Abstract
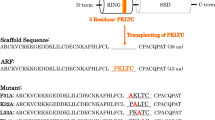
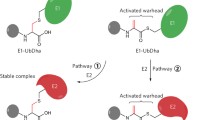
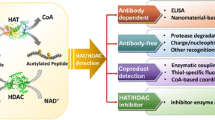
Ubiquitination is a sequential cascade consisting of ubiquitin-activating (E1), ubiquitin-conjugating (E2), and ubiquitin-ligating (E3) enzymes. It controls numerous processes such as protein degradation, DNA repair, and signal transduction pathways. E2 enzymes are associated with a variety of diseases such as leukemia, breast cancer, lung cancer, and colorectal cancer. To date, the monitoring of E2 activity for cancer diagnosis is challenging due to its intricate cascade reaction. To surmount this hurdle, we have recently developed a novel strategy for monitoring E2 activities. Here, we describe the concise machinery of ubiquitination with artificial RING finger proteins (ARFs) functioning as E3 enzymes. This machinery enables the simplified monitoring of E2 activities. Furthermore, our system combines a signal accumulation ion-sensitive field-effect transistor biosensor with ARFs, allowing for real-time monitoring of the pathological conditions of cancer cells. The present methodology may lead to novel diagnostic techniques for cancers.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Haas AL, Siepmann TJ (1997) Pathways of ubiquitin conjugation. FASEB J 11:1257–1268
Hershko A, Ciechanover A (1998) The ubiquitin system. Annu Rev Biochem 67:425–479
Weissman AM (2001) Themes and variations on ubiquitylation. Nat Rev Mol Cell Biol 2:169–178
Varshavsky A (1997) The ubiquitin system. Trends Biochem Sci 22:383–387
Deshaies RJ, Joazeiro CA (2009) RING domain E3 ubiquitin ligases. Annu Rev Biochem 78:399–434
Borden KL (2000) RING domains: master builders of molecular scaffolds? J Mol Biol 295:1103–1112
Freemont PS (1993) The RING finger. A novel protein sequence motif related to the zinc finger. Ann N Y Acad Sci 684:174–192
Takenokuchi M, Miyamoto K, Saigo K, Taniguchi T (2015) Bortezomib causes ER stress-related death of acute promyelocytic leukemia cells through excessive accumulation of PML–RARA. Anticancer Res 35:3307–3316
Ueki T, Park JH, Nishidate T, Kijima K, Hirata K, Nakamura Y, Katagiri T (2009) Ubiquitination and downregulation of BRCA1 by ubiquitin-conjugating enzyme E2T overexpression in human breast cancer cells. Cancer Res 69(22):8752–8760
Snoek BC, de Wilt LH, Jansen G, Peters GJ (2013) Role of E3 ubiquitin ligases in lung cancer. World J Clin Oncol 4(3):58–69
Chen S, Chen Y, Hu C, Jing H, Cao Y, Liu X (2010) Association of clinicopathological features with UbcH10 expression in colorectal cancer. J Cancer Res Clin Oncol 136:419–426
Liani E, Eyal A, Avraham E, Shemer R, Szargel R, Berg D, Bornemann A, Riess O, Ross CA, Rott R, Engelender S (2004) Ubiquitylation of synphilin-1 and alpha-synuclein by SIAH and its presence in cellular inclusions and Lewy bodies imply a role in Parkinson’s disease. Proc Natl Acad Sci U S A 101:5500–5505
Kramer OH, Muller S, Buchwald M, Reichardt S, Heinzel T (2008) Mechanism for ubiquitylation of the leukemia fusion proteins AML1-ETO and PML-RARalpha. FASEB J 22:1369–1379
Lee JT, Wheeler TC, Li L, Chin LS (2008) Ubiquitination of alpha-synuclein by Siah-1 promotes alpha-synuclein aggregation and apoptotic cell death. Hum Mol Genet 17:906–917
Brown NG, Watson ER, Weissmann F, Jarvis MA, VanderLinden R, Grace CR, Frye JJ, Qiao R, Dube P, Petzold G, Cho SE, Alsharif O, Bao J, Davidson IF, Zheng JJ, Nourse A, Kurinov I, Peters JM, Stark H, Schulman BA (2014) Mechanism of polyubiquitination by human anaphase-promoting complex: RING repurposing for ubiquitin chain assembly. Mol Cell 56:246–260
Miyamoto K (2012) Ubiquitination of an artificial RING finger without a substrate and a tag. J Pept Sci 18:135–139
Miyamoto K, Togiya K (2010) The creation of the artificial RING finger from the cross-brace zinc finger by alpha-helical region substitution. Biochem Biophys Res Commun 394:972–975
Pascual J, Martinez-Yamout M, Dyson HJ, Wright PE (2000) Structure of the PHD zinc finger from human Williams-Beuren syndrome transcription factor. J Mol Biol 304:723–729
Matsuzawa S, Li C, Ni CZ, Takayama S, Reed JC, Ely KR (2003) Structural analysis of Siah1 and its interactions with Siah-interacting protein (SIP). J Biol Chem 278:1837–1840
Miyamoto K (2014) Structural model of ubiquitin transfer onto an artificial RING finger as an E3 ligase. Sci Rep 4:6574
