Abstract
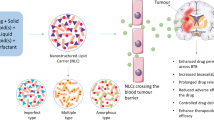
Brain disorders, a diverse range of conditions comprising of neurological and psychiatric conditions, are the leading cause of disability, severely affect the quality of life, and in many cases lead to mortality. The prime challenge in treatment of brain disorders is to deliver therapeutics by overcoming the blood–brain barrier (BBB), a unique anatomical and physiological barrier which restricts the passage of a number of molecules, proteins, and cells from the bloodstream. Lipid nanoparticles have emerged as promising drug delivery systems primarily because of biodegradability, low toxicity potential, and the ability to cross physiological barriers especially the BBB even without surface modifications.
In this chapter we discuss the preparation and characterization of nanostructured lipid carriers of temozolomide, a chemotherapeutic drug. Evaluation of pharmacokinetics and biodistribution of the nanocarrier system in rats revealed improved delivery of the chemotherapeutic agent to the brain with the potential of lesser side effects.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jaul E, Barron J (2017) Age-related diseases and clinical and public health implications for the 85 years old and over population. Front Public Health 5:335
Dong X (2018) Current strategies for brain drug delivery. Theranostics 8:1481–1493
Banks WA (2016) From blood-brain barrier to blood-brain interface: new opportunities for CNS drug delivery. Nat Rev Drug Discov 15:275–292
Allhenn D, Shetab Boushehri MA, Lamprecht A (2012) Drug delivery strategies for the treatment of malignant gliomas. Int J Pharm 436:299–310
Wolinsky JB, Colson YL, Grinstaff MW (2012) Local drug delivery strategies for cancer treatment: gels, nanoparticles, polymeric films, rods, and wafers. J Control Release 159:14–26
Saraiva C, Praça C, Ferreira R et al (2016) Nanoparticle-mediated brain drug delivery: overcoming blood–brain barrier to treat neurodegenerative diseases. J Control Release 235:34–47
Martins S, Sarmento B, Ferreira DC et al (2007) Lipid-based colloidal carriers for peptide and protein delivery - liposomes versus lipid nanoparticles. Int J Nanomedicine 2:595–607
Sarmento B, Martins S, Ferreira D et al (2007) Oral insulin delivery by means of solid lipid nanoparticles. Int J Nanomedicine 2:743–749
Neves AR, Lúcio M, Martins S et al (2013) Novel resveratrol nanodelivery systems based on lipid nanoparticles to enhance its oral bioavailability. Int J Nanomedicine 8:177–187
Shah B, Khunt D, Bhatt H et al (2015) Application of quality by design approach for intranasal delivery of rivastigmine loaded solid lipid nanoparticles: effect on formulation and characterization parameters. Eur J Pharm Sci 78:54–66
Finn JD, Smith AR, Patel MC et al (2018) A single administration of CRISPR/Cas9 lipid nanoparticles achieves robust and persistent in vivo genome editing. Cell Rep 22:2227–2235
Apaolaza PS, del Pozo-Rodríguez A, Solinís MA et al (2016) Structural recovery of the retina in a retinoschisin-deficient mouse after gene replacement therapy by solid lipid nanoparticles. Biomaterials 90:40–49
Khosa A, Reddi S, Saha RN (2018) Nanostructured lipid carriers for site-specific drug delivery. Biomed Pharmacother 103:598–613
Gastaldi L, Battaglia L, Peira E et al (2014) Solid lipid nanoparticles as vehicles of drugs to the brain: current state of the art. Eur J Pharm Biopharm 87:433–444
Wesolowski JR, Rajdev P, Mukherji SK (2010) Temozolomide (Temodar). Am J Neuroradiol 31:1383
Farid RM, El-Salamouni NS, El-Kamel AH et al (2017) Lipid-based nanocarriers for ocular drug delivery. In: Andronescu E, Grumezescu AM (eds) Nanostructures for drug delivery, 1st edn. William Andrew, New York
