Summary
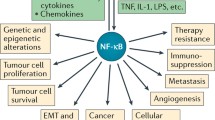
Activation of the transcription factor, nuclear factor of κB (NF-κB), has recently emerged as the critical link between inflammation and cancer. NF-κB is activated by many signals including DNA damage and microbial pathogen recognition. Although microbial infections account for an estimated 15% of all cancer related deaths, NF-κB is constitutively activated in various types of cancer as a direct result of chromosomal translocations, deletions and mutations affecting both NF-κB and its regulators. NF-κB exerts the transcriptional activation and repression of inflammatory and immune response genes as well as genes mediating cell survival and proliferation, and thus stands to impact various cellular processes. The decision is made through the integration of the pathway of NF-κB activation with a complex array of cell signaling networks that are at present poorly understood. Here, two methods are presented, protein co-immunoprecipitation and subcellular co-localization by immunofluorescence microscopy, to help investigations into the links between NF-κB-induced inflammation and cancer. Understanding the details of these pathways will aid in the development of new diagnostic and therapeutic applications.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Medzhitov, R. (2007) Recognition of microorganisms and activation of the immune response. Nature 449, 819–826.
Karin, M., and Greten, F.R. (2005) NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 5, 749–759.
Balkwill, F., and Mantovani, A. (2001) Inflammation and cancer: back to Virchow? Lancet 357, 539–545.
Kuper, H., Adami, H.O., and Trichopoulos, D. (2000) Infections as a major preventable cause of human cancer. J Intern Med 248, 171–183.
Coussens, L.M., and Werb, Z. (2002) Inflammation and cancer. Nature 420, 860–867.
Karin, M., Cao, Y., Greten, F.R., and Li, Z.W. (2002) NF-kappaB in cancer: from innocent bystander to major culprit. Nat Rev Cancer 2, 301–310.
Hayden, M.S., and Ghosh, S. (2004) Signaling to NF-kappaB. Genes Dev 18, 2195–2224.
Janssens, S., and Tschopp, J. (2006) Signals from within: the DNA-damage-induced NF-kappaB response. Cell Death Differ 13, 773–784.
Karin, M., and Ben-Neriah, Y. (2000) Phos-phorylation meets ubiquitination: the control of NF-[kappa]B activity. Annu Rev Immunol 18, 621–663.
Wood, L.D., Parsons, D.W., Jones, S., Lin, J., Sjoblom, T., Leary, R.J., Shen, D., Boca, S.M., Barber, T., Ptak, J., Silliman, N., Szabo, S., Dezso, Z., Ustyanksky, V., Nikolskaya, T., Nikol-sky, Y., Karchin, R., Wilson, P.A., Kaminker, J.S., Zhang, Z., Croshaw, R., Willis, J., Daw-son, D., Shipitsin, M., Willson, J.K., Sukumar, S., Polyak, K., Park, B.H., Pethiyagoda, C.L., Pant, P.V., Ballinger, D.G., Sparks, A.B., Har-tigan, J., Smith, D.R., Suh, E., Papadopoulos, N., Buckhaults, P., Markowitz, S.D., Parmi-giani, G., Kinzler, K.E., Velculescu, V.E., and Vogelstein, B. (2007) The genomic landscapes of human breast and colorectal cancers. Science 318, 1108–1113.
Boehm, J.S., Zhao, J.J., Yao, J., Kim, S.Y., Firestein, R., Dunn, I.F., Sjostrom, S.K., Garr-away, L.A., Weremowicz, S., Richardson, A.L., Greulich, H., Stewart, C.J., Mulvey, L.A., Shen, R.R., Ambrogio, L., Hirozane-Kishikawa, T., Hill, D.E., Vidal, M., Meyerson, M., Grenier, J.K., Hinkle, G., Root, D.E., Roberts, T.M., Lander, E.S., Polyak, K., and Hahn, W.C. (2007) Integrative genomic approaches identify IKBKE as a breast cancer oncogene. Cell 129, 1065–1079.
Kinzler, K.W., and Vogelstein, B. (1996) Lessons from hereditary colorectal cancer. Cell 87, 159–170.
Hayden, M.S., West, A.P., and Ghosh, S. (2006) SnapShot: NF-kappaB signaling pathways. Cell 127, 1286–1287.
Perkins, N.D. (2007) Integrating cell-signalling pathways with NF-kappaB and IKK function. Nat Rev Mol Cell Biol 8, 49–62.
Shimazu, R., Akashi, S., Ogata, H., Nagai, Y., Fukudome, K., Miyake, K., and Kimoto, M. (1999) MD-2, a molecule that confers lipopol-ysaccharide responsiveness on Toll-like receptor 4. J Exp Med 189, 1777–1782.