Markson G, Kiel C, Hyde R, Brown S, Charalabous P, Bremm A, Semple J, Woodsmith J, Duley S, Salehi-Ashtiani K, Vidal M, Komander D, Serrano L, Lehner P, Sanderson CM (2009) Analysis of the human E2 ubiquitin conjugating enzyme protein interaction network. Genome Res 19:1905–1911
van Wijk SJ, de Vries SJ, Kemmeren P, Huang A, Boelens R, Bonvin AM, Timmers HT (2009) A comprehensive framework of E2-RING E3 interactions of the human ubiquitin-proteasome system. Mol Syst Biol 5:295
Miyamoto K, Sumida M, Yuasa-Sunagawa M, Saito K (2017) Highly sensitive detection of E2 activity in ubiquitination using an artificial RING finger. J Pept Sci 23:222–227
Gu Y, Bouwman P, Greco D, Saarela J, Yadav B, Jonkers J, Kuznetsov SG (2014) Suppression of BRCA1 sensitizes cells to proteasome inhibitors. Cell Death Dis 5:e1580
Niewerth D, Dingjan I, Cloos J, Jansen G, Kaspers G (2013) Proteasome inhibitors in acute leukemia. Expert Rev Anticancer Ther 13:327–337
Ying M, Zhou X, Zhong L, Lin N, Jing H, Luo P, Yang X, Song H, Yang B, He Q (2012) Bortezomib sensitizes human acute myeloid leukemia cells to all-trans-retinoic acid-induced differentiation by modifying the RARalpha/STAT1 axis. Mol Cancer Ther 12:195–206
Finkel R, Clark MA, Cubeddu LX (2009) Lippincott’s illustrated reviews: pharmacology, fourth edition pharmacology. Lippincott Williams & Wilkins, Philadelphia
van Ree JH, Jeganathan KB, Malureanu L, van Deursen JM (2010) Overexpression of the E2 ubiquitin-conjugating enzyme UbcH10 causes chromosome missegregation and tumor formation. J Cell Biol 188:83–100
Wu X, Zhang W, Font-Burgada J, Palmer T, Hamil AS, Biswas SK, Poidinger M, Borcherding N, Xie Q, Ellies LG, Lytle NK, Wu LW, Fox RG, Yang J, Dowdy SF, Reya T, Karin M (2014) Ubiquitin-conjugating enzyme Ubc13 controls breast cancer metastasis through a TAK1-p38 MAP kinase cascade. Proc Natl Acad Sci U S A 111:13870–13875
Brzovic PS, Rajagopal P, Hoyt DW, King MC, Klevit RE (2001) Structure of a BRCA1-BARD1 heterodimeric RING-RING complex. Nat Struct Biol 8:833–837
Katoh S, Hong C, Tsunoda Y, Murata K, Takai R, Minami E, Yamazaki T, Katoh E (2003) High precision NMR structure and function of the RING-H2 finger domain of EL5, a rice protein whose expression is increased upon exposure to pathogen-derived oligosaccharides. J Biol Chem 278:15341–15348
Kostic M, Matt T, Martinez-Yamout MA, Dyson HJ, Wright PE (2006) Solution structure of the Hdm2 C2H2C4 RING, a domain critical for ubiquitination of p53. J Mol Biol 363:433–450
Miyamoto K, Uechi A, Saito K (2017) The zinc finger domain of RING finger protein 141 reveals a unique RING fold. Protein Sci 26:1681–1686
Katoh S, Tsunoda Y, Murata K, Minami E, Katoh E (2005) Active site residues and amino acid specificity of the ubiquitin carrier protein-binding RING-H2 finger domain. J Biol Chem 280:41015–41024
Scheel H, Hofmann K (2003) No evidence for PHD fingers as ubiquitin ligases. Trends Cell Biol 13:285–287; author reply 287–288
Aravind L, Iyer LM, Koonin EV (2003) Scores of RINGS but no PHDs in ubiquitin signaling. Cell Cycle 2:123–126
Chakravarty S, Zeng L, Zhou MM (2009) Structure and site-specific recognition of histone H3 by the PHD finger of human autoimmune regulator. Structure 17:670–679
Pena PV, Davrazou F, Shi X, Walter KL, Verkhusha VV, Gozani O, Zhao R, Kutateladze TG (2006) Molecular mechanism of histone H3K4me3 recognition by plant homeodomain of ING2. Nature 442:100–103
Szargel R, Rott R, Eyal A, Haskin J, Shani V, Balan L, Wolosker H, Engelender S (2009) Synphilin-1A inhibits seven in absentia homolog (SIAH) and modulates alpha-synuclein monoubiquitylation and inclusion formation. J Biol Chem 284:11706–11716
Acknowledgments
This research was supported by A-STEP from Japan Science and Technology Agency (JST), a Grant-in-Aid for Scientific Research (KAKENHI 26430147), Takeda Science Foundation, Sanyo Chemical Industries Foundation, and Nakatani Foundation.
Author contributions
K.M. designed this study. K.M. and K.S. wrote the main manuscript text and prepared all the figures.
Competing interests
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Miyamoto, K., Saito, K. (2018). Design of a System for Monitoring Ubiquitination Activities of E2 Enzymes Using Engineered RING Finger Proteins. In: Liu, J. (eds) Zinc Finger Proteins. Methods in Molecular Biology, vol 1867. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8799-3_6
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8799-3_6
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8798-6
Online ISBN: 978-1-4939-8799-3
eBook Packages: Springer Protocols