Choulis NH (2011) Miscellaneous drugs, materials, medical devices, and techniques. In: Aronson JK (ed) Side effects of drugs annual, vol 33, 1st edn. Elsevier Science, New York
Moloughney JG, Weisleder N (2012) Poloxamer 188 (P188) as a membrane resealing reagent in biomedical applications. Recent Pat Biotechnol 6:200–211
Moghimi SM, Hunter AC (2000) Poloxamers and poloxamines in nanoparticle engineering and experimental medicine. Trends Biotechnol 18:412–420
Sinhmar GK, Shah NN, Chokshi NV et al (2018) Process, optimization and characterization of budesonide-loaded nanostructured lipid carriers for the treatment of inflammatory bowel disease. Drug Dev Ind Pharm 44:1078–1089
Rocha KAD, Krawczyk-Santos AP, Andrade LM et al (2017) Voriconazole-loaded nanostructured lipid carriers (NLC) for drug delivery in deeper regions of the nail plate. Int J Pharm 531:292–298
Cirri M, Maestrini L, Maestrelli F et al (2018) Design, characterization and in vivo evaluation of nanostructured lipid carriers (NLC) as a new drug delivery system for hydrochlorothiazide oral administration in pediatric therapy. Drug Deliv 25:1910–1921
Amasya G, Badilli U, Aksu B et al (2016) Quality by design case study 1: design of 5-fluorouracil loaded lipid nanoparticles by the w/o/w double emulsion-solvent evaporation method. Eur J Pharm Sci 84:92–102
Schubert MA, Müller-Goymann CC (2003) Solvent injection as a new approach for manufacturing lipid nanoparticles—evaluation of the method and process parameters. Eur J Pharm Biopharm 55:125–131
Oliveira DRB, Michelon M, de Figueiredo Furtado G et al (2016) β-Carotene-loaded nanostructured lipid carriers produced by solvent displacement method. Food Res Int 90:139–146
Gomes GVL, Sola MR, Rochetti AL, Fukumasu H et al (2019) β-Carotene and α-tocopherol coencapsulated in nanostructured lipid carriers of murumuru (Astrocaryum murumuru) butter produced by phase inversion temperature method: characterisation, dynamic in vitro digestion and cell viability study. J Microencapsul 36:43–52
Liu C-H, Wu C-T (2010) Optimization of nanostructured lipid carriers for lutein delivery. Colloids Surf A Physicochem Eng Asp 353:149–156
Charcosset C, El-Harati A, Fessi H (2005) Preparation of solid lipid nanoparticles using a membrane contactor. J Control Release 108:112–120
Schwarz JC, Weixelbaum A, Pagitsch E et al (2012) Nanocarriers for dermal drug delivery: influence of preparation method, carrier type and rheological properties. Int J Pharm 437:83–88
Moghimi SM, Hunter AC, Andresen TL (2012) Factors controlling nanoparticle pharmacokinetics: an integrated analysis and perspective. Annu Rev Pharmacol Toxicol 52:481–503
Li S-D, Huang L (2008) Pharmacokinetics and biodistribution of nanoparticles. Mol Pharm 5:496–504
Betzer O, Shilo M, Opochinsky R et al (2017) The effect of nanoparticle size on the ability to cross the blood–brain barrier: an in vivo study. Nanomedicine (Lond) 12:1533–1546
Hirst TC, Vesterinen HM, Sena ES et al (2013) Systematic review and meta-analysis of temozolomide in animal models of glioma: was clinical efficacy predicted? Br J Cancer 108:64
Agency EMEA (2011) CHMP assessment report Temozolomide Sun, Procedure No. EMEA/H/C/002198. https://www.ema.europa.eu/en/documents/assessment-report/temozolomide-sun-epar-public-assessment-report_en.pdf 1–20. Accessed 21 April 2019
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Khosa, A., Krishna, K.V., Dubey, S.K., Saha, R.N. (2020). Lipid Nanocarriers for Enhanced Delivery of Temozolomide to the Brain. In: Jain, K. (eds) Drug Delivery Systems. Methods in Molecular Biology, vol 2059. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9798-5_15
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9798-5_15
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9797-8
Online ISBN: 978-1-4939-9798-5
eBook Packages: Springer Protocols