Abstract
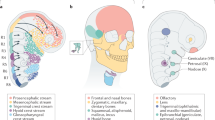
For decades, the quail-chick system has been a gold standard approach to track cells and their progenies over complex morphogenetic movements and long-range migrations as well as to unravel their dialogue and interplays in varied processes of cell induction. More specifically, this model became decisive for the systematic explorations of the neural crest and its lineages and allowed a tremendous stride in understanding the wealth and complexity of this fascinating cell population. Much of our knowledge on craniofacial morphogenesis and vertebrate organogenesis was first gained in avian chimeras and later extended to mammalian models and humans. In addition, this system permits tissue and gene manipulations to be performed at once in the same cell population. Through the use of in ovo electroporation, this model became tractable for functional genomics, hence being even more resourceful for functional studies. Due to the ease of access and the possibility to combine micromanipulation of tissue anlagen and gene expression, this model offers the prospect of decrypting instructive versus permissive tissue interactions, to identify and crack the molecular codes underlying cell positioning and differentiation, with an unparalleled spatiotemporal accuracy.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
His W (1868) Untersuchungen über die erste Anlage des Wirbeltierleibes. Die erste Entwickelung des Hühnchens im Ei. FCW Vogel, Leipzig
Hörstadius S (1950) The neural crest: its properties and derivatives in the light of experimental research. Oxford University Press, London
Le Douarin NM (1969) Particularitiés du noyau interphasique chez la caille japonaise (Coturnix coturnix japonica). Utilisation de ces particularités comme “marquage biologique” dans les recherches sur les interactions tissulaires et les migrations cellulaires au cours de l’ontogenèse. Bull Biol Fr Belg 103:435–452
Le Lièvre C, Le Douarin N (1974) Ectodermic origin of the derma of the face and neck, demonstrated by interspecific combinations in the bird embryo. C R Acad Sci Hebd Seances Acad Sci D 278:517–520
Le Lièvre C, Le Douarin NM (1975) Mesenchymal derivatives of the neural crest: analysis of chimaeric quail and chick embryos. J Embryol Exp Morphol 34:125–154
D'Amico-Martel A, Noden DM (1983) Contributions of placodal and neural crest cells to avian cranial peripheral ganglia. Am J Anat 166:445–468
Couly GF, Coltey PM, Le Douarin NM (1993) The triple origin of skull in higher vertebrates—a study in quail-chick chimeras. Development (Cambridge, UK) 117:409–429
Couly G, Grapin-Botton A, Coltey P, Le Douarin NM (1996) The regeneration of the cephalic neural crest, a problem revisited: the regenerating cells originate from the contralateral or from the anterior and posterior neural fold. Development 122:3393–3407
Koentges G, Lumsden A (1996) Rhombencephalic neural crest segmentation is preserved throughout craniofacial ontogeny. Development 122:3229–3242
Etchevers HC, Couly G, Vincent C, Le Douarin NM (1999) Anterior cephalic neural crest is required for forebrain viability. Development 26:3533–3543
Etchevers HC, Vincent C, Le Douarin NM, Couly GF (2001) The cephalic neural crest provides pericytes and smooth muscle cells to all blood vessels of the face and forebrain. Development 128:1059–1068
Gans C, Northcutt RG (1983) Neural crest and the origin of vertebrates. A new head. Science 220:268–274
Hall BK (2000) The neural crest as a fourth germ layer and vertebrate as quadroblastic not triploblastic. Evol Dev 2:3–5
Muramatsu T, Shibata O, Ryoki S, Ohmori Y, Okumura J (1997) Foreign gene expression in the mouse testis by localized in vivo gene transfer. Biochem Biophys Res Commun 233:45–49
Creuzet SE (2009a) Regulation of pre-otic brain development by the cephalic neural crest. Proc Natl Acad Sci U S A 106:15774–15779
Creuzet S, Schuler B, Couly G, Le Douarin NM (2004) Reciprocal relationships between Fgf8 and neural crest cells in facial and forebrain development. Proc Natl Acad Sci USA 101:4843–4847
Creuzet S, Martinez S, Le Douarin NM (2006) The cephalic neural crest exerts a critical effect on forebrain and midbrain development. Proc Natl Acad Sci U S A 103:14033–14038
Creuzet SE (2009b) Neural crest contribution to forebrain development. Development of patterning of the vertebrate forebrain. Sem Cell Dev Biol 20:751–759
Cases O, Perea-Gomez A, Aguiar DP, Nykjaer A, Lelièvre V, Amsellem S, Chandellier J, Pedersen G, Cereghini S, Collignon J, Verroust P, Umbhauer M, Madsen M, Riou JF, Creuzet SE, Kozyraki R (2013) Cubilin is required for FgF-promoted cell survival in the developing vertebrate head. J Biol Chem 288:16655–16670
Garcez RC, Le Douarin NM, Creuzet SE (2014) Combinatorial activity of Six1-2-4 genes in cephalic neural crest cells controls craniofacial and brain development. Cell Mol Life Sci 71:2149–2164
