Abstract
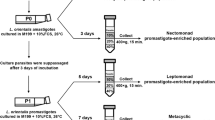
This chapter describes, in detail, the method our laboratory developed to differentiate L. donovani promastigotes into amastigotes in a host-free culture. This method is based on previous observations that Leishmania promastigotes can combine two environmental signals, typical to lysosomes, acidic pH (~5.5) and body temperature (37 °C), into a signal that induces differentiation. Based on this concept, we have modified medium 199 to make it into an amastigote-specific medium. Shifting promastigotes to this medium, followed by incubation in a CO2 incubator, induced differentiation. Axenic amastigotes reach maturation within 5 days, resembling the time it takes in vivo. This chapter provides a complete protocol that should be useful for both Old and New World species of Leishmania.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zilberstein D (2018) Nutrient transport and sensing as pharmacological targets for leishmaniasis. In: Rivas L, Carmen G (eds) Drug discovery for leishmaniasis, Drug discovery, vol 60. The Royal Society of Chemistry, Cambridge, UK, pp 282–296
Harms E, Gochman N, Schneider JA (1981) Lysosomal pool of free-amino acids. Biochem Biophys Res Commun 99(3):830–836
Burchmore RJ, Barrett MP (2001) Life in vacuoles--nutrient acquisition by Leishmania amastigotes. Int J Parasitol 31(12):1311–1320
Zilberstein D, Shapira M (1994) The role of pH and temperature in the development of Leishmania parasites. Annu Rev Microbiol 48:449–470. https://doi.org/10.1146/annurev.mi.48.100194.002313
Zilberstein D, Blumenfeld N, Liveanu V, Gepstein A, Jaffe CL (1991) Growth at acidic pH induces an amastigote stage-specific protein in Leishmania promastigotes. Mol Biochem Parasitol 45(1):175–178. doi:0166-6851(91)90040-D [pii]
Doyle PS, Engel JC, Pimenta PF, da Silva PP, Dwyer DM (1991) Leishmania donovani: long-term culture of axenic amastigotes at 37 degrees C. Exp Parasitol 73(3):326–334. https://doi.org/0014-4894(91)90104-5 [pii]
Barak E, Amin-Spector S, Gerliak E, Goyard S, Holland N, Zilberstein D (2005) Differentiation of Leishmania donovani in host-free system: analysis of signal perception and response. Mol Biochem Parasitol 141(1):99–108
Saar Y, Ransford A, Waldman E, Mazareb S, Amin-Spector S, Plumblee J, Turco SJ, Zilberstein D (1998) Characterization of developmentally-regulated activities in axenic amastigotes of Leishmania donovani. Mol Biochem Parasitol 95(1):9–20
Bates PA, Robertson CD, Tetley L, Coombs GH (1992) Axenic cultivation and characterization of Leishmania mexicana amastigote-like forms. Parasitology 105(Pt 2):193–202
Debrabant A, Joshi MB, Pimenta PF, dwyer DM (2004) Generation of Leishmania donovani axenic amastigotes: their growth and biological characteristics. Int J Parasitol 34(2):205–217
Goyard S, Segawa H, Gordon J, Showalter M, Duncan R, Turco SJ, Beverley SM (2003) An in vitro system for developmental and genetic studies of Leishmania donovani phosphoglycans. Mol Biochem Parasitol 130(1):31–42
Zilberstein D (2008) Physiological and biochemical aspects of Leishmania develpment. In: Myler PJ, Fasel N (eds) Leishmania after the genome: biology and control. Horizon Scientific Press and Caiser Academic Press, New York, pp 107–122
Rosenzweig D, Smith D, Opperdoes F, Stern S, Olafson RW, Zilberstein D (2008) Retooling Leishmania metabolism: from sand fly gut to human macrophage. FASEB J 22(2):590–602. https://doi.org/10.1096/fj.07-9254com
Bachmaier S, Witztum R, Tsigankov P, Koren R, Boshart M, Zilberstein D (2016) Protein kinase a signaling during bidirectional axenic differentiation in Leishmania. Int J Parasitol 46(2):75–82. https://doi.org/10.1016/j.ijpara.2015.09.003
Saxena A, Lahav T, Holland N, Aggarwal G, Anupama A, Huang Y, Volpin H, Myler PJ, Zilberstein D (2007) Analysis of the Leishmania donovani transcriptome reveals an ordered progression of transient and permanent changes in gene expression during differentiation. Mol Biochem Parasitol 152(1):53–65. https://doi.org/10.1016/j.molbiopara.2006.11.011. S0166-6851(06)00332-X [pii]
Mengeling BJ, Zilberstein D, Turco SJ (1997) Biosynthesis of Leishmania lipophosphoglycan: solubilization and partial characterization of the initiating mannosylphosphoryltransferase. Glycobiology 7(6):847–853
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Zilberstein, D., Nitzan Koren, R. (2019). Host-Free Systems for Differentiation of Axenic Leishmania. In: Clos, J. (eds) Leishmania. Methods in Molecular Biology, vol 1971. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-9210-2_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9210-2_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-9209-6
Online ISBN: 978-1-4939-9210-2
eBook Packages: Springer Protocols