Alexopoulou, L., Holt, A.C., Medzhitov, R., and Flavell, R.A. (2001) Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 413, 732–738.
Takeuchi, O., Hoshino, K., Kawai, T., Sanjo, H., Takada, H., Ogawa, T., Takeda, K., and Akira, S. (1999) Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11, 443–451.
Takeuchi, O., Kaufmann, A., Grote, K., Kawai, T., Hoshino, K., Morr, M., Muhlradt, P.F., and Akira, S. (2000) Cutting edge: preferentially the R-stereoisomer of the mycoplasmal lipopeptide macrophage-activating lipopeptide-2 activates immune cells through a toll-like receptor 2- and MyD88-dependent signaling pathway. J Immunol 164, 554–557.
Hirschfeld, M., Ma, Y., Weis, J.H., Vogel, S.N., and Weis, J.J. (2000) Cutting edge: repurifi-cation of lipopolysaccharide eliminates signaling through both human and murine toll-like receptor 2. J Immunol 165, 618–622.
Muzio, M., Bosisio, D., Polentarutti, N., D'Amico, G., Stoppacciaro, A., Mancinelli, R., van't Veer, C., Penton-Rol, G., Ruco, L.P., Allavena, P., and Mantovani, A. (2000) Differential expression and regulation of toll-like receptors (TLR) in human leukocytes: selective expression of TLR3 in dendritic cells. J Immunol 164, 5998–6004.
Miyake, K., Yamashita, Y., Ogata, M., Sudo, T., and Kimoto, M. (1995) RP105, a novel B cell surface molecule implicated in B cell activation, is a member of the leucine-rich repeat protein family. J Immunol 154, 3333–3340.
Miyake, K., Shimazu, R., Kondo, J., Niki, T., Akashi, S., Ogata, H., Yamashita, Y., Miura, Y., and Kimoto, M. (1998) Mouse MD-1, a molecule that is physically associated with RP105 and positively regulates its expression. J Immunol 161, 1348–1353.
Rock, F.L. Hardiman, G., Timans, J.C., Kastelein, R.A., and Bazan, J.F. (1998) A family of human receptors structurally related to Drosophila Toll. Proc Natl Acad Sci U S A 95, 588–593.
Akira, S., Uematsu, S., and Takeuchi, O. (2006) Pathogen recognition and innate immunity. Cell 124, 783–801.
Underhill, D.M., Ozinsky, A., Hajjar, A.M., Stevens, A., Wilson, C.B., Bassetti, M., and Aderem, A. (1999) The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401, 811–815.
Barton, G.M., Kagan, J.C., and Medzhitov, R. (2006) Intracellular localization of Toll-like receptor 9 prevents recognition of self DNA but facilitates access to viral DNA. Nat Immunol 7, 49–56.
Flo, T.H., Halaas, O., Torp, S., Ryan, L., Lien, E., Dybdahl, B., Sundan, A., and Espe-vik, T. (2001) Differential expression of Tolllike receptor 2 in human cells. J Leukoc Biol 69, 474–481.
Hausmann, M., Kiessling, S., Mestermann, S., Webb, G., Spottl, T., Andus, T., Scholmerich, J., Herfarth, H., Ray, K., Falk, W., and Rogler, G (2002) Toll-like receptors 2 and 4 are up-regulated during intestinal inflammation. Gastroenterology 122, 1987–2000.
Li, C., Wang, Y., Gao, L., Zhang, J., Shao, J., Wang, S., Feng, W., Wang, X., Li, M., and Chang, Z. (2002) Expression of toll-like receptors 2 and 4 and CD14 during differentiation of HL-60 cells induced by phorbol 12-myr-istate 13-acetate and 1 alpha, 25-dihydroxy-vitamin D(3). Cell Growth Differ 13, 27–38.
Mueller, T., Terada, T., Rosenberg, I.M., Shi-bolet, O., and Podolsky, D.K. (2006) Th2 cytokines down-regulate TLR expression and function in human intestinal epithelial cells. J Immunol 176, 5805–5814.
Akashi, S., Shimazu, R., Ogata, H., Nagai, Y., Takeda, K., Kimoto, M., and Miyake, K. (2000) Cutting edge: cell surface expression and lipopolysaccharide signaling via the tolllike receptor 4-MD-2 complex on mouse peritoneal macrophages. J Immunol 164, 3471–3475.
Matsumoto, M., Funami, K., Tanabe, A., Oshiumi, H., Shingai, A., Seto, Y., Yamamoto, A., and Seya, T. (2003) Subcellular localization of Toll-like receptor 3 in human dendritic cells. J Immunol 171, 3154–3162.
Hoene, V., Peiser, M., and Wanner, R. (2006) Human monocyte-derived dendritic cells express TLR9 and react directly to the CpG-A oligo-nucleotide D19. J Leukoc Biol 80, 1328–1336.
