Abstract
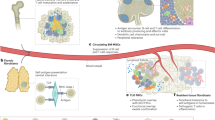
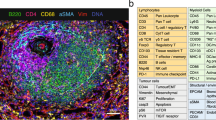
Genetically engineered mouse models (GEMMs), in which autochthonous tumors develop into advanced-stage disease in the presence of a functional immune system, have contributed significantly to the understanding of most types of cancer. Using a GEMM of lung adenocarcinoma, we have found that immune cells are present in complex, highly organized, lymph node (LN)-like structures known as the tumor-associated tertiary lymphoid structures (TA-TLS). TA-TLS have been characterized in human lung cancer patients, but not in animal tumor models, and hence remain untapped targets for therapeutic interventions. We have shown that TA-TLS emerge as a result of tumor growth and that therapeutically depleting regulatory T cells (Tregs) from TA-TLS results in tumor elimination. Hence, a strong antitumor immune response exists but is suppressed in TA-TLS. Here, we describe a high-throughput immunofluorescence (IF) analysis pipeline for visualization and quantification of TA-TLS. Imaging the relatively small size of TA-TLS within tumor-bearing lung lobes using confocal microscopy is a labor-intensive process that can take up to 1 month. We have optimized this process and reduced the time required per lung lobe to 1–2 weeks using automated microscopy methods. Combining IF with multicolor fluorescence-activated cell sorting (FACS), we are able to interrogate not only the size and location of TA-TLS but also the activation status of immune cells within these structures. Using these techniques, investigation of TLS in lung adenocarcinoma combines cutting-edge technological tools in cancer biology and immunology to interrogate a fundamental, but poorly understood, tumor-associated immune structure.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Dieu-Nosjean MC, Antoine M, Danel C et al (2008) Long-term survival for patients with non-small-cell lung cancer with intratumoral lymphoid structures. J Clin Oncol 26(27):4410–4417. https://doi.org/10.1200/JCO.2007.15.0284
Ogino S, Nosho K, Irahara N et al (2009) Lymphocytic reaction to colorectal cancer is associated with longer survival, independent of lymph node count, microsatellite instability, and CpG island methylator phenotype. Clin Cancer Res 15(20):6412–6420. https://doi.org/10.1158/1078-0432.CCR-09-1438
Hiraoka N, Ino Y, Yamazaki-Itoh R et al (2015) Intratumoral tertiary lymphoid organ is a favourable prognosticator in patients with pancreatic cancer. Br J Cancer 112(11):1782–1790. https://doi.org/10.1038/bjc.2015.145
Kroeger DR, Milne K, Nelson BH (2016) Tumor-infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-cell responses, and superior prognosis in ovarian cancer. Clin Cancer Res 22(12):3005–3015. https://doi.org/10.1158/1078-0432.CCR-15-2762
Germain C, Gnjatic S, Tamzalit F et al (2014) Presence of B cells in tertiary lymphoid structures is associated with a protective immunity in patients with lung cancer. Am J Respir Crit Care Med 189(7):832–844. https://doi.org/10.1164/rccm.201309-1611OC
Sanmamed MF, Chester C, Melero I, Kohrt H (2016) Defining the optimal murine models to investigate immune checkpoint blockers and their combination with other immunotherapies. Ann Oncol 27(7):1190–1198. https://doi.org/10.1093/annonc/mdw041
Politi K, Pao W (2011) How genetically engineered mouse tumor models provide insights into human cancers. J Clin Oncol 29(16):2273–2281. https://doi.org/10.1200/JCO.2010.30.8304
McFadden DG, Politi K, Bhutkar A et al (2016) Mutational landscape of EGFR-, MYC-, and Kras-driven genetically engineered mouse models of lung adenocarcinoma. Proc Natl Acad Sci U S A 113(42):E6409–E6417. https://doi.org/10.1073/pnas.1613601113
DuPage M, Mazumdar C, Schmidt LM et al (2012) Expression of tumour-specific antigens underlies cancer immunoediting. Nature 482(7385):405–409. https://doi.org/10.1038/nature10803
Joshi NS, Akama-Garren EH, Lu Y et al (2015) Regulatory T cells in tumor-associated tertiary lymphoid structures suppress anti-tumor T cell responses. Immunity 43(3):579–590. https://doi.org/10.1016/j.immuni.2015.08.006
Caswell DR, Chuang CH, Yang D et al (2014) Obligate progression precedes lung adenocarcinoma dissemination. Cancer Discov 4(7):781–789. https://doi.org/10.1158/2159-8290.CD-13-0862
Madisen L, Zwingman TA, Sunkin SM et al (2010) A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci 13(1):133–140. https://doi.org/10.1038/nn.2467
Nakamura N, Miyagi E, Murata S et al (2002) Expression of thyroid transcription factor-1 in normal and neoplastic lung tissues. Mod Pathol 15(10):1058–1067. https://doi.org/10.1097/01.MP.0000028572.44247.CF
Acknowledgements
We thank Professor Tyler Jacks (Massachusetts Institute of Technology) and Jacks lab members for their generous gift of reagents, advice, and time.
Funding: This work was supported by the National Cancer Institute K22 Transition Career Award.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Connolly, K.A., Nader, M., Joshi, N. (2018). Investigating Tumor-Associated Tertiary Lymphoid Structures in Murine Lung Adenocarcinoma. In: Dieu-Nosjean, MC. (eds) Tertiary Lymphoid Structures. Methods in Molecular Biology, vol 1845. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8709-2_15
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8709-2_15
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8708-5
Online ISBN: 978-1-4939-8709-2
eBook Packages: Springer Protocols