Aguiar DP, Sghari S, Creuzet S (2014) The facial neural crest controls fore- and midbrain patterning by regulating Foxg1 expression through Smad1 activity. Development 141:2494–2505
Creuzet S, Viallet J, Ghawitian M, Thélu J, Alrajeh M, Costagliola F, Le Borgne M, Buchet-Poyau K, Aznar N, Buschlen S, Hosoya H, Thibert C, Billaud M (2016) LKB1 signaling in cephalic neural crest is essential for vertebrate head development. Dev Biol 418(2):283–296
Creuzet S, Couly G, Vincent C, Le Douarin NM (2002) Negative effect of Hox gene expression on the development of neural crest derived facial skeleton. Development (Cambridge, UK) 129:4301–4313
Le Douarin N, Dieterlen-Lièvre F, Creuzet S, Teillet M-A (2008) Quail–Chick transplantations. Methods Cell Biol 87:19–58
Conrad GW, Bee JA, Roche SM, Teillet M-A (1993) Fabrication of microscalpels by electrolysis of tungsten wire in a meniscus. J Neurosci Methods 50:123–127
Couly G, Creuzet S, Bennaceur S, Vincent C, Le Douarin NM (2002) Interactions between Hox-negative cephalic neural crest cells and the foregut endoderm in patterning the facial skeleton in the vertebrate head. Development 129:1061–1073
Ruhin B, Creuzet S, Vincent C, Benouaiche L, Le Douarin NM, Couly G (2003) Patterning of the hyoid cartilage depends upon signals arising from the ventral foregut endoderm. Dev Dyn 228:239–246
Lwigale PY, Schneider RA (2008) Other chimeras: quail-duck and mouse-chick. Methods Cell Biol 87:59–74
Schneider RA (1999) Neural crest can form cartilages normally derived from mesoderm during development of the avian head skeleton. Dev Biol 208:441–455
Fish JL, Schneider RA (2014) Assessing species-specific contributions to craniofacial development using quail-duck chimeras. J Vis Exp (87):51534
Hamburger V, Hamilton HL (1951) A series of normal stages in the development of chick embryo. J Morphol 88:49–92
Zacchei AM (1961) Lo sviluppo embrionale della quaglia giaponese. Archivi Anatomica 66:36–62
Eyal-Giladi H, Kochav S (1976) From cleavage to primitive streak formation: a complementary normal table and a new look at the first stages of the development of the chick. I. General morphology. Dev Biol 49:321–337
Ainsworth SJ, Stanley RL, Evans DJ (2009) Developmental stages of the Japanese quail. J Anat 216:3–15
Couly GF, Le Douarin NM (1985) Mapping of the early neural primordium in quail-chick chimeras. I. Developmental relationships between placodes, facial ectoderm, and prosencephalon. Dev Biol 110:422–439
Couly GF, Le Douarin NM (1987) Mapping of the early neural primordium in quail-chick chimeras. II. The prosencephalic neural plate and neural folds: implications for the genesis of cephalic human congenital abnormalities. Dev Biol 120:198–214
Couly GF, Le Douarin NM (1988) The fate map of the cephalic neural primordium at the presomitic to the 3-somite stage in the avian embryo. Development (Cambridge, UK) 103(Suppl):101–113
Catala M, Ziller C, Lapointe F, Le Douarin NM (2000) The developmental potentials of the caudalmost part of the neural crest are restricted to melanocytes and glia. Mech Dev 95:77–87
Feulgen R, Rossenbeck H (1924) Mikroskopisch-chemischer Nachweiss einer Nucleinsaüre von Typus der Thymonucleinsaüre und die darauf beruhende elektive Farbung von Zellkernen in microskopischen Präparaten. Hoppe Seylers Z Physiol Chem 135:203–252
Pekarik V, Bourikas D, Miglino N, Joset P, Stoeckli E (2003) Screening for gene function in chicken embryo using RNAi and electroporation. Nat Biotechnol 21:93–96
Stoeckli ET (2005) RNAi in avian embryos. In: Gannon GH (ed) RNAi: a guide of gene silencing. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Ogino H, Yasuda K (1996) Induction of lens differentiation by activation of a bZIP transcription factor, L-Maf. Science 280(5360):115–118
Momose T, Tonegawa A, Takeuchi J, Ogawa H, Umesono K, Yasuda K (1999) Efficient targeting of gene expression in chick embryos by microelectroporation. Develop Growth Differ 41:335–344
Acknowledgments
This work is supported by the Centre National de la Recherche Scientifique (CNRS) and the Fondation pour la Recherche Médicale (FRM-, DEQ20170839116 grant). MA and ZV are recipients of fellowships from the FRM (FRM-MedicoSci program) and the National Institutes of Health (Richard A. Schneider’s lab, University of California, San Francisco, USA), respectively.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Alrajeh, M., Vavrusova, Z., Creuzet, S.E. (2019). Deciphering the Neural Crest Contribution to Cephalic Development with Avian Embryos. In: Schwarz, Q., Wiszniak, S. (eds) Neural Crest Cells. Methods in Molecular Biology, vol 1976. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-9412-0_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9412-0_5
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-9411-3
Online ISBN: 978-1-4939-9412-0
eBook Packages: Springer Protocols