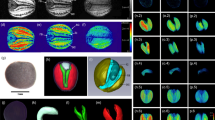
Summary
Plant oil has become an important component in the search for a replacement for non-renewable energy sources, as well as being used for a wide range of industrial purposes, all in addition to its vital importance for human diet. Detailed knowledge of the lipid distribution in plants is fundamental for the understanding of local regulatory networks covering storage metabolism, and for the development of new approaches for plant breeding and transgenic research. We here review a measurement protocol or “tool” based on magnetic resonance imaging (MRI), which allows the non-invasive detection and quantitative visualization of lipid in living plant tissue. The method provides quantitative lipid maps with a resolution close to the cellular level and can be used on a wide range of plants and is applicable at the level of individual tissues, organs, or entire plants during ontogeny. Lipid imaging is designed for both biotechnology and basic science and can be combined with histological, biochemical, and gene expression analysis. Here we present the method as practiced in our group, and discuss unique advantages and limitations of the lipid-imaging tool. Seeds of barley and rapeseed were used as a model for visualization of local oil accumulation at the organ- and tissue-specific scale.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Breithaupt H. (2006) Seeing is understanding. EMBO; Rep. 7: 467–470.
Kano H, Ishida N, Kobayashi T, Koizuml M. (1990) 1H-NMR imaging analysis of changes of free water distribution in barley and soybean seeds during maturation. Jpn. J. Crop Sci.; 59: 503–509.
Pope JM, Jonas D, Walker RR. (1993) Applications of NMR micro-imaging to the study of water, lipid, and carbohydrate distribution in grape berries. Protoplasma; 173: 357–363.
Baeten V, Aparicio R. (2000) Edible oils and fats authentication by Fourier transform Raman spectrometry. Biotechnol. Agron. Soc. Environ.; 4: 196–203.
Baranska M, Schulz H, Reitzenstein S, Uhlemann U, Strehle MA, Kruger H, Quilitzsch R, Foley W, Popp J. (2005) Vibrational spectroscopic studies to acquire a quality control method o f Eucalyptus essential oils. Biopolymers; 78: 237–248.
Borisjuk L, Nguyen TH, Neuberger T, Rutten T, Tschiersch H, Claus B, Feussner I, Webb AG, Jakob P, Weber H, Wobus U. and Rolletschek H. (2005) Gradients of lipid storage, photosynthesis and plastid differentiation in developing soybean seeds. New Phytol.; 167: 761–776.
Adlof, R.O. (2003) Advances in Lipid Methodology – Five. Bridgwater, Somerset: The Oily Press.
Ratcliffe RG, Shachar-Hill Y. (2001) Probing plant metabolism with NMR. Annu. Rev. Plant Physiol. Plant Mol. Biol.; 52: 499–526.
Knothe G, Kenar JA. (2004) Determination of the fatty acid profile by 1H-NMR spectroscopy. Eur. J. Lipid Sci. Technol.; 106: 88–96.
Débarre D, Supatto W, Pena AM, Fabre A, Tordjmann T, Combettes L, Schanne-Klein MC, Beaurepaire E. (2006) Imaging lipid bodies in cells and tissues using third-harmonic generation microscopy. Nat. Methods; 3: 47–53.
Débarre D, Beaurepaire E. (2007) Quantitative characterization of biological liquids for third-harmonic generation microscopy. Biophys. J.; 92: 603–612.
Köckenberger W, DePanfilis C, Santoro D, Dahiva P, Rawsthorne S. (2004) High resolution NMR microscopy of plants and fungi. J. Microsc.; 214: 182–189.
Neuberger T, Sreenivasulu N, Rokitta M, Rolletschek H, Göbel C, Rutten T, Radchuk V, Feussner I, Wobus U, Jakob P, Webb A, Borisjuk L. (2008) Quantitative imaging of oil storage in developing crop seeds. Plant Biotechnol. J.; 6: 31–45
Ishida N, Koizumi M, Kano H. (2000) The NMR microscope: a unique and promising tool for plant science. Ann. Bot.; 86: 259–278.
Van der Weerd L, Claessens MAE, Ruttink T, Vergeldt FJ, Schaafsma TJ, Van As H. (2001) Quantitative NMR microscopy of osmotic stress responses in maize and pearl millet. J. Exp. Bot.; 52: 2333–2343.
Van As H. (2007) Intact plant MRI for the study of water relations, membrane, permeability, cell-to-cell and long distance water transport. J. Exp. Bot.; 58: 743–756
Huang AHC. (1992) Oil bodies and oleosins in seeds. Annu. Rev. Plant Physiol. Mol. Biol.; 43: 177–200.
Webb AG. (1997) Radiofrequency microcoils in magnetic resonance. Prog. NMR Spectrosc.; 31: 1–42.
Acknowledgments
We are grateful to P. Jakob and U. Wobus for support and critical discussion, G. Melkus, A. Purea, and V. C. Behr for help with the MRI. I. Feussner and C. Göbel are acknowledged for help with gas chromatography. Our special thanks to T. Rutten for electron microscopy, U. Tiemann and K. Lipfert for artwork and A. Stegmann for excellent technical assistance. We are grateful for funding by the Deutsche Forschungsgemeinschaft (Project number RO 2411/2–1/2–2 and BO 1917/2–1), and the Federal Ministry of Education and Research (BMBF; GABI SEED II grant). Andrew G. Webb and Thomas Neuberger acknowledge support of the Alexander von Humboldt Stiftung, Wolfgang Paul Preis.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Humana Press, a part of Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Neuberger, T., Rolletschek, H., Webb, A., Borisjuk, L. (2009). Non-invasive Mapping of Lipids in Plant Tissue Using Magnetic Resonance Imaging. In: Armstrong, D. (eds) Lipidomics. Methods in Molecular Biology, vol 579. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-60761-322-0_24
Download citation
DOI: https://doi.org/10.1007/978-1-60761-322-0_24
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-60761-321-3
Online ISBN: 978-1-60761-322-0
eBook Packages: Springer Protocols