolism Engineering. Methods in Molecular Biology, vol 643. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-60761-723-5_3
Download citation
DOI: https://doi.org/10.1007/978-1-60761-723-5_3
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-60761-722-8
Online ISBN: 978-1-60761-723-5
eBook Packages: Springer Protocols