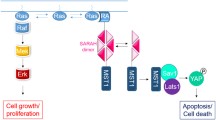
Abstract
The Bcl-2 proteins control cell death via interchanging interactions within the Bcl-2 family. Fluorescence lifetime imaging microscopy (FLIM) is used to detect Förster resonance energy transfer (FRET) between two fluorescent-fusion proteins in live cells. FLIM-FRET has been used to detect specific interactions and their disruption, for Bcl-2 family proteins. To date, this has been possible only in low throughput, due to the time required for serial data acquisition. We developed an automated optical system with eight parallel detectors for rapid and efficient data collection. Here we describe how to use this system for FLIM-FRET imaging of Bcl-2 family protein interactions in a 384-well plate format.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kale J, Osterlund EJ, Andrews DW (2018) BCL-2 family proteins: changing partners in the dance towards death. Cell Death Differ 25:65–80
Brahmbhatt H, Oppermann S, Osterlund EJ et al (2015) Molecular pathways: leveraging the BCL-2 interactome to kill cancer cells-mitochondrial outer membrane permeabilization and beyond. Clin Cancer Res 21:2671–2676
Certo M, Del Gaizo Moore V, Nishino M et al (2006) Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell 9:351–365
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Soderquist R S, Eastman A (2016) BCL2 inhibitors as anticancer drugs: a plethora of misleading BH3 mimetics. Mol Cancer Ther 15:2011–2017
Opydo-Chanek M, Gonzalo O, Marzo I (2017) Multifaceted anticancer activity of BH3 mimetics: current evidence and future prospects. Biochem Pharmacol 136:12–23
Vogler M (2014) Targeting BCL2-proteins for the treatment of solid tumours
Liu Q, Leber B, Andrews DW (2012) Interactions of pro-apoptotic BH3 proteins with anti-apoptotic Bcl-2 family proteins measured in live MCF-7 cells using FLIM FRET. Cell Cycle 11:3536–3542
Aranovich A, Liu Q, Collins T et al (2012) Differences in the mechanisms of proapoptotic BH3 proteins binding to Bcl-XL and Bcl-2 quantified in live MCF-7 cells. Mol Cell 45:754–763
Lakowicz JR (2006) Principles of fluorescence spectroscopy. Springer, New York
Becker W (2005) Advanced time-correlated single photon counting techniques. Springer, New York
Osterlund EJ, Liu Q, Andrews DW (2015) The use of FLIM-FRET for the detection of mitochondria-associated protein interactions. Methods Mol Biol 1264:395–419
Time-resolved Microscopy Instrumentation | ISS, http://www.iss.com/microscopy/index.html
Liu M, Jia M, Pan H et al (2014) Instrument response standard in time-resolved fluorescence spectroscopy at visible wavelength: quenched fluorescein sodium. j 68(5):577–583
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Osterlund, E.J., Hirmiz, N., Tardif, C., Andrews, D.W. (2019). Rapid Imaging of BCL-2 Family Interactions in Live Cells Using FLIM-FRET. In: Gavathiotis, E. (eds) BCL-2 Family Proteins. Methods in Molecular Biology, vol 1877. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8861-7_19
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8861-7_19
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8860-0
Online ISBN: 978-1-4939-8861-7
eBook Packages: Springer Protocols