Bergeron, J.J., Brenner, M.B., Thomas, D.Y., and Williams, D.B. (1994) Calnexin: a membrane-bound chaperone of the endoplasmic reticulum. Trends Biochem Sci 19, 124–128.
Molinari, M., Eriksson, K.K., Calanca, V., Galli, C., Cresswell, P. , Michalak, M., and Helenius (2004) Contrasting functions of cal-reticulin and calnexin in glycoprotein folding and ER quality control. Mol Cell 13, 125–135.
Muller-Taubenberger, A., Lupas, A.N., Li, H., Ecke, M., Simmeth, E., and Gerisch, G. (2001) Calreticulin and calnexin in the endoplasmic reticulum are important for phagocytosis. EMBO J 20, 6772–6782.
Ortmann, B., Copeman, J., Lehner, P.J., Sada-sivan, B., Herberg, J.A., Grandea, A.G., Rid-dell, S.R., Tampe, R., Spies, T., Trowsdale, J., and Cresswell, P. (1997) A critical role for tapasin in the assembly and function of mul-timeric MHC class I-TAP complexes. Science 277, 1306–1309.
Hughes, E.A., and Cresswell, P. (1998) The thiol oxidoreductase ERp57 is a component of the MHC class I peptide-loading complex. Curr Biol 8, 709–712.
Sonnichsen, B., Lowe, M., Levine, T., Jamsa, E., Dirac-Svejstrup, B., and Warren, G. (1998) A role for giantin in docking COPI vesicles to Golgi membranes. J Cell Biol 140, 1013–1021.
Woodman, P.G. (2000) Biogenesis of the sorting endosome: the role of Rab5. Traffic 1, 695–701.
Gorvel, J.P., Chavrier, P. , Zerial, M., and Gruenberg, J. (1991) rab5 controls early endosome fusion in vitro. Cell 64, 915–925.
Nielsen, E., Christoforidis, S., Uttenweiler-Joseph, S., Miaczynska, M., Dewitte, F., Wilm, M., Hoflack, B., and Zerial, M. (2000) Rabenosyn-5, a novel Rab5 effector, is com-plexed with hVPS45 and recruited to endo-somes through a FYVE finger domain. J Cell Biol 151, 601–612.
Bivona, T.G., Wiener, H.H., Ahearn, I.M., Silletti, J., Chiu, V.K., and Philips, M.R. (2004) Rap1 up-regulation and activation on plasma membrane regulates T cell adhesion. J Cell Biol 164, 461–470.
Huynh, K.K., Eskelinen, E.L., Scott, C.C., Malevanets, A., Saftig, P., and Grinstein, S. (2007) LAMP proteins are required for fusion of lysosomes with phagosomes. EMBO J 26, 313–324.
Harter, C., and Mellman, I. (1992) Transport of the lysosomal membrane glycoprotein lgp120 (lgp-A) to lysosomes does not require appearance on the plasma membrane. J Cell Biol 117, 311–325.
Granger, B.L., Green, S.A., Gabel, C.A., Howe, C.L., Mellman, I., and Helenius., A. (1990) Characterization and cloning of lgp110, a lysosomal membrane glycoprotein from mouse and rat cells. J Biol Chem 265, 12036–12043.
de Saint-Vis, B., Vincent, J., Vandenabeele, S., Vanbervliet, B., Pin, J.J., Ait-Yahia, S., Patel, S., Mattei, M.G., Banchereau, J., Zurawski, S., Davoust, J., Caux, C., and Lebecque, S. (1998) A novel lysosome-associated membrane glyco-protein, DC-LAMP, induced upon DC maturation, is transiently expressed in MHC class II compartment. Immunity 9:325–336.
Blander, J.M., and Medzhitov, R. (2004) Regulation of phagosome maturation by signals from toll-like receptors. Science 304, 1014–1018.
Kang, B.H., Plescia, J., Dohi, T., Rosa, J., Doxsey, S.J., and Altieri, D.C. (2007) Regulation of tumor cell mitochondrial homeosta-sis by an organelle-specific Hsp90 chaperone network. Cell 131, 257–270.
Blander, J.M., and Medzhitov, R. (2006) Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Blander, J.M. (2009). Analysis of the TLR/NF-κB Pathway in Antigen-Presenting Cells in Malignancies Promoted by Inflammation. In: Kozlov, S.V. (eds) Inflammation and Cancer. Methods in Molecular Biology™, vol 512. Humana Press. https://doi.org/10.1007/978-1-60327-530-9_7
Download citation
DOI: https://doi.org/10.1007/978-1-60327-530-9_7
Publisher Name: Humana Press
Print ISBN: 978-1-60327-529-3
Online ISBN: 978-1-60327-530-9
eBook Packages: Springer